200cac4710947773ca16bf1c07af760c.ppt
- Количество слайдов: 19
 Ideas ASA The Medical Technology Company 1
Ideas ASA The Medical Technology Company 1
 Ideas Vision and Mission Vision To become the world’s leading supplier of Detection Modules for Functional Imaging. Mission To improve patient care and save lives through delivery of improved and more efficient imaging detection capabilities that can detect cancer and diagnose diseases earlier with use of less radiation. 3
Ideas Vision and Mission Vision To become the world’s leading supplier of Detection Modules for Functional Imaging. Mission To improve patient care and save lives through delivery of improved and more efficient imaging detection capabilities that can detect cancer and diagnose diseases earlier with use of less radiation. 3
 Company Background • Established in 1992 based on research at Cern (Switzerland) • 10 Technology Patents • 27 employees from 7 countries • Hereof 6 with Ph. D • Experienced management team 4
Company Background • Established in 1992 based on research at Cern (Switzerland) • 10 Technology Patents • 27 employees from 7 countries • Hereof 6 with Ph. D • Experienced management team 4
 How is this possible ? • Slim and small detectors allow dedicated cameras (analogy to flat screen PC vs table PC) x * * Anger Camera New technology Nuclear Medicine Computer Tomograph (SPECT - Old technology) • Ideas recognized as the best vendor to this level * • Extremely talented people with world wide reputation • Innovative and unique patented technology * (References GE, Philips, Siemens, as well as Glenn Knoll) 5
How is this possible ? • Slim and small detectors allow dedicated cameras (analogy to flat screen PC vs table PC) x * * Anger Camera New technology Nuclear Medicine Computer Tomograph (SPECT - Old technology) • Ideas recognized as the best vendor to this level * • Extremely talented people with world wide reputation • Innovative and unique patented technology * (References GE, Philips, Siemens, as well as Glenn Knoll) 5
 What do Ideas want to achieve ? • Find an industry partner for the Prostate Probe • Develop an Ideas Detector Module for PET 7
What do Ideas want to achieve ? • Find an industry partner for the Prostate Probe • Develop an Ideas Detector Module for PET 7
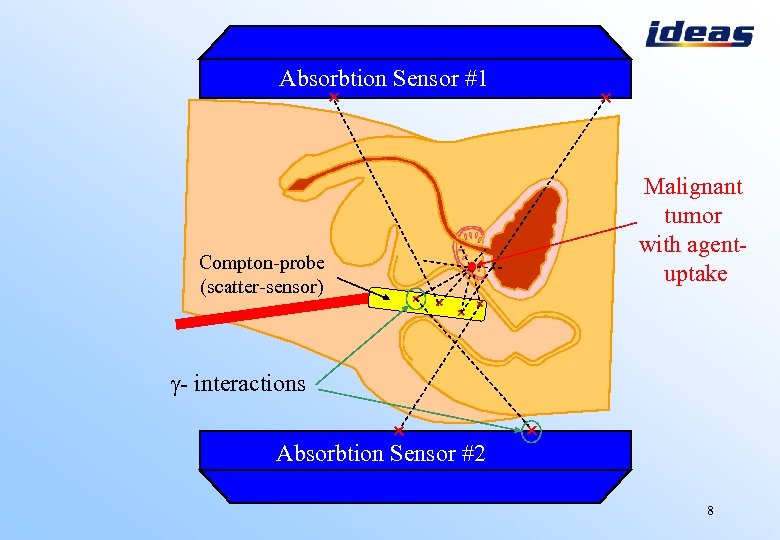 Absorbtion Sensor #1 How it works Compton-probe (scatter-sensor) Malignant tumor with agentuptake - interactions Absorbtion Sensor #2 8
Absorbtion Sensor #1 How it works Compton-probe (scatter-sensor) Malignant tumor with agentuptake - interactions Absorbtion Sensor #2 8
 Ongoing investments SPECT Ideas Detector Module for SPECT: • Stand. Segemented Modules as an alternative to similar CZT Modules and PMT • Known and available scintillator and detector technology • New and unique version of ASIC • Under developed jointly together with customer (50% co-ownership) • Ideas bus. pot. for IDM SPECT = 6 mill. cm 2 * 25$ = 150 mill. $ 9
Ongoing investments SPECT Ideas Detector Module for SPECT: • Stand. Segemented Modules as an alternative to similar CZT Modules and PMT • Known and available scintillator and detector technology • New and unique version of ASIC • Under developed jointly together with customer (50% co-ownership) • Ideas bus. pot. for IDM SPECT = 6 mill. cm 2 * 25$ = 150 mill. $ 9
 Ongoing investments PET Ideas Detector Module for PET • Standard Segemented Modules as an alternative to traditional PMT • Known and available scintillator and detector technology • New and unique version of ASIC and Module handling Depth Of Interaction • Ideas started development based on market requirements and customer feedback • Ideas bus. pot. for IDM PET = 2, 15 mill. cm 2 * 100$ = 215 mill. $ (high growth) 11
Ongoing investments PET Ideas Detector Module for PET • Standard Segemented Modules as an alternative to traditional PMT • Known and available scintillator and detector technology • New and unique version of ASIC and Module handling Depth Of Interaction • Ideas started development based on market requirements and customer feedback • Ideas bus. pot. for IDM PET = 2, 15 mill. cm 2 * 100$ = 215 mill. $ (high growth) 11
 Why an Integrated Project? • EC FP 6 (2003 -2007) introduced two new tools: Networks of Excellence (No. E) and Integrated Projects (IP) • IP’s meant for real delivery of products • IP’s have higher funding than No. E, but strictly related to achievements • New compared to FP 5: large flexibility in project management, now entirely carried on inside the project (to the level of making bids for new partners) 12
Why an Integrated Project? • EC FP 6 (2003 -2007) introduced two new tools: Networks of Excellence (No. E) and Integrated Projects (IP) • IP’s meant for real delivery of products • IP’s have higher funding than No. E, but strictly related to achievements • New compared to FP 5: large flexibility in project management, now entirely carried on inside the project (to the level of making bids for new partners) 12
 Euro. Med. Im and Emi-Care 13
Euro. Med. Im and Emi-Care 13
 Euro. Med. Im Objectives • Proof of Concept, Early Demonstrators and Clinical Prototypes for: – Prostate Cancer – Breast Cancer – Brain Imaging – Whole-Body Multimodal Imaging • Provide adequate set of radiotracers for each device, to be tested with the clinical prototypes • Overall time scale: 5 years 14
Euro. Med. Im Objectives • Proof of Concept, Early Demonstrators and Clinical Prototypes for: – Prostate Cancer – Breast Cancer – Brain Imaging – Whole-Body Multimodal Imaging • Provide adequate set of radiotracers for each device, to be tested with the clinical prototypes • Overall time scale: 5 years 14
 Euro. Med. Im 22 Participants in the proposal • • • Organisation of European Cancer Institutes (OECI) European Organisation for Nuclear Research (CERN) Karolinska Hospital/Institute (Karolinska) Vrije Universiteit Brussel (VUB) Centre Leon Berard (CLB-LCU) Deutsche Krebsforschungszentrum (DKFZ) Heidelberg (DKFZ) La Sapienza Università degli Studi di Roma (Uni Roma) Laboratório de Instrumentação e Física Experimental de Partículas (LIP) Consejo Superior de Investigaciones Científicas (CSIC) Ecole Polytechnique Fédérale de Lausanne (EPFL) Università di Pisa- Dipartimento di Fisica (Uni Pisa) • • • Istituto Nazionale per lo Studio e la Cura dei Tumori, Milano (ITM) Hospital Clínico San Carlos (SCH) Commissariat à l'énergie atomique (CEA) The University of Manchester (Uo. M) General Electric Medical Systems Israel LTD (GEMSI) SIEMENS AG (SIEMENS) Forschungszentrum Juelich Gmb. H (FZJ) VITAMIB Sarl (VITAMIB) Centre Nationale de la Recherche Scientifique (CNRS/IN 2 P 3) Tagusparque, S. P. D. PCT de Lisboa, S. A (Taguspark ) Advanced Accelerator Applications AAA) 15
Euro. Med. Im 22 Participants in the proposal • • • Organisation of European Cancer Institutes (OECI) European Organisation for Nuclear Research (CERN) Karolinska Hospital/Institute (Karolinska) Vrije Universiteit Brussel (VUB) Centre Leon Berard (CLB-LCU) Deutsche Krebsforschungszentrum (DKFZ) Heidelberg (DKFZ) La Sapienza Università degli Studi di Roma (Uni Roma) Laboratório de Instrumentação e Física Experimental de Partículas (LIP) Consejo Superior de Investigaciones Científicas (CSIC) Ecole Polytechnique Fédérale de Lausanne (EPFL) Università di Pisa- Dipartimento di Fisica (Uni Pisa) • • • Istituto Nazionale per lo Studio e la Cura dei Tumori, Milano (ITM) Hospital Clínico San Carlos (SCH) Commissariat à l'énergie atomique (CEA) The University of Manchester (Uo. M) General Electric Medical Systems Israel LTD (GEMSI) SIEMENS AG (SIEMENS) Forschungszentrum Juelich Gmb. H (FZJ) VITAMIB Sarl (VITAMIB) Centre Nationale de la Recherche Scientifique (CNRS/IN 2 P 3) Tagusparque, S. P. D. PCT de Lisboa, S. A (Taguspark ) Advanced Accelerator Applications AAA) 15
 Euro. Med. Im What went wrong • Too ambigiouse, driven by physicists • Too short time to understand what was required • Users (doctors) and industry came to late into the project • Meetings at CERN with 100 people from 7 -8 different nationalities 16
Euro. Med. Im What went wrong • Too ambigiouse, driven by physicists • Too short time to understand what was required • Users (doctors) and industry came to late into the project • Meetings at CERN with 100 people from 7 -8 different nationalities 16
 Euro. Med. Im What did we learn • • EU do have a business focus – think product Look for the Industry-partner – that’s your market The need for SME opens up possibilities Be careful how you commit yourself – Be sure you have control of your part – It must be a core part of your development strategy • You must be strong enough to say no to limit your responsibilities 17
Euro. Med. Im What did we learn • • EU do have a business focus – think product Look for the Industry-partner – that’s your market The need for SME opens up possibilities Be careful how you commit yourself – Be sure you have control of your part – It must be a core part of your development strategy • You must be strong enough to say no to limit your responsibilities 17
 EMI-CARE European Molecular Imaging Consortium for Advanced Research (Integrated Project) Integration of nanotechnologies 18
EMI-CARE European Molecular Imaging Consortium for Advanced Research (Integrated Project) Integration of nanotechnologies 18
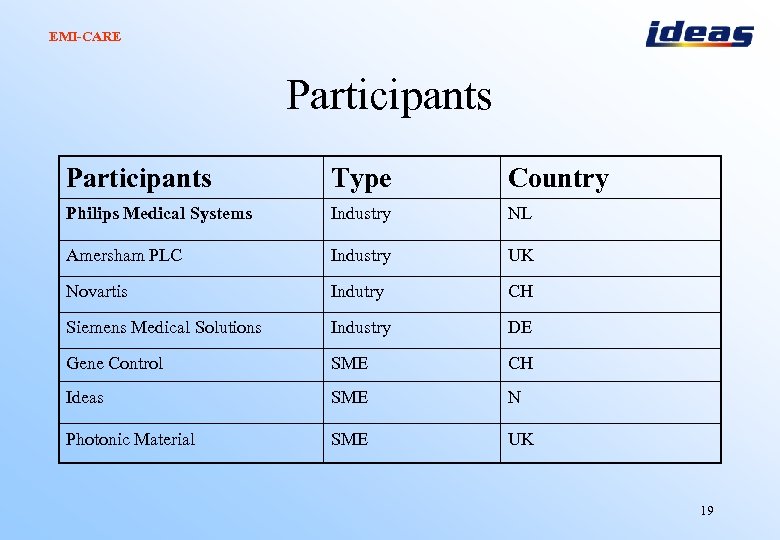 EMI-CARE Participants Type Country Philips Medical Systems Industry NL Amersham PLC Industry UK Novartis Indutry CH Siemens Medical Solutions Industry DE Gene Control SME CH Ideas SME N Photonic Material SME UK 19
EMI-CARE Participants Type Country Philips Medical Systems Industry NL Amersham PLC Industry UK Novartis Indutry CH Siemens Medical Solutions Industry DE Gene Control SME CH Ideas SME N Photonic Material SME UK 19
 EMI-CARE What went wrong • 2 weeks prior to closing: – Novartis and Amersham withdraw because of IP-rights • The rules from EU were ready much too late ! 20
EMI-CARE What went wrong • 2 weeks prior to closing: – Novartis and Amersham withdraw because of IP-rights • The rules from EU were ready much too late ! 20
 EMI-CARE and Euro. Med. Im What has Ideas achieved ? • Worked together with Philips for 3 months: – Explaining and marketing our technology – Made them think our way for future directions – Made their R&D want to continue with Ideas • New EU STREP project defined together with Philips and Photonics (May 2004) • CERN is asked by Brussel to apply for a ”Network Of Excellence”. Ideas is asked to follow 21
EMI-CARE and Euro. Med. Im What has Ideas achieved ? • Worked together with Philips for 3 months: – Explaining and marketing our technology – Made them think our way for future directions – Made their R&D want to continue with Ideas • New EU STREP project defined together with Philips and Photonics (May 2004) • CERN is asked by Brussel to apply for a ”Network Of Excellence”. Ideas is asked to follow 21
 Why is it worth fighting for • Networking with your most important customers and partners • Industry and R&D are forced to incorporate SME – Your chance in a life time !! • From promising technology to products supported by the industry • NFR grant for application support is very helpful • Joint projects with the giants have a company value 22
Why is it worth fighting for • Networking with your most important customers and partners • Industry and R&D are forced to incorporate SME – Your chance in a life time !! • From promising technology to products supported by the industry • NFR grant for application support is very helpful • Joint projects with the giants have a company value 22


