794cae2d74570a487ea6cb7f46f83c43.ppt
- Количество слайдов: 30
 ©i. Stockphoto. com/Günay Mutlu Caries Formation and the Effects of Various Fluorides in Treatment www. aquafreshscienceacademy. com ©i. Stockphoto. com/Nina Shannon
©i. Stockphoto. com/Günay Mutlu Caries Formation and the Effects of Various Fluorides in Treatment www. aquafreshscienceacademy. com ©i. Stockphoto. com/Nina Shannon
 Learning objectives To summarise the caries formation process and fluoride mode of action To discuss the different types of fluoride available and their relative efficacies
Learning objectives To summarise the caries formation process and fluoride mode of action To discuss the different types of fluoride available and their relative efficacies
 ©i. Stockphoto. com ©Sciencephotolibrary. com Caries development process ©i. Stockphoto. com. Peter Nguyen
©i. Stockphoto. com ©Sciencephotolibrary. com Caries development process ©i. Stockphoto. com. Peter Nguyen
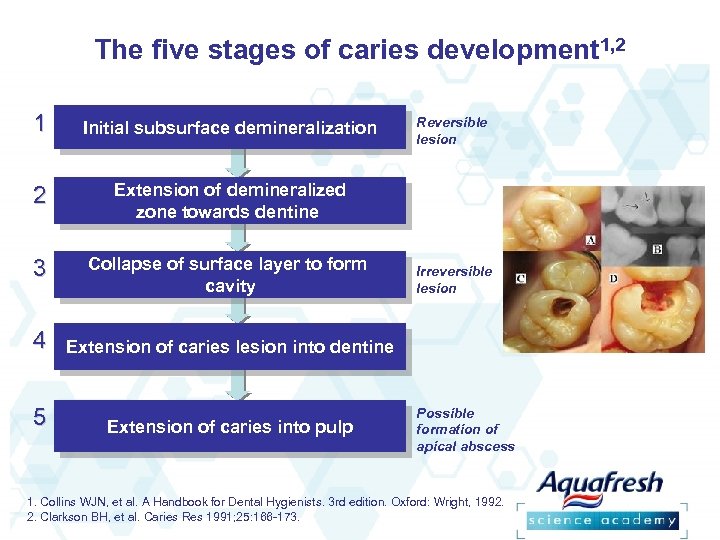 The five stages of caries development 1, 2 1 Initial subsurface demineralization 1. Initial subsurface demineralization 2 3 4 5 Reversible lesion Extension of demineralized zone towards dentine Collapse of surface layer to form Irreversible 3. Collapse of surface layer to form cavity lesion Extension of caries lesion into dentine Extension of caries into pulp Possible formation of apical abscess 1. Collins WJN, et al. A Handbook for Dental Hygienists. 3 rd edition. Oxford: Wright, 1992. 2. Clarkson BH, et al. Caries Res 1991; 25: 166 -173.
The five stages of caries development 1, 2 1 Initial subsurface demineralization 1. Initial subsurface demineralization 2 3 4 5 Reversible lesion Extension of demineralized zone towards dentine Collapse of surface layer to form Irreversible 3. Collapse of surface layer to form cavity lesion Extension of caries lesion into dentine Extension of caries into pulp Possible formation of apical abscess 1. Collins WJN, et al. A Handbook for Dental Hygienists. 3 rd edition. Oxford: Wright, 1992. 2. Clarkson BH, et al. Caries Res 1991; 25: 166 -173.
![The five stages of caries development [Insert animation ‘Caries development’] The five stages of caries development [Insert animation ‘Caries development’]](https://present5.com/presentation/794cae2d74570a487ea6cb7f46f83c43/image-5.jpg) The five stages of caries development [Insert animation ‘Caries development’]
The five stages of caries development [Insert animation ‘Caries development’]
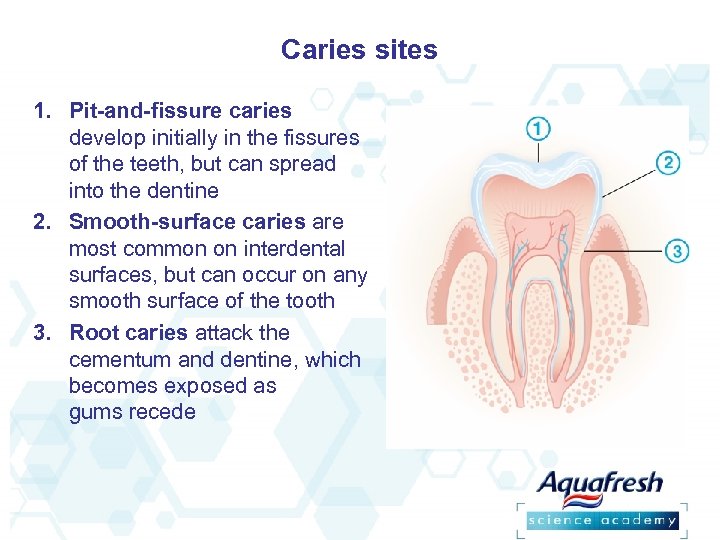 Caries sites 1. Pit-and-fissure caries develop initially in the fissures of the teeth, but can spread into the dentine 2. Smooth-surface caries are most common on interdental surfaces, but can occur on any smooth surface of the tooth 3. Root caries attack the cementum and dentine, which becomes exposed as gums recede
Caries sites 1. Pit-and-fissure caries develop initially in the fissures of the teeth, but can spread into the dentine 2. Smooth-surface caries are most common on interdental surfaces, but can occur on any smooth surface of the tooth 3. Root caries attack the cementum and dentine, which becomes exposed as gums recede
 Caries lesions Figure 1. Sectioned, extracted tooth with three caries lesions Figure 2. A single caries lesion showing bacterial invasion in dentinal tubules
Caries lesions Figure 1. Sectioned, extracted tooth with three caries lesions Figure 2. A single caries lesion showing bacterial invasion in dentinal tubules
 Demineralization and remineralization Tooth enamel is involved in continuous demineralization and remineralization in the oral environment The progression or reversal of caries depends upon the balance of demineralization and remineralization Salivary [Ca 2+] Demineralization Low p. H Salivary [PO 43 -] Salivary [F-] Remineralization
Demineralization and remineralization Tooth enamel is involved in continuous demineralization and remineralization in the oral environment The progression or reversal of caries depends upon the balance of demineralization and remineralization Salivary [Ca 2+] Demineralization Low p. H Salivary [PO 43 -] Salivary [F-] Remineralization
 The natural demineralization and remineralization process Cyclical changes in the oral environment result in alternating periods of demineralization and remineralization at the tooth-plaque interface 1 Adapted from Aoba T. Oral Dis 2004; 10: 249 -257. 1. Gao XJ, et al. J Dent Res 2001; 80: 1834 -1839.
The natural demineralization and remineralization process Cyclical changes in the oral environment result in alternating periods of demineralization and remineralization at the tooth-plaque interface 1 Adapted from Aoba T. Oral Dis 2004; 10: 249 -257. 1. Gao XJ, et al. J Dent Res 2001; 80: 1834 -1839.
 The natural demineralization and remineralization process [Insert animation ‘The action of demineralization and remineralization in the mouth’]
The natural demineralization and remineralization process [Insert animation ‘The action of demineralization and remineralization in the mouth’]
 Fluoride mode of action
Fluoride mode of action
 Sources of fluoride Topical agents ©i. Stockphoto. com Fluoridated water ©i. Stockphoto. com/Adam Gryko Other ingested sources ©i. Stockphoto. com/ Wolfgang Amri ©i. Stockphoto. com
Sources of fluoride Topical agents ©i. Stockphoto. com Fluoridated water ©i. Stockphoto. com/Adam Gryko Other ingested sources ©i. Stockphoto. com/ Wolfgang Amri ©i. Stockphoto. com
 Fluoride effect on remineralization and demineralization of enamel Promote remineralization 1 F- Reduce demineralization 2, 3 1. Silverstone LM. Clinical uses of fluoride 1985; 153 -175. 2. Featherstone JD, et al. J Dent Res 1990; 69: 620 -625. 3. Aoba T. Crit Rev Oral Biol Med 1997; 8: 136 -153. 4. Briner WW & Francis MD. Arch Oral Biol 1962; 7: 541 -550. Inhibit acid generation from plaque bacteria 4
Fluoride effect on remineralization and demineralization of enamel Promote remineralization 1 F- Reduce demineralization 2, 3 1. Silverstone LM. Clinical uses of fluoride 1985; 153 -175. 2. Featherstone JD, et al. J Dent Res 1990; 69: 620 -625. 3. Aoba T. Crit Rev Oral Biol Med 1997; 8: 136 -153. 4. Briner WW & Francis MD. Arch Oral Biol 1962; 7: 541 -550. Inhibit acid generation from plaque bacteria 4
 Fluoride inhibits demineralization: Formation of FAP Fluoride prevents demineralization through formation of fluorohydroxyapatite (FAP) [Insert animation ‘Fluoride prevents demineralization: The formation of FAP’]
Fluoride inhibits demineralization: Formation of FAP Fluoride prevents demineralization through formation of fluorohydroxyapatite (FAP) [Insert animation ‘Fluoride prevents demineralization: The formation of FAP’]
 Fluoride inhibits demineralization: Helps prevent mineral loss Fluoride prevents demineralization through inhibition of mineral loss from enamel [Insert animation ‘Fluoride prevents demineralization min loss’]
Fluoride inhibits demineralization: Helps prevent mineral loss Fluoride prevents demineralization through inhibition of mineral loss from enamel [Insert animation ‘Fluoride prevents demineralization min loss’]
 Fluoride promotes remineralization: Formation of a fluoride reservoir Fluoride promotes remineralization through formation of a fluoride reservoir [Insert animation ‘Fluoride promotes remineralization: Formation of a fluoride reservoir’]
Fluoride promotes remineralization: Formation of a fluoride reservoir Fluoride promotes remineralization through formation of a fluoride reservoir [Insert animation ‘Fluoride promotes remineralization: Formation of a fluoride reservoir’]
 Fluoride promotes remineralization: Creation of supersaturated solutions Fluoride promotes remineralization through creation of supersaturated solutions [Insert animation ‘Fluoride promotes remineralization: Creation of supersaturated solutions’]
Fluoride promotes remineralization: Creation of supersaturated solutions Fluoride promotes remineralization through creation of supersaturated solutions [Insert animation ‘Fluoride promotes remineralization: Creation of supersaturated solutions’]
 Fluoride inhibits plaque bacteria in vitro 1 -4 At low p. H, fluoride combines with hydrogen ions and diffuses into oral bacteria as hydrogen fluoride (HF) Inside the cell HF dissociates, acidifying the cell and releasing fluoride ions Fluoride ions inhibit glycolysis As fluoride is trapped inside the cell this becomes a cumulative process 1. Hamilton IR, et al. Fluoride in dentistry. Copenhagen: Munksgaard; 1996. p 23 -51. 2. Whitford GM, et al. Infect Immun 1977; 18: 680 -687. 3. Van Loveren C. J Dent Res 1990; 69: 676 -681. 4. ten Cate JM. Acta Odontol Scand 1999; 57: 325 -329.
Fluoride inhibits plaque bacteria in vitro 1 -4 At low p. H, fluoride combines with hydrogen ions and diffuses into oral bacteria as hydrogen fluoride (HF) Inside the cell HF dissociates, acidifying the cell and releasing fluoride ions Fluoride ions inhibit glycolysis As fluoride is trapped inside the cell this becomes a cumulative process 1. Hamilton IR, et al. Fluoride in dentistry. Copenhagen: Munksgaard; 1996. p 23 -51. 2. Whitford GM, et al. Infect Immun 1977; 18: 680 -687. 3. Van Loveren C. J Dent Res 1990; 69: 676 -681. 4. ten Cate JM. Acta Odontol Scand 1999; 57: 325 -329.
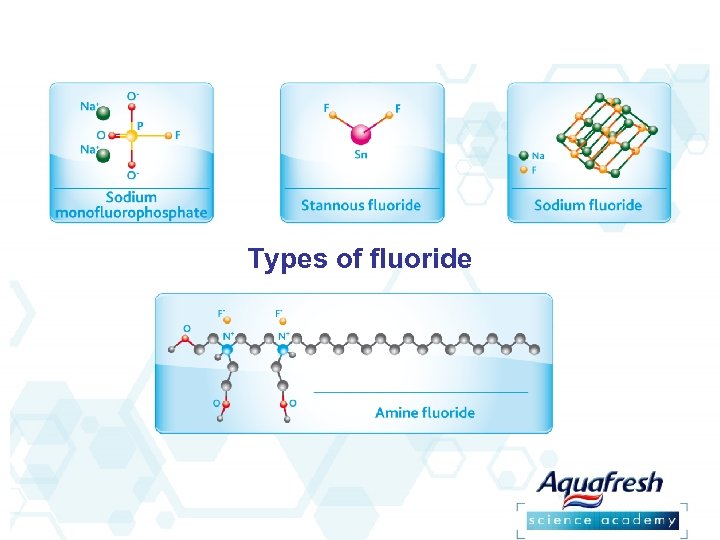 Types of fluoride
Types of fluoride
 Types of fluoride overview The use of fluoride dentifrices has reduced the incidence of caries by 9. 7%– 24. 9%1 Sodium fluoride (Na. F) and sodium monofluorophosphate (MFP) are the most common sources of fluoride in dentifrices – These can be used alone or in combination 1. Twetman S, et al. Acta Odontologica Scandinavica 2003; 61; 6: 347 -355. 2. Volpe AR, et al. Am J Dent. 1993; 6: S 13 -S 42. 3. Sullivan RJ, et al. J Clin Dent. 1995; 6: 135 -138.
Types of fluoride overview The use of fluoride dentifrices has reduced the incidence of caries by 9. 7%– 24. 9%1 Sodium fluoride (Na. F) and sodium monofluorophosphate (MFP) are the most common sources of fluoride in dentifrices – These can be used alone or in combination 1. Twetman S, et al. Acta Odontologica Scandinavica 2003; 61; 6: 347 -355. 2. Volpe AR, et al. Am J Dent. 1993; 6: S 13 -S 42. 3. Sullivan RJ, et al. J Clin Dent. 1995; 6: 135 -138.
 Fluoride formulation factors and mode of action Not all fluoride toothpastes are the same – Different fluoride source, p. H and choice of formulation can affect fluoride uptake 1, 2 Fluoride needs to be deposited and slowly released to be effective following brushing 3 – The amount of fluoride released into saliva and adsorbed by enamel during the period after brushing is critical 1. Friberger P. Scand J Dent Res 1975: 83; 339 -344. 2. White DJ, et al. Caries Res 1986; 20: 332 -336. 3. ten Cate JM. Eur J Oral Sci 1997; 105: 461 -465.
Fluoride formulation factors and mode of action Not all fluoride toothpastes are the same – Different fluoride source, p. H and choice of formulation can affect fluoride uptake 1, 2 Fluoride needs to be deposited and slowly released to be effective following brushing 3 – The amount of fluoride released into saliva and adsorbed by enamel during the period after brushing is critical 1. Friberger P. Scand J Dent Res 1975: 83; 339 -344. 2. White DJ, et al. Caries Res 1986; 20: 332 -336. 3. ten Cate JM. Eur J Oral Sci 1997; 105: 461 -465.
 Factors that influence fluoride delivery Fluoride source (Na. F, MFP, stannous fluoride) – For example, MFP requires activation by hydrolysis by salivary phosphatase to release active F- Fluoride concentration in formulation Formulation properties – p. H will drive different fluoride modes of action – Ingredients such as divalent cations (eg, Ca 2+) can reduce the amount of available fluoride – Ingredients such as high levels of phosphates can reduce fluoride uptake
Factors that influence fluoride delivery Fluoride source (Na. F, MFP, stannous fluoride) – For example, MFP requires activation by hydrolysis by salivary phosphatase to release active F- Fluoride concentration in formulation Formulation properties – p. H will drive different fluoride modes of action – Ingredients such as divalent cations (eg, Ca 2+) can reduce the amount of available fluoride – Ingredients such as high levels of phosphates can reduce fluoride uptake
 Na. F vs MFP: Supporting studies Fluoride ions are freely available in Na. F whereas MFP requires hydrolysis by salivary phosphatase to release free fluoride, the biologically active species 1, 2 In vitro, in situ, animal and clinical studies all support that Na. F has superior anti-caries efficacy to MFP in an equivalent silica base formulation – A calcium carbonate-based MFP formulation contains abrasive particles which are thought to complement or enhance fluoride efficacy 2 – Meta-analysis of 12 clinical studies: 6. 8% clinically and statistically significant greater benefit with Na. F vs MFP 3 – Study in adolescents: 7% greater benefit with Na. F than MFP 4 1. Newby CS, et al. J Clin Dent 2006; 17: 94 -99. 2. Lynch RJ, et al. Int Dent J 2005; 55: 175 -178. 3. Bowen WH. J Royal Soc Med 1995; 88: 505 -507.
Na. F vs MFP: Supporting studies Fluoride ions are freely available in Na. F whereas MFP requires hydrolysis by salivary phosphatase to release free fluoride, the biologically active species 1, 2 In vitro, in situ, animal and clinical studies all support that Na. F has superior anti-caries efficacy to MFP in an equivalent silica base formulation – A calcium carbonate-based MFP formulation contains abrasive particles which are thought to complement or enhance fluoride efficacy 2 – Meta-analysis of 12 clinical studies: 6. 8% clinically and statistically significant greater benefit with Na. F vs MFP 3 – Study in adolescents: 7% greater benefit with Na. F than MFP 4 1. Newby CS, et al. J Clin Dent 2006; 17: 94 -99. 2. Lynch RJ, et al. Int Dent J 2005; 55: 175 -178. 3. Bowen WH. J Royal Soc Med 1995; 88: 505 -507.
 Comparison of fluoride performance in dentifrices
Comparison of fluoride performance in dentifrices
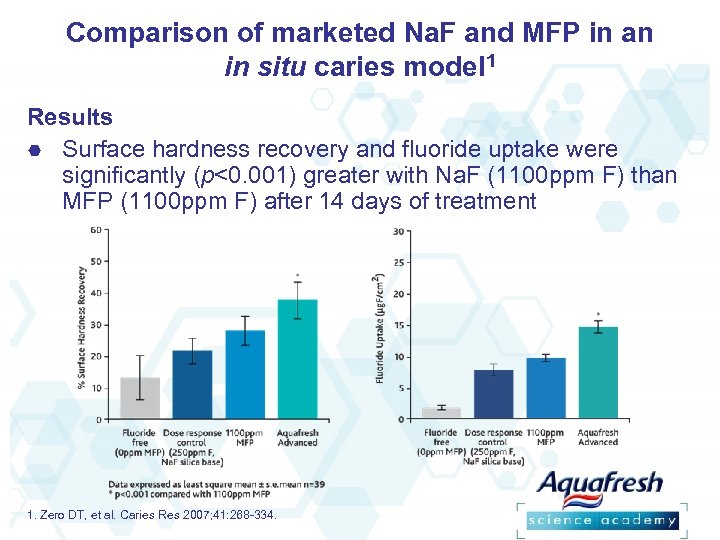 Comparison of marketed Na. F and MFP in an in situ caries model 1 Results Surface hardness recovery and fluoride uptake were significantly (p<0. 001) greater with Na. F (1100 ppm F) than MFP (1100 ppm F) after 14 days of treatment 1. Zero DT, et al. Caries Res 2007; 41: 268 -334.
Comparison of marketed Na. F and MFP in an in situ caries model 1 Results Surface hardness recovery and fluoride uptake were significantly (p<0. 001) greater with Na. F (1100 ppm F) than MFP (1100 ppm F) after 14 days of treatment 1. Zero DT, et al. Caries Res 2007; 41: 268 -334.
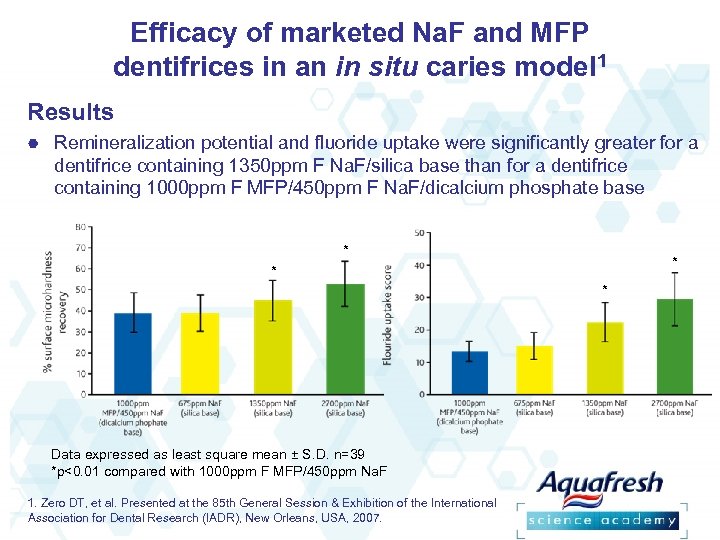 Efficacy of marketed Na. F and MFP dentifrices in an in situ caries model 1 Results Remineralization potential and fluoride uptake were significantly greater for a dentifrice containing 1350 ppm F Na. F/silica base than for a dentifrice containing 1000 ppm F MFP/450 ppm F Na. F/dicalcium phosphate base * * Data expressed as least square mean ± S. D. n=39 *p<0. 01 compared with 1000 ppm F MFP/450 ppm Na. F 1. Zero DT, et al. Presented at the 85 th General Session & Exhibition of the International Association for Dental Research (IADR), New Orleans, USA, 2007.
Efficacy of marketed Na. F and MFP dentifrices in an in situ caries model 1 Results Remineralization potential and fluoride uptake were significantly greater for a dentifrice containing 1350 ppm F Na. F/silica base than for a dentifrice containing 1000 ppm F MFP/450 ppm F Na. F/dicalcium phosphate base * * Data expressed as least square mean ± S. D. n=39 *p<0. 01 compared with 1000 ppm F MFP/450 ppm Na. F 1. Zero DT, et al. Presented at the 85 th General Session & Exhibition of the International Association for Dental Research (IADR), New Orleans, USA, 2007.
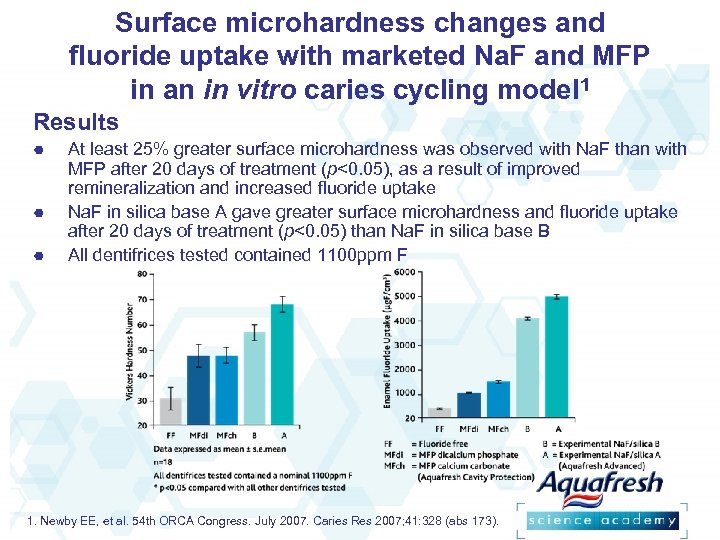 Surface microhardness changes and fluoride uptake with marketed Na. F and MFP in an in vitro caries cycling model 1 Results At least 25% greater surface microhardness was observed with Na. F than with MFP after 20 days of treatment (p<0. 05), as a result of improved remineralization and increased fluoride uptake Na. F in silica base A gave greater surface microhardness and fluoride uptake after 20 days of treatment (p<0. 05) than Na. F in silica base B All dentifrices tested contained 1100 ppm F 1. Newby EE, et al. 54 th ORCA Congress. July 2007. Caries Res 2007; 41: 328 (abs 173).
Surface microhardness changes and fluoride uptake with marketed Na. F and MFP in an in vitro caries cycling model 1 Results At least 25% greater surface microhardness was observed with Na. F than with MFP after 20 days of treatment (p<0. 05), as a result of improved remineralization and increased fluoride uptake Na. F in silica base A gave greater surface microhardness and fluoride uptake after 20 days of treatment (p<0. 05) than Na. F in silica base B All dentifrices tested contained 1100 ppm F 1. Newby EE, et al. 54 th ORCA Congress. July 2007. Caries Res 2007; 41: 328 (abs 173).
 Conclusions from in situ and in vitro studies comparing Na. F with MFP 1– 3 Na. F (1100 ppm F/silica base) showed greater efficacy with regard to surface hardness recovery and fluoride uptake than MFP (1100 ppm F/dicalcium phosphate base) in an in situ caries model 1 A second in situ study similarly demonstrated that Na. F (1350 ppm F) in a silica base provided greater remineralization potential and fluoride uptake than a combination of MFP (1000 ppm F) and Na. F (450 ppm F) in a dicalcium phosphate base 2 This in situ evidence is supported by data from an vitro study. 3 Taken together, these results suggest: – Na. F in silica base provides superior anti-caries potential to MFP formulations – Different formulations of Na. F in silica base have different remineralization potentials and fluoride uptake 1. Zero DT, et al. Caries Res 2007; 41: 268 -334. 2. Zero DT, et al. Presented at the 85 th General Session & Exhibition of the International Association for Dental Research (IADR), New Orleans, USA, 2007. 3. Newby EE, et al. 54 th ORCA Congress. July 2007. Caries Res 2007; 41: 328 (abs 173).
Conclusions from in situ and in vitro studies comparing Na. F with MFP 1– 3 Na. F (1100 ppm F/silica base) showed greater efficacy with regard to surface hardness recovery and fluoride uptake than MFP (1100 ppm F/dicalcium phosphate base) in an in situ caries model 1 A second in situ study similarly demonstrated that Na. F (1350 ppm F) in a silica base provided greater remineralization potential and fluoride uptake than a combination of MFP (1000 ppm F) and Na. F (450 ppm F) in a dicalcium phosphate base 2 This in situ evidence is supported by data from an vitro study. 3 Taken together, these results suggest: – Na. F in silica base provides superior anti-caries potential to MFP formulations – Different formulations of Na. F in silica base have different remineralization potentials and fluoride uptake 1. Zero DT, et al. Caries Res 2007; 41: 268 -334. 2. Zero DT, et al. Presented at the 85 th General Session & Exhibition of the International Association for Dental Research (IADR), New Orleans, USA, 2007. 3. Newby EE, et al. 54 th ORCA Congress. July 2007. Caries Res 2007; 41: 328 (abs 173).
 Summary Dental caries is a progressive disease characterised by demineralization (dissolution) and destruction of enamel and dentine Fluoride can reduce caries by preventing demineralization and promoting remineralization of tooth surfaces and can also inhibit plaque acid production Four fluoride sources are used routinely in dentifrices: sodium fluoride (Na. F); sodium monofluorophosphate (MFP); amine fluoride (Am. F) and stannous fluoride (Sn. F 2) While investigations continue to reveal the relative benefits and mode of action of these different fluoride sources, there is a strong set of data which indicates that Na. F is a superior anti-caries agent to MFP Optimizing the base formulation can increase fluoride bioactivity without altering the fluoride level, with the potential to enhance anticaries efficacy
Summary Dental caries is a progressive disease characterised by demineralization (dissolution) and destruction of enamel and dentine Fluoride can reduce caries by preventing demineralization and promoting remineralization of tooth surfaces and can also inhibit plaque acid production Four fluoride sources are used routinely in dentifrices: sodium fluoride (Na. F); sodium monofluorophosphate (MFP); amine fluoride (Am. F) and stannous fluoride (Sn. F 2) While investigations continue to reveal the relative benefits and mode of action of these different fluoride sources, there is a strong set of data which indicates that Na. F is a superior anti-caries agent to MFP Optimizing the base formulation can increase fluoride bioactivity without altering the fluoride level, with the potential to enhance anticaries efficacy
 For more information visit: www. Aquafresh. Science. Academy. com
For more information visit: www. Aquafresh. Science. Academy. com


