b3908fdfb3f63aeac18e576d57a9e512.ppt
- Количество слайдов: 27
 Integrated Medical Care for Children who are Deaf/HOH Quality Control Assessment Ellis Arjmand, MD, Ph. D Susan Wiley, MD Cincinnati Children’s Hospital Medical Center University of Cincinnati
Integrated Medical Care for Children who are Deaf/HOH Quality Control Assessment Ellis Arjmand, MD, Ph. D Susan Wiley, MD Cincinnati Children’s Hospital Medical Center University of Cincinnati
 Objectives • Present a rationale for interdisciplinary medical evaluations for children who are deaf/hard of hearing • Describe an integrated model of medical care for children who are deaf/hard of hearing • Present preliminary clinical outcomes seen in this setting
Objectives • Present a rationale for interdisciplinary medical evaluations for children who are deaf/hard of hearing • Describe an integrated model of medical care for children who are deaf/hard of hearing • Present preliminary clinical outcomes seen in this setting
 Premise Behind Clinic Model • Little consensus behind standardized medical evaluation (no evidence based guidelines) • Variable approach to work-up – Evaluation protocol often more standardized for children who are considering a cochlear implant • Etiology of hearing loss often unknown • Attempt to standardize care protocols • Provide quality of care to all children who are deaf/hoh
Premise Behind Clinic Model • Little consensus behind standardized medical evaluation (no evidence based guidelines) • Variable approach to work-up – Evaluation protocol often more standardized for children who are considering a cochlear implant • Etiology of hearing loss often unknown • Attempt to standardize care protocols • Provide quality of care to all children who are deaf/hoh
 Pre-clinic Data • CCHMC otology and audiology serve approximately 150 newly identified children with hearing loss annually • This includes approximately 50 cochlear implant evaluations per year • Prior to the clinic model, many children had long waits for appointments with subspecialists such as ophthalmology, neurology, developmental pediatrics, genetics • Estimate of clinician time possible based on baseline data
Pre-clinic Data • CCHMC otology and audiology serve approximately 150 newly identified children with hearing loss annually • This includes approximately 50 cochlear implant evaluations per year • Prior to the clinic model, many children had long waits for appointments with subspecialists such as ophthalmology, neurology, developmental pediatrics, genetics • Estimate of clinician time possible based on baseline data
 Clinic Needs • Service coordination (scheduling) • Buy-in from specialists (ophthalmology, genetics, etc) • Standardized evaluation protocol • Comprehensive evaluations for all children irrespective of degree and type of hearing loss • Improved access for families/patients • Outcome data/QA • Support from institution (start-up costs)
Clinic Needs • Service coordination (scheduling) • Buy-in from specialists (ophthalmology, genetics, etc) • Standardized evaluation protocol • Comprehensive evaluations for all children irrespective of degree and type of hearing loss • Improved access for families/patients • Outcome data/QA • Support from institution (start-up costs)
 Clinic Participants • Clinic coordination – One 0. 5 FTE scheduler – Schedules appointments, collects reports from evaluations • Medical Sub-specialists – – – Pediatric Otologists Genetics Ophthalmology Radiology/Laboratory Medicine Developmental Pediatrics (as needed) Neurology (as needed)
Clinic Participants • Clinic coordination – One 0. 5 FTE scheduler – Schedules appointments, collects reports from evaluations • Medical Sub-specialists – – – Pediatric Otologists Genetics Ophthalmology Radiology/Laboratory Medicine Developmental Pediatrics (as needed) Neurology (as needed)
 Clinic Participants • Allied Health Specialists – – Pediatric Audiologists Aural Rehabilitation Specialists Speech/Language Pathologists Social Work • Community Collaboration – Part C (Regional Infant Hearing Programs) – Part B (Local school programs)
Clinic Participants • Allied Health Specialists – – Pediatric Audiologists Aural Rehabilitation Specialists Speech/Language Pathologists Social Work • Community Collaboration – Part C (Regional Infant Hearing Programs) – Part B (Local school programs)
 Entry Criteria • Any child with a newly identified hearing loss irrespective of degree, side, or type of hearing loss • Any child with an identified hearing loss who needed collaborative approach to care (often at the discretion of ENT and Audiology)
Entry Criteria • Any child with a newly identified hearing loss irrespective of degree, side, or type of hearing loss • Any child with an identified hearing loss who needed collaborative approach to care (often at the discretion of ENT and Audiology)
 Referral System • Referral by community physicians, audiologists, or otolaryngologists • Scheduler contacts families to confirm demographic information, contact information • Nurse intake by telephone • Pediatric otologist reviews intake information, develops treatment plan • Scheduler contact families to arrange appointments
Referral System • Referral by community physicians, audiologists, or otolaryngologists • Scheduler contacts families to confirm demographic information, contact information • Nurse intake by telephone • Pediatric otologist reviews intake information, develops treatment plan • Scheduler contact families to arrange appointments
 Day of Appointments • Goal: Appointments range from occurring from a 1 -2 day period to within a 2 week period • Results shared with families by specialists • Brief summary of findings from specialist faxed to managing otologist
Day of Appointments • Goal: Appointments range from occurring from a 1 -2 day period to within a 2 week period • Results shared with families by specialists • Brief summary of findings from specialist faxed to managing otologist
 Clinic Collaboration • Follow-up appointment with otologist after evaluations are complete • Summary letter sent to family, other providers, and referring physician • Format for discussion among providers at weekly team meetings
Clinic Collaboration • Follow-up appointment with otologist after evaluations are complete • Summary letter sent to family, other providers, and referring physician • Format for discussion among providers at weekly team meetings
 Clinic Data • Random selection of 100 children referred to the HDRC clinic between July 2005 and December 2006 • 18 were evaluated for a cochlear implant (CI) • 82 were referred for hearing loss in general • Of the 100, 10 did not complete the evaluation process (none were CI candidates)
Clinic Data • Random selection of 100 children referred to the HDRC clinic between July 2005 and December 2006 • 18 were evaluated for a cochlear implant (CI) • 82 were referred for hearing loss in general • Of the 100, 10 did not complete the evaluation process (none were CI candidates)
 Time to Completion of Appointments Among patients who completed the full evaluation process (n=90) Population Time Entire Group Median time (range) Mean time (SD) CI group* 2. 8 mos (19 – 319 days) 3. 6 mos (2. 8 mos) Mean time (SD) 4 mos (3. 2 mos) Mean time (SD) 3. 5 mos (2. 8 mos) HI group CI group calculated based on date of surgery, thus increasing the length of time through the process. *
Time to Completion of Appointments Among patients who completed the full evaluation process (n=90) Population Time Entire Group Median time (range) Mean time (SD) CI group* 2. 8 mos (19 – 319 days) 3. 6 mos (2. 8 mos) Mean time (SD) 4 mos (3. 2 mos) Mean time (SD) 3. 5 mos (2. 8 mos) HI group CI group calculated based on date of surgery, thus increasing the length of time through the process. *
 Time from Identification to Amplification Among those receiving amplification (n=60) Population Time to amplification Entire Group Median (range) Mean (SD) 2 mos (at diagnosis – 13 years) 1 year ( 2 years) Median (range) Mean (SD) 1 month (at diagnosis - 4 mos) 1. 2 month (1 month) Median (range) Mean (SD) 2 mos (at diagnosis – 13 years) 1. 2 years (2 years) CI group HI group
Time from Identification to Amplification Among those receiving amplification (n=60) Population Time to amplification Entire Group Median (range) Mean (SD) 2 mos (at diagnosis – 13 years) 1 year ( 2 years) Median (range) Mean (SD) 1 month (at diagnosis - 4 mos) 1. 2 month (1 month) Median (range) Mean (SD) 2 mos (at diagnosis – 13 years) 1. 2 years (2 years) CI group HI group
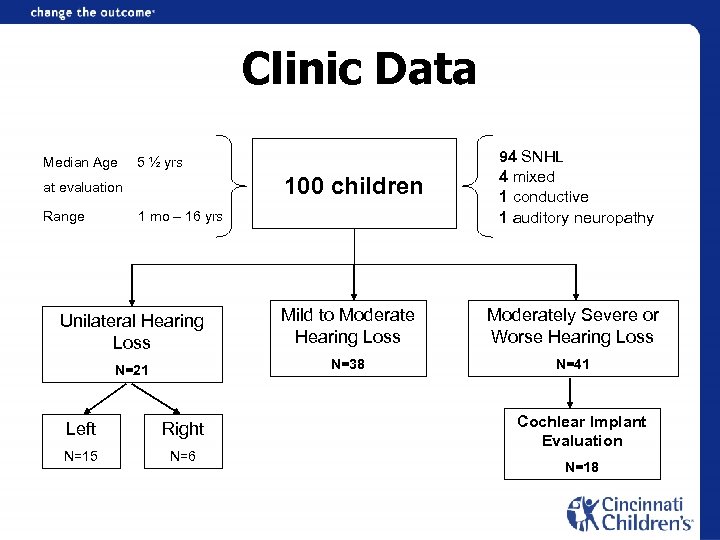 Clinic Data Median Age 5 ½ yrs 100 children at evaluation Range 1 mo – 16 yrs 94 SNHL 4 mixed 1 conductive 1 auditory neuropathy Unilateral Hearing Loss Mild to Moderate Hearing Loss Moderately Severe or Worse Hearing Loss N=21 N=38 N=41 Left Right N=15 N=6 Cochlear Implant Evaluation N=18
Clinic Data Median Age 5 ½ yrs 100 children at evaluation Range 1 mo – 16 yrs 94 SNHL 4 mixed 1 conductive 1 auditory neuropathy Unilateral Hearing Loss Mild to Moderate Hearing Loss Moderately Severe or Worse Hearing Loss N=21 N=38 N=41 Left Right N=15 N=6 Cochlear Implant Evaluation N=18
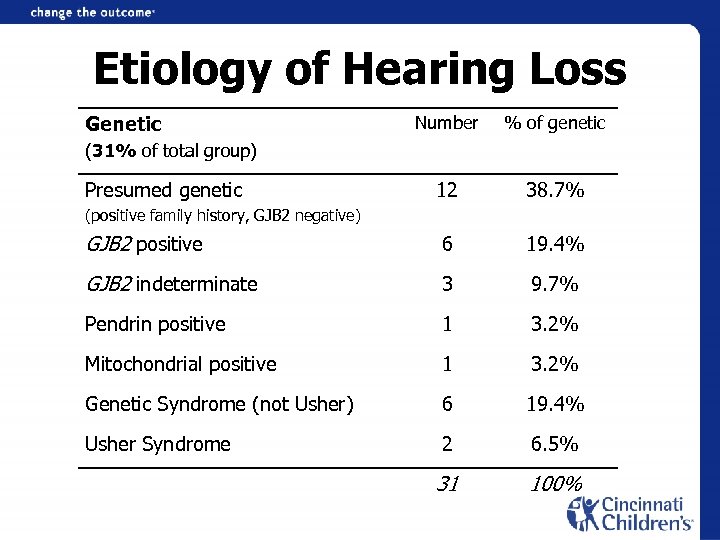 Etiology of Hearing Loss Genetic Number % of genetic 12 38. 7% GJB 2 positive 6 19. 4% GJB 2 indeterminate 3 9. 7% Pendrin positive 1 3. 2% Mitochondrial positive 1 3. 2% Genetic Syndrome (not Usher) 6 19. 4% Usher Syndrome 2 6. 5% 31 100% (31% of total group) Presumed genetic (positive family history, GJB 2 negative)
Etiology of Hearing Loss Genetic Number % of genetic 12 38. 7% GJB 2 positive 6 19. 4% GJB 2 indeterminate 3 9. 7% Pendrin positive 1 3. 2% Mitochondrial positive 1 3. 2% Genetic Syndrome (not Usher) 6 19. 4% Usher Syndrome 2 6. 5% 31 100% (31% of total group) Presumed genetic (positive family history, GJB 2 negative)
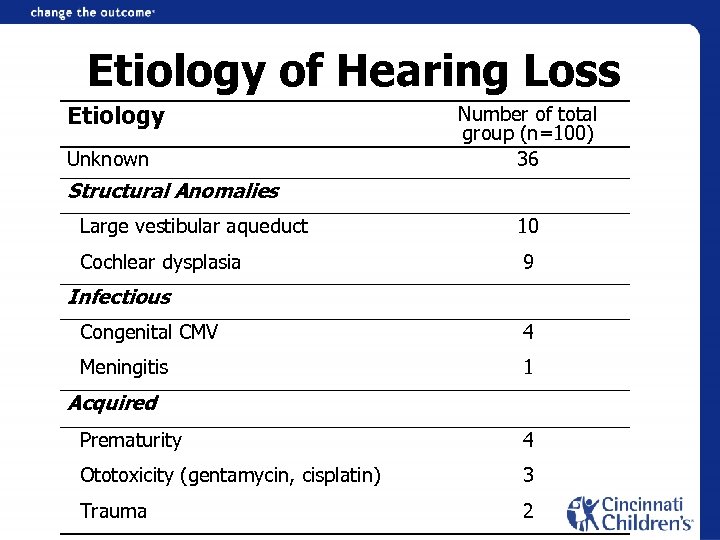 Etiology of Hearing Loss Etiology Unknown Number of total group (n=100) 36 Structural Anomalies Large vestibular aqueduct 10 Cochlear dysplasia 9 Infectious Congenital CMV 4 Meningitis 1 Acquired Prematurity 4 Ototoxicity (gentamycin, cisplatin) 3 Trauma 2
Etiology of Hearing Loss Etiology Unknown Number of total group (n=100) 36 Structural Anomalies Large vestibular aqueduct 10 Cochlear dysplasia 9 Infectious Congenital CMV 4 Meningitis 1 Acquired Prematurity 4 Ototoxicity (gentamycin, cisplatin) 3 Trauma 2
 Medical Evaluations Suggested in Literature • All children with hearing loss should see the following 3 medical specialists: – Pediatric Otolaryngology – Ophthalmology – Genetics
Medical Evaluations Suggested in Literature • All children with hearing loss should see the following 3 medical specialists: – Pediatric Otolaryngology – Ophthalmology – Genetics
 Pediatric Otolaryngology • 100% (by definition) saw a pediatric otolaryngologist • 76% with GJB 2 testing (51/67 with indications) • Not indicated in 33 (unilateral, known syndrome, CMV, meningitis, CMV) – 51 subjects completed GJB 2 testing – 6 positive (8% of those tested) – 3 indeterminate • CT results – 76 % completed – 42% of those tested had findings (32/76 patients)
Pediatric Otolaryngology • 100% (by definition) saw a pediatric otolaryngologist • 76% with GJB 2 testing (51/67 with indications) • Not indicated in 33 (unilateral, known syndrome, CMV, meningitis, CMV) – 51 subjects completed GJB 2 testing – 6 positive (8% of those tested) – 3 indeterminate • CT results – 76 % completed – 42% of those tested had findings (32/76 patients)
 Ophthalmology • 46% of entire group completed an eye exam • 54% of those with an eye exam (25/46 patients) had a significant finding – 18 with glasses – 7 with more significant vision problems • RP, coloboma, etc. • 47% of those with mild or unilateral HI completed an eye exam (28/59) – 53% (15/28) with findings
Ophthalmology • 46% of entire group completed an eye exam • 54% of those with an eye exam (25/46 patients) had a significant finding – 18 with glasses – 7 with more significant vision problems • RP, coloboma, etc. • 47% of those with mild or unilateral HI completed an eye exam (28/59) – 53% (15/28) with findings
 Genetics • 21 completed a genetics evaluation other than for genetic testing for hearing loss • 9 had a genetic syndrome, 1 was not the cause of the hearing loss. – – – – Beckwith Wiedemann and connexin CHARGE and XXY Partial trisomy 3 q and congenital CMV Miller Syndrome Campomelic dysplasia (2 sibs) Usher Syndrome (2 patients)
Genetics • 21 completed a genetics evaluation other than for genetic testing for hearing loss • 9 had a genetic syndrome, 1 was not the cause of the hearing loss. – – – – Beckwith Wiedemann and connexin CHARGE and XXY Partial trisomy 3 q and congenital CMV Miller Syndrome Campomelic dysplasia (2 sibs) Usher Syndrome (2 patients)
 Developmental Pediatrics • 58 patients completed a neurodevelopmental evaluation • 4 additional charts indicated issues, 2 did not keep appointment, 2 were not referred • 40 patients had findings which could impact education/development – 40% among entire population (n=100) – 69% among those referred and evaluated (n=58)
Developmental Pediatrics • 58 patients completed a neurodevelopmental evaluation • 4 additional charts indicated issues, 2 did not keep appointment, 2 were not referred • 40 patients had findings which could impact education/development – 40% among entire population (n=100) – 69% among those referred and evaluated (n=58)
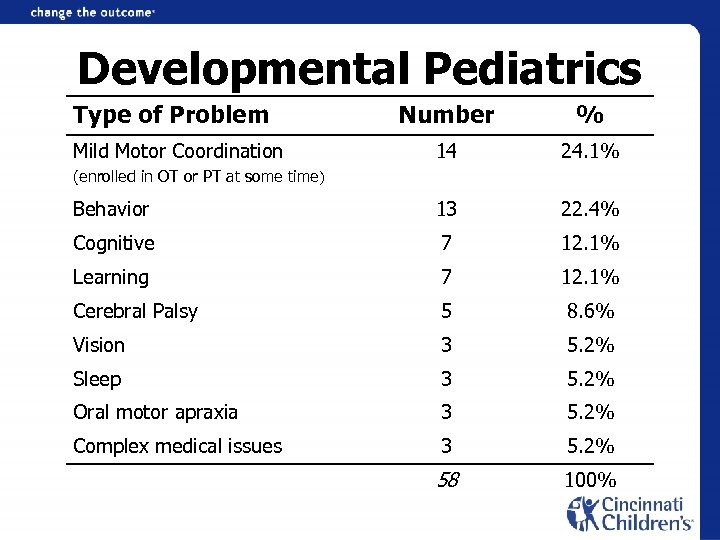 Developmental Pediatrics Type of Problem Number % 14 24. 1% Behavior 13 22. 4% Cognitive 7 12. 1% Learning 7 12. 1% Cerebral Palsy 5 8. 6% Vision 3 5. 2% Sleep 3 5. 2% Oral motor apraxia 3 5. 2% Complex medical issues 3 5. 2% 58 100% Mild Motor Coordination (enrolled in OT or PT at some time)
Developmental Pediatrics Type of Problem Number % 14 24. 1% Behavior 13 22. 4% Cognitive 7 12. 1% Learning 7 12. 1% Cerebral Palsy 5 8. 6% Vision 3 5. 2% Sleep 3 5. 2% Oral motor apraxia 3 5. 2% Complex medical issues 3 5. 2% 58 100% Mild Motor Coordination (enrolled in OT or PT at some time)
 Therapy Appointments • 52 patients completed a speech/language evaluation – 63% of those evaluated (33/52 patients) had an identified delay/issue requiring therapy • 42 patients completed an aural rehabilitation evaluation – 55% of those evaluated (23/42 patients) had an identified delay/issue requiring therapy
Therapy Appointments • 52 patients completed a speech/language evaluation – 63% of those evaluated (33/52 patients) had an identified delay/issue requiring therapy • 42 patients completed an aural rehabilitation evaluation – 55% of those evaluated (23/42 patients) had an identified delay/issue requiring therapy
 Unilateral/Mild HL 59 subjects had unilateral/mild hearing loss • 25 subjects (42%) saw DBP – 68% had findings (17/25) • 23 subjects (39%) saw SLP, 3 missing reports – 60% were suggested intervention (14/23) • 14 subjects (24%) saw AR, 1 missing report – 35% were suggested intervention (5/14)
Unilateral/Mild HL 59 subjects had unilateral/mild hearing loss • 25 subjects (42%) saw DBP – 68% had findings (17/25) • 23 subjects (39%) saw SLP, 3 missing reports – 60% were suggested intervention (14/23) • 14 subjects (24%) saw AR, 1 missing report – 35% were suggested intervention (5/14)
 Implications • Quality Improvement data to identify appropriate benchmarks or goals of care • Information to support the development of evidence-based clinical guidelines for the management of SNHL
Implications • Quality Improvement data to identify appropriate benchmarks or goals of care • Information to support the development of evidence-based clinical guidelines for the management of SNHL



