db4d5b418b9ea5787ea1cc6b185c16e8.ppt
- Количество слайдов: 34

Influenza Neuraminidase Inhibitor IC 50 Data: Calculation, Interpretation and Statistical Analyses

Presentation Outline • Determining IC 50 values Curve-fitting methods Sources of variation • Identifying IC 50 outliers Determining cut-offs/thresholds Outlier values versus resistant viruses • Monitoring trends over time in IC 50 data

Abbreviations NA: Neuraminidase NI: Neuraminidase Inhibitor RFU: Relative Fluorescence Units RLU: Relative Luminescence Units VC: Virus Control

Determining NI IC 50 Values IC 50: The concentration of NI which reduces NA activity by 50% of the virus-control, or upper asymptote Estimated by: Measuring the NA activity (RFU or RLU) of an isolate against a range of dilutions of the drug, as well as without drug (virus-control)

Determining IC 50 Values: Calculation Options • Curve fitting-Statistical software Graph Pad Prism (www. graphpad. com) • $595 Grafit (www. erithacus. com/grafit/) • $400 -500 Jaspr (Developed by CDC) • Contact CDC for suitability and further details • Point-to-Point calculation Excel templates (Created by Health Protection Agency, UK)
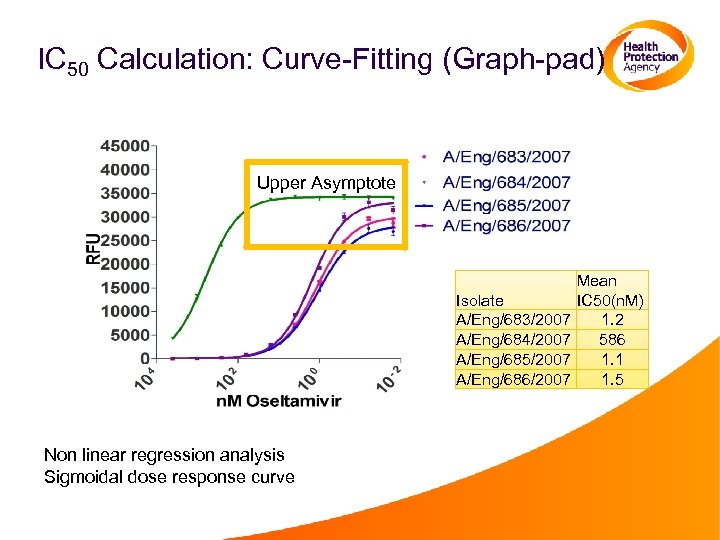
IC 50 Calculation: Curve-Fitting (Graph-pad) Upper Asymptote Isolate A/Eng/683/2007 A/Eng/684/2007 A/Eng/685/2007 A/Eng/686/2007 Non linear regression analysis Sigmoidal dose response curve Mean IC 50(n. M) 1. 2 586 1. 1 1. 5
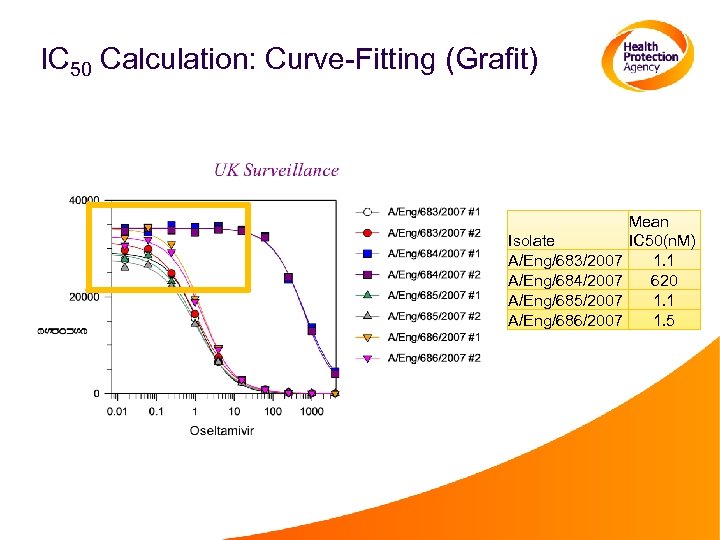
IC 50 Calculation: Curve-Fitting (Grafit) Isolate A/Eng/683/2007 A/Eng/684/2007 A/Eng/685/2007 A/Eng/686/2007 Mean IC 50(n. M) 1. 1 620 1. 1 1. 5
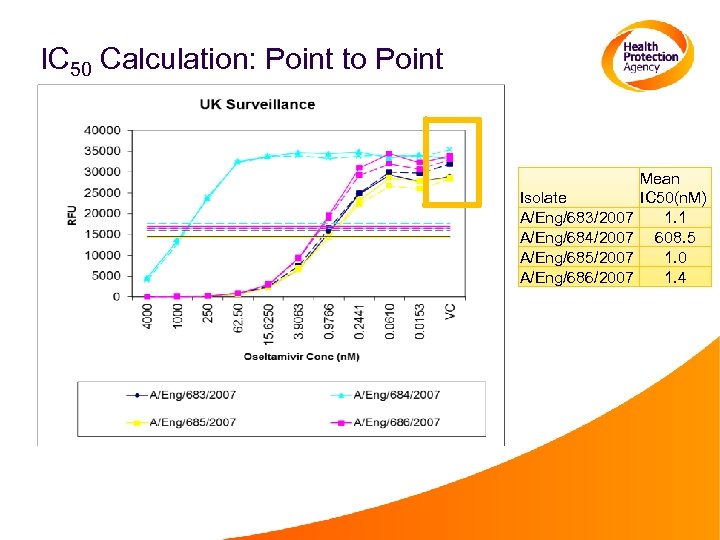
IC 50 Calculation: Point to Point Isolate A/Eng/683/2007 A/Eng/684/2007 A/Eng/685/2007 A/Eng/686/2007 Mean IC 50(n. M) 1. 1 608. 5 1. 0 1. 4
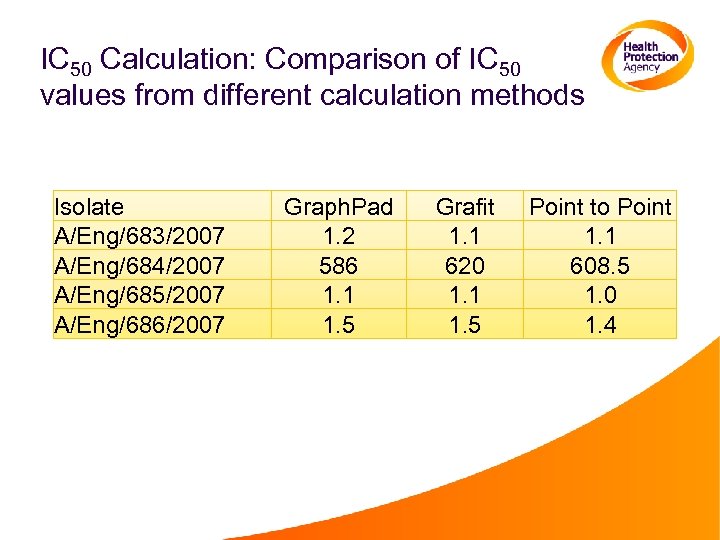
IC 50 Calculation: Comparison of IC 50 values from different calculation methods Isolate A/Eng/683/2007 A/Eng/684/2007 A/Eng/685/2007 A/Eng/686/2007 Graph. Pad 1. 2 586 1. 1 1. 5 Grafit 1. 1 620 1. 1 1. 5 Point to Point 1. 1 608. 5 1. 0 1. 4

Determining IC 50 Values: Sources of Variation • Method of calculation Using point-to-point or curve fitting software Choice of curve fitting software used • Intra-assay variation Difference between 2 or more replicates • Inter-assay variation Difference in calculated value for a given isolate in multiple assays
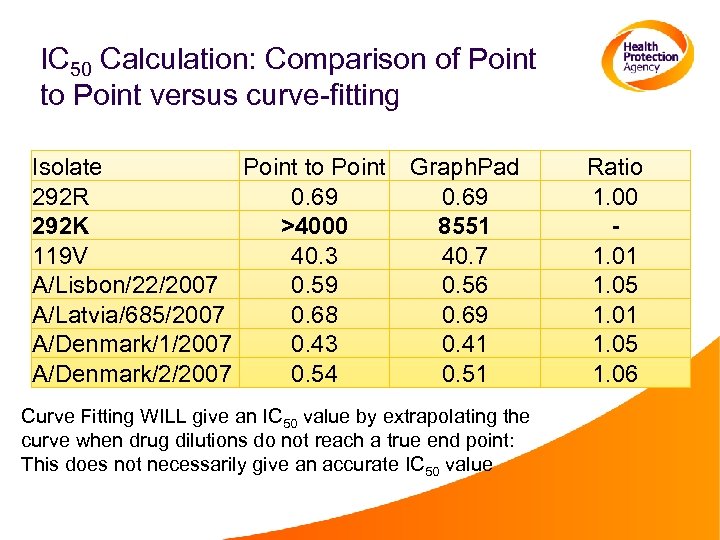
IC 50 Calculation: Comparison of Point to Point versus curve-fitting Isolate Point to Point 292 R 0. 69 292 K >4000 119 V 40. 3 A/Lisbon/22/2007 0. 59 A/Latvia/685/2007 0. 68 A/Denmark/1/2007 0. 43 A/Denmark/2/2007 0. 54 Graph. Pad 0. 69 8551 40. 7 0. 56 0. 69 0. 41 0. 51 Curve Fitting WILL give an IC 50 value by extrapolating the curve when drug dilutions do not reach a true end point: This does not necessarily give an accurate IC 50 value Ratio 1. 00 1. 01 1. 05 1. 06
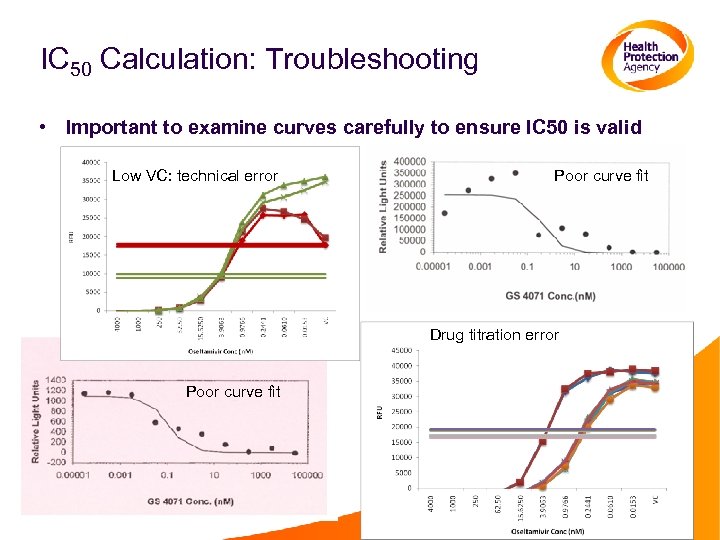
IC 50 Calculation: Troubleshooting • Important to examine curves carefully to ensure IC 50 is valid Low VC: technical error Poor curve fit Drug titration error Poor curve fit

Comments: Choice of IC 50 Calculation Method • Choice of IC 50 calculation method will make no more than about 5% difference to IC 50 value, for most samples • Must be clear exactly how the curve fitting and calculation of IC 50 is working Is IC 50 based on 50% of RFU/RLU of VC or 50% of fitted upper asymptote. • Regardless of method used, careful examination of the curve produced is required to identify technical issues. See presentation on validation and troubleshooting of IC 50 testing methods
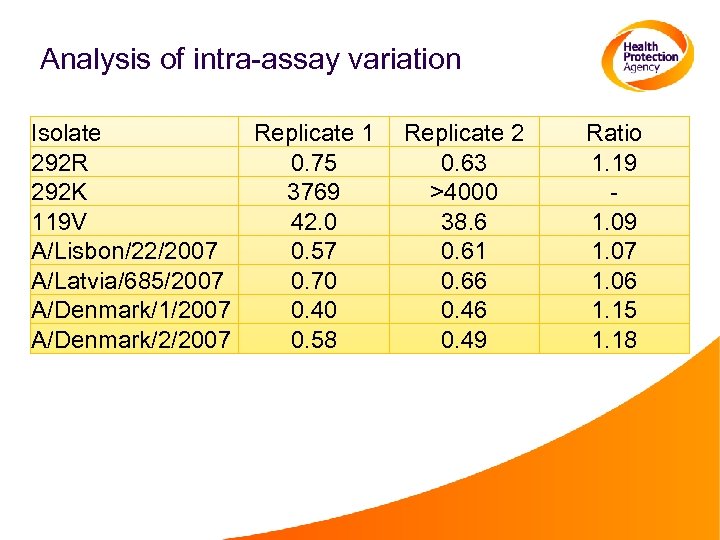
Analysis of intra-assay variation Isolate Replicate 1 292 R 0. 75 292 K 3769 119 V 42. 0 A/Lisbon/22/2007 0. 57 A/Latvia/685/2007 0. 70 A/Denmark/1/2007 0. 40 A/Denmark/2/2007 0. 58 Replicate 2 0. 63 >4000 38. 6 0. 61 0. 66 0. 49 Ratio 1. 19 1. 07 1. 06 1. 15 1. 18

Comments: Intra-assay Variation • Variation between replicates in the same assay can be 15%-20%. • This variation is greater than that seen with changes to curve-fitting method • Using replicates and taking the average reduces this effect. • A large difference between replicates (e. g. >30%) of a given virus indicates a technical issue In these instances repeat testing should be performed
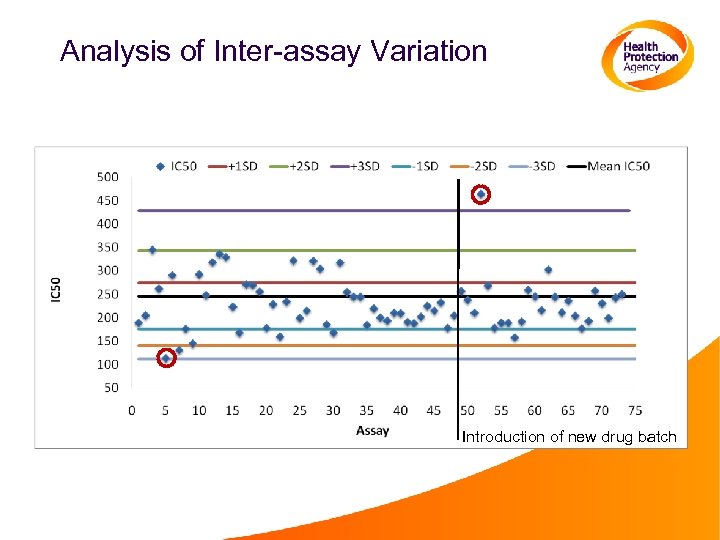
Analysis of Inter-assay Variation Introduction of new drug batch
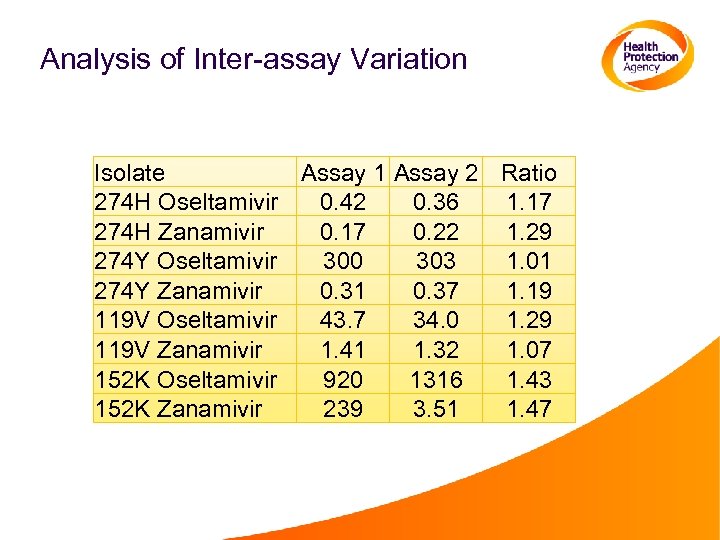
Analysis of Inter-assay Variation Isolate Assay 1 Assay 2 Ratio 274 H Oseltamivir 0. 42 0. 36 1. 17 274 H Zanamivir 0. 17 0. 22 1. 29 274 Y Oseltamivir 300 303 1. 01 274 Y Zanamivir 0. 31 0. 37 1. 19 119 V Oseltamivir 43. 7 34. 0 1. 29 119 V Zanamivir 1. 41 1. 32 1. 07 152 K Oseltamivir 920 1316 1. 43 152 K Zanamivir 239 3. 51 1. 47

Comments: Inter-assay Variation • Variation in IC 50 values for a virus in multiple assays can be 50%. • Control viruses should be included in every assay to identify technical issues. • Control viruses should be validated, and have a defined range between which the IC 50 is valid. Assays in which the control virus IC 50 falls outside the accepted range should be reaped in their entirety.

Conclusions: Determining IC 50 Values • Several methods for IC 50 calculation available at a range of price and sophistication • Variation due to choice of IC 50 calculation method is minimal (510%) in comparison with intra-assay (20%) and inter-assay (50%) variation. • Choice of curve-fitting method should be made based on individual laboratory circumstances • All variation can be minimised using appropriate assay controls (reference/control viruses, validation of curves generated) • Consistency in methodology used (statistical and laboratory) is important for long term analysis (time trends)

Identifying IC 50 outliers • Aim: identify isolates with higher (or lower) than expected IC 50 values (outliers) • First determine the ‘normal range’ of IC 50 values • Each • Various statistical methods may be used • Critical to ensure that any outliers do not unduly affect the cut-off/threshold • Outlier does not equal resistant Identifies isolates that may be worth further investigation (retesting/sequencing)

Identifying IC 50 outliers: Commonly Used Statistical Methods • SMAD Robust estimate of the standard deviation based on the median absolute deviation from the median • Box and Whisker plots Graphical representation of the 5 number summary of the data (the sample minimum, the lower or first quartile, the median, the upper or third quartile, the sample maximum) • Both methods require a minimum dataset to perform robust analyses (>20) Cut offs can be calculated mid-season, once a reasonable number of samples has been tested, to monitor outliers At the end of the season, cut offs can be updated and a retrospective analysis of all season data performed.

Using SMAD Analysis • Create a scatter plot of all data • Useful to see the spread and trend of the data • Log transform the data • Calculate a robust estimate of the standard deviation based on the median absolute deviation from the median using log 10 data Templates for this analyses are available from HPA, UK • Major outliers: all those with values more than 3 SD above the median • Minor Outliers: all those more than 1. 65 SD above the median
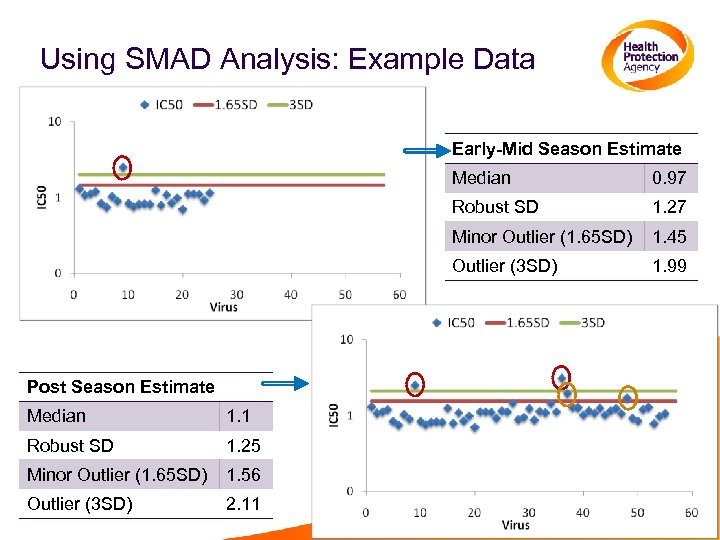
Using SMAD Analysis: Example Data Early-Mid Season Estimate Median Robust SD 1. 1 Robust SD 1. 25 Minor Outlier (1. 65 SD) 1. 56 Outlier (3 SD) 2. 11 1. 45 Outlier (3 SD) Median 1. 27 Minor Outlier (1. 65 SD) Post Season Estimate 0. 97 1. 99

Using Box and Whisker Analysis • This analysis can be performed in Graphpad Prism, with the box and whisker plots drawn automatically • Calculations can be done in excel, but drawing the box and whisker plots is more complicated A template for plotting the graphs is available from Adam Meijer • Log transform the data • Calculate the median, upper quartile, lower quartile, interquartile range, upper minor and major fences, and lower major and minor fences • Mild outliers lie between the minor and major fences • Extreme outliers lie outside the major fence
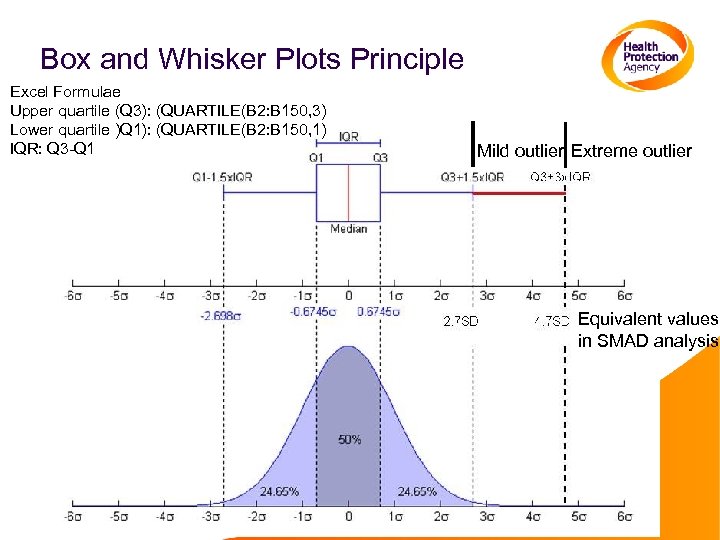
Box and Whisker Plots Principle Excel Formulae Upper quartile (Q 3): (QUARTILE(B 2: B 150, 3) Lower quartile )Q 1): (QUARTILE(B 2: B 150, 1) IQR: Q 3 -Q 1 Mild outlier Extreme outlier Equivalent values in SMAD analysis
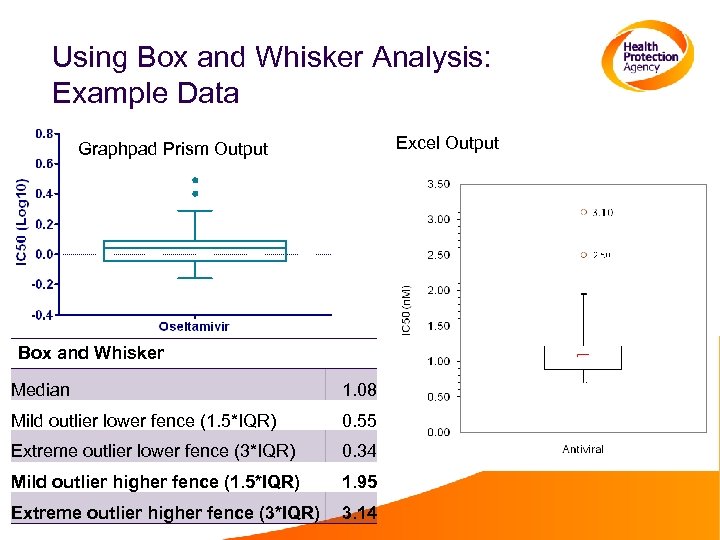
Using Box and Whisker Analysis: Example Data Excel Output Graphpad Prism Output Box and Whisker Median 1. 08 Mild outlier lower fence (1. 5*IQR) 0. 55 Extreme outlier lower fence (3*IQR) 0. 34 Mild outlier higher fence (1. 5*IQR) 1. 95 Extreme outlier higher fence (3*IQR) 3. 14

Do we need to log-transform? • Most results from dilution assays produce ‘geometric results’ so likely to be sensible • Sometimes data are skewed. (e. g. lower quartile much closer to median than upper quartile) • Important to log transform as robust methods assume data are normal once outliers are removed.
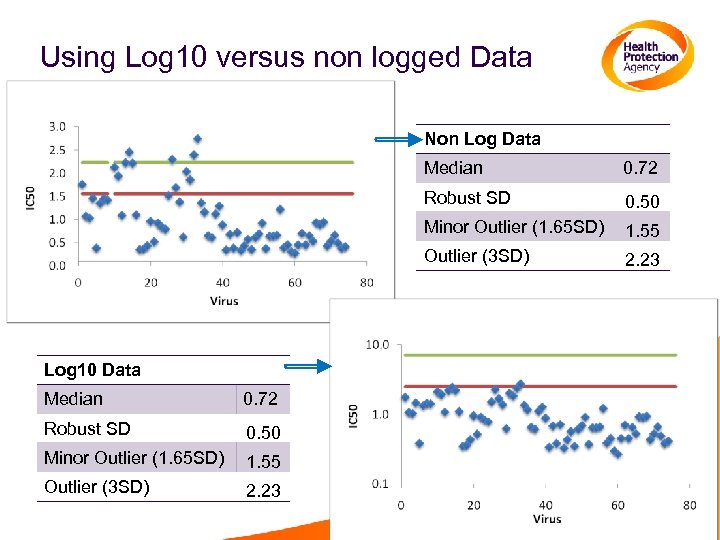
Using Log 10 versus non logged Data Non Log Data Median Robust SD 0. 72 Robust SD 0. 50 Minor Outlier (1. 65 SD) 1. 55 Outlier (3 SD) 2. 23 1. 55 Outlier (3 SD) Median 0. 50 Minor Outlier (1. 65 SD) Log 10 Data 0. 72 2. 23
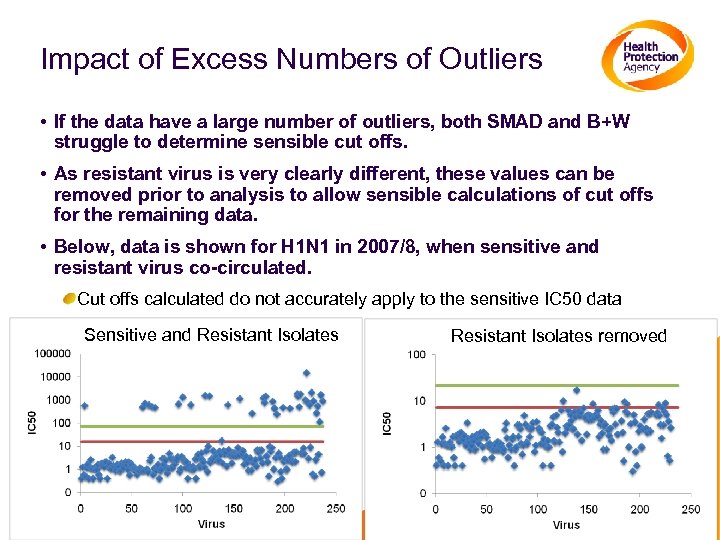
Impact of Excess Numbers of Outliers • If the data have a large number of outliers, both SMAD and B+W struggle to determine sensible cut offs. • As resistant virus is very clearly different, these values can be removed prior to analysis to allow sensible calculations of cut offs for the remaining data. • Below, data is shown for H 1 N 1 in 2007/8, when sensitive and resistant virus co-circulated. Cut offs calculated do not accurately apply to the sensitive IC 50 data Sensitive and Resistant Isolates removed

Conclusions: Identifying Outliers • Determining cut offs/thresholds identifies those isolates with IC 50 values higher than the normal range Cut offs/thresholds need to be subtype specific Season specific cut offs are useful, if enough data is generated in one season, but data from multiple seasons can be merged to perform a more reliable analyses • Box and whisker plots and SMAD analyses generate slightly different cut offs Q 3+1. 5 x. IQR (mild outlier cut off) is equivalent to 2. 7 SD from SMAD Q 3+3 x. ICR is equivalent to 4. 7 SD from SMAD. Cut offs calculated by box and whisker analyses are higher than those from SMAD analyses. • Choice depends on individual laboratory preference Box and whisker plots present the data well • Both methods minimise the impact of outlier values on the analyses, but both will fail once too many outliers are present Data begins to have two populations

Monitoring Trends Over Time • The normal range of IC 50 values for a particular subtype can change over time • This could be seen by an increase in the number of outliers, or by changes in the median • Simple to monitor, using the methods already described for identifying outliers • scatter plots/box-whisker • Other statistical methods can be used to further analyse data from several seasons
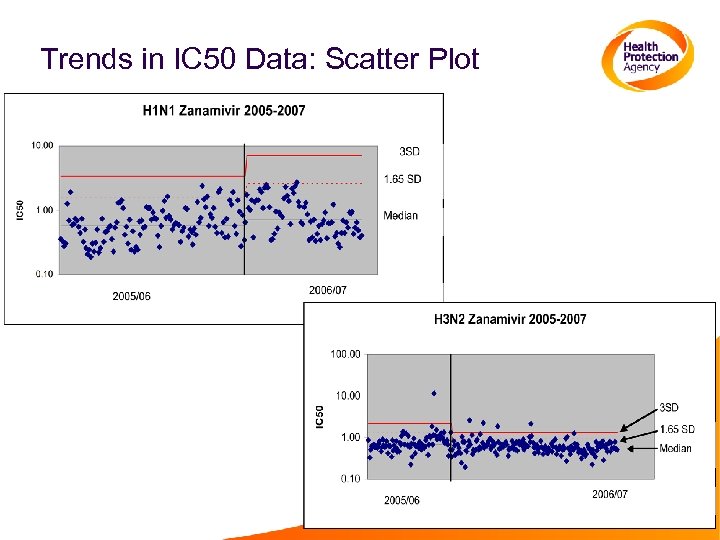
Trends in IC 50 Data: Scatter Plot
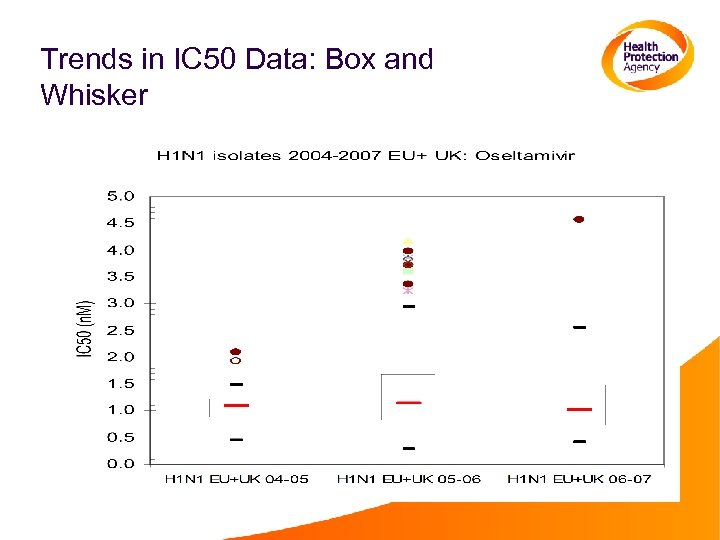
Trends in IC 50 Data: Box and Whisker

Summary • Good use of statistical methods can help interpret the IC 50 results and ensure assay results are reliable. • Analyses of data not only identifies individual outliers, but allows continuous monitoring of trends • Retrospective analyses of multiple seasons of data can identify changes in viral characteristics and susceptibilities • Do not use statistics without first looking at the data by scatter plot to find obvious deviations which require an adapted statistical approach • Challenge is to find explanations for trends
db4d5b418b9ea5787ea1cc6b185c16e8.ppt