edb7385bda18b97a42bbf1f09dc5aeeb.ppt
- Количество слайдов: 45
 Initiation of precipitation via the ice phase Part I: Microphysical processes COPS summer school July 2007 SFB 641: The tropospheric ice phase Karoline Diehl University of Mainz, Germany Institute of Atmospheric Physics Max-Planck Institute of Chemistry, Mainz
Initiation of precipitation via the ice phase Part I: Microphysical processes COPS summer school July 2007 SFB 641: The tropospheric ice phase Karoline Diehl University of Mainz, Germany Institute of Atmospheric Physics Max-Planck Institute of Chemistry, Mainz
 Initiation of precipitation • cloud droplet sizes: 2 to 100 µm • rain drop sizes: 100 µm to 8 mm → cloud drops have to grow to larger sizes to fall out from the cloud → growth by diffusion of water vapor is not sufficient two main processes: 1. collision and coalescence (warm process) larger cloud drops collect smaller droplets
Initiation of precipitation • cloud droplet sizes: 2 to 100 µm • rain drop sizes: 100 µm to 8 mm → cloud drops have to grow to larger sizes to fall out from the cloud → growth by diffusion of water vapor is not sufficient two main processes: 1. collision and coalescence (warm process) larger cloud drops collect smaller droplets
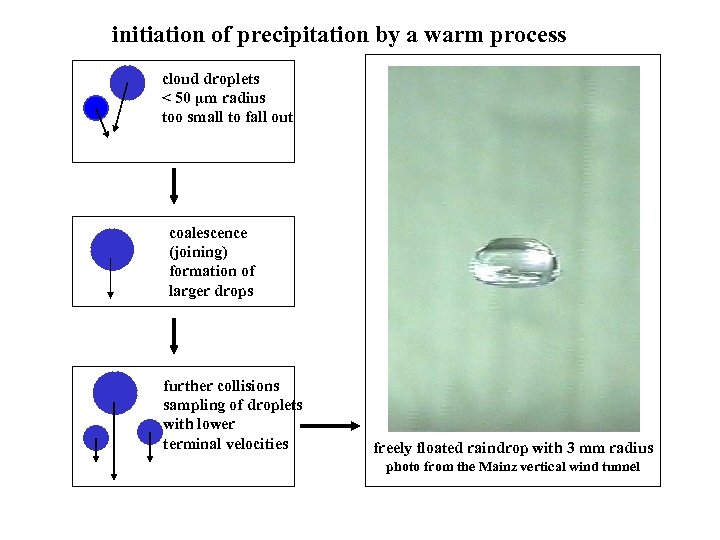 initiation of precipitation by a warm process cloud droplets < 50 µm radius too small to fall out coalescence (joining) formation of larger drops further collisions sampling of droplets with lower terminal velocities freely floated raindrop with 3 mm radius photo from the Mainz vertical wind tunnel
initiation of precipitation by a warm process cloud droplets < 50 µm radius too small to fall out coalescence (joining) formation of larger drops further collisions sampling of droplets with lower terminal velocities freely floated raindrop with 3 mm radius photo from the Mainz vertical wind tunnel
 Initiation of precipitation • cloud droplet sizes: 2 to 100 µm • rain drop sizes: 100 µm to 8 mm → cloud drops have to grow to larger sizes to fall out from the cloud → growth by diffusion of water vapor is not sufficient two main processes: 1. collision and coalescence (warm process) larger cloud drops collect smaller droplets 2. ice particles (cold process) ice particles once formed grow at the expense of liquid drops 3.
Initiation of precipitation • cloud droplet sizes: 2 to 100 µm • rain drop sizes: 100 µm to 8 mm → cloud drops have to grow to larger sizes to fall out from the cloud → growth by diffusion of water vapor is not sufficient two main processes: 1. collision and coalescence (warm process) larger cloud drops collect smaller droplets 2. ice particles (cold process) ice particles once formed grow at the expense of liquid drops 3.
 Initiation of precipitation via the ice phase major fraction of precipitation is initialized by a cold process troposphere: mostly mixed phase clouds → coexistence of super-cooled liquid drops and ice particles ▪ super-cooled liquid drops down to -40°C ▪ incidence of ice around -4°C
Initiation of precipitation via the ice phase major fraction of precipitation is initialized by a cold process troposphere: mostly mixed phase clouds → coexistence of super-cooled liquid drops and ice particles ▪ super-cooled liquid drops down to -40°C ▪ incidence of ice around -4°C
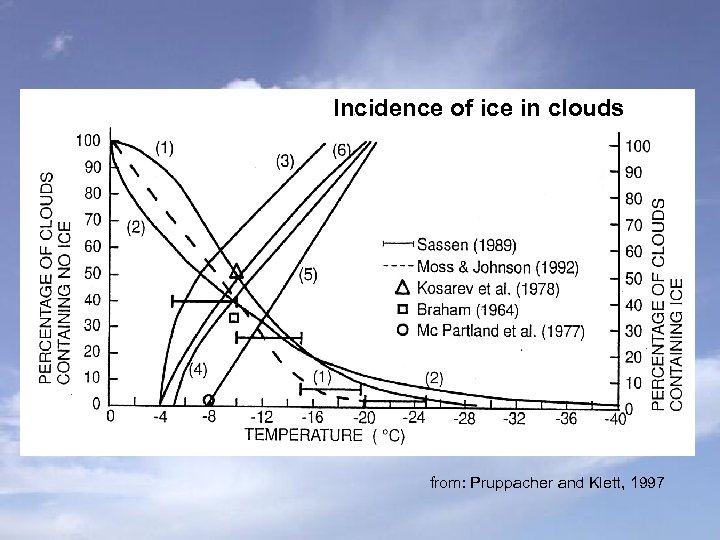 Incidence of ice in clouds from: Pruppacher and Klett, 1997
Incidence of ice in clouds from: Pruppacher and Klett, 1997
 Initiation of precipitation via the ice phase • ice nuclei • formation of ice • growth of ice particles
Initiation of precipitation via the ice phase • ice nuclei • formation of ice • growth of ice particles
 Definition of ice nuclei are aerosol particles which trigger the transition from vapor to ice phase requirements: * insolubility * size > 0. 1 µm * ice active sites on the surface * crystallographic shape similar to ice atmospheric concentrations of ice nuclei: < 10 per liter well-known types of atmospheric ice nuclei: mineral dust, soot particles, biological particles
Definition of ice nuclei are aerosol particles which trigger the transition from vapor to ice phase requirements: * insolubility * size > 0. 1 µm * ice active sites on the surface * crystallographic shape similar to ice atmospheric concentrations of ice nuclei: < 10 per liter well-known types of atmospheric ice nuclei: mineral dust, soot particles, biological particles
 Formation of ice * sublimation of water vapor on ice nuclei * condensation of water vapor on ice nuclei, followed by freezing * freezing of super-cooled drops 1. homogeneously without ice nuclei 2. heterogeneously with ice nuclei a) ice nuclei suspended in drops b) ice nuclei collide with drops
Formation of ice * sublimation of water vapor on ice nuclei * condensation of water vapor on ice nuclei, followed by freezing * freezing of super-cooled drops 1. homogeneously without ice nuclei 2. heterogeneously with ice nuclei a) ice nuclei suspended in drops b) ice nuclei collide with drops
 Formation of ice: Deposition freezing sublimation of water vapor on ice nuclei → deposition freezing requirements: • dry ice nucleus • super-saturation with respect to ice • critical temperature to initiate ice nucleation result: ice crystals in various habits, dependant on 1. temperature 2. super-saturation
Formation of ice: Deposition freezing sublimation of water vapor on ice nuclei → deposition freezing requirements: • dry ice nucleus • super-saturation with respect to ice • critical temperature to initiate ice nucleation result: ice crystals in various habits, dependant on 1. temperature 2. super-saturation
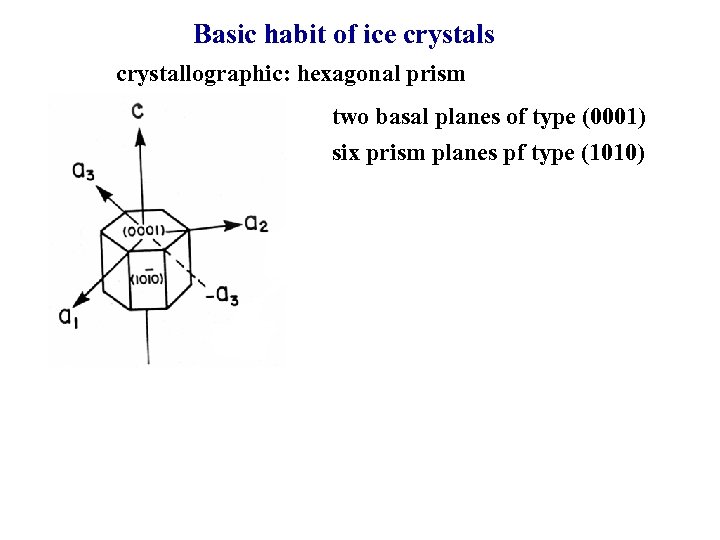 Basic habit of ice crystals crystallographic: hexagonal prism two basal planes of type (0001) six prism planes pf type (1010)
Basic habit of ice crystals crystallographic: hexagonal prism two basal planes of type (0001) six prism planes pf type (1010)
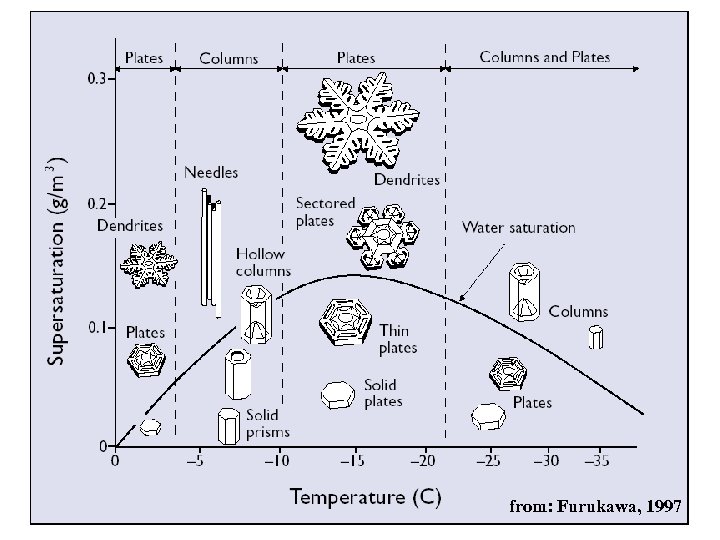 from: Furukawa, 1997
from: Furukawa, 1997
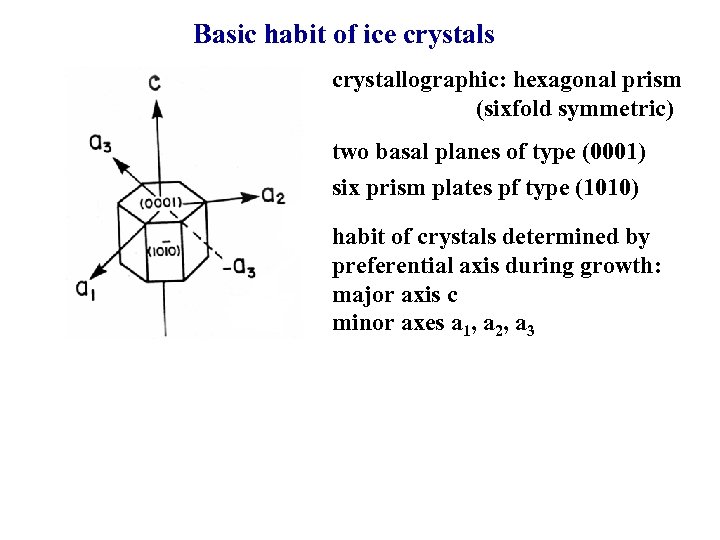 Basic habit of ice crystals crystallographic: hexagonal prism (sixfold symmetric) two basal planes of type (0001) six prism plates pf type (1010) habit of crystals determined by preferential axis during growth: major axis c minor axes a 1, a 2, a 3
Basic habit of ice crystals crystallographic: hexagonal prism (sixfold symmetric) two basal planes of type (0001) six prism plates pf type (1010) habit of crystals determined by preferential axis during growth: major axis c minor axes a 1, a 2, a 3
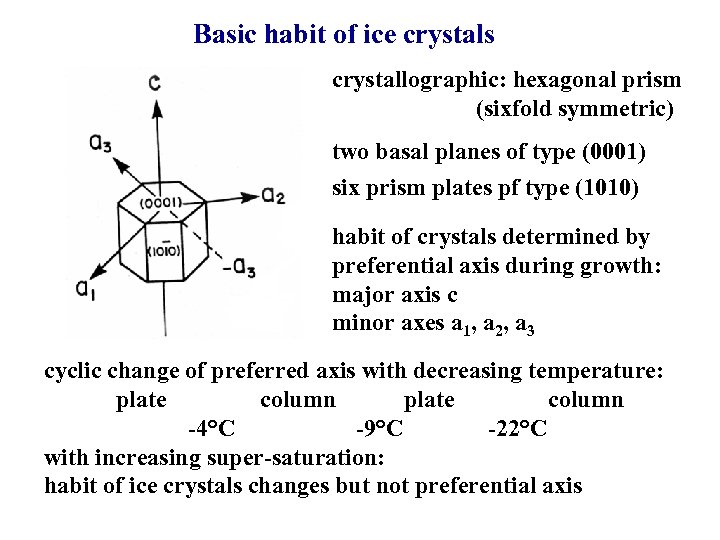 Basic habit of ice crystals crystallographic: hexagonal prism (sixfold symmetric) two basal planes of type (0001) six prism plates pf type (1010) habit of crystals determined by preferential axis during growth: major axis c minor axes a 1, a 2, a 3 cyclic change of preferred axis with decreasing temperature: plate column -4°C -9°C -22°C with increasing super-saturation: habit of ice crystals changes but not preferential axis
Basic habit of ice crystals crystallographic: hexagonal prism (sixfold symmetric) two basal planes of type (0001) six prism plates pf type (1010) habit of crystals determined by preferential axis during growth: major axis c minor axes a 1, a 2, a 3 cyclic change of preferred axis with decreasing temperature: plate column -4°C -9°C -22°C with increasing super-saturation: habit of ice crystals changes but not preferential axis
 Photos of elemental ice crystals from: Kobayashi and Kuroda, 1987
Photos of elemental ice crystals from: Kobayashi and Kuroda, 1987
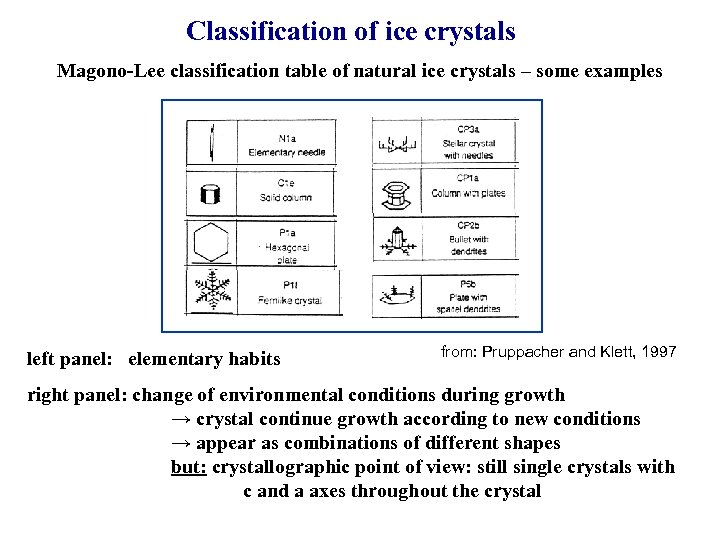 Classification of ice crystals Magono-Lee classification table of natural ice crystals – some examples left panel: elementary habits from: Pruppacher and Klett, 1997 right panel: change of environmental conditions during growth → crystal continue growth according to new conditions → appear as combinations of different shapes but: crystallographic point of view: still single crystals with c and a axes throughout the crystal
Classification of ice crystals Magono-Lee classification table of natural ice crystals – some examples left panel: elementary habits from: Pruppacher and Klett, 1997 right panel: change of environmental conditions during growth → crystal continue growth according to new conditions → appear as combinations of different shapes but: crystallographic point of view: still single crystals with c and a axes throughout the crystal
 Formation of ice: Condensation freezing condensation of water vapor on ice nuclei, followed by freezing of the drop → condensation freezing requirements: • dry ice nucleus which is suited as condensation nucleus • super-saturation with respect to water • critical temperature to initiate ice nucleation result: frozen drop
Formation of ice: Condensation freezing condensation of water vapor on ice nuclei, followed by freezing of the drop → condensation freezing requirements: • dry ice nucleus which is suited as condensation nucleus • super-saturation with respect to water • critical temperature to initiate ice nucleation result: frozen drop
 Formation of ice: Condensation freezing Experiments at the Institute of Atmospheric Research, Mainz: • super-saturation chamber, temperature < 0°C • scattering of particles into the chamber • sampling of ice particles on formvar plates 1. biological particles (pollen) drop diameters between 110 and 170 µm from: Diehl et al. , 2001
Formation of ice: Condensation freezing Experiments at the Institute of Atmospheric Research, Mainz: • super-saturation chamber, temperature < 0°C • scattering of particles into the chamber • sampling of ice particles on formvar plates 1. biological particles (pollen) drop diameters between 110 and 170 µm from: Diehl et al. , 2001
 Formation of ice: Condensation freezing Experiments at the Institute of Atmospheric Research, Mainz: 2. soot particles from a kerosene burner ice crystal diameters between 40 and 100 µm from: Diehl and Mitra, 1998
Formation of ice: Condensation freezing Experiments at the Institute of Atmospheric Research, Mainz: 2. soot particles from a kerosene burner ice crystal diameters between 40 and 100 µm from: Diehl and Mitra, 1998
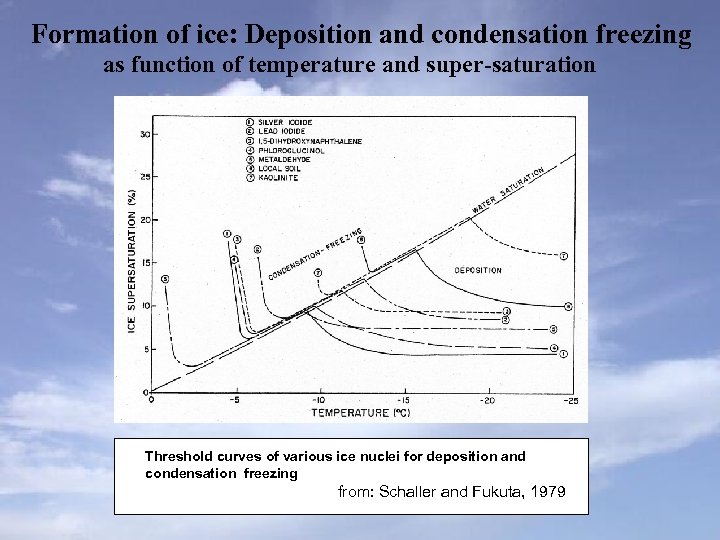 Formation of ice: Deposition and condensation freezing as function of temperature and super-saturation Threshold curves of various ice nuclei for deposition and condensation freezing from: Schaller and Fukuta, 1979
Formation of ice: Deposition and condensation freezing as function of temperature and super-saturation Threshold curves of various ice nuclei for deposition and condensation freezing from: Schaller and Fukuta, 1979
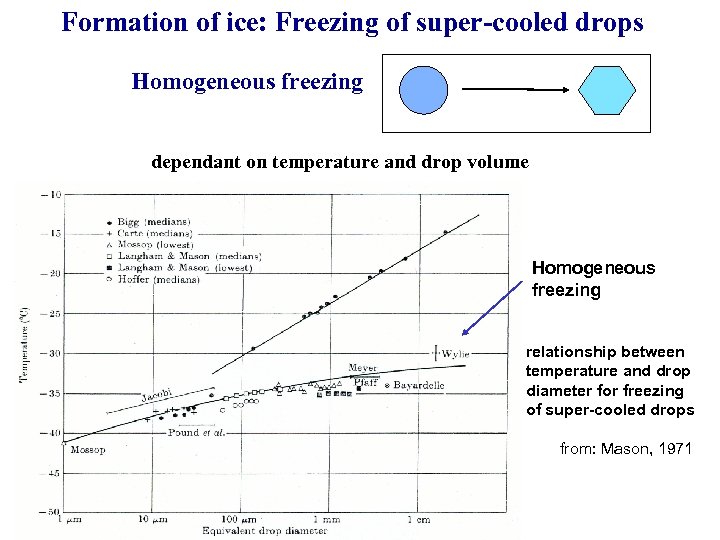 Formation of ice: Freezing of super-cooled drops Homogeneous freezing dependant on temperature and drop volume Homogeneous freezing relationship between temperature and drop diameter for freezing of super-cooled drops from: Mason, 1971
Formation of ice: Freezing of super-cooled drops Homogeneous freezing dependant on temperature and drop volume Homogeneous freezing relationship between temperature and drop diameter for freezing of super-cooled drops from: Mason, 1971
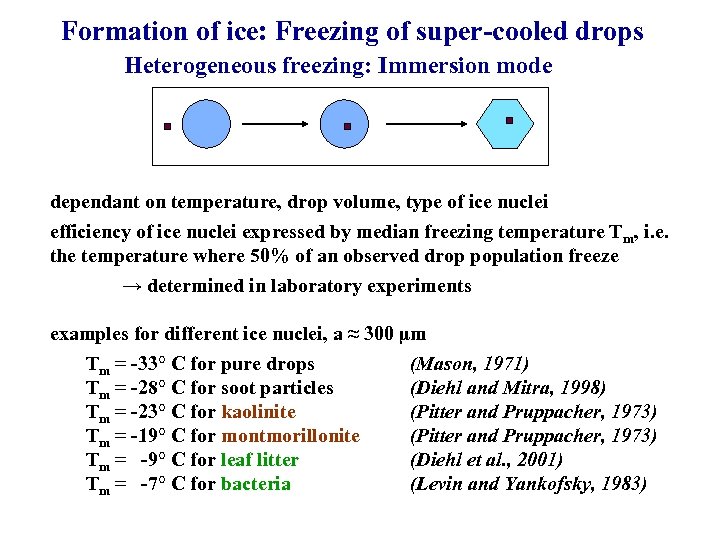 Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Immersion mode dependant on temperature, drop volume, type of ice nuclei efficiency of ice nuclei expressed by median freezing temperature Tm, i. e. the temperature where 50% of an observed drop population freeze → determined in laboratory experiments examples for different ice nuclei, a ≈ 300 µm Tm = -33° C for pure drops (Mason, 1971) Tm = -28° C for soot particles (Diehl and Mitra, 1998) Tm = -23° C for kaolinite (Pitter and Pruppacher, 1973) Tm = -19° C for montmorillonite (Pitter and Pruppacher, 1973) Tm = -9° C for leaf litter (Diehl et al. , 2001) Tm = -7° C for bacteria (Levin and Yankofsky, 1983)
Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Immersion mode dependant on temperature, drop volume, type of ice nuclei efficiency of ice nuclei expressed by median freezing temperature Tm, i. e. the temperature where 50% of an observed drop population freeze → determined in laboratory experiments examples for different ice nuclei, a ≈ 300 µm Tm = -33° C for pure drops (Mason, 1971) Tm = -28° C for soot particles (Diehl and Mitra, 1998) Tm = -23° C for kaolinite (Pitter and Pruppacher, 1973) Tm = -19° C for montmorillonite (Pitter and Pruppacher, 1973) Tm = -9° C for leaf litter (Diehl et al. , 2001) Tm = -7° C for bacteria (Levin and Yankofsky, 1983)
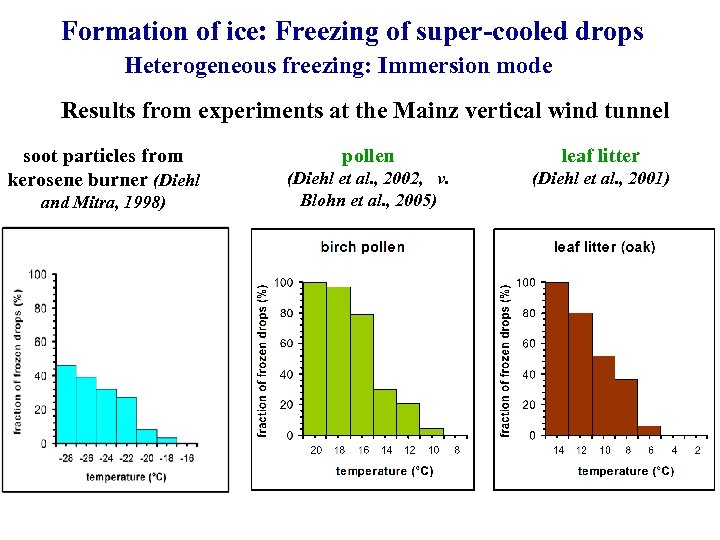 Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Immersion mode Results from experiments at the Mainz vertical wind tunnel soot particles from kerosene burner (Diehl and Mitra, 1998) pollen leaf litter (Diehl et al. , 2002, v. Blohn et al. , 2005) (Diehl et al. , 2001)
Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Immersion mode Results from experiments at the Mainz vertical wind tunnel soot particles from kerosene burner (Diehl and Mitra, 1998) pollen leaf litter (Diehl et al. , 2002, v. Blohn et al. , 2005) (Diehl et al. , 2001)
 Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Immersion mode dependant on temperature, drop volume, type of ice nuclei examples for different drop radii, ice nuclei are montmorillonite Tm = -24° C for a = 50 µm (Hoffer, 1961) Tm = -19° C for a = 350 µm (Pitter and Pruppacher, 1973) median freezing temperature ~ ln (drop volume)
Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Immersion mode dependant on temperature, drop volume, type of ice nuclei examples for different drop radii, ice nuclei are montmorillonite Tm = -24° C for a = 50 µm (Hoffer, 1961) Tm = -19° C for a = 350 µm (Pitter and Pruppacher, 1973) median freezing temperature ~ ln (drop volume)
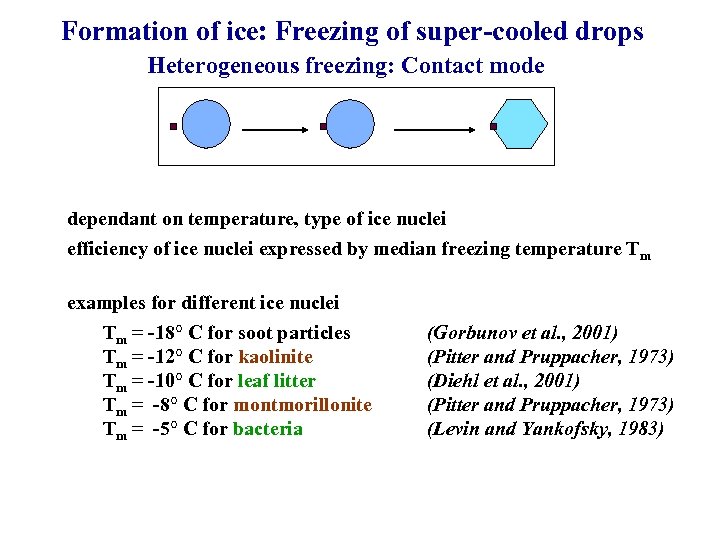 Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Contact mode dependant on temperature, type of ice nuclei efficiency of ice nuclei expressed by median freezing temperature Tm examples for different ice nuclei Tm = -18° C for soot particles Tm = -12° C for kaolinite Tm = -10° C for leaf litter Tm = -8° C for montmorillonite Tm = -5° C for bacteria (Gorbunov et al. , 2001) (Pitter and Pruppacher, 1973) (Diehl et al. , 2001) (Pitter and Pruppacher, 1973) (Levin and Yankofsky, 1983)
Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Contact mode dependant on temperature, type of ice nuclei efficiency of ice nuclei expressed by median freezing temperature Tm examples for different ice nuclei Tm = -18° C for soot particles Tm = -12° C for kaolinite Tm = -10° C for leaf litter Tm = -8° C for montmorillonite Tm = -5° C for bacteria (Gorbunov et al. , 2001) (Pitter and Pruppacher, 1973) (Diehl et al. , 2001) (Pitter and Pruppacher, 1973) (Levin and Yankofsky, 1983)
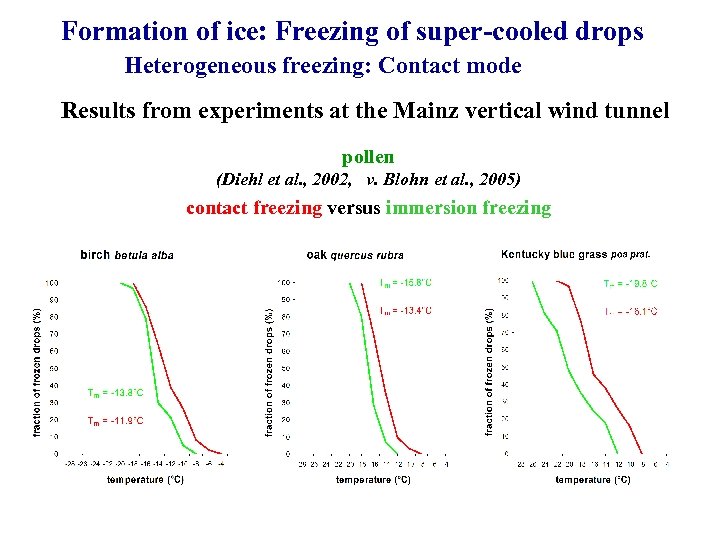 Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Contact mode Results from experiments at the Mainz vertical wind tunnel pollen (Diehl et al. , 2002, v. Blohn et al. , 2005) contact freezing versus immersion freezing
Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Contact mode Results from experiments at the Mainz vertical wind tunnel pollen (Diehl et al. , 2002, v. Blohn et al. , 2005) contact freezing versus immersion freezing
 Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Contact mode → seems to be a very efficient process, but: it is restricted by the collision efficiency between drops and aerosol particles, which is dependant on: ▪ Brown diffusion ▪ thermo-phoretic and diffusio-phoretic forces ▪ inertia effects ▪ electrical forces collision kernel dependant on terminal velocities and cross sections of colliding partners:
Formation of ice: Freezing of super-cooled drops Heterogeneous freezing: Contact mode → seems to be a very efficient process, but: it is restricted by the collision efficiency between drops and aerosol particles, which is dependant on: ▪ Brown diffusion ▪ thermo-phoretic and diffusio-phoretic forces ▪ inertia effects ▪ electrical forces collision kernel dependant on terminal velocities and cross sections of colliding partners:
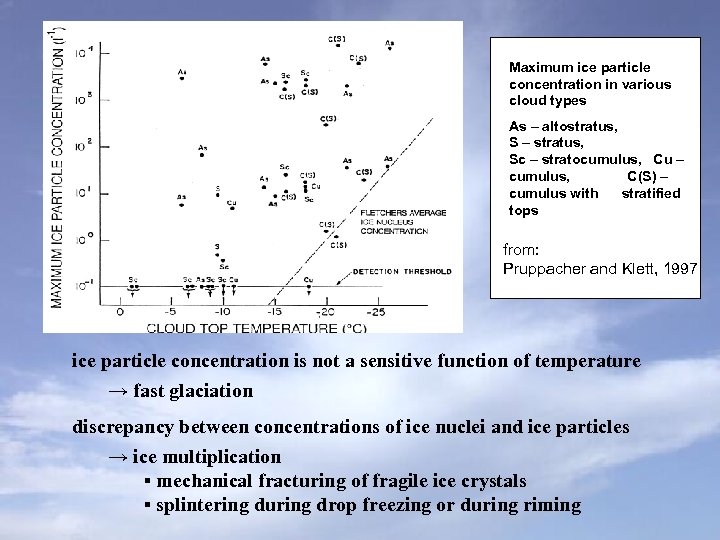 Maximum ice particle concentration in various cloud types As – altostratus, S – stratus, Sc – stratocumulus, Cu – cumulus, C(S) – cumulus with stratified tops from: Pruppacher and Klett, 1997 ice particle concentration is not a sensitive function of temperature → fast glaciation discrepancy between concentrations of ice nuclei and ice particles → ice multiplication ▪ mechanical fracturing of fragile ice crystals ▪ splintering during drop freezing or during riming
Maximum ice particle concentration in various cloud types As – altostratus, S – stratus, Sc – stratocumulus, Cu – cumulus, C(S) – cumulus with stratified tops from: Pruppacher and Klett, 1997 ice particle concentration is not a sensitive function of temperature → fast glaciation discrepancy between concentrations of ice nuclei and ice particles → ice multiplication ▪ mechanical fracturing of fragile ice crystals ▪ splintering during drop freezing or during riming
 Growth of ice particles • deposition of water vapor • riming • agglomeration
Growth of ice particles • deposition of water vapor • riming • agglomeration
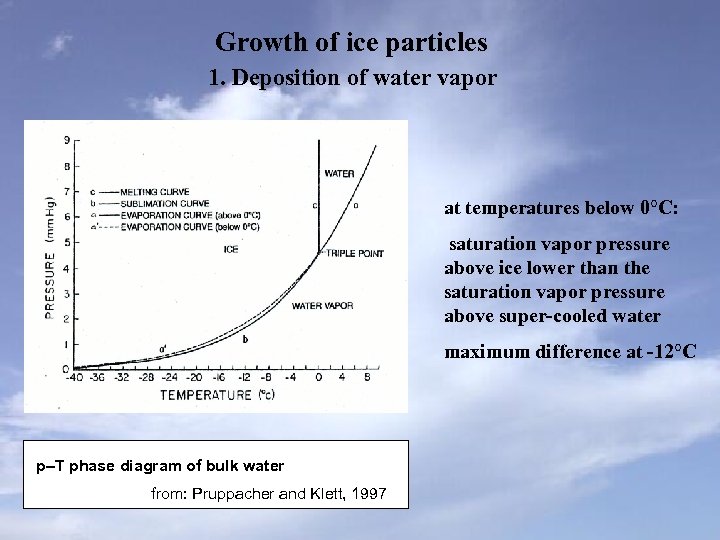 Growth of ice particles 1. Deposition of water vapor at temperatures below 0°C: saturation vapor pressure above ice lower than the saturation vapor pressure above super-cooled water maximum difference at -12°C p–T phase diagram of bulk water from: Pruppacher and Klett, 1997
Growth of ice particles 1. Deposition of water vapor at temperatures below 0°C: saturation vapor pressure above ice lower than the saturation vapor pressure above super-cooled water maximum difference at -12°C p–T phase diagram of bulk water from: Pruppacher and Klett, 1997
 Growth of ice particles 1. Deposition of water vapor at temperatures between 0 and -30°C: the saturation vapor pressure above ice is lower than the saturation vapor pressure above super-cooled water • no equilibrium between super-cooled water drops and ice particles • ice particles grow at the expense of liquid drops → Bergeron-Findeisen process
Growth of ice particles 1. Deposition of water vapor at temperatures between 0 and -30°C: the saturation vapor pressure above ice is lower than the saturation vapor pressure above super-cooled water • no equilibrium between super-cooled water drops and ice particles • ice particles grow at the expense of liquid drops → Bergeron-Findeisen process
 Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → collision process → dependant on collision partners
Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → collision process → dependant on collision partners
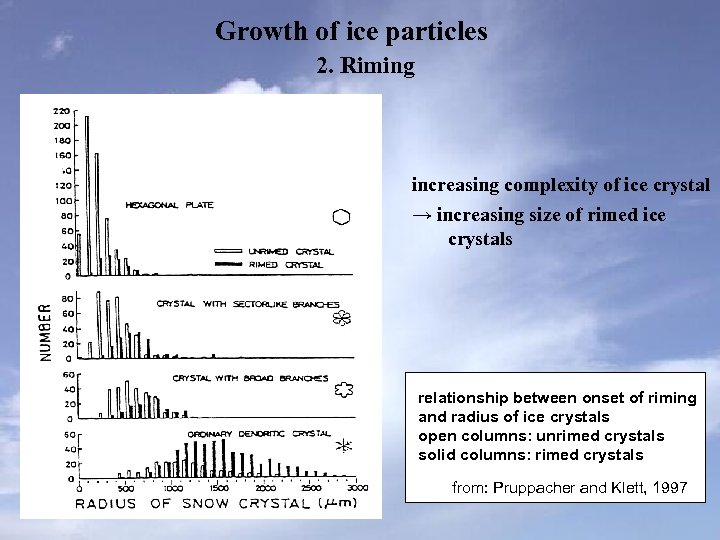 Growth of ice particles 2. Riming increasing complexity of ice crystal → increasing size of rimed ice crystals relationship between onset of riming and radius of ice crystals open columns: unrimed crystals solid columns: rimed crystals from: Pruppacher and Klett, 1997
Growth of ice particles 2. Riming increasing complexity of ice crystal → increasing size of rimed ice crystals relationship between onset of riming and radius of ice crystals open columns: unrimed crystals solid columns: rimed crystals from: Pruppacher and Klett, 1997
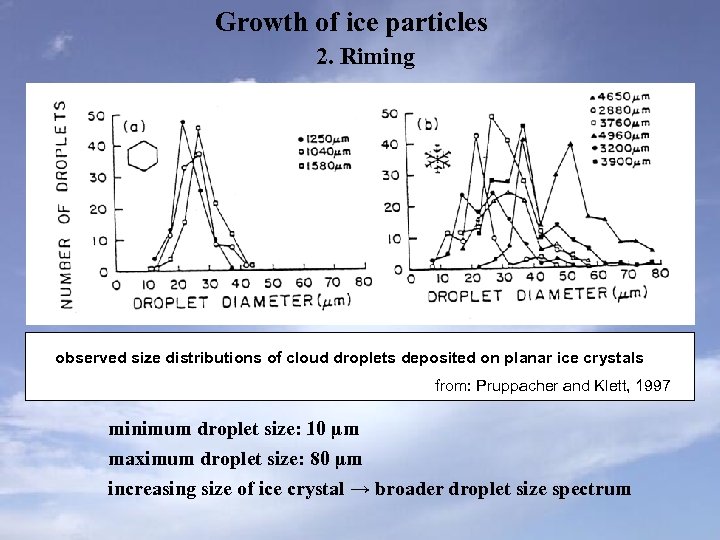 Growth of ice particles 2. Riming observed size distributions of cloud droplets deposited on planar ice crystals from: Pruppacher and Klett, 1997 minimum droplet size: 10 µm maximum droplet size: 80 µm increasing size of ice crystal → broader droplet size spectrum
Growth of ice particles 2. Riming observed size distributions of cloud droplets deposited on planar ice crystals from: Pruppacher and Klett, 1997 minimum droplet size: 10 µm maximum droplet size: 80 µm increasing size of ice crystal → broader droplet size spectrum
 Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → rimed ice crystals original habit still visible
Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → rimed ice crystals original habit still visible
 Growth of ice particles 2. Riming Rimed planar ice crystals of 2 to 3 mm diameter, rimed column ice crystal of 1. 5 mm length from: Pruppacher and Klett, 1997
Growth of ice particles 2. Riming Rimed planar ice crystals of 2 to 3 mm diameter, rimed column ice crystal of 1. 5 mm length from: Pruppacher and Klett, 1997
 Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → rimed ice crystals original habit still visible → graupel original habit no more visible conical graupel or lump graupel
Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → rimed ice crystals original habit still visible → graupel original habit no more visible conical graupel or lump graupel
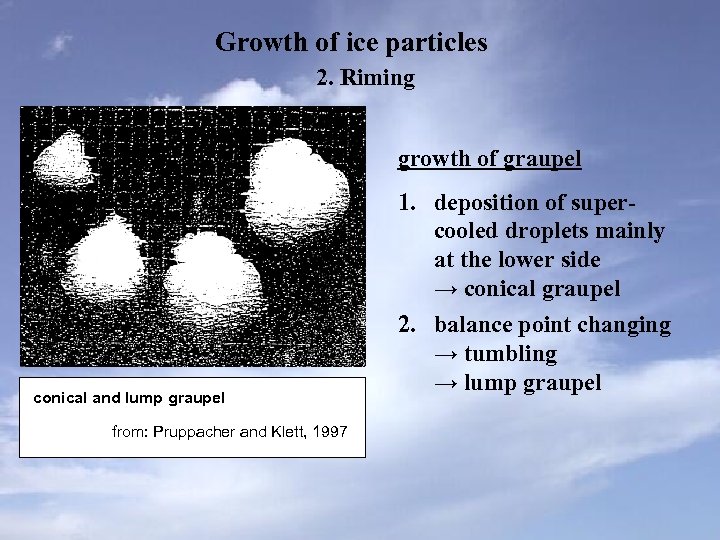 Growth of ice particles 2. Riming growth of graupel conical and lump graupel from: Pruppacher and Klett, 1997 1. deposition of supercooled droplets mainly at the lower side → conical graupel 2. balance point changing → tumbling → lump graupel
Growth of ice particles 2. Riming growth of graupel conical and lump graupel from: Pruppacher and Klett, 1997 1. deposition of supercooled droplets mainly at the lower side → conical graupel 2. balance point changing → tumbling → lump graupel
 Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → rimed ice crystals original habit still visible → graupel original habit not visible conical graupel or lump graupel → hailstones diameter > 5 mm in extreme cases: sizes up to 10 cm
Growth of ice particles 2. Riming deposition of super-cooled liquid droplets on frozen drops or ice crystals → rimed ice crystals original habit still visible → graupel original habit not visible conical graupel or lump graupel → hailstones diameter > 5 mm in extreme cases: sizes up to 10 cm
 Growth of ice particles 2. Riming ▪ visible germ in the hailstone center → frozen drop or graupel thin section of hailstone diameter 4 cm from: Pruppacher and Klett, 1997 ▪ onion-like structure: clear and opaque layers clear layers: temperature slightly below 0°C, much super-cooled water → fast freezing hinders crystallization → solidified melt opaque layers: low temperatures, little super-cooled water → slow freezing, crystallization, inclusion of air bubbles
Growth of ice particles 2. Riming ▪ visible germ in the hailstone center → frozen drop or graupel thin section of hailstone diameter 4 cm from: Pruppacher and Klett, 1997 ▪ onion-like structure: clear and opaque layers clear layers: temperature slightly below 0°C, much super-cooled water → fast freezing hinders crystallization → solidified melt opaque layers: low temperatures, little super-cooled water → slow freezing, crystallization, inclusion of air bubbles
 Growth of ice particles 3. Agglomeration agglomeration of ice crystals → snow flake * crystal shape complex habit with branches favored * temperature quasi-liquid layer at ice surface 1 × 10 -2 µm at -1°C 1 × 10 -4 µm at -20°C maximum dimensions: 0. 5 to 12 mm
Growth of ice particles 3. Agglomeration agglomeration of ice crystals → snow flake * crystal shape complex habit with branches favored * temperature quasi-liquid layer at ice surface 1 × 10 -2 µm at -1°C 1 × 10 -4 µm at -20°C maximum dimensions: 0. 5 to 12 mm
 Terminal velocities of hydrometeors size terminal velocity cloud droplets < 100 µm < 0. 25 m/s rain drops 100 µm – 8 mm 0. 25 m/s – 4 m/s ice crystals < 1. 5 mm < 0. 6 m/s snow flakes 1 mm – 12 mm 0. 5 m/s – 1. 5 m/s graupel 0. 5 mm – 4 mm 0. 75 m/s – 3 m/s hailstones 5 mm – 8 cm 5 m/s – 50 m/s
Terminal velocities of hydrometeors size terminal velocity cloud droplets < 100 µm < 0. 25 m/s rain drops 100 µm – 8 mm 0. 25 m/s – 4 m/s ice crystals < 1. 5 mm < 0. 6 m/s snow flakes 1 mm – 12 mm 0. 5 m/s – 1. 5 m/s graupel 0. 5 mm – 4 mm 0. 75 m/s – 3 m/s hailstones 5 mm – 8 cm 5 m/s – 50 m/s
 Initiation of precipitation via the ice phase mixed phase clouds: * ice particles are present at temperatures as high as -4°C * ice particles grow mainly at the expense of liquid drops → fast glaciation of clouds → ice particles reach large terminal velocities * efficient formation of precipitation-sized ice particles
Initiation of precipitation via the ice phase mixed phase clouds: * ice particles are present at temperatures as high as -4°C * ice particles grow mainly at the expense of liquid drops → fast glaciation of clouds → ice particles reach large terminal velocities * efficient formation of precipitation-sized ice particles
 Initiation of precipitation via the ice phase mixed phase clouds: * ice particles are present at temperatures as high as -4°C * ice particles grow mainly at the expense of liquid drops → fast glaciation of clouds → ice particles reach large terminal velocities * efficient formation of precipitation-sized ice particles highly important for special cases: suppression of warm rain • examples: biomass burning, strong pollution • small aerosol particles → formation of small drops • reduced formation of precipitation sized drops via collision and coalescence
Initiation of precipitation via the ice phase mixed phase clouds: * ice particles are present at temperatures as high as -4°C * ice particles grow mainly at the expense of liquid drops → fast glaciation of clouds → ice particles reach large terminal velocities * efficient formation of precipitation-sized ice particles highly important for special cases: suppression of warm rain • examples: biomass burning, strong pollution • small aerosol particles → formation of small drops • reduced formation of precipitation sized drops via collision and coalescence
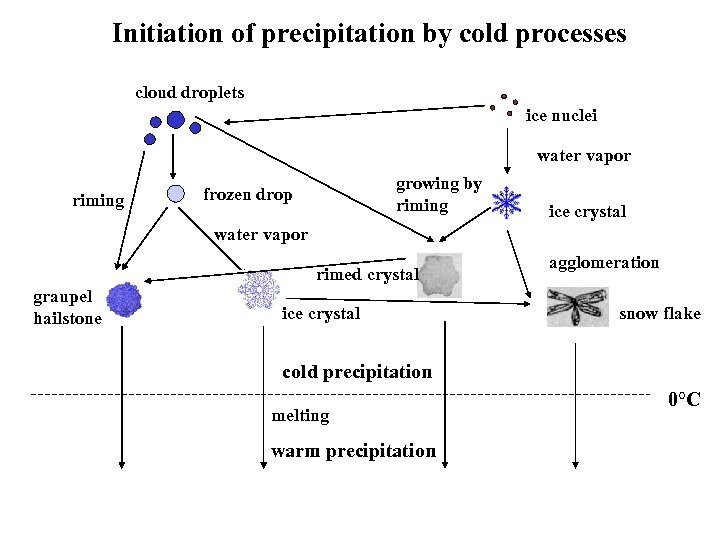 Initiation of precipitation by cold processes cloud droplets ice nuclei water vapor riming growing by riming frozen drop ice crystal water vapor rimed crystal graupel hailstone ice crystal agglomeration snow flake cold precipitation melting warm precipitation 0°C
Initiation of precipitation by cold processes cloud droplets ice nuclei water vapor riming growing by riming frozen drop ice crystal water vapor rimed crystal graupel hailstone ice crystal agglomeration snow flake cold precipitation melting warm precipitation 0°C


