c06136e2ead7d9b0633bd646a5bec263.ppt
- Количество слайдов: 55


 In 1989 along the coast of Emilia Romagna…
In 1989 along the coast of Emilia Romagna…
 Mytilus galloprovincialis
Mytilus galloprovincialis
 Mouse Bioassay for DSP Toxins • Intraperitoneal (IP) injection of lipophylic extract of shellfish digestive glands into 1820 g mouse • Measurement time to death; one mouse unit = amount that kills mouse in 24 hours • Regulatory level: 0. 05 MU per • Rapid and inexpensive g of edible tissue (equivalent • Poor reproducibility and long assay to 200 ng/g of okadaic acid) time • A validated method accepted by several countries • False positives mostly due to the presence of high levels of fatty acids • No information on toxins profile
Mouse Bioassay for DSP Toxins • Intraperitoneal (IP) injection of lipophylic extract of shellfish digestive glands into 1820 g mouse • Measurement time to death; one mouse unit = amount that kills mouse in 24 hours • Regulatory level: 0. 05 MU per • Rapid and inexpensive g of edible tissue (equivalent • Poor reproducibility and long assay to 200 ng/g of okadaic acid) time • A validated method accepted by several countries • False positives mostly due to the presence of high levels of fatty acids • No information on toxins profile
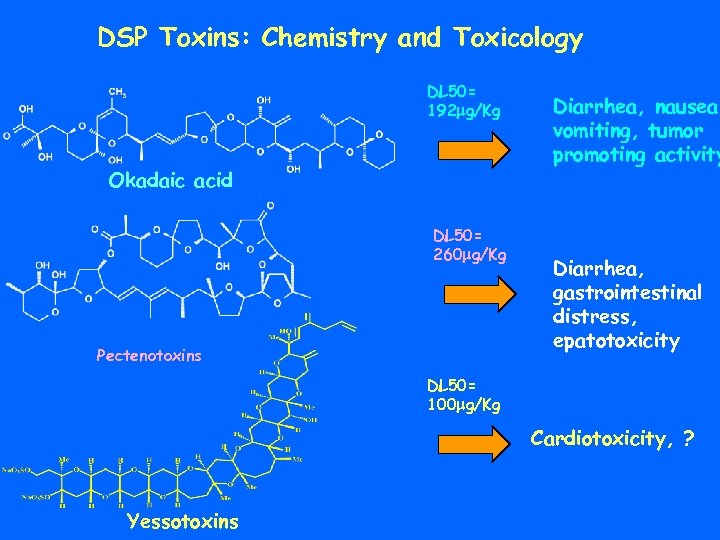 DSP Toxins: Chemistry and Toxicology DL 50= 192 mg/Kg Okadaic acid DL 50= 260 mg/Kg Pectenotoxins Diarrhea, nausea, vomiting, tumor promoting activity Diarrhea, gastrointestinal distress, epatotoxicity DL 50= 100 mg/Kg Cardiotoxicity, ? Yessotoxins
DSP Toxins: Chemistry and Toxicology DL 50= 192 mg/Kg Okadaic acid DL 50= 260 mg/Kg Pectenotoxins Diarrhea, nausea, vomiting, tumor promoting activity Diarrhea, gastrointestinal distress, epatotoxicity DL 50= 100 mg/Kg Cardiotoxicity, ? Yessotoxins
 ~200 Kg of mussels extraction hundreds g of extract chromatografic separation Fats Sterols Contaminants Toxins (mg)
~200 Kg of mussels extraction hundreds g of extract chromatografic separation Fats Sterols Contaminants Toxins (mg)
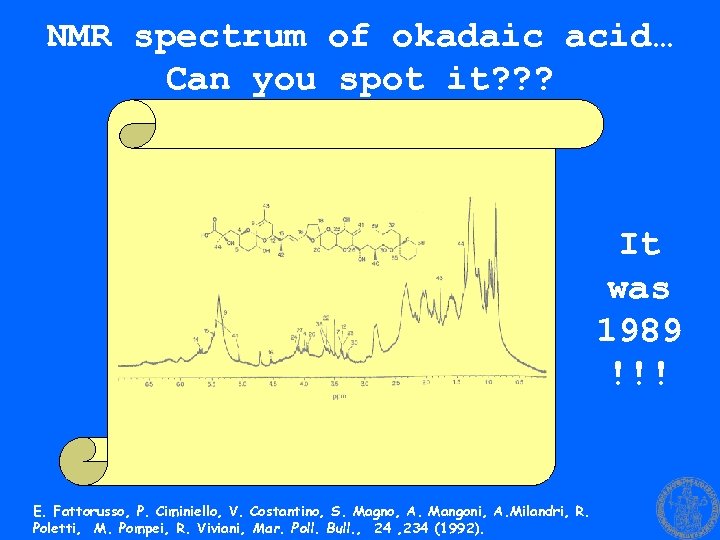 NMR spectrum of okadaic acid… Can you spot it? ? ? It was 1989 !!! E. Fattorusso, P. Ciminiello, V. Costantino, S. Magno, A. Mangoni, A. Milandri, R. Poletti, M. Pompei, R. Viviani, Mar. Poll. Bull. , 24 , 234 (1992).
NMR spectrum of okadaic acid… Can you spot it? ? ? It was 1989 !!! E. Fattorusso, P. Ciminiello, V. Costantino, S. Magno, A. Mangoni, A. Milandri, R. Poletti, M. Pompei, R. Viviani, Mar. Poll. Bull. , 24 , 234 (1992).
 Adriatic toxin profile: chronicle 1989: for the very first time okadaic acid was spotted in the Adriatic Sea From 1989 on: Okadaic Acid has been the main Adriatic toxin
Adriatic toxin profile: chronicle 1989: for the very first time okadaic acid was spotted in the Adriatic Sea From 1989 on: Okadaic Acid has been the main Adriatic toxin
 ce n e L ’s t ow n ou b i to n !! 5! 99 1
ce n e L ’s t ow n ou b i to n !! 5! 99 1
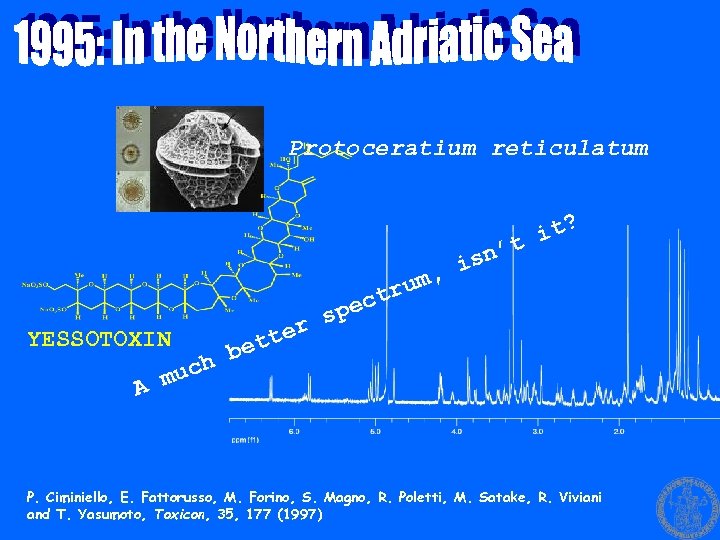 Protoceratium reticulatum it? n’t is um, ctr spe ter YESSOTOXIN bet uch A m P. Ciminiello, E. Fattorusso, M. Forino, S. Magno, R. Poletti, M. Satake, R. Viviani and T. Yasumoto, Toxicon, 35, 177 (1997)
Protoceratium reticulatum it? n’t is um, ctr spe ter YESSOTOXIN bet uch A m P. Ciminiello, E. Fattorusso, M. Forino, S. Magno, R. Poletti, M. Satake, R. Viviani and T. Yasumoto, Toxicon, 35, 177 (1997)
 Known Yessotoxin analogues 1995: A RE A Yessotoxin T 1997 : X O C I Yessotoxin homo. Yessotoxin 45 -hydroxy-homo. Yessotoxin
Known Yessotoxin analogues 1995: A RE A Yessotoxin T 1997 : X O C I Yessotoxin homo. Yessotoxin 45 -hydroxy-homo. Yessotoxin
 New Yessotoxin analogues n=1 Adriatoxin (1997) Carboxy YTX (1997) n=2 Carboxyhomo. YTX (1998) P. Ciminiello, E. Fattorusso, M. Forino, S. Magno, R. Poletti, R. Viviani. Tetrahedron Lett. 39, 8897 (1998). n=1 Oxo. YTX (2001) n=2 Oxohomo. YTX (1998) P. Ciminiello, E. Fattorusso, M. Forino, R. Poletti and R. Viviani. Eur. J. Org. Chem. 291 -294 (2000). P. Ciminiello, E. Fattorusso, M. Forino, R. Poletti and R. Viviani. Chem. Res. Toxicol. 13, 770 -774 (2000). P. Ciminiello, E. Fattorusso, M. Forino e R. Poletti Chem. Res. Toxicol. 14, 596 -599 (2001).
New Yessotoxin analogues n=1 Adriatoxin (1997) Carboxy YTX (1997) n=2 Carboxyhomo. YTX (1998) P. Ciminiello, E. Fattorusso, M. Forino, S. Magno, R. Poletti, R. Viviani. Tetrahedron Lett. 39, 8897 (1998). n=1 Oxo. YTX (2001) n=2 Oxohomo. YTX (1998) P. Ciminiello, E. Fattorusso, M. Forino, R. Poletti and R. Viviani. Eur. J. Org. Chem. 291 -294 (2000). P. Ciminiello, E. Fattorusso, M. Forino, R. Poletti and R. Viviani. Chem. Res. Toxicol. 13, 770 -774 (2000). P. Ciminiello, E. Fattorusso, M. Forino e R. Poletti Chem. Res. Toxicol. 14, 596 -599 (2001).
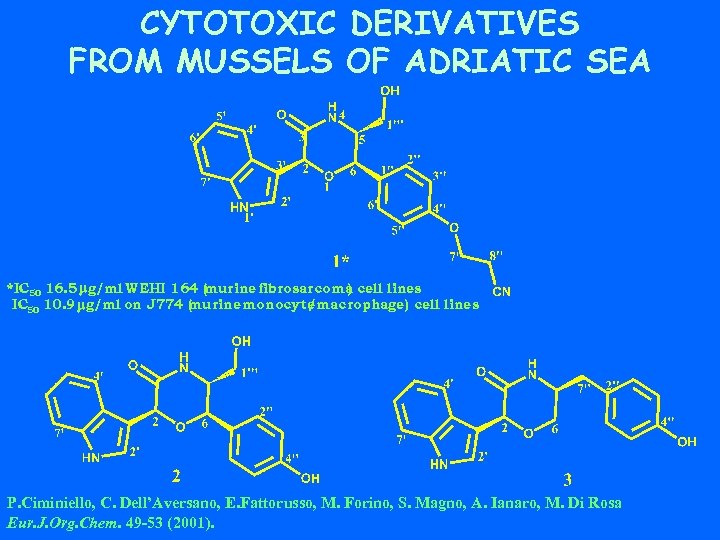 CYTOTOXIC DERIVATIVES FROM MUSSELS OF ADRIATIC SEA *IC 50 16. 5 mg/ml WEHI 164 ( urine fibrosarcoma cell lines m ) IC 50 10. 9 mg/ml on J 774 ( urine monocyte m /macrophage) cell lines P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, A. Ianaro, M. Di Rosa Eur. J. Org. Chem. 49 -53 (2001).
CYTOTOXIC DERIVATIVES FROM MUSSELS OF ADRIATIC SEA *IC 50 16. 5 mg/ml WEHI 164 ( urine fibrosarcoma cell lines m ) IC 50 10. 9 mg/ml on J 774 ( urine monocyte m /macrophage) cell lines P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, A. Ianaro, M. Di Rosa Eur. J. Org. Chem. 49 -53 (2001).
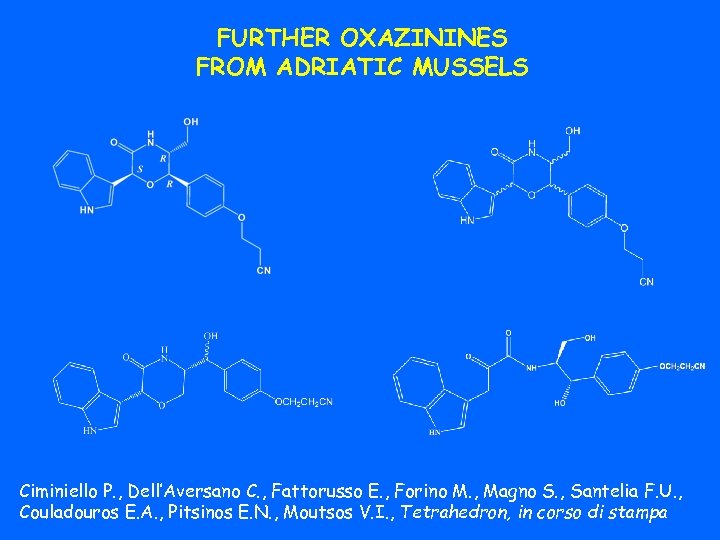 FURTHER OXAZININES FROM ADRIATIC MUSSELS Ciminiello P. , Dell’Aversano C. , Fattorusso E. , Forino M. , Magno S. , Santelia F. U. , Couladouros E. A. , Pitsinos E. N. , Moutsos V. I. , Tetrahedron, in corso di stampa
FURTHER OXAZININES FROM ADRIATIC MUSSELS Ciminiello P. , Dell’Aversano C. , Fattorusso E. , Forino M. , Magno S. , Santelia F. U. , Couladouros E. A. , Pitsinos E. N. , Moutsos V. I. , Tetrahedron, in corso di stampa
 IC 50 (mg/ml) CHLOROSULFOLIPID FROM MUSSELS OF ADRIATIC SEA P. Ciminiello, M. Di Rosa, E. Fattorusso, M. Forino, A. Ianaro, and R. Poletti. J. Org. Chem. 66(2), 578 -582 (2001).
IC 50 (mg/ml) CHLOROSULFOLIPID FROM MUSSELS OF ADRIATIC SEA P. Ciminiello, M. Di Rosa, E. Fattorusso, M. Forino, A. Ianaro, and R. Poletti. J. Org. Chem. 66(2), 578 -582 (2001).
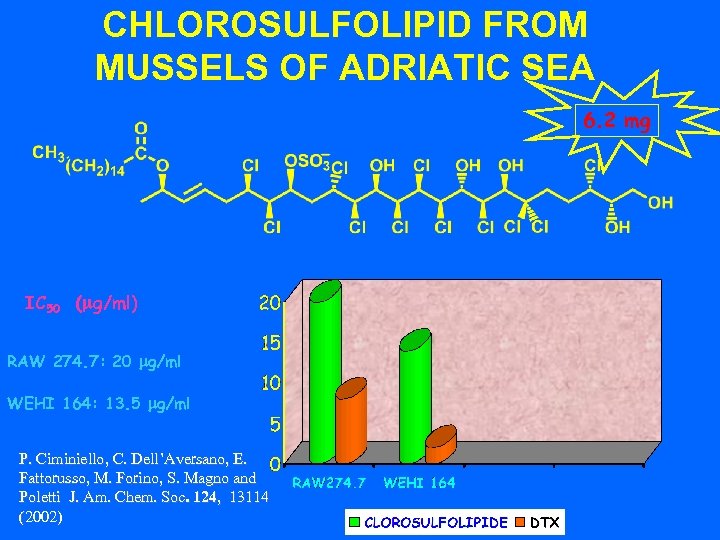 CHLOROSULFOLIPID FROM MUSSELS OF ADRIATIC SEA 6. 2 mg IC 50 (mg/ml) RAW 274. 7: 20 mg/ml WEHI 164: 13. 5 mg/ml P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno and Poletti J. Am. Chem. Soc. 124, 13114 (2002)
CHLOROSULFOLIPID FROM MUSSELS OF ADRIATIC SEA 6. 2 mg IC 50 (mg/ml) RAW 274. 7: 20 mg/ml WEHI 164: 13. 5 mg/ml P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno and Poletti J. Am. Chem. Soc. 124, 13114 (2002)
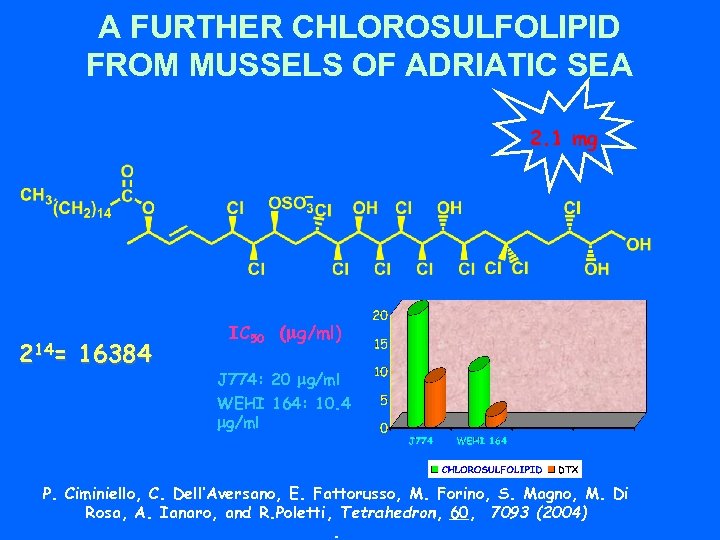 A FURTHER CHLOROSULFOLIPID FROM MUSSELS OF ADRIATIC SEA 2. 1 mg 214= 16384 IC 50 (mg/ml) J 774: 20 mg/ml WEHI 164: 10. 4 mg/ml P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, M. Di Rosa, A. Ianaro, and R. Poletti, Tetrahedron, 60, 7093 (2004).
A FURTHER CHLOROSULFOLIPID FROM MUSSELS OF ADRIATIC SEA 2. 1 mg 214= 16384 IC 50 (mg/ml) J 774: 20 mg/ml WEHI 164: 10. 4 mg/ml P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, M. Di Rosa, A. Ianaro, and R. Poletti, Tetrahedron, 60, 7093 (2004).
 Ø High variation in toxins composition Ø New Biotoxins ØNew Cytotoxins
Ø High variation in toxins composition Ø New Biotoxins ØNew Cytotoxins
 • Monitoring program • qualitative analysis • quantitative analysis
• Monitoring program • qualitative analysis • quantitative analysis
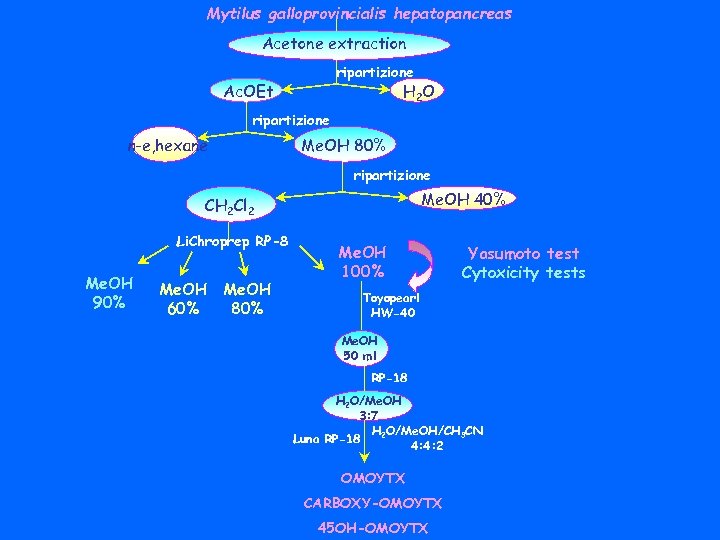 Mytilus galloprovincialis hepatopancreas Acetone extraction ripartizione Ac. OEt H 2 O ripartizione n-e, hexane Me. OH 80% ripartizione Me. OH 40% CH 2 Cl 2 Li. Chroprep RP-8 Me. OH 90% Me. OH 60% Me. OH 80% Me. OH 100% Yasumoto test Cytoxicity tests Toyopearl HW-40 Me. OH 50 ml RP-18 H 2 O/Me. OH 3: 7 H 2 O/Me. OH/CH 3 CN Luna RP-18 4: 4: 2 OMOYTX CARBOXY-OMOYTX 45 OH-OMOYTX
Mytilus galloprovincialis hepatopancreas Acetone extraction ripartizione Ac. OEt H 2 O ripartizione n-e, hexane Me. OH 80% ripartizione Me. OH 40% CH 2 Cl 2 Li. Chroprep RP-8 Me. OH 90% Me. OH 60% Me. OH 80% Me. OH 100% Yasumoto test Cytoxicity tests Toyopearl HW-40 Me. OH 50 ml RP-18 H 2 O/Me. OH 3: 7 H 2 O/Me. OH/CH 3 CN Luna RP-18 4: 4: 2 OMOYTX CARBOXY-OMOYTX 45 OH-OMOYTX
 Analysis Methods: LC-MS • Molecular weight & elemental composition (HRMS) • Structural information (MS/MS) • • • Wide range of analytes possible Universal AND selective High sensitivity Excellent quantitation Automated analysis • Legally-acceptable confirmation • Discovery of new toxins, analogs and metabolites • Multiple toxin determinations
Analysis Methods: LC-MS • Molecular weight & elemental composition (HRMS) • Structural information (MS/MS) • • • Wide range of analytes possible Universal AND selective High sensitivity Excellent quantitation Automated analysis • Legally-acceptable confirmation • Discovery of new toxins, analogs and metabolites • Multiple toxin determinations
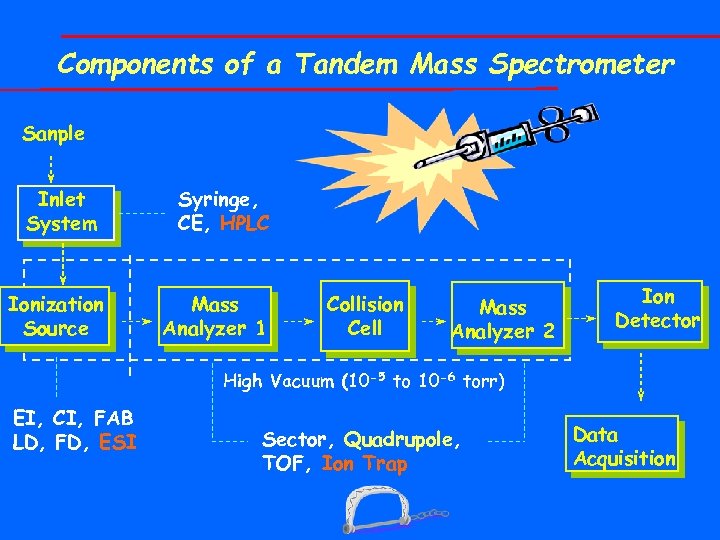 Components of a Tandem Mass Spectrometer Sanple Inlet System Ionization Source Syringe, CE, HPLC Mass Analyzer 1 Collision Cell Mass Analyzer 2 Ion Detector High Vacuum (10 -5 to 10 -6 torr) EI, CI, FAB LD, FD, ESI Sector, Quadrupole, TOF, Ion Trap Data Acquisition
Components of a Tandem Mass Spectrometer Sanple Inlet System Ionization Source Syringe, CE, HPLC Mass Analyzer 1 Collision Cell Mass Analyzer 2 Ion Detector High Vacuum (10 -5 to 10 -6 torr) EI, CI, FAB LD, FD, ESI Sector, Quadrupole, TOF, Ion Trap Data Acquisition
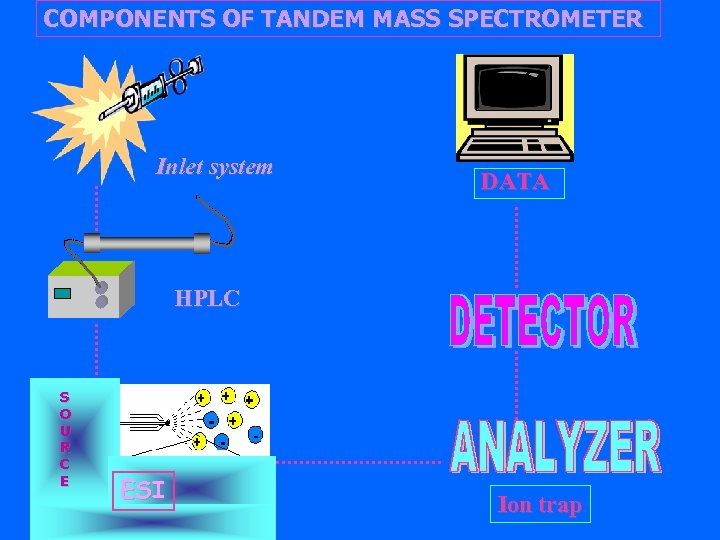 COMPONENTS OF TANDEM MASS SPECTROMETER Inlet system DATA HPLC S O U R C E ESI Ion trap
COMPONENTS OF TANDEM MASS SPECTROMETER Inlet system DATA HPLC S O U R C E ESI Ion trap
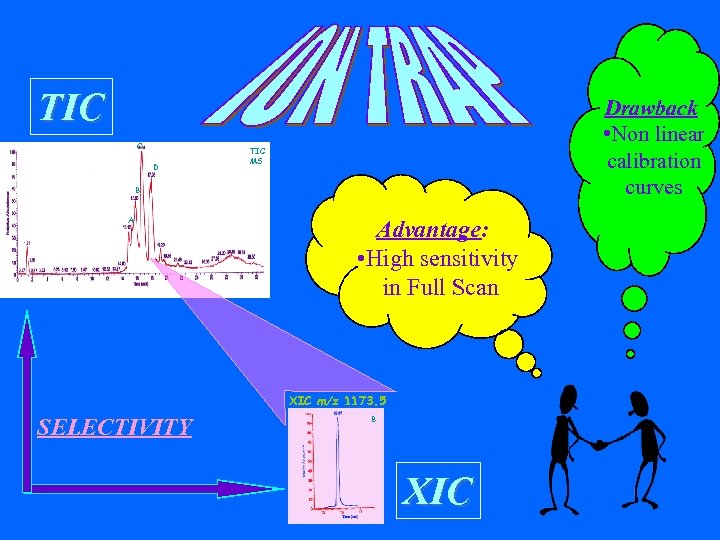 TIC C D Drawback • Non linear calibration curves TIC MS B A Advantage: • High sensitivity in Full Scan XIC m/z 1173. 5 SELECTIVITY B XIC
TIC C D Drawback • Non linear calibration curves TIC MS B A Advantage: • High sensitivity in Full Scan XIC m/z 1173. 5 SELECTIVITY B XIC
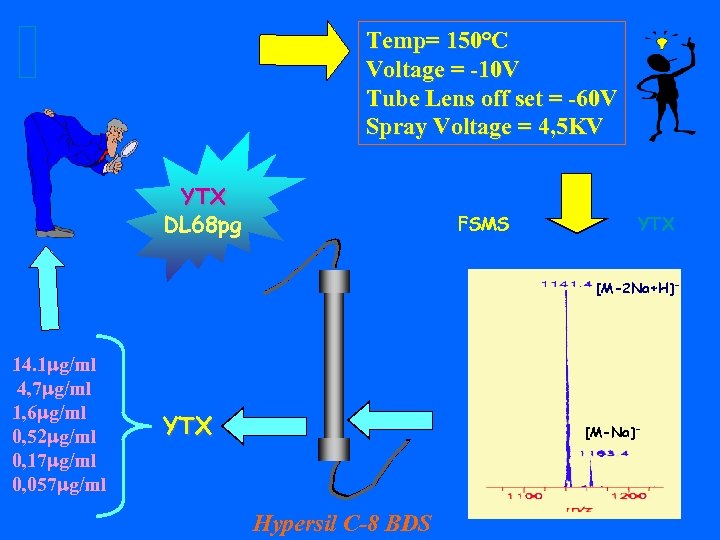 Temp= 150°C Voltage = -10 V Tube Lens off set = -60 V Spray Voltage = 4, 5 KV YTX DL 68 pg FSMS YTX [M-2 Na+H]- 14. 1 mg/ml 4, 7 mg/ml 1, 6 mg/ml 0, 52 mg/ml 0, 17 mg/ml 0, 057 mg/ml YTX [M-Na]- Hypersil C-8 BDS
Temp= 150°C Voltage = -10 V Tube Lens off set = -60 V Spray Voltage = 4, 5 KV YTX DL 68 pg FSMS YTX [M-2 Na+H]- 14. 1 mg/ml 4, 7 mg/ml 1, 6 mg/ml 0, 52 mg/ml 0, 17 mg/ml 0, 057 mg/ml YTX [M-Na]- Hypersil C-8 BDS
 HPLC-ESI-ion trap-MS Method for Direct Detection of Yessotoxins DL 70 pg FINNIGAN LCQ Mass Spectrometer Hypersil 3 mm C 8 BDS, 50 x 2. 0 mm Gradient H 2 O/ACN 2 m. M AF/50 m. M FA P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, and R. Poletti J. Chromat. A, 968, 61 (2002).
HPLC-ESI-ion trap-MS Method for Direct Detection of Yessotoxins DL 70 pg FINNIGAN LCQ Mass Spectrometer Hypersil 3 mm C 8 BDS, 50 x 2. 0 mm Gradient H 2 O/ACN 2 m. M AF/50 m. M FA P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, and R. Poletti J. Chromat. A, 968, 61 (2002).
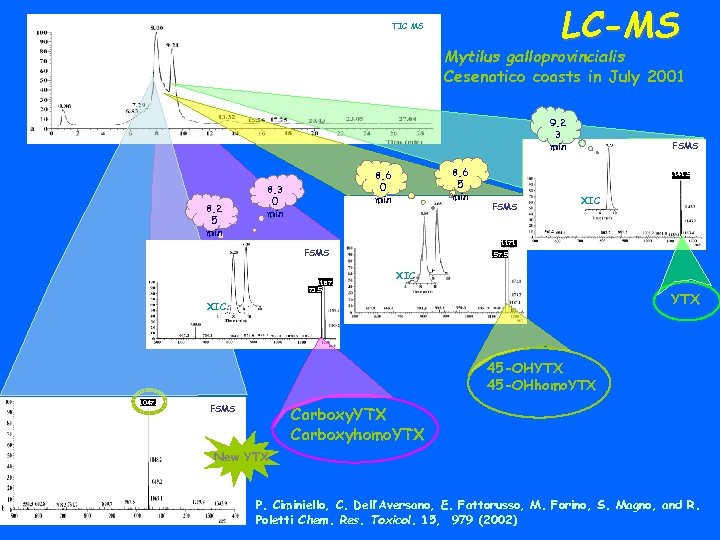 LC-MS TIC MS Mytilus galloprovincialis Cesenatico coasts in July 2001 9. 2 3 min 8. 2 5 min 8. 6 0 min 8. 3 0 min 1141. 5 FSMS XIC 1171. 5 FSMS 1187. 5 1173. 5 FSMS 1157. 5 XIC YTX XIC 45 -OHYTX 45 -OHhomo. YTX 1047. 5 FSMS Carboxy. YTX Carboxyhomo. YTX New YTX P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, and R. Poletti Chem. Res. Toxicol. 15, 979 (2002)
LC-MS TIC MS Mytilus galloprovincialis Cesenatico coasts in July 2001 9. 2 3 min 8. 2 5 min 8. 6 0 min 8. 3 0 min 1141. 5 FSMS XIC 1171. 5 FSMS 1187. 5 1173. 5 FSMS 1157. 5 XIC YTX XIC 45 -OHYTX 45 -OHhomo. YTX 1047. 5 FSMS Carboxy. YTX Carboxyhomo. YTX New YTX P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, S. Magno, and R. Poletti Chem. Res. Toxicol. 15, 979 (2002)
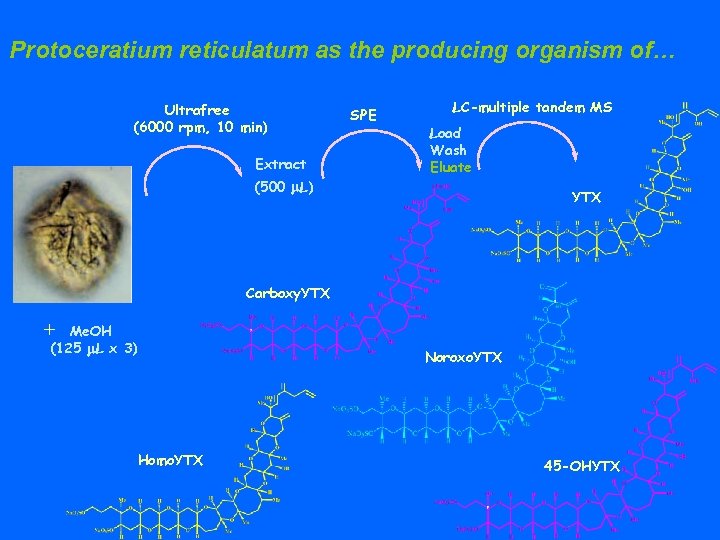 Protoceratium reticulatum as the producing organism of… Ultrafree (6000 rpm, 10 min) Extract SPE LC-multiple tandem MS Load Wash Eluate (500 m. L) YTX Carboxy. YTX + Me. OH (125 m. L x 3) Noroxo. YTX Homo. YTX 45 -OHYTX
Protoceratium reticulatum as the producing organism of… Ultrafree (6000 rpm, 10 min) Extract SPE LC-multiple tandem MS Load Wash Eluate (500 m. L) YTX Carboxy. YTX + Me. OH (125 m. L x 3) Noroxo. YTX Homo. YTX 45 -OHYTX
 Conclusions Some YTX analogues present in large quantities in contaminated shellfish – 45 -hydroxy. YTX, carboxy. YTX, noroxo. YTX could to derive from oxidative modification of the absorbed YTX within the mollusk.
Conclusions Some YTX analogues present in large quantities in contaminated shellfish – 45 -hydroxy. YTX, carboxy. YTX, noroxo. YTX could to derive from oxidative modification of the absorbed YTX within the mollusk.
 In 1995: Yessotoxin was Adriatic toxin profile: detected for the first time chronicle In the Adriatic Sea 1989: 1995 the very first time From for on: Yessotoxins have become okadaic acid was spotted the main Adriatic toxins While Okadaic Adriatic Sea in the acid has slowly subsided From 1989 on: Okadaic Acid At the beginning of the new millennium: has been Okadaic Acid had the main Adriatic toxin virtually disappeared
In 1995: Yessotoxin was Adriatic toxin profile: detected for the first time chronicle In the Adriatic Sea 1989: 1995 the very first time From for on: Yessotoxins have become okadaic acid was spotted the main Adriatic toxins While Okadaic Adriatic Sea in the acid has slowly subsided From 1989 on: Okadaic Acid At the beginning of the new millennium: has been Okadaic Acid had the main Adriatic toxin virtually disappeared
 Anomalous toxicity of Adriatic mussels cultivated in November 2004
Anomalous toxicity of Adriatic mussels cultivated in November 2004
 ALLOWANCE LEVELS SUGGESTED BY EU TOXIN OKADAIC ACID YESSOTOXIN 100 g SHELLFISH 16 mg 100 mg
ALLOWANCE LEVELS SUGGESTED BY EU TOXIN OKADAIC ACID YESSOTOXIN 100 g SHELLFISH 16 mg 100 mg
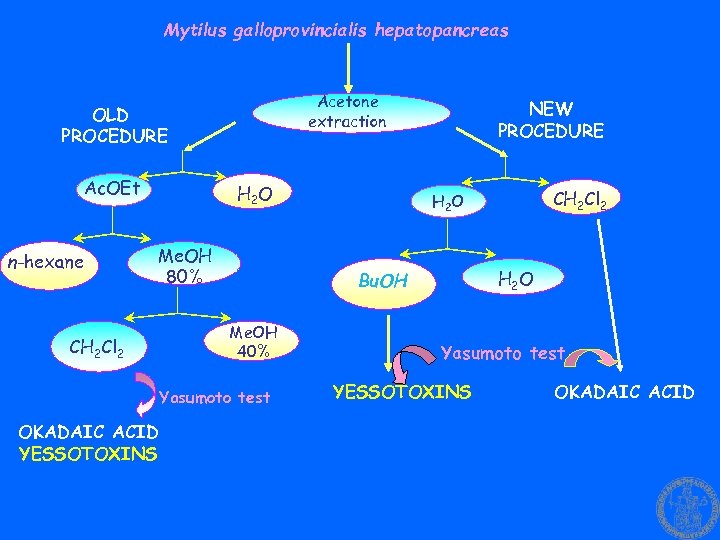 Mytilus galloprovincialis hepatopancreas Acetone extraction OLD PROCEDURE Ac. OEt n-hexane H 2 O Me. OH 80% CH 2 Cl 2 Me. OH 40% CH 2 Cl 2 H 2 O Bu. OH Yasumoto test OKADAIC ACID YESSOTOXINS NEW PROCEDURE Yasumoto test YESSOTOXINS OKADAIC ACID
Mytilus galloprovincialis hepatopancreas Acetone extraction OLD PROCEDURE Ac. OEt n-hexane H 2 O Me. OH 80% CH 2 Cl 2 Me. OH 40% CH 2 Cl 2 H 2 O Bu. OH Yasumoto test OKADAIC ACID YESSOTOXINS NEW PROCEDURE Yasumoto test YESSOTOXINS OKADAIC ACID
 In summer 2004 CH 2 Cl 2 phase deriving From Adriatic mussels Was found TOXIC!!!
In summer 2004 CH 2 Cl 2 phase deriving From Adriatic mussels Was found TOXIC!!!
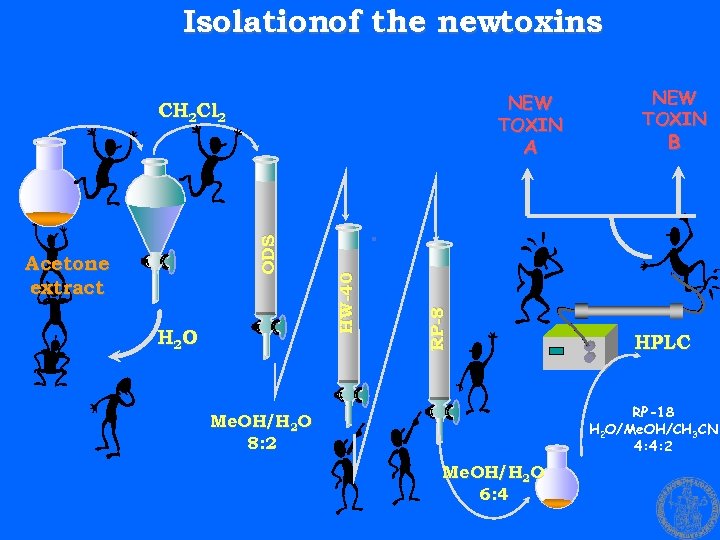 Isolationof the newtoxins NEW TOXIN CH 2 Cl 2 H 2 O RP-8 Acetone extract B . HW-40 ODS A NEW TOXIN HPLC RP-18 H 2 O/Me. OH/CH 3 CN 4: 4: 2 Me. OH/H 2 O 8: 2 Me. OH/H 2 O 6: 4
Isolationof the newtoxins NEW TOXIN CH 2 Cl 2 H 2 O RP-8 Acetone extract B . HW-40 ODS A NEW TOXIN HPLC RP-18 H 2 O/Me. OH/CH 3 CN 4: 4: 2 Me. OH/H 2 O 8: 2 Me. OH/H 2 O 6: 4
 New Toxin- A: Carboxyhomo-1 -desulfo. YTX 1008 HOOC 44 Me 940 HO H 40 869 O H H 727 O H RO 1 H H OH O O E 2 a 3 A 7 B C D 6 R'O 4 O O H H H 2 1 H H O Me Me H O Me OH H O H Me H 2 -1 H-4 5. 0 H-5 4. 0 3. 0 2. 0 1. 0
New Toxin- A: Carboxyhomo-1 -desulfo. YTX 1008 HOOC 44 Me 940 HO H 40 869 O H H 727 O H RO 1 H H OH O O E 2 a 3 A 7 B C D 6 R'O 4 O O H H H 2 1 H H O Me Me H O Me OH H O H Me H 2 -1 H-4 5. 0 H-5 4. 0 3. 0 2. 0 1. 0
 Toxin A Spin system C-1/C-2 a
Toxin A Spin system C-1/C-2 a
 New Toxin- B : Carboxyhomo-4 -desulfo. YTX 1008 HOOC 44 Me 940 HO H 40 869 O H H 727 O H RO 1 H H OH O O E 2 a 3 A 7 B C D 6 R'O 4 O O H H H 2 H H O Me 4 O Me Me H O Me OH H O H Me H 2 -1 H-4 5. 0 H-5 4. 0 3. 0 2. 0 1. 0
New Toxin- B : Carboxyhomo-4 -desulfo. YTX 1008 HOOC 44 Me 940 HO H 40 869 O H H 727 O H RO 1 H H OH O O E 2 a 3 A 7 B C D 6 R'O 4 O O H H H 2 H H O Me 4 O Me Me H O Me OH H O H Me H 2 -1 H-4 5. 0 H-5 4. 0 3. 0 2. 0 1. 0
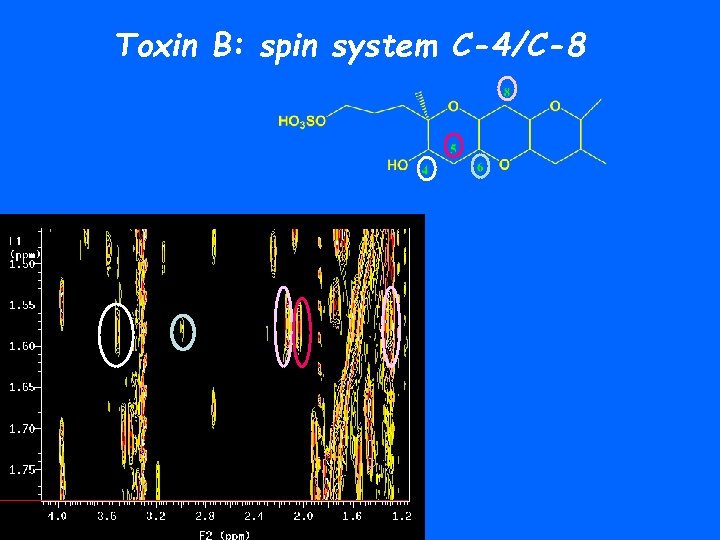 Toxin B: spin system C-4/C-8
Toxin B: spin system C-4/C-8
 Two new toxins in Adriatic mussels: Desulfohomoyessotoxins P. Ciminiello, C. Dell'Aversano, E. Fattorusso, M. Forino, L. Grauso, S. Magno, R. Poletti, L. Tartaglione Chem. Res. in Toxicol. , 20, 95 (2007)
Two new toxins in Adriatic mussels: Desulfohomoyessotoxins P. Ciminiello, C. Dell'Aversano, E. Fattorusso, M. Forino, L. Grauso, S. Magno, R. Poletti, L. Tartaglione Chem. Res. in Toxicol. , 20, 95 (2007)
 What issues do Carboxyhomodesulfo. YTXs Raise? Mytilus galloprovincialis hepatopancreas EU official protocol for extracting Marine Biotoxins needs to be updated Acetone extraction NEW PROCEDURE Carboxyhomodesulfo. YTX toxicology H 2 O should be investigated Bu. OH CH 2 Cl 2 H 2 O Who is responsible for the lack of the sulfate ester in carboxyhomodesulfo. YTXs? YESSOTOXINS OKADAIC ACID
What issues do Carboxyhomodesulfo. YTXs Raise? Mytilus galloprovincialis hepatopancreas EU official protocol for extracting Marine Biotoxins needs to be updated Acetone extraction NEW PROCEDURE Carboxyhomodesulfo. YTX toxicology H 2 O should be investigated Bu. OH CH 2 Cl 2 H 2 O Who is responsible for the lack of the sulfate ester in carboxyhomodesulfo. YTXs? YESSOTOXINS OKADAIC ACID
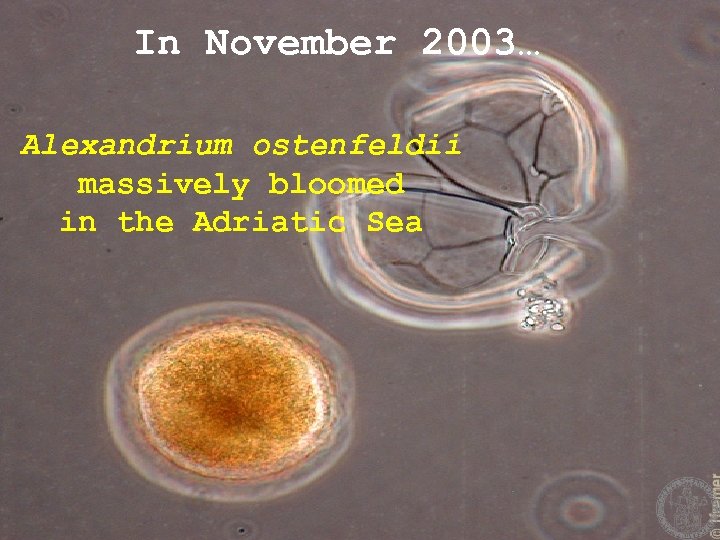 In November 2003… Alexandrium ostenfeldii massively bloomed in the Adriatic Sea
In November 2003… Alexandrium ostenfeldii massively bloomed in the Adriatic Sea
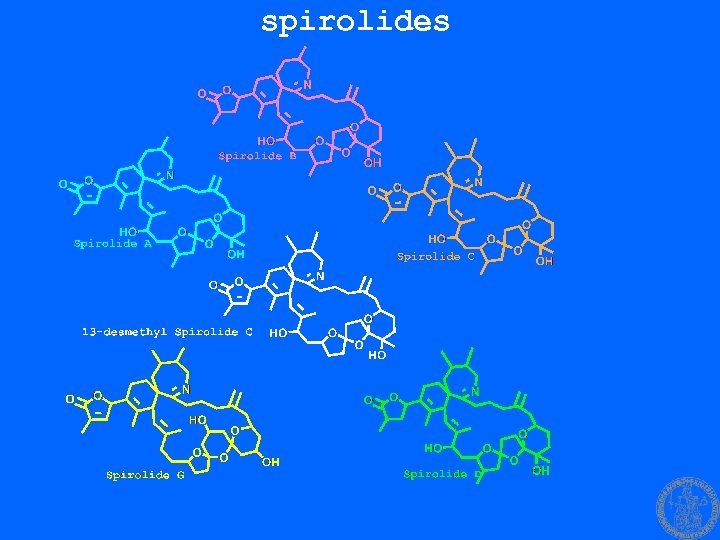 spirolides
spirolides
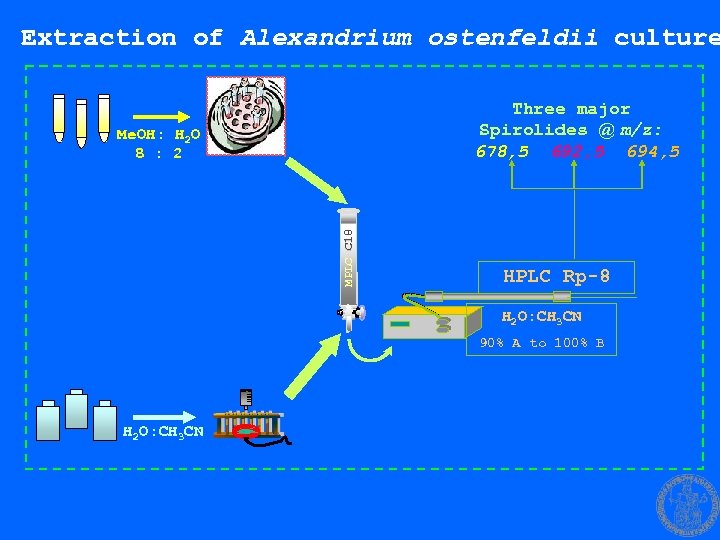 Extraction of Alexandrium ostenfeldii culture Three major Spirolides @ m/z: 678, 5 692, 5 694, 5 MPLC C 18 Me. OH: H 2 O 8 : 2 HPLC Rp-8 H 2 O: CH 3 CN 90% A to 100% B H 2 O: CH 3 CN
Extraction of Alexandrium ostenfeldii culture Three major Spirolides @ m/z: 678, 5 692, 5 694, 5 MPLC C 18 Me. OH: H 2 O 8 : 2 HPLC Rp-8 H 2 O: CH 3 CN 90% A to 100% B H 2 O: CH 3 CN
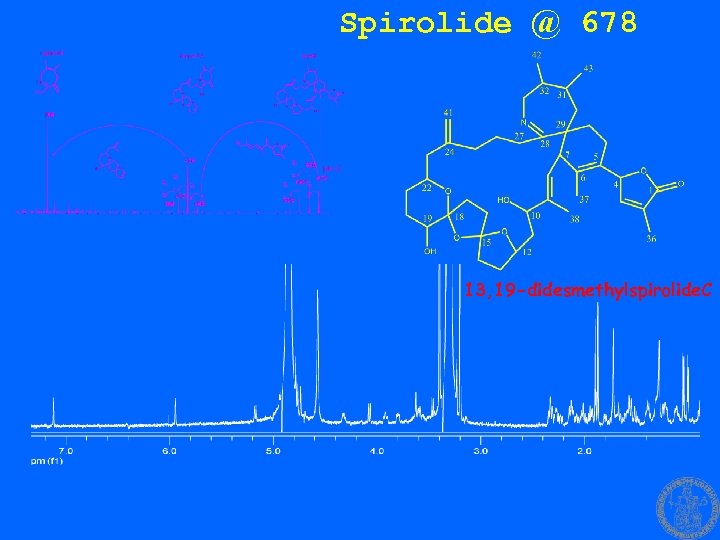 Spirolide @ 678 13, 19 -didesmethylspirolide. C
Spirolide @ 678 13, 19 -didesmethylspirolide. C
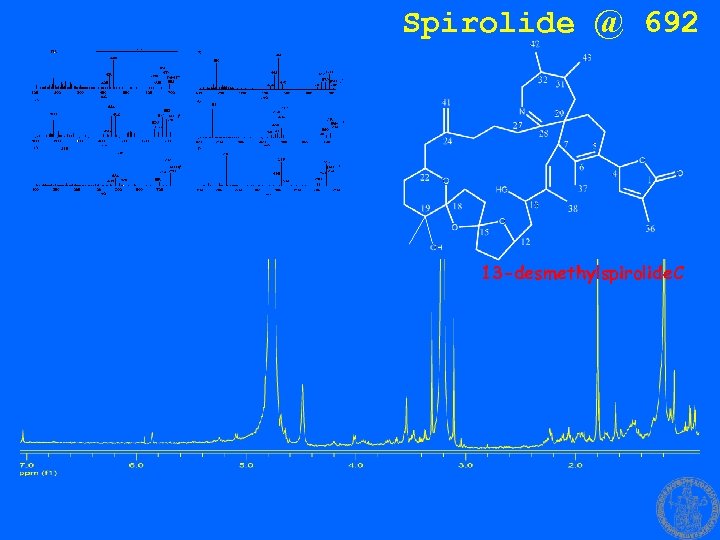 Spirolide @ 692 13 -desmethylspirolide. C
Spirolide @ 692 13 -desmethylspirolide. C
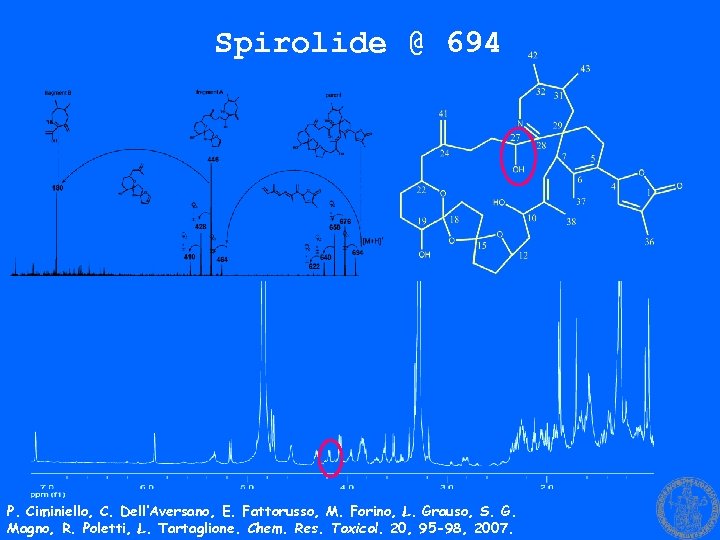 Spirolide @ 694 P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, L. Grauso, S. G. Magno, R. Poletti, L. Tartaglione. Chem. Res. Toxicol. 20, 95 -98, 2007.
Spirolide @ 694 P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, L. Grauso, S. G. Magno, R. Poletti, L. Tartaglione. Chem. Res. Toxicol. 20, 95 -98, 2007.
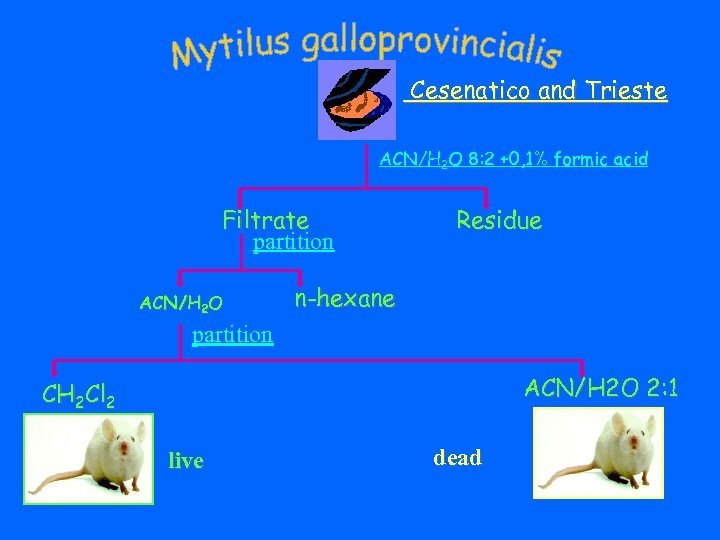 Cesenatico and Trieste ACN/H 2 O 8: 2 +0, 1% formic acid Filtrate partition ACN/H 2 O Residue n-hexane partition ACN/H 2 O 2: 1 CH 2 Cl 2 live dead
Cesenatico and Trieste ACN/H 2 O 8: 2 +0, 1% formic acid Filtrate partition ACN/H 2 O Residue n-hexane partition ACN/H 2 O 2: 1 CH 2 Cl 2 live dead
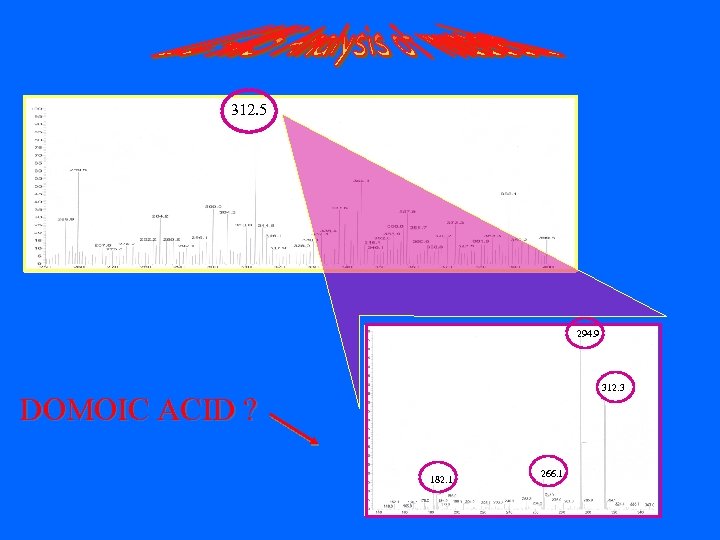 312. 5 294. 9 312. 3 DOMOIC ACID ? 182. 1 266. 1
312. 5 294. 9 312. 3 DOMOIC ACID ? 182. 1 266. 1
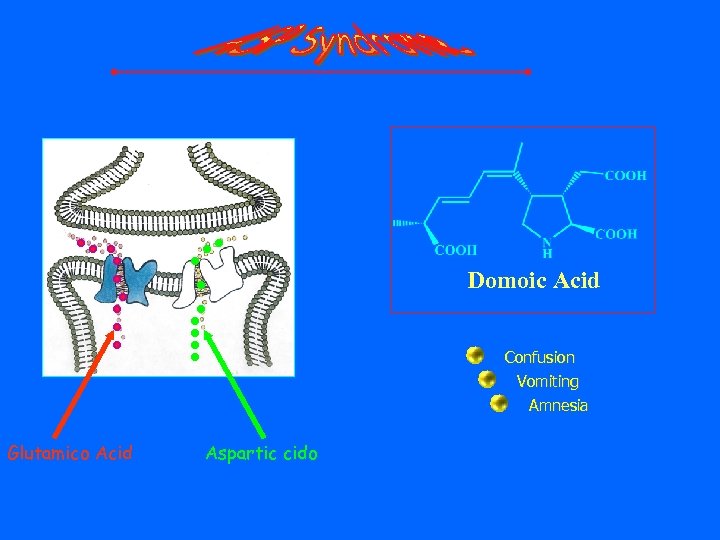 Domoic Acid Confusion Vomiting Amnesia Glutamico Acid Aspartic cido
Domoic Acid Confusion Vomiting Amnesia Glutamico Acid Aspartic cido
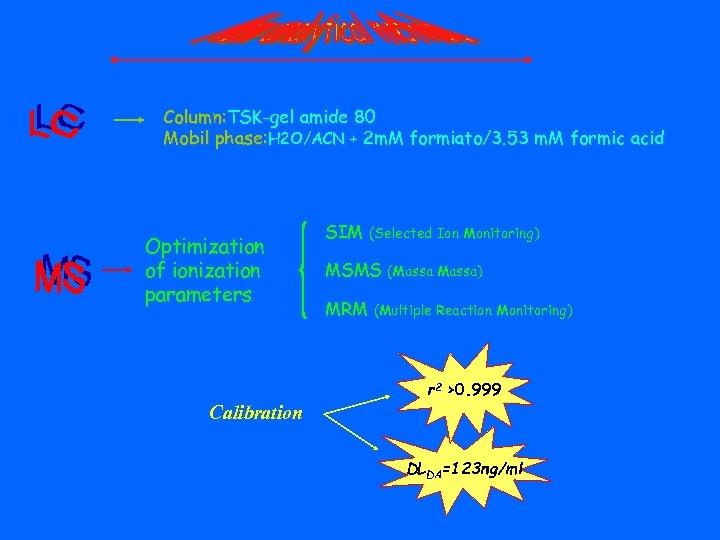 Column: TSK-gel amide 80 Mobil phase: H 2 O/ACN + 2 m. M formiato/3. 53 m. M formic acid Optimization of ionization parameters SIM (Selected Ion Monitoring) MSMS MRM (Massa) (Multiple Reaction Monitoring) r 2 >0. 999 Calibration DLDA=123 ng/ml
Column: TSK-gel amide 80 Mobil phase: H 2 O/ACN + 2 m. M formiato/3. 53 m. M formic acid Optimization of ionization parameters SIM (Selected Ion Monitoring) MSMS MRM (Massa) (Multiple Reaction Monitoring) r 2 >0. 999 Calibration DLDA=123 ng/ml
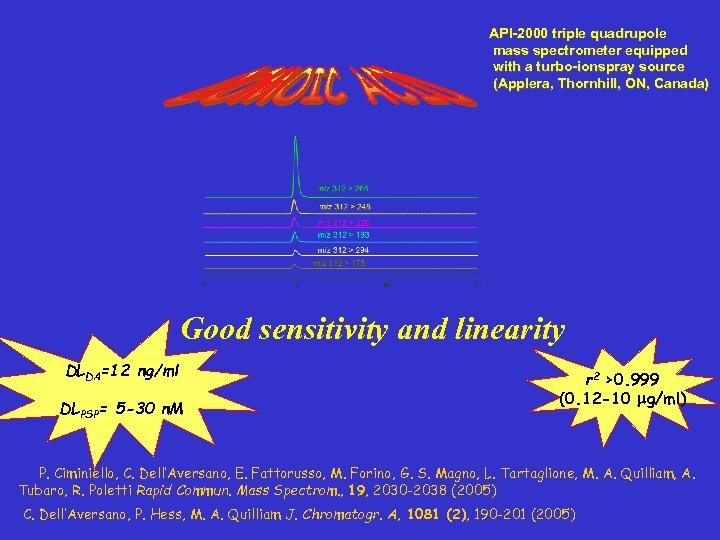 API-2000 triple quadrupole mass spectrometer equipped with a turbo-ionspray source (Applera, Thornhill, ON, Canada) Good sensitivity and linearity DLDA=12 ng/ml DLPSP= 5 -30 n. M r 2 >0. 999 (0. 12 -10 mg/ml) P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, G. S. Magno, L. Tartaglione, M. A. Quilliam, A. Tubaro, R. Poletti Rapid Commun. Mass Spectrom. , 19, 2030 -2038 (2005) C. Dell’Aversano, P. Hess, M. A. Quilliam J. Chromatogr. A, 1081 (2), 190 -201 (2005)
API-2000 triple quadrupole mass spectrometer equipped with a turbo-ionspray source (Applera, Thornhill, ON, Canada) Good sensitivity and linearity DLDA=12 ng/ml DLPSP= 5 -30 n. M r 2 >0. 999 (0. 12 -10 mg/ml) P. Ciminiello, C. Dell’Aversano, E. Fattorusso, M. Forino, G. S. Magno, L. Tartaglione, M. A. Quilliam, A. Tubaro, R. Poletti Rapid Commun. Mass Spectrom. , 19, 2030 -2038 (2005) C. Dell’Aversano, P. Hess, M. A. Quilliam J. Chromatogr. A, 1081 (2), 190 -201 (2005)
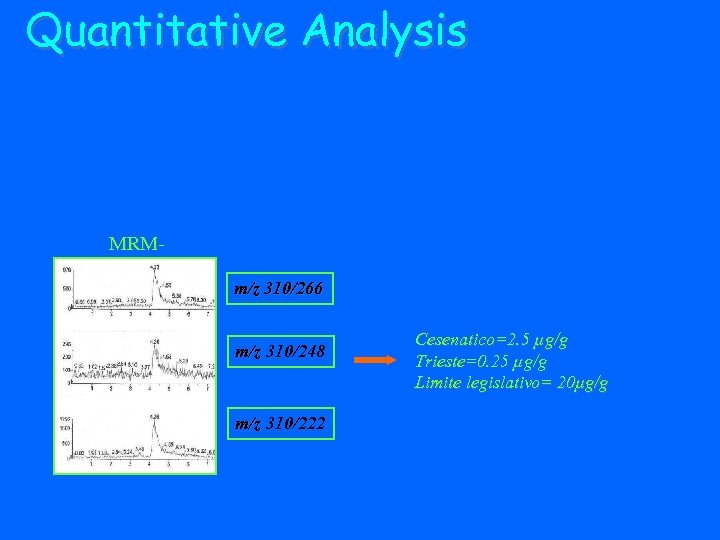 Quantitative Analysis MRMm/z 310/266 m/z 310/248 m/z 310/222 Cesenatico=2. 5 µg/g Trieste=0. 25 µg/g Limite legislativo= 20µg/g
Quantitative Analysis MRMm/z 310/266 m/z 310/248 m/z 310/222 Cesenatico=2. 5 µg/g Trieste=0. 25 µg/g Limite legislativo= 20µg/g


