ab3dc07fbe67673395de83cbb008e2c9.ppt
- Количество слайдов: 56
 Immunoprecipitation 1
Immunoprecipitation 1
 Definition of Immunology Ø The study of the physiological mechanisms which enable the body to recognize materials as foreign and to neutralize, eliminate, or metabolize them without injury to the host tissue. 2
Definition of Immunology Ø The study of the physiological mechanisms which enable the body to recognize materials as foreign and to neutralize, eliminate, or metabolize them without injury to the host tissue. 2
 Overview of Immunodiagnostic Studies Ø Antigen-antibody reactions for diagnosis of: • • infectious disease, autoimmune disorders, immune allergies, and neoplastic disease. Ø These modalities also test for: • blood groups and types, • tissue and graft transplant matching. 3
Overview of Immunodiagnostic Studies Ø Antigen-antibody reactions for diagnosis of: • • infectious disease, autoimmune disorders, immune allergies, and neoplastic disease. Ø These modalities also test for: • blood groups and types, • tissue and graft transplant matching. 3
 Types of Tests Ø Many methods of varying sophistication are used for immunodiagnostic studies 4
Types of Tests Ø Many methods of varying sophistication are used for immunodiagnostic studies 4
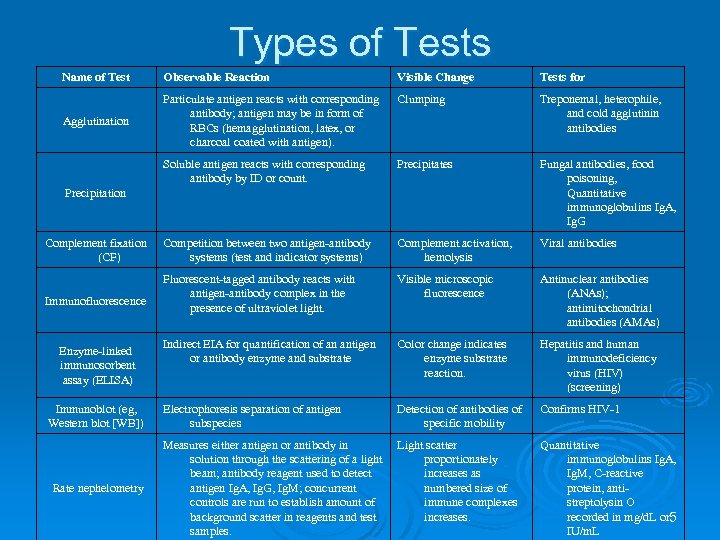 Types of Tests Name of Test Observable Reaction Visible Change Tests for Clumping Agglutination Particulate antigen reacts with corresponding antibody; antigen may be in form of RBCs (hemagglutination, latex, or (hemagglutination, charcoal coated with antigen). Treponemal, heterophile, and cold agglutinin antibodies Soluble antigen reacts with corresponding antibody by ID or count. Precipitates Fungal antibodies, food poisoning, Quantitative immunoglobulins Ig. A, Ig. G Competition between two antigen-antibody systems (test and indicator systems) Complement activation, hemolysis Viral antibodies Fluorescent-tagged antibody reacts with antigen-antibody complex in the presence of ultraviolet light. Visible microscopic fluorescence Antinuclear antibodies (ANAs); antimitochondrial antibodies (AMAs) Indirect EIA for quantification of an antigen or antibody enzyme and substrate Color change indicates enzyme substrate reaction. Hepatitis and human immunodeficiency virus (HIV) (screening) Electrophoresis separation of antigen subspecies Detection of antibodies of specific mobility Confirms HIV-1 Measures either antigen or antibody in solution through the scattering of a light beam; antibody reagent used to detect antigen Ig. A, Ig. G, Ig. M; concurrent Ig. A, Ig. G, Ig. M; controls are run to establish amount of background scatter in reagents and test samples. Light scatter proportionately increases as numbered size of immune complexes increases. Quantitative immunoglobulins Ig. A, Ig. M, C-reactive Ig. M, protein, antistreptolysin O recorded in mg/d. L or 5 mg/d. L IU/m. L Precipitation Complement fixation (CF) Immunofluorescence Enzyme-linked immunosorbent assay (ELISA) Immunoblot (eg, Western blot [WB]) Rate nephelometry
Types of Tests Name of Test Observable Reaction Visible Change Tests for Clumping Agglutination Particulate antigen reacts with corresponding antibody; antigen may be in form of RBCs (hemagglutination, latex, or (hemagglutination, charcoal coated with antigen). Treponemal, heterophile, and cold agglutinin antibodies Soluble antigen reacts with corresponding antibody by ID or count. Precipitates Fungal antibodies, food poisoning, Quantitative immunoglobulins Ig. A, Ig. G Competition between two antigen-antibody systems (test and indicator systems) Complement activation, hemolysis Viral antibodies Fluorescent-tagged antibody reacts with antigen-antibody complex in the presence of ultraviolet light. Visible microscopic fluorescence Antinuclear antibodies (ANAs); antimitochondrial antibodies (AMAs) Indirect EIA for quantification of an antigen or antibody enzyme and substrate Color change indicates enzyme substrate reaction. Hepatitis and human immunodeficiency virus (HIV) (screening) Electrophoresis separation of antigen subspecies Detection of antibodies of specific mobility Confirms HIV-1 Measures either antigen or antibody in solution through the scattering of a light beam; antibody reagent used to detect antigen Ig. A, Ig. G, Ig. M; concurrent Ig. A, Ig. G, Ig. M; controls are run to establish amount of background scatter in reagents and test samples. Light scatter proportionately increases as numbered size of immune complexes increases. Quantitative immunoglobulins Ig. A, Ig. M, C-reactive Ig. M, protein, antistreptolysin O recorded in mg/d. L or 5 mg/d. L IU/m. L Precipitation Complement fixation (CF) Immunofluorescence Enzyme-linked immunosorbent assay (ELISA) Immunoblot (eg, Western blot [WB]) Rate nephelometry
 Antigen-antibody interaction Ø Antigens are substances that stimulate and subsequently react with the products of an immune response. They may be: l l l Ø enzymes, toxins, microorganisms (eg, bacterial, viral, parasitic, fungal), tumors, or autoimmune factors. Antibodies are proteins produced by the body's immune system in response to an antigen or antigens. The antigen-antibody response is the body's natural defense against invading organisms. 6
Antigen-antibody interaction Ø Antigens are substances that stimulate and subsequently react with the products of an immune response. They may be: l l l Ø enzymes, toxins, microorganisms (eg, bacterial, viral, parasitic, fungal), tumors, or autoimmune factors. Antibodies are proteins produced by the body's immune system in response to an antigen or antigens. The antigen-antibody response is the body's natural defense against invading organisms. 6
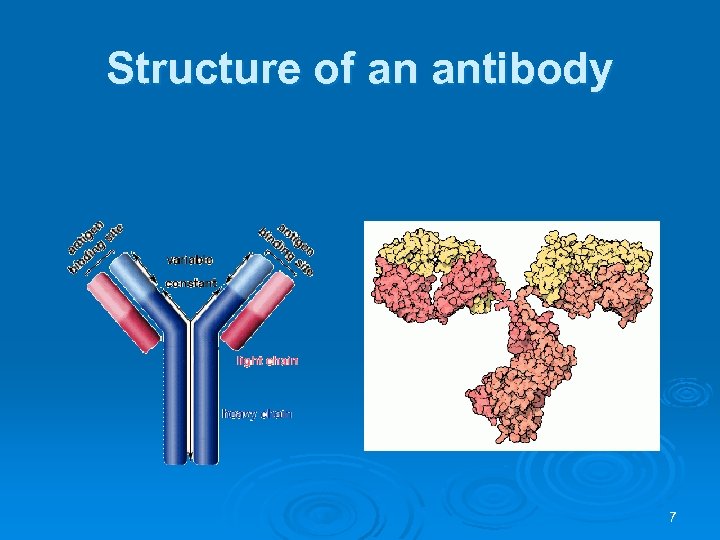 Structure of an antibody 7
Structure of an antibody 7
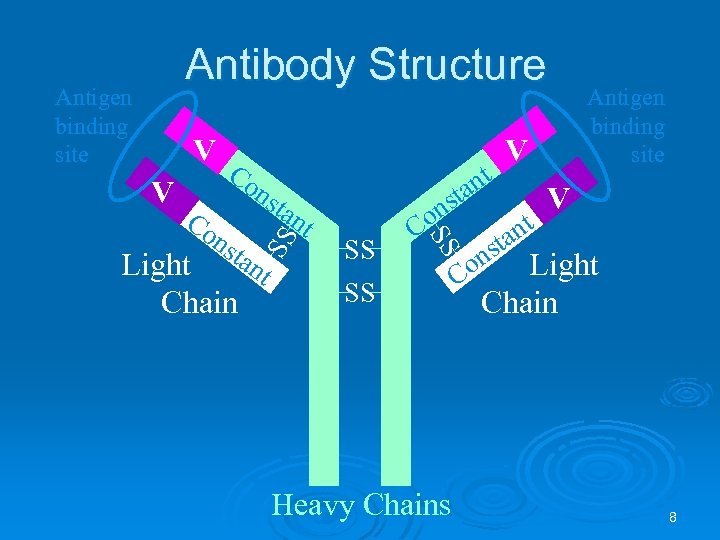 Antibody Structure V V Co nt ta ns tan t tan Light t Chain SS ns Co SS Co ns SS Antigen binding site SS Heavy Chains V nt ta Antigen binding site V ns Light Co Chain 8
Antibody Structure V V Co nt ta ns tan t tan Light t Chain SS ns Co SS Co ns SS Antigen binding site SS Heavy Chains V nt ta Antigen binding site V ns Light Co Chain 8
 Classes of antibodies Ig. G - A monomer - Most abundant antibody in blood. Ig. G easily leaves the circulatory system to fight infection and crosses the placenta conferring passive immunity to a fetus. Ig. D - A monomer - Found on the surface of B cells probably allowing recognition of antigens thus triggering differentiation into plasma and memory B cells Ig. E - A monomer - The least common antibody. The tails attach to mast cells and basophils. When antigens bind, they signal release of histamine. Ig. A - A dimer - Produced by cells in the mucus membranes to prevent attachment of pathogens. Ig. A is also found in many body secretions including milk. Ig. M - A pentamer - First antibody to appear following exposure to an antigen. Because it declines rapidly in the 9 blood, high Ig. M levels indicate a current infection.
Classes of antibodies Ig. G - A monomer - Most abundant antibody in blood. Ig. G easily leaves the circulatory system to fight infection and crosses the placenta conferring passive immunity to a fetus. Ig. D - A monomer - Found on the surface of B cells probably allowing recognition of antigens thus triggering differentiation into plasma and memory B cells Ig. E - A monomer - The least common antibody. The tails attach to mast cells and basophils. When antigens bind, they signal release of histamine. Ig. A - A dimer - Produced by cells in the mucus membranes to prevent attachment of pathogens. Ig. A is also found in many body secretions including milk. Ig. M - A pentamer - First antibody to appear following exposure to an antigen. Because it declines rapidly in the 9 blood, high Ig. M levels indicate a current infection.
 Three Distinct Phases of Antigen/Antibody Reactions Ø Primary Phenomenon – l involves the combination of an individual binding site on an antibody molecule with a single epitope or determinant site on an antigen Ø Secondary Phenomenon – l Lattice formation include precipitation, agglutination and complement fixation Ø Tertiary Phenomenon – l Detected by affect on tissues or cells, Inflammation, phagocytosis, deposition of immune complexes, immune adherence and chemotaxis 10
Three Distinct Phases of Antigen/Antibody Reactions Ø Primary Phenomenon – l involves the combination of an individual binding site on an antibody molecule with a single epitope or determinant site on an antigen Ø Secondary Phenomenon – l Lattice formation include precipitation, agglutination and complement fixation Ø Tertiary Phenomenon – l Detected by affect on tissues or cells, Inflammation, phagocytosis, deposition of immune complexes, immune adherence and chemotaxis 10
 Detection of antigen-antibody interactions: Primary Phenomenon e. g. 1. l l l Enzyme linked immuono sorben assay (ELISA) Immuno flurescent antibody technique (IFAT) Radio immuno assay (RIA) Secondary Phenomenon : e. g. 2. l l l Agglutination tests Complement fixation tests (CFT) Precipitation tests Tertiary Phenomenon : e. g. 3. l Reaction not visible, detected by affect of reaction on tissues or cells 11
Detection of antigen-antibody interactions: Primary Phenomenon e. g. 1. l l l Enzyme linked immuono sorben assay (ELISA) Immuno flurescent antibody technique (IFAT) Radio immuno assay (RIA) Secondary Phenomenon : e. g. 2. l l l Agglutination tests Complement fixation tests (CFT) Precipitation tests Tertiary Phenomenon : e. g. 3. l Reaction not visible, detected by affect of reaction on tissues or cells 11
 Antigen Binding An ab ri y a v V ea H le ab ri t a V gh Li Antigen 3 2 le en tig Antigen 1 12
Antigen Binding An ab ri y a v V ea H le ab ri t a V gh Li Antigen 3 2 le en tig Antigen 1 12
 Ag-Ab interactions Bonds: Ø Ø Hydrogen Ionic Hydrophobic interactions Van der Waals forces Each bond is weak; many are strong To “hold” they must be close and complementary 13
Ag-Ab interactions Bonds: Ø Ø Hydrogen Ionic Hydrophobic interactions Van der Waals forces Each bond is weak; many are strong To “hold” they must be close and complementary 13
 Features of the Antigen-Antibody Interaction • Reversibility Non-covalent Interactions • Affinity Measure of the strength of the binding Ease of association or dissociation • Avidity Increase in affinity due to multivalent binding The summation of multiple affinities 15
Features of the Antigen-Antibody Interaction • Reversibility Non-covalent Interactions • Affinity Measure of the strength of the binding Ease of association or dissociation • Avidity Increase in affinity due to multivalent binding The summation of multiple affinities 15
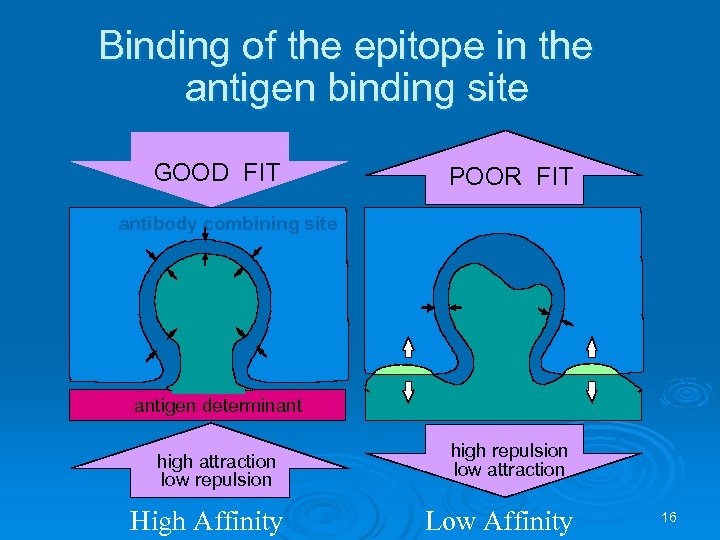 Binding of the epitope in the antigen binding site GOOD FIT POOR FIT antibody combining site antigen determinant high attraction low repulsion High Affinity high repulsion low attraction Low Affinity 16
Binding of the epitope in the antigen binding site GOOD FIT POOR FIT antibody combining site antigen determinant high attraction low repulsion High Affinity high repulsion low attraction Low Affinity 16
 Antibody Affinity Ø Affinity – the strength of the total noncovalent interactions between a single Ag-binding site on an Ab and a single epitope l l Low affinity Abs bind Ag weakly and dissociate readily High affinity Abs bind Ag more tightly and remain bound longer Ag + Ab k 1 k-1 Ab-Ag K 1 = forward (association) constant K-1 = reverse (dissociation) constant 17
Antibody Affinity Ø Affinity – the strength of the total noncovalent interactions between a single Ag-binding site on an Ab and a single epitope l l Low affinity Abs bind Ag weakly and dissociate readily High affinity Abs bind Ag more tightly and remain bound longer Ag + Ab k 1 k-1 Ab-Ag K 1 = forward (association) constant K-1 = reverse (dissociation) constant 17
 Antibody Affinity K can be determined using equilibrium dialysis 18
Antibody Affinity K can be determined using equilibrium dialysis 18
 Antibody Avidity Ø Avidity – the strength of the total noncovalent interactions between a multivalent Ab and Ag l l The avidity of an antibody is a better measure of its binding capacity within biological systems than is the affinity of its individual binding sites High avidity can compensate for low affinity; eg. pentameric Ig. M often has lower affinity than Ig. G but its higher avidity (due to presence of 10 binding sites) may enable it to bind Ag more effectively 20
Antibody Avidity Ø Avidity – the strength of the total noncovalent interactions between a multivalent Ab and Ag l l The avidity of an antibody is a better measure of its binding capacity within biological systems than is the affinity of its individual binding sites High avidity can compensate for low affinity; eg. pentameric Ig. M often has lower affinity than Ig. G but its higher avidity (due to presence of 10 binding sites) may enable it to bind Ag more effectively 20
 Antibody Avidity Keq = 104 Affinity 106 Avidity 1010 Avidity 21
Antibody Avidity Keq = 104 Affinity 106 Avidity 1010 Avidity 21
 Effect of multivalent interactions antibody Ig. G Ig. M effective antibody valence 1 1 2 up to 10 antigen valence 1 n n equilibrium constant (L/M) 104 107 1011 advantage of multi-valence - - 103 -fold 107 -fold affinity avidity definition of bindng Fab 1 affinity intrinsic affinity functional affinity 22
Effect of multivalent interactions antibody Ig. G Ig. M effective antibody valence 1 1 2 up to 10 antigen valence 1 n n equilibrium constant (L/M) 104 107 1011 advantage of multi-valence - - 103 -fold 107 -fold affinity avidity definition of bindng Fab 1 affinity intrinsic affinity functional affinity 22
 Factors Affecting Measurement of Ag/Ab Reactions • Affinity • Avidity Ab excess Ag excess • Ag: Ab ratio • Physical form of Ag Equivalence – Lattice formation 23
Factors Affecting Measurement of Ag/Ab Reactions • Affinity • Avidity Ab excess Ag excess • Ag: Ab ratio • Physical form of Ag Equivalence – Lattice formation 23
 Tests Based on Ag/Ab Reactions Ø All tests based on Ag/Ab reactions will have to depend on lattice formation or they will have to utilize ways to detect small immune complexes Ø All tests based on Ag/Ab reactions can be used to detect either Ag or Ab 24
Tests Based on Ag/Ab Reactions Ø All tests based on Ag/Ab reactions will have to depend on lattice formation or they will have to utilize ways to detect small immune complexes Ø All tests based on Ag/Ab reactions can be used to detect either Ag or Ab 24
 Immunoprecipitation Ø Immunoprecipitation is the technique of precipitating an antigen out of solution using an antibody that specifically binds to that particular antigen. This process can be used to isolate and concentrate a particular antigen from a sample containing many thousands of different antigens. 25
Immunoprecipitation Ø Immunoprecipitation is the technique of precipitating an antigen out of solution using an antibody that specifically binds to that particular antigen. This process can be used to isolate and concentrate a particular antigen from a sample containing many thousands of different antigens. 25
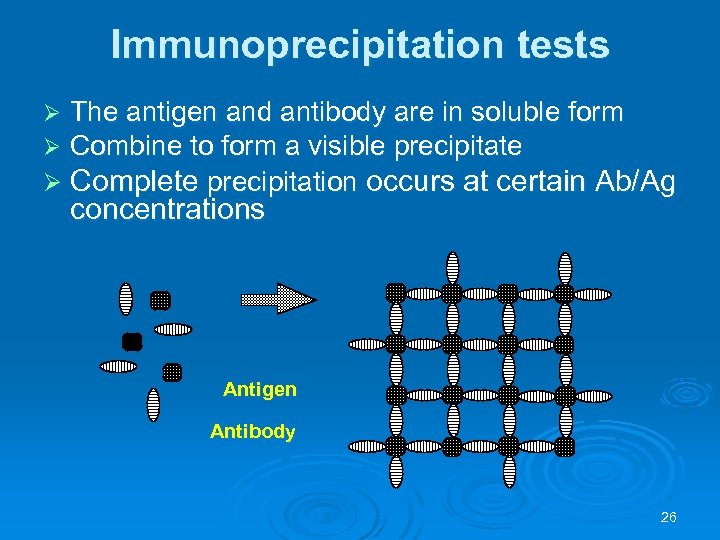 Immunoprecipitation tests The antigen and antibody are in soluble form Combine to form a visible precipitate Ø Complete precipitation occurs at certain Ab/Ag Ø Ø concentrations Antigen Antibody 26
Immunoprecipitation tests The antigen and antibody are in soluble form Combine to form a visible precipitate Ø Complete precipitation occurs at certain Ab/Ag Ø Ø concentrations Antigen Antibody 26
 Precipitation Curve Ø Prozone – antibody excess, many antibodies coat all antigen sites- results in false negative Ø Postzone – antigen excess, antibody coats antigen but cannot get lattice formation, results in false negative Ø Zone of Equivalence – antigen and antibody present in optimal proportions to bind and give visible reaction 27
Precipitation Curve Ø Prozone – antibody excess, many antibodies coat all antigen sites- results in false negative Ø Postzone – antigen excess, antibody coats antigen but cannot get lattice formation, results in false negative Ø Zone of Equivalence – antigen and antibody present in optimal proportions to bind and give visible reaction 27
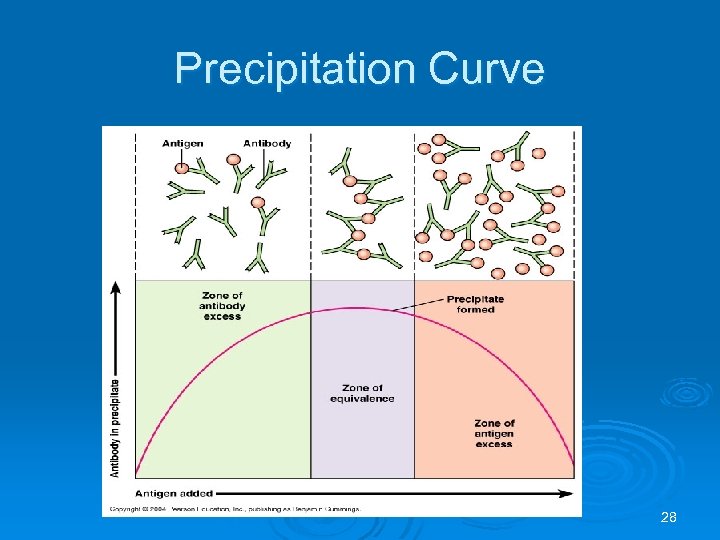 Precipitation Curve 28
Precipitation Curve 28
 Precipitation Reactions Ab + soluble Ag Ø Lattice Precipitation techniques l l Tube precipitation test Gel diffusion • Single radial • Double l l Immunoelectrophoresis Countercurrent electrophoresis (CEP), 29
Precipitation Reactions Ab + soluble Ag Ø Lattice Precipitation techniques l l Tube precipitation test Gel diffusion • Single radial • Double l l Immunoelectrophoresis Countercurrent electrophoresis (CEP), 29
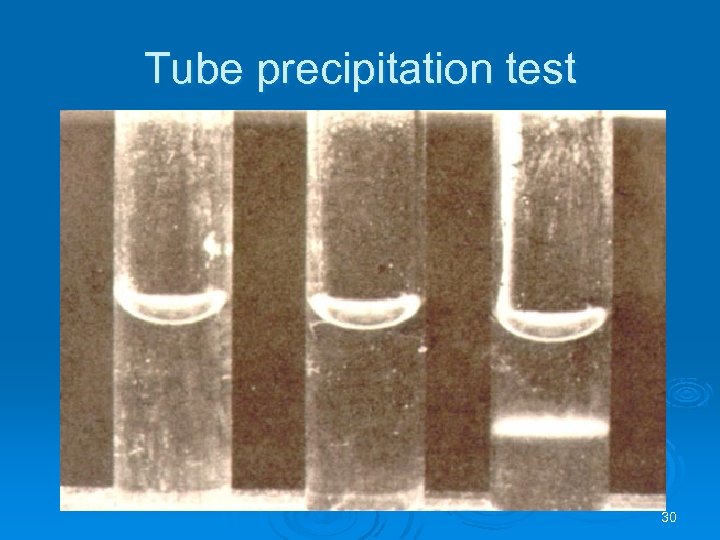 Tube precipitation test 30
Tube precipitation test 30
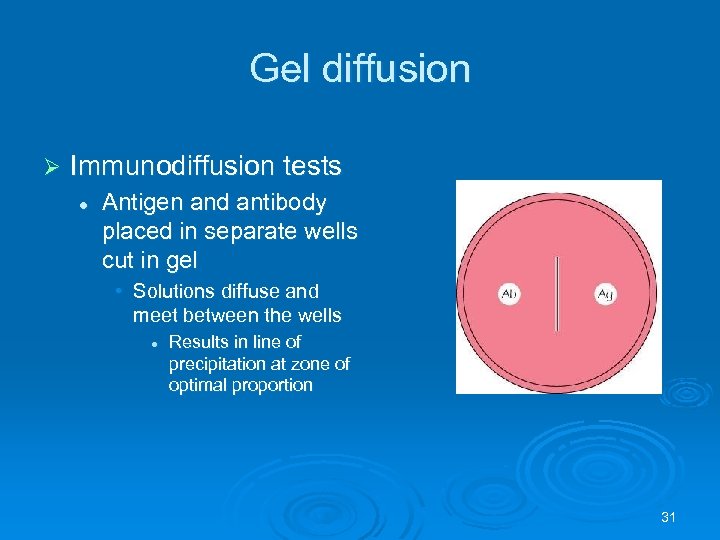 Gel diffusion Ø Immunodiffusion tests l Antigen and antibody placed in separate wells cut in gel • Solutions diffuse and meet between the wells l Results in line of precipitation at zone of optimal proportion 31
Gel diffusion Ø Immunodiffusion tests l Antigen and antibody placed in separate wells cut in gel • Solutions diffuse and meet between the wells l Results in line of precipitation at zone of optimal proportion 31
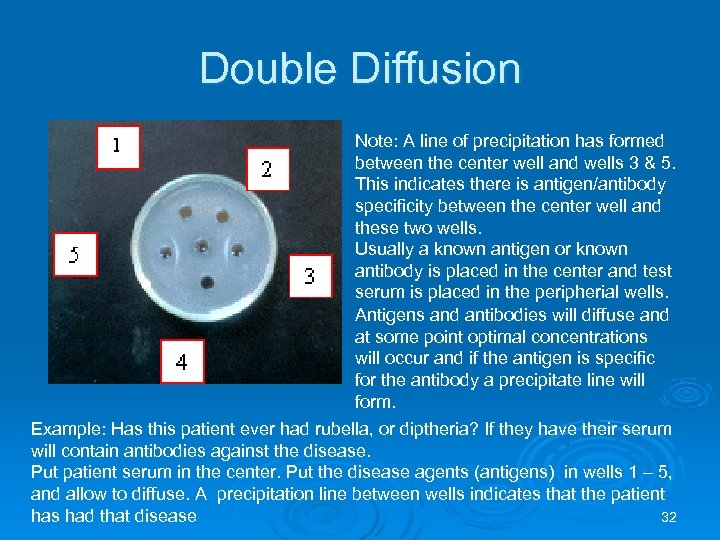 Double Diffusion Note: A line of precipitation has formed between the center well and wells 3 & 5. This indicates there is antigen/antibody specificity between the center well and these two wells. Usually a known antigen or known antibody is placed in the center and test serum is placed in the peripherial wells. Antigens and antibodies will diffuse and at some point optimal concentrations will occur and if the antigen is specific for the antibody a precipitate line will form. Example: Has this patient ever had rubella, or diptheria? If they have their serum will contain antibodies against the disease. Put patient serum in the center. Put the disease agents (antigens) in wells 1 – 5, and allow to diffuse. A precipitation line between wells indicates that the patient has had that disease 32
Double Diffusion Note: A line of precipitation has formed between the center well and wells 3 & 5. This indicates there is antigen/antibody specificity between the center well and these two wells. Usually a known antigen or known antibody is placed in the center and test serum is placed in the peripherial wells. Antigens and antibodies will diffuse and at some point optimal concentrations will occur and if the antigen is specific for the antibody a precipitate line will form. Example: Has this patient ever had rubella, or diptheria? If they have their serum will contain antibodies against the disease. Put patient serum in the center. Put the disease agents (antigens) in wells 1 – 5, and allow to diffuse. A precipitation line between wells indicates that the patient has had that disease 32
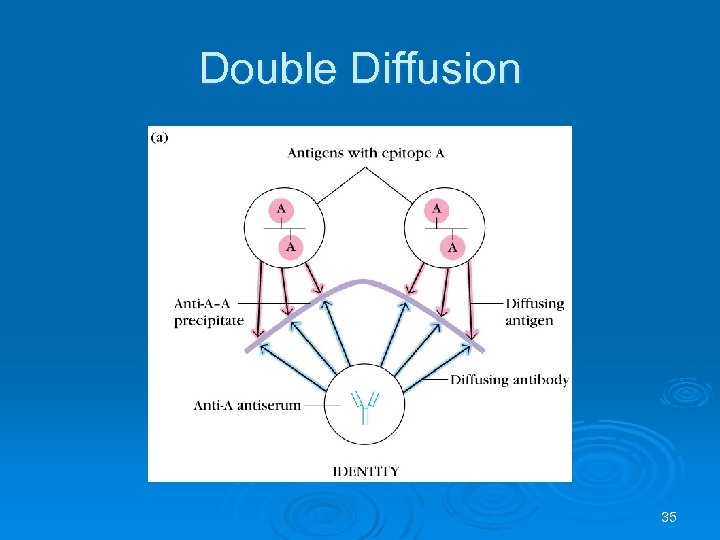
 Double Diffusion Ab and Ag diffuse towards each other Used for determining antigenic relationship between substances Identity Non-identity Partial identity 33
Double Diffusion Ab and Ag diffuse towards each other Used for determining antigenic relationship between substances Identity Non-identity Partial identity 33
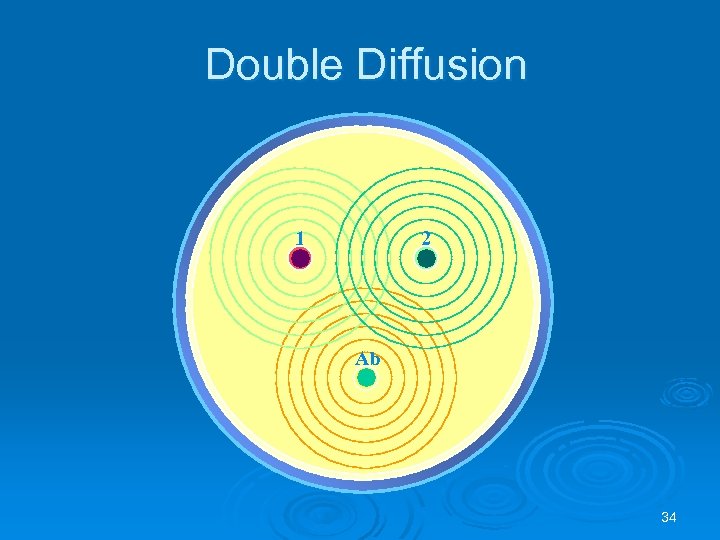 Double Diffusion 1 2 Ab 34
Double Diffusion 1 2 Ab 34
 Double Diffusion 35
Double Diffusion 35
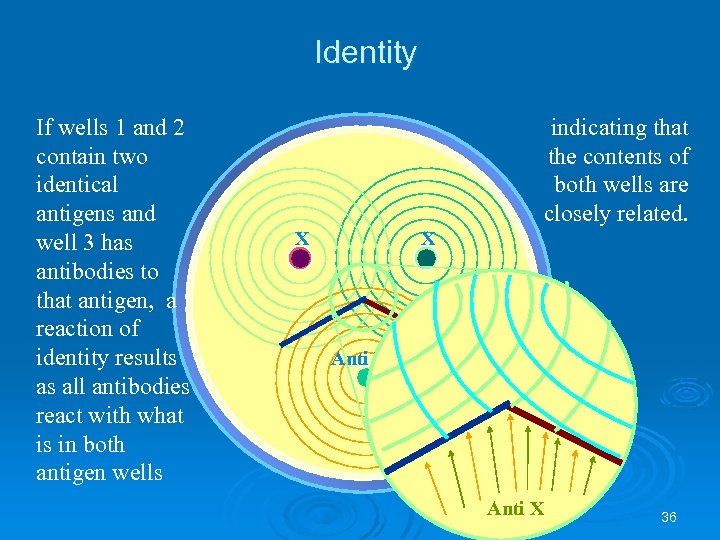 Identity If wells 1 and 2 contain two identical antigens and well 3 has antibodies to that antigen, a reaction of identity results as all antibodies react with what is in both antigen wells indicating that the contents of both wells are closely related. X X Anti X Y Anti X 36
Identity If wells 1 and 2 contain two identical antigens and well 3 has antibodies to that antigen, a reaction of identity results as all antibodies react with what is in both antigen wells indicating that the contents of both wells are closely related. X X Anti X Y Anti X 36
 Identity: What You Really See 37
Identity: What You Really See 37
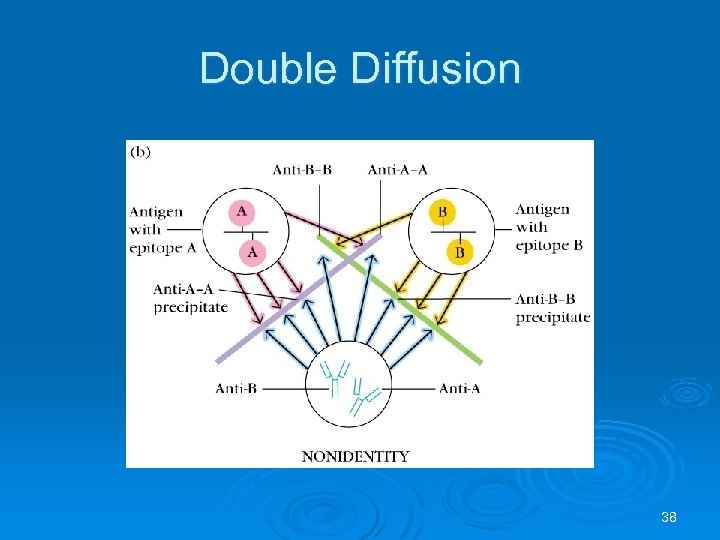 Double Diffusion 38
Double Diffusion 38
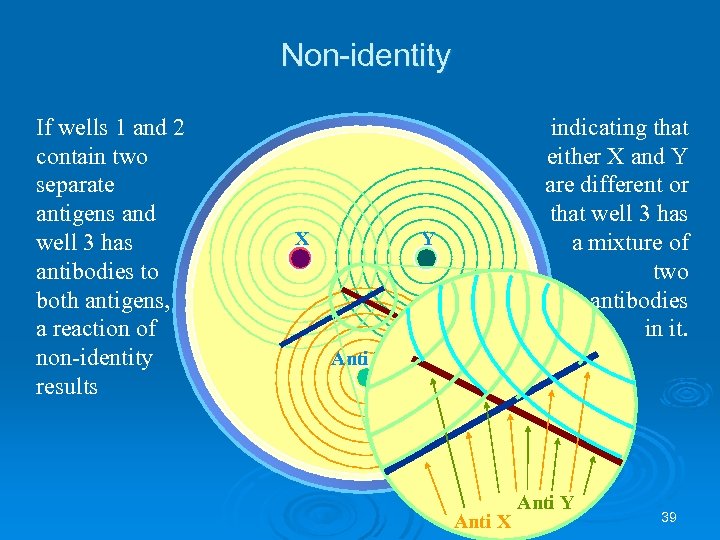 Non-identity If wells 1 and 2 contain two separate antigens and well 3 has antibodies to both antigens, a reaction of non-identity results X indicating that either X and Y are different or that well 3 has a mixture of two antibodies in it. Y Anti X Anti Y 39
Non-identity If wells 1 and 2 contain two separate antigens and well 3 has antibodies to both antigens, a reaction of non-identity results X indicating that either X and Y are different or that well 3 has a mixture of two antibodies in it. Y Anti X Anti Y 39
 Non-identity: What You Really See 40
Non-identity: What You Really See 40
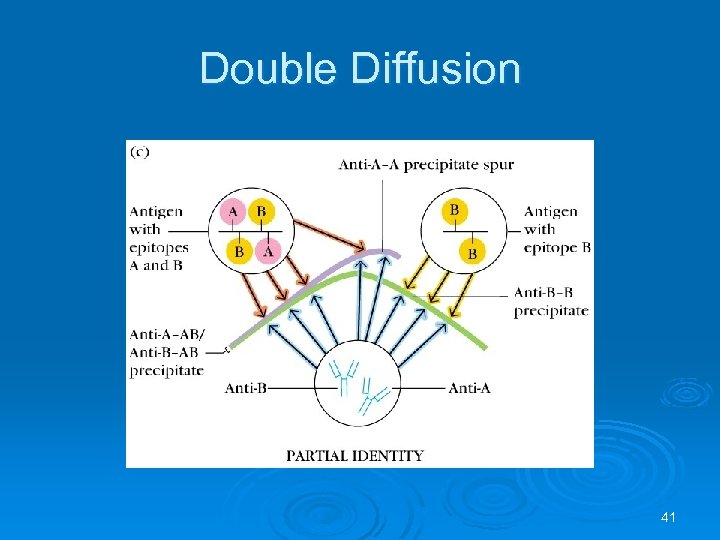 Double Diffusion 41
Double Diffusion 41
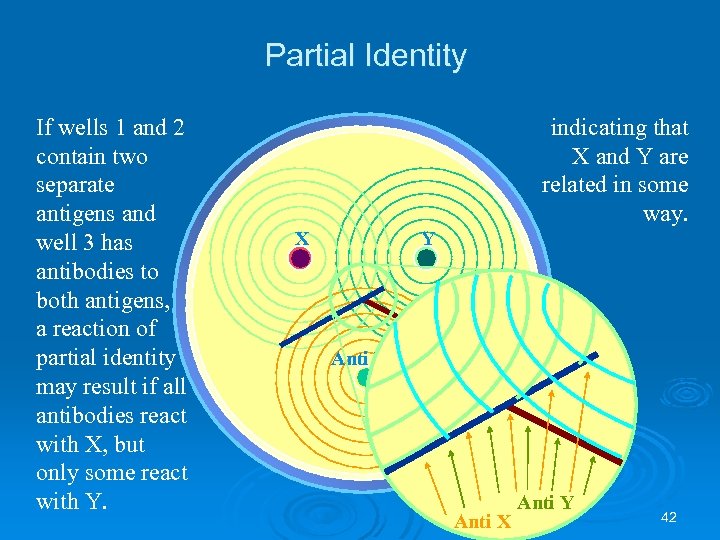 Partial Identity If wells 1 and 2 contain two separate antigens and well 3 has antibodies to both antigens, a reaction of partial identity may result if all antibodies react with X, but only some react with Y. indicating that X and Y are related in some way. X Y Anti X Anti Y 42
Partial Identity If wells 1 and 2 contain two separate antigens and well 3 has antibodies to both antigens, a reaction of partial identity may result if all antibodies react with X, but only some react with Y. indicating that X and Y are related in some way. X Y Anti X Anti Y 42
 Non-identity: What You Really See 43
Non-identity: What You Really See 43
 Radial immunodiffusion test Ø Radial immunodiffusion test is quantitative l l Antibody is added to liquid agar that is allowed to harden Created a uniform antibody concentration Antigen added to wells cut in gel • Diffusion outward forms concentration gradient • Ring forms at antigen-antibody precipitation Standards can be used to construct standard curve to establish concentration 44
Radial immunodiffusion test Ø Radial immunodiffusion test is quantitative l l Antibody is added to liquid agar that is allowed to harden Created a uniform antibody concentration Antigen added to wells cut in gel • Diffusion outward forms concentration gradient • Ring forms at antigen-antibody precipitation Standards can be used to construct standard curve to establish concentration 44
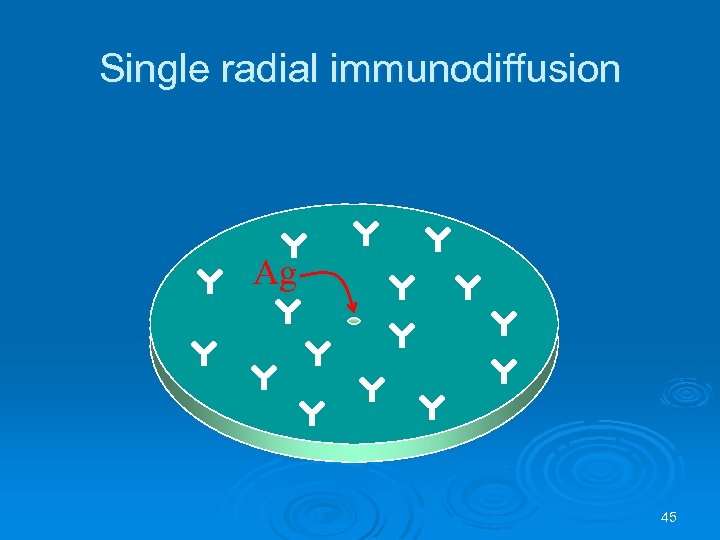 Single radial immunodiffusion Ag 45
Single radial immunodiffusion Ag 45
 Single radial immunodiffusion 46
Single radial immunodiffusion 46
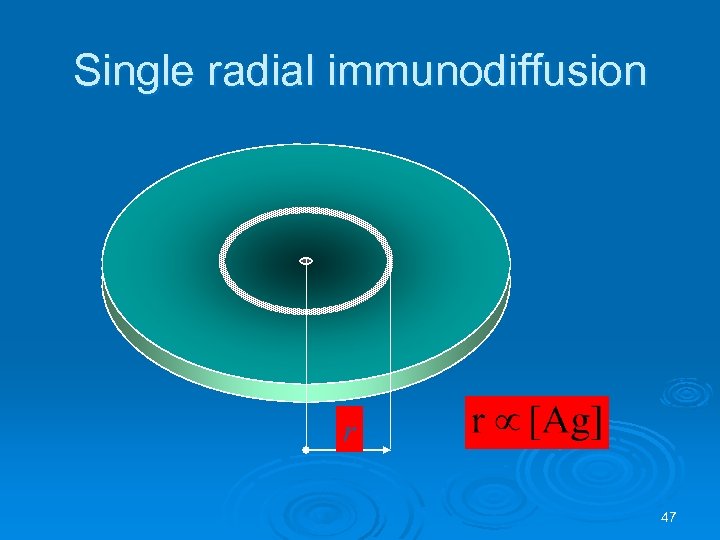 Single radial immunodiffusion r 47
Single radial immunodiffusion r 47
 Single radial immunodiffusion Ø Interpretation Ø Quantitative l Ag Diameter of ring is proportional to the concentration Ig levels Ag Ag Ag Diameter 2 l Ab in gel Ag Concentration 48
Single radial immunodiffusion Ø Interpretation Ø Quantitative l Ag Diameter of ring is proportional to the concentration Ig levels Ag Ag Ag Diameter 2 l Ab in gel Ag Concentration 48
 Single radial immunodiffusion 49
Single radial immunodiffusion 49
 Single radial immunodiffusion Exercise 1 50
Single radial immunodiffusion Exercise 1 50
 Materials Ø Standard Ø Single radial immunodifusion plates Ø Moisture chamber Ø Micropipettes 51
Materials Ø Standard Ø Single radial immunodifusion plates Ø Moisture chamber Ø Micropipettes 51
 Specimen Ø Collect whole blood without anticoagulant and allow to clot at room temperature. Separate serum by centrifugation within 23 hours after collection. 52
Specimen Ø Collect whole blood without anticoagulant and allow to clot at room temperature. Separate serum by centrifugation within 23 hours after collection. 52
 Method Remove plates from refrigerator to room temperature approximately 30 minutes before filling wells. Do not open bag until ready for use. Ø If excess moisture is present, remove plate from its bag and remove cover until evaporation has dried the surface and wells. Replace cover until used. Ø For best results, three wells should be filled with reference sera for each plate. Location of each should be noted. Ø 53
Method Remove plates from refrigerator to room temperature approximately 30 minutes before filling wells. Do not open bag until ready for use. Ø If excess moisture is present, remove plate from its bag and remove cover until evaporation has dried the surface and wells. Replace cover until used. Ø For best results, three wells should be filled with reference sera for each plate. Location of each should be noted. Ø 53
 Preparing the standards (serial dilution) Ø Label four microtest tubes: 1: 2, 1: 4, 1: 8, and 1: 16. Ø Using a micropipette, add 50 microliters of Buffer to each tube. Ø With a fresh pipet tip, add 50 microliters of “Standard” to the tube labeled 1: 2, Mix. Transfer 50 microliters of the 1: 2 dilution to the tube labeled 1: 4, Mix, and so on. 54
Preparing the standards (serial dilution) Ø Label four microtest tubes: 1: 2, 1: 4, 1: 8, and 1: 16. Ø Using a micropipette, add 50 microliters of Buffer to each tube. Ø With a fresh pipet tip, add 50 microliters of “Standard” to the tube labeled 1: 2, Mix. Transfer 50 microliters of the 1: 2 dilution to the tube labeled 1: 4, Mix, and so on. 54
 Performance of Test Deliver specimen or standard (5 µl) to well by placing the pipette tip at the bottom of the well. Allow the well to fill to the top of the agar surface. Avoid bubbles to ensure proper volume and diffusion of sample. Visualization may be aided by placing the plate on dark background. Ø Mark time of completion on plate cover and replace cover. Ø Replace plate in bag and reseal carefully. Ø Incubate plates upright on a flat surface at room temperature (20 to 24 o. C) over 48 hours for End Point readings. Ø 55
Performance of Test Deliver specimen or standard (5 µl) to well by placing the pipette tip at the bottom of the well. Allow the well to fill to the top of the agar surface. Avoid bubbles to ensure proper volume and diffusion of sample. Visualization may be aided by placing the plate on dark background. Ø Mark time of completion on plate cover and replace cover. Ø Replace plate in bag and reseal carefully. Ø Incubate plates upright on a flat surface at room temperature (20 to 24 o. C) over 48 hours for End Point readings. Ø 55
 Calibration Curve Ø Using the reference sera determine their ring diameters to the nearest 0. 1 mm. Ø Using regular graph paper plot the concentration on the X axis and the zone diameters squared on the Y axis for each protein for End point readings. Ø Draw a straight line of "best fit" between the four points. 56
Calibration Curve Ø Using the reference sera determine their ring diameters to the nearest 0. 1 mm. Ø Using regular graph paper plot the concentration on the X axis and the zone diameters squared on the Y axis for each protein for End point readings. Ø Draw a straight line of "best fit" between the four points. 56
 Results Measure diameters of precipitin zones to within 0. 1 mm. Variations in incubation times of more than 30 minutes will produce changes in diameters, especially those at higher levels of antigen except when plates have reached endpoint. Ø Determine the concentration of each unknown or specimen protein from the reference curve find the corresponding concentration. Ø 57
Results Measure diameters of precipitin zones to within 0. 1 mm. Variations in incubation times of more than 30 minutes will produce changes in diameters, especially those at higher levels of antigen except when plates have reached endpoint. Ø Determine the concentration of each unknown or specimen protein from the reference curve find the corresponding concentration. Ø 57
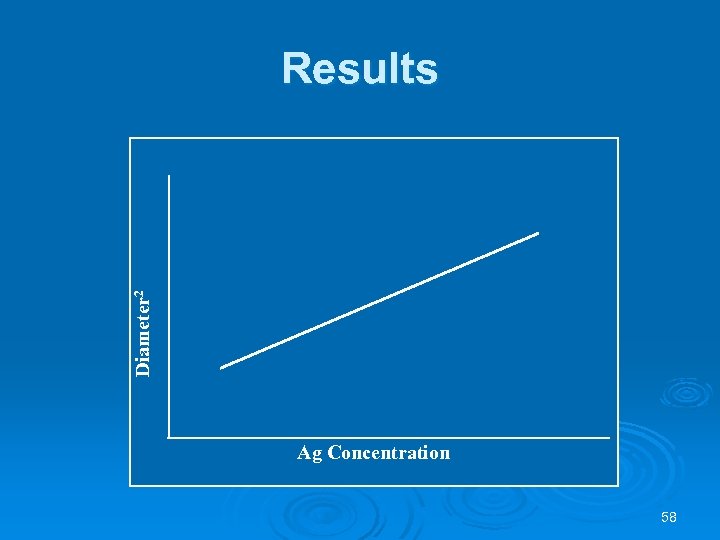 Diameter 2 Results Ag Concentration 58
Diameter 2 Results Ag Concentration 58


