a407a1c00cb1077d800f7f022738bfa3.ppt
- Количество слайдов: 1
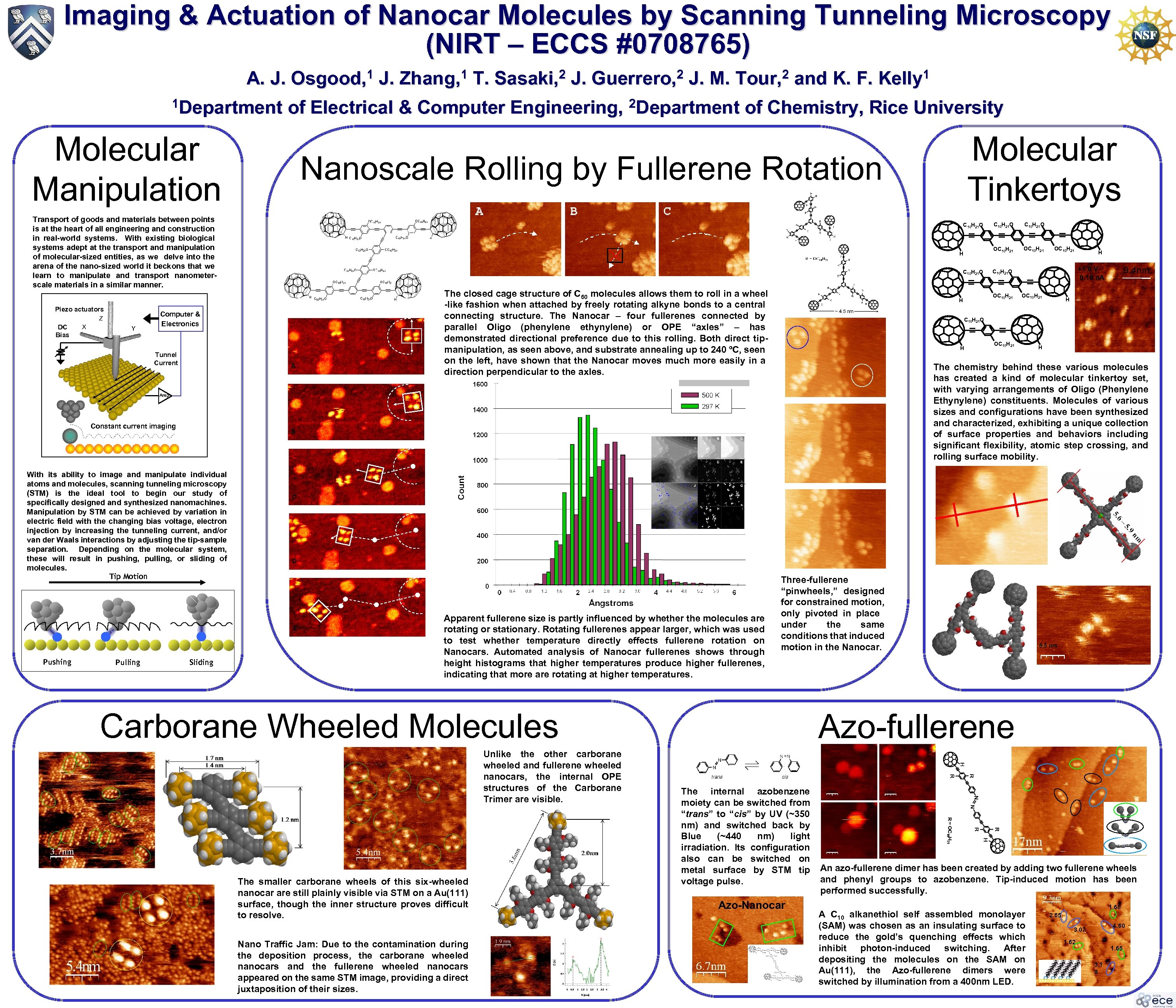
Imaging & Actuation of Nanocar Molecules by Scanning Tunneling Microscopy (NIRT – ECCS #0708765) A. J. 1 Department 1 Osgood, DC Bias X Y J. M. 2 Tour, and K. F. 1 Kelly Molecular Tinkertoys H C 10 H 21 O A The closed cage structure of C 60 molecules allows them to roll in a wheel -like fashion when attached by freely rotating alkyne bonds to a central connecting structure. The Nanocar – four fullerenes connected by parallel Oligo (phenylene ethynylene) or OPE “axles” – has demonstrated directional preference due to this rolling. Both direct tipmanipulation, as seen above, and substrate annealing up to 240 ºC, seen on the left, have shown that the Nanocar moves much more easily in a direction perpendicular to the axles. Pulling Sliding OC 10 H 21 H 9. 4 nm +1. 0 V 0. 10 n. A C 10 H 21 O OC 10 H 21 H H H The chemistry behind these various molecules has created a kind of molecular tinkertoy set, with varying arrangements of Oligo (Phenylene Ethynylene) constituents. Molecules of various sizes and configurations have been synthesized and characterized, exhibiting a unique collection of surface properties and behaviors including significant flexibility, atomic step crossing, and rolling surface mobility. C D E Pushing OC 10 H 21 O B With its ability to image and manipulate individual atoms and molecules, scanning tunneling microscopy (STM) is the ideal tool to begin our study of specifically designed and synthesized nanomachines. Manipulation by STM can be achieved by variation in electric field with the changing bias voltage, electron injection by increasing the tunneling current, and/or van der Waals interactions by adjusting the tip-sample separation. Depending on the molecular system, these will result in pushing, pulling, or sliding of molecules. Tip Motion C 10 H 21 O OC 10 H 21 H Amp Constant current imaging C 10 H 21 O OC 10 H 21 O Computer & Electronics Tunnel Current J. 2 Guerrero, Nanoscale Rolling by Fullerene Rotation Transport of goods and materials between points is at the heart of all engineering and construction in real-world systems. With existing biological systems adept at the transport and manipulation of molecular-sized entities, as we delve into the arena of the nano-sized world it beckons that we learn to manipulate and transport nanometerscale materials in a similar manner. Z T. 2 Sasaki, of Electrical & Computer Engineering, 2 Department of Chemistry, Rice University Molecular Manipulation Piezo actuators J. 1 Zhang, Apparent fullerene size is partly influenced by whether the molecules are rotating or stationary. Rotating fullerenes appear larger, which was used to test whether temperature directly effects fullerene rotation on Nanocars. Automated analysis of Nanocar fullerenes shows through height histograms that higher temperatures produce higher fullerenes, indicating that more are rotating at higher temperatures. Three-fullerene “pinwheels, ” designed for constrained motion, only pivoted in place under the same conditions that induced motion in the Nanocar. Carborane Wheeled Molecules Unlike the other carborane wheeled and fullerene wheeled nanocars, the internal OPE structures of the Carborane Trimer are visible. The smaller carborane wheels of this six-wheeled nanocar are still plainly visible via STM on a Au(111) surface, though the inner structure proves difficult to resolve. Nano Traffic Jam: Due to the contamination during the deposition process, the carborane wheeled nanocars and the fullerene wheeled nanocars appeared on the same STM image, providing a direct juxtaposition of their sizes. 6. 5 nm Azo-fullerene R R 1 2 The internal azobenzene moiety can be switched from “trans” to “cis” by UV (~350 nm) and switched back by Blue (~440 nm) light irradiation. Its configuration also can be switched on metal surface by STM tip voltage pulse. Azo-Nanocar 2. 6 nm 3. 8 nm R R 1 1 2 2 2. 8 nm An azo-fullerene dimer has been created by adding two fullerene wheels and phenyl groups to azobenzene. Tip-induced motion has been performed successfully. A C 10 alkanethiol self assembled monolayer (SAM) was chosen as an insulating surface to reduce the gold’s quenching effects which inhibit photon-induced switching. After depositing the molecules on the SAM on Au(111), the Azo-fullerene dimers were switched by illumination from a 400 nm LED. 1. 60 2. 65 4. 60 3. 07 1. 62 2. 00 1. 65 3. 1 8
a407a1c00cb1077d800f7f022738bfa3.ppt