9e7b182742ecda3cdcbe95a1e815743f.ppt
- Количество слайдов: 14

ICC and AIPLA Paris, September 13, 2002 The Future of TRIPS: Impact of the Doha Public Health Declaration Felix Addor Chief Legal Officer and Deputy Director General Swiss Federal Institute of Intellectual Property Ministry of Justice 1
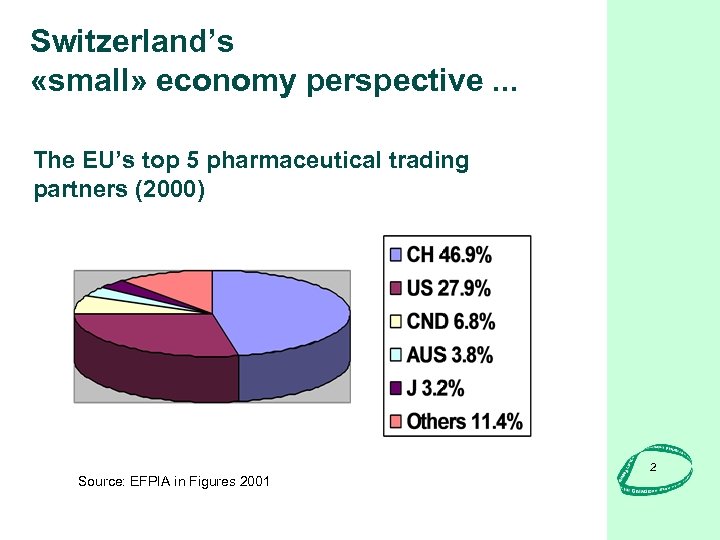
Switzerland’s «small» economy perspective. . . The EU’s top 5 pharmaceutical trading partners (2000) Source: EFPIA in Figures 2001 2

Switzerland’s pharmaceutical industry (2001) Ü Exports: CHF 28 billion, 20% of all Swiss exports Ü Export surplus: CHF 14 billion Ü Worldwide first exporter of pharmaceutical products ! Ü Employees: approx. 28’ 000 / 156’ 000 Ü R&D expenditure: 40% of all Swiss private sector R&D expenditure (CHF 3, 5 billion) Ü Results: 70 new, innovative drugs to market between 1981 and 2001 Patents protect Switzerland’s only «raw material» : Knowledge ! 3
The importance of patent law n Each countries’ patent law is proof of its technological strength 11 1 1 n Patents serve as a key indicator in all rankings on the economic growth potential 4
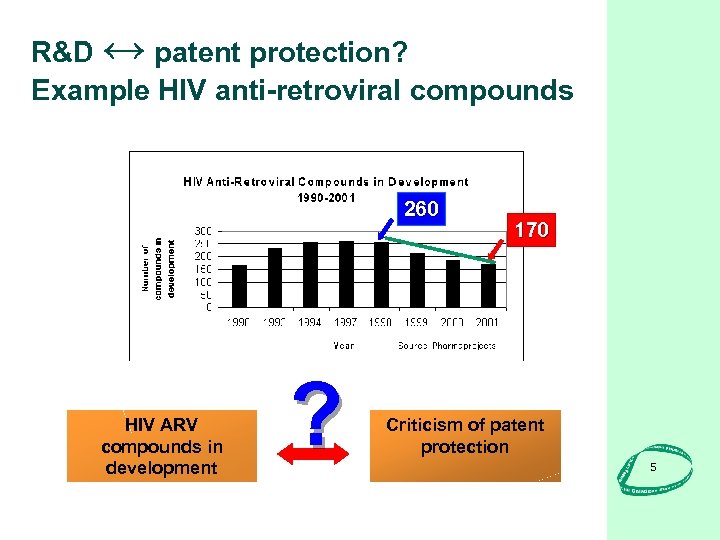
R&D ↔ patent protection? Example HIV anti-retroviral compounds 260 HIV ARV compounds in development ? 170 Criticism of patent protection 5

WTO Declaration on TRIPS and Public Health Ü Commitment to the TRIPS Agreement Ü Clarification of the relationship between the TRIPS Agreement and public health policies of WTO Members Ü Recognition of gravity of public health problems afflicting DCs and LDCs Members have the right to use, to the full, the flexibility of the TRIPS Agreement! Ü «Definition» of some main points of TRIPS flexibility 6

WTO Declaration on TRIPS and Public Health THE TRIPS AGREEMENT RECONCILES INTELLECTUAL PROPERTY WITH PUBLIC HEALTH POLICIES Ü PATENTS ARE NOT AN OBSTACLE TO ACCESS TO AFFORDABLE MEDICINES, BUT ARE PART OF THE SOLUTION Ü CONSENSUS IS POSSIBLE Ü WHICH CONSENSUS FOR THE PARA 6 QUESTION ? 7

One outstanding issue = para. 6 Doha Declaration What are the possibilities open to Members with insufficient or no manufacturing capacities in the pharmaceutical sector ? 8

Scope and coverage of any solution Scope = All diseases causing public health problems especially those resulting from HIV/AIDS, tuberculosis, malaria and other epidemics Product coverage = All patented pharmaceutical products and pharmaceutical products manufactured through a patented process, required when dealing with a public health problem Open questions = Ü Diagnostic kits ? Ü Medical equipment ? 9

Scope and coverage of any solution Beneficiary recipient countries = ÜAll LDCs without any further examination ÜNo OECD country ÜOther DCs on a case by case examination on the basis of clear objective criteria re “sufficient production capacities” Eligible supplying countries = ? 10

Conditions for any solution Whatever the legal mechanism: clarity, adequacy and transparency are needed Transparency Ü Allows for monitoring and reviewing of the efficiency of the system Ü Helps identify and inform potential suppliers Ü increases competition among potential suppliers Involvement of the right holder Ü Delivery by the right holder = most expeditious Ü Requires same rights and obligations as any potential supplier 11

Safeguards against product diversion 1. Production of the quantity needed by beneficiary Member(s) 2. Entirety of production to be exported to the beneficiary Member(s) 3. No (re-)exportation 4. labelling, packaging, coloring and shaping 12

Possible legal mechanisms Ü Interpretation of TRIPS under Art IX WTO ? Ü Waiver under Art IX WTO ? Ü Amendment of TRIPS under Art X WTO ? Ü Art 31 f TRIPS deletion / modification ? Ü Art 30 TRIPS authoritative interpretation ? Ü Dispute Settlement Moratorium ? Ü Solutions outside TRIPS (e. g. public tender) ? „We need to combine incentives for research with access to medication for the poor. “ (Kofi Annan, UN Secretary General, Statement in Amsterdam, 5 April 2001) 13

Work Ahead: Goals and Objectives Any solution should Ü be transparent, legally secure, practical and economically viable Ü serve the interests of those really in need Ü include the patent holder Ü keep / enhance the incentive for research into diseases of the developing world (“Don’t kill IP”!) Ü keep / create incentives for technology transfer and foreign investment 14 Yes, Drugs for the Poor and Patents as well ! (Mike Moor, former Director-General to WTO, International Herald Tribune, 22. 02. 2001)
9e7b182742ecda3cdcbe95a1e815743f.ppt