1b5ab962bce15cd8cda17f79cd3add6e.ppt
- Количество слайдов: 142
 Hypertension: New Concepts, Guidelines, and Clinical Management Nathan D. Wong, Ph. D, FACC Associate Professor and Director Heart Disease Prevention Program Division of Cardiology, Department of Medicine College of Medicine, University of California, Irvine
Hypertension: New Concepts, Guidelines, and Clinical Management Nathan D. Wong, Ph. D, FACC Associate Professor and Director Heart Disease Prevention Program Division of Cardiology, Department of Medicine College of Medicine, University of California, Irvine
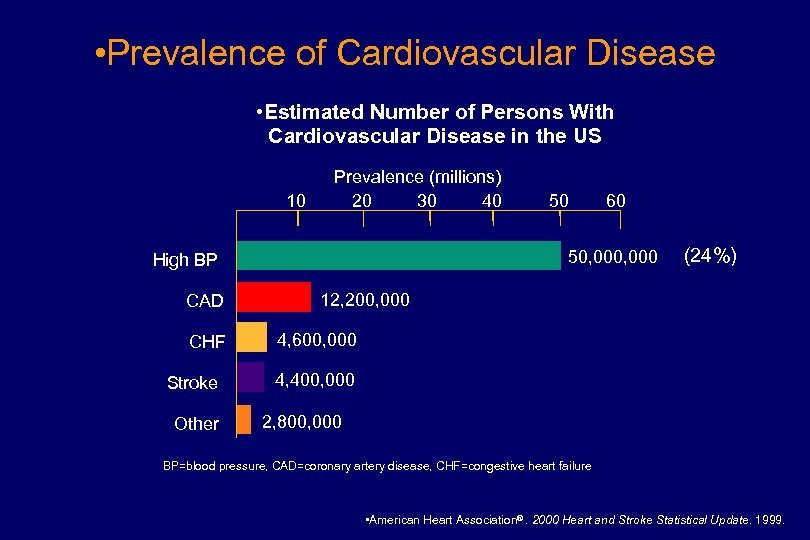 • Prevalence of Cardiovascular Disease • Estimated Number of Persons With Cardiovascular Disease in the US 10 Prevalence (millions) 20 30 40 CHF Stroke Other 60 50, 000 High BP CAD 50 (24%) 12, 200, 000 4, 600, 000 4, 400, 000 2, 800, 000 BP=blood pressure, CAD=coronary artery disease, CHF=congestive heart failure • American Heart Association®. 2000 Heart and Stroke Statistical Update. 1999.
• Prevalence of Cardiovascular Disease • Estimated Number of Persons With Cardiovascular Disease in the US 10 Prevalence (millions) 20 30 40 CHF Stroke Other 60 50, 000 High BP CAD 50 (24%) 12, 200, 000 4, 600, 000 4, 400, 000 2, 800, 000 BP=blood pressure, CAD=coronary artery disease, CHF=congestive heart failure • American Heart Association®. 2000 Heart and Stroke Statistical Update. 1999.
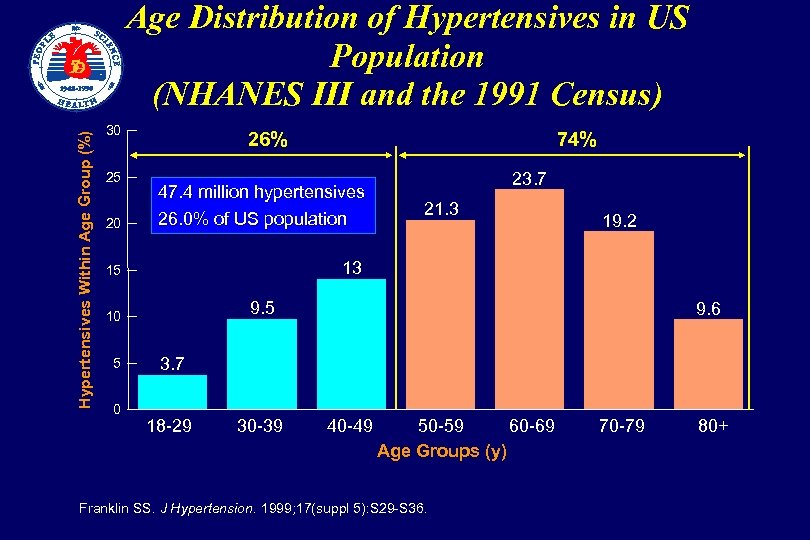 Age Distribution of Hypertensives in US Population (NHANES III and the 1991 Census) Hypertensives Within Age Group (%) 30 25 26% 74% 20 47. 4 million hypertensives 26. 0% of US population 15 23. 7 13 0 19. 2 9. 5 10 5 21. 3 9. 6 3. 7 18 -29 30 -39 40 -49 50 -59 60 -69 Age Groups (y) Franklin SS. J Hypertension. 1999; 17(suppl 5): S 29 -S 36. 70 -79 80+
Age Distribution of Hypertensives in US Population (NHANES III and the 1991 Census) Hypertensives Within Age Group (%) 30 25 26% 74% 20 47. 4 million hypertensives 26. 0% of US population 15 23. 7 13 0 19. 2 9. 5 10 5 21. 3 9. 6 3. 7 18 -29 30 -39 40 -49 50 -59 60 -69 Age Groups (y) Franklin SS. J Hypertension. 1999; 17(suppl 5): S 29 -S 36. 70 -79 80+
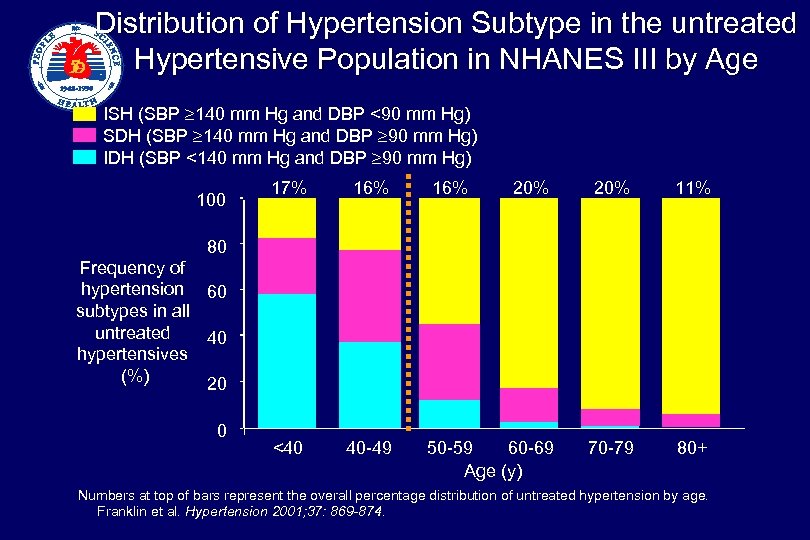 Distribution of Hypertension Subtype in the untreated Hypertensive Population in NHANES III by Age ISH (SBP 140 mm Hg and DBP <90 mm Hg) SDH (SBP 140 mm Hg and DBP 90 mm Hg) IDH (SBP <140 mm Hg and DBP 90 mm Hg) 100 17% 16% <40 40 -49 16% 20% 11% 50 -59 60 -69 Age (y) 70 -79 80+ 80 Frequency of hypertension 60 subtypes in all untreated 40 hypertensives (%) 20 0 Numbers at top of bars represent the overall percentage distribution of untreated hypertension by age. Franklin et al. Hypertension 2001; 37: 869 -874.
Distribution of Hypertension Subtype in the untreated Hypertensive Population in NHANES III by Age ISH (SBP 140 mm Hg and DBP <90 mm Hg) SDH (SBP 140 mm Hg and DBP 90 mm Hg) IDH (SBP <140 mm Hg and DBP 90 mm Hg) 100 17% 16% <40 40 -49 16% 20% 11% 50 -59 60 -69 Age (y) 70 -79 80+ 80 Frequency of hypertension 60 subtypes in all untreated 40 hypertensives (%) 20 0 Numbers at top of bars represent the overall percentage distribution of untreated hypertension by age. Franklin et al. Hypertension 2001; 37: 869 -874.
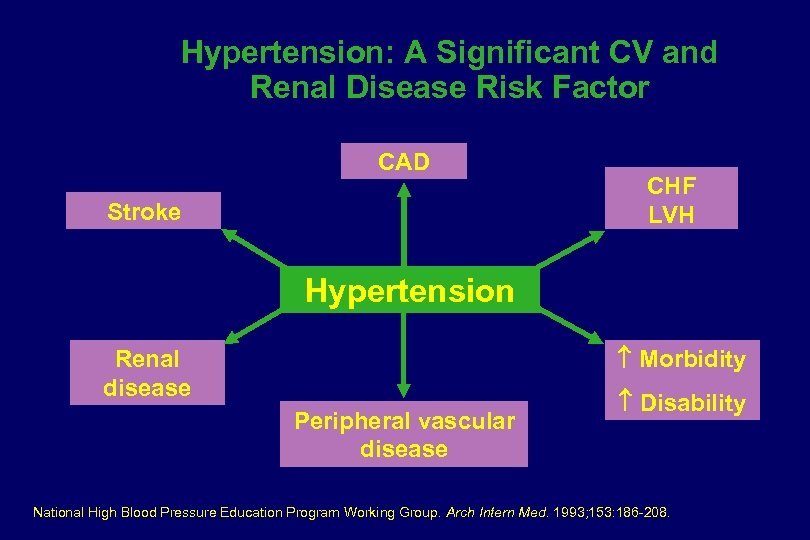 Hypertension: A Significant CV and Renal Disease Risk Factor CAD Stroke CHF LVH Hypertension Morbidity Renal disease Peripheral vascular disease Disability National High Blood Pressure Education Program Working Group. Arch Intern Med. 1993; 153: 186 -208.
Hypertension: A Significant CV and Renal Disease Risk Factor CAD Stroke CHF LVH Hypertension Morbidity Renal disease Peripheral vascular disease Disability National High Blood Pressure Education Program Working Group. Arch Intern Med. 1993; 153: 186 -208.
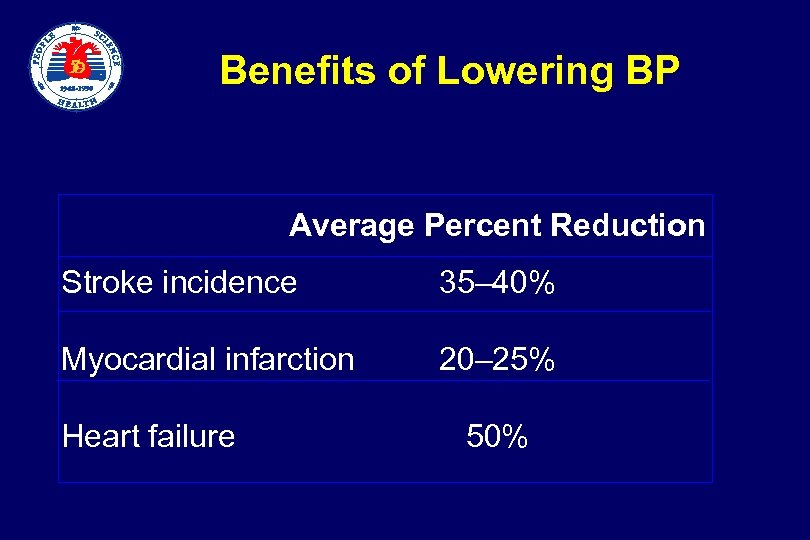 Benefits of Lowering BP Average Percent Reduction Stroke incidence Myocardial infarction Heart failure 35– 40% 20– 25% 50%
Benefits of Lowering BP Average Percent Reduction Stroke incidence Myocardial infarction Heart failure 35– 40% 20– 25% 50%
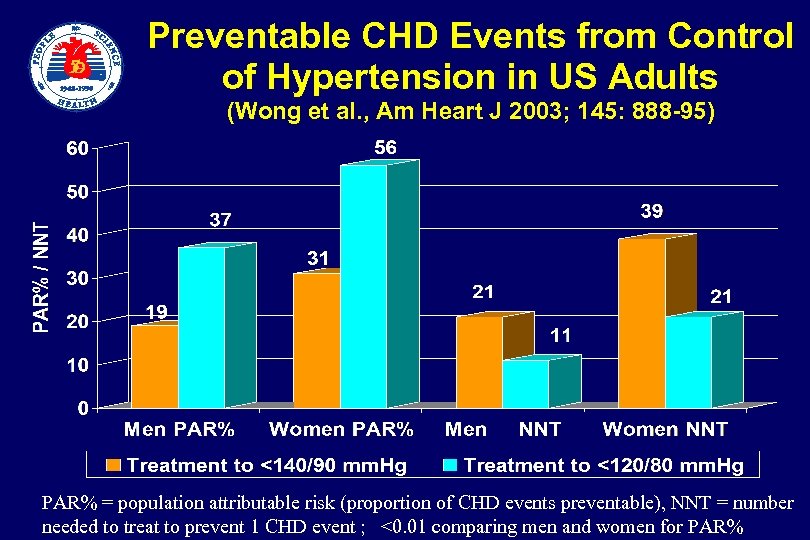 Preventable CHD Events from Control of Hypertension in US Adults (Wong et al. , Am Heart J 2003; 145: 888 -95) PAR% = population attributable risk (proportion of CHD events preventable), NNT = number needed to treat to prevent 1 CHD event ; <0. 01 comparing men and women for PAR%
Preventable CHD Events from Control of Hypertension in US Adults (Wong et al. , Am Heart J 2003; 145: 888 -95) PAR% = population attributable risk (proportion of CHD events preventable), NNT = number needed to treat to prevent 1 CHD event ; <0. 01 comparing men and women for PAR%
 Preventable CHD Events from Control of Hypertension in US Adults (Wong et al. , Am Heart J 2003; 145: 888 -95) (cont. ) • The greatest impact (absolute numbers) from control of hypertension occurs in men, older persons, and those with isolated systolic hypertension • The greatest proportion of preventable CHD events from control of hypertension occurs in women • Optimal control of blood pressure could prevent more than one third of CHD events in men and more than half of CHD events in women
Preventable CHD Events from Control of Hypertension in US Adults (Wong et al. , Am Heart J 2003; 145: 888 -95) (cont. ) • The greatest impact (absolute numbers) from control of hypertension occurs in men, older persons, and those with isolated systolic hypertension • The greatest proportion of preventable CHD events from control of hypertension occurs in women • Optimal control of blood pressure could prevent more than one third of CHD events in men and more than half of CHD events in women
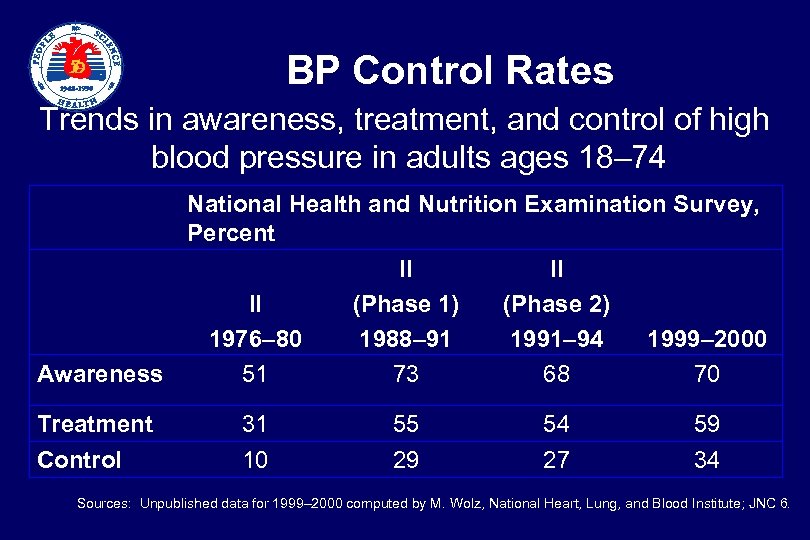 BP Control Rates Trends in awareness, treatment, and control of high blood pressure in adults ages 18– 74 National Health and Nutrition Examination Survey, Percent Awareness II 1976– 80 51 II (Phase 1) 1988– 91 73 II (Phase 2) 1991– 94 68 1999– 2000 70 Treatment Control 31 10 55 29 54 27 59 34 Sources: Unpublished data for 1999– 2000 computed by M. Wolz, National Heart, Lung, and Blood Institute; JNC 6.
BP Control Rates Trends in awareness, treatment, and control of high blood pressure in adults ages 18– 74 National Health and Nutrition Examination Survey, Percent Awareness II 1976– 80 51 II (Phase 1) 1988– 91 73 II (Phase 2) 1991– 94 68 1999– 2000 70 Treatment Control 31 10 55 29 54 27 59 34 Sources: Unpublished data for 1999– 2000 computed by M. Wolz, National Heart, Lung, and Blood Institute; JNC 6.
 U. S. Department of Health and Human Services National Institutes of Health National Heart, Lung, and Blood Institute National High Blood Pressure Education Program The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7)
U. S. Department of Health and Human Services National Institutes of Health National Heart, Lung, and Blood Institute National High Blood Pressure Education Program The Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure (JNC 7)
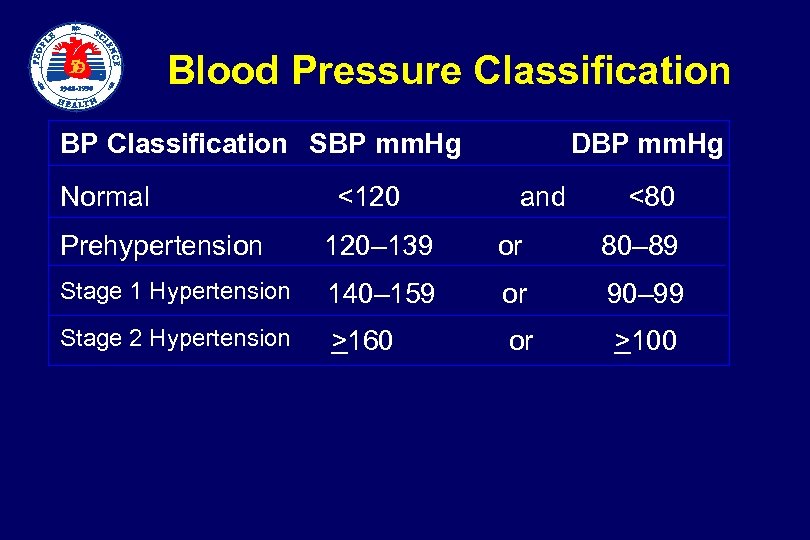 Blood Pressure Classification BP Classification SBP mm. Hg Normal <120 DBP mm. Hg and <80 Prehypertension 120– 139 or 80– 89 Stage 1 Hypertension 140– 159 or 90– 99 Stage 2 Hypertension >160 or >100
Blood Pressure Classification BP Classification SBP mm. Hg Normal <120 DBP mm. Hg and <80 Prehypertension 120– 139 or 80– 89 Stage 1 Hypertension 140– 159 or 90– 99 Stage 2 Hypertension >160 or >100
 New Features and Key Messages § For persons over age 50, SBP is a more important than DBP as CVD risk factor § Starting at 115/75 mm. Hg, CVD risk doubles with each increment of 20/10 mm. Hg throughout the BP range. § Persons who are normotensive at age 55 have a 90% lifetime risk for developing HTN. § Those with SBP 120– 139 mm. Hg or DBP 80– 89 mm. Hg should be considered prehypertensive who require health-promoting lifestyle modifications to prevent CVD.
New Features and Key Messages § For persons over age 50, SBP is a more important than DBP as CVD risk factor § Starting at 115/75 mm. Hg, CVD risk doubles with each increment of 20/10 mm. Hg throughout the BP range. § Persons who are normotensive at age 55 have a 90% lifetime risk for developing HTN. § Those with SBP 120– 139 mm. Hg or DBP 80– 89 mm. Hg should be considered prehypertensive who require health-promoting lifestyle modifications to prevent CVD.
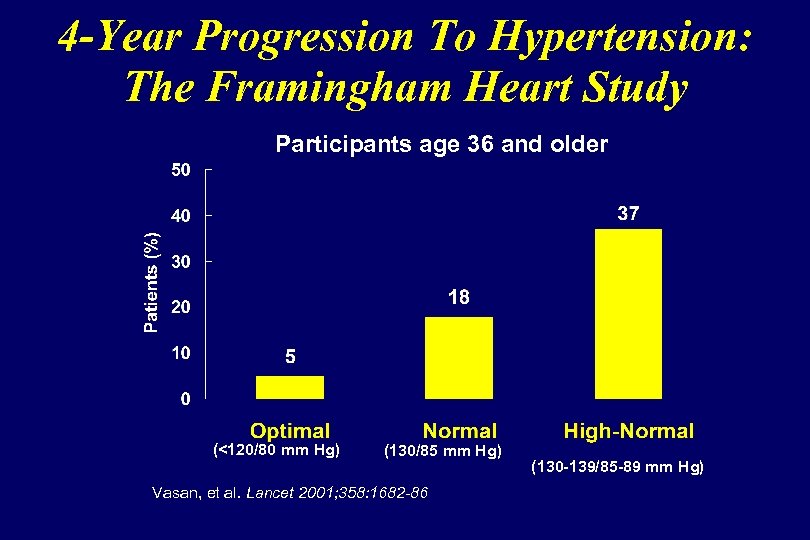 4 -Year Progression To Hypertension: The Framingham Heart Study Participants age 36 and older (<120/80 mm Hg) (130/85 mm Hg) Vasan, et al. Lancet 2001; 358: 1682 -86 (130 -139/85 -89 mm Hg)
4 -Year Progression To Hypertension: The Framingham Heart Study Participants age 36 and older (<120/80 mm Hg) (130/85 mm Hg) Vasan, et al. Lancet 2001; 358: 1682 -86 (130 -139/85 -89 mm Hg)
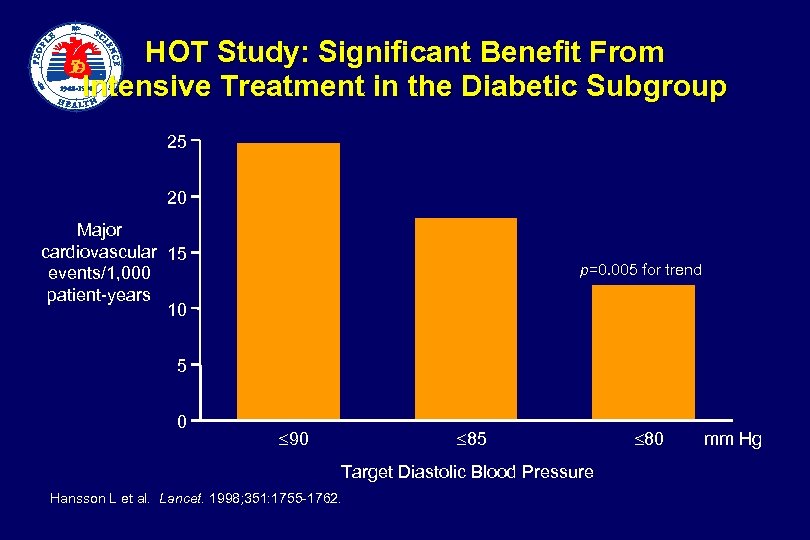 HOT Study: Significant Benefit From Intensive Treatment in the Diabetic Subgroup 25 20 Major cardiovascular 15 events/1, 000 patient-years 10 p=0. 005 for trend 5 0 90 85 Target Diastolic Blood Pressure Hansson L et al. Lancet. 1998; 351: 1755 -1762. 80 mm Hg
HOT Study: Significant Benefit From Intensive Treatment in the Diabetic Subgroup 25 20 Major cardiovascular 15 events/1, 000 patient-years 10 p=0. 005 for trend 5 0 90 85 Target Diastolic Blood Pressure Hansson L et al. Lancet. 1998; 351: 1755 -1762. 80 mm Hg
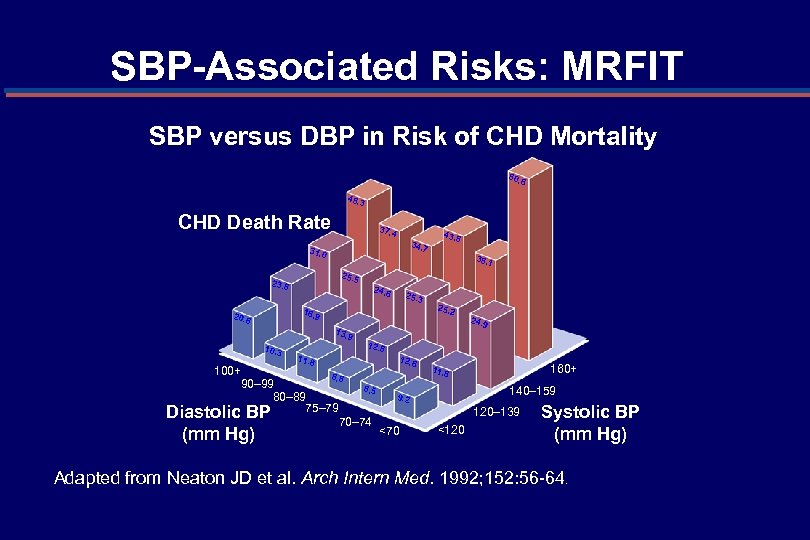 SBP-Associated Risks: MRFIT SBP versus DBP in Risk of CHD Mortality 80. 6 48. 3 CHD Death Rate 37. 4 34. 7 31. 0 25. 5 23. 8 24. 6 16. 9 20. 6 13. 9 10. 3 11. 8 38. 1 25. 2 24. 9 12. 8 100+ 8. 8 90– 99 8. 5 80– 89 75– 79 Diastolic BP 70– 74 (mm Hg) 25. 3 43. 8 12. 6 160+ 11. 8 140– 159 9. 2 120– 139 <70 <120 Systolic BP (mm Hg) Adapted from Neaton JD et al. Arch Intern Med. 1992; 152: 56 -64.
SBP-Associated Risks: MRFIT SBP versus DBP in Risk of CHD Mortality 80. 6 48. 3 CHD Death Rate 37. 4 34. 7 31. 0 25. 5 23. 8 24. 6 16. 9 20. 6 13. 9 10. 3 11. 8 38. 1 25. 2 24. 9 12. 8 100+ 8. 8 90– 99 8. 5 80– 89 75– 79 Diastolic BP 70– 74 (mm Hg) 25. 3 43. 8 12. 6 160+ 11. 8 140– 159 9. 2 120– 139 <70 <120 Systolic BP (mm Hg) Adapted from Neaton JD et al. Arch Intern Med. 1992; 152: 56 -64.
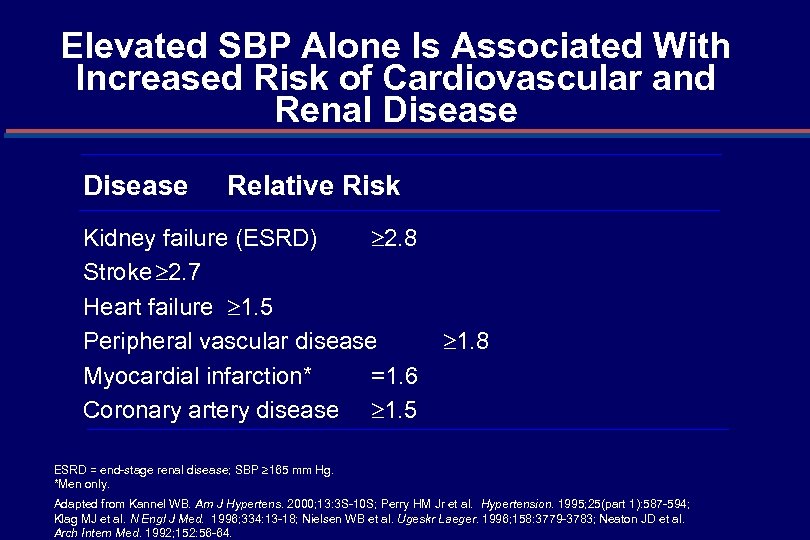 Elevated SBP Alone Is Associated With Increased Risk of Cardiovascular and Renal Disease Relative Risk Kidney failure (ESRD) 2. 8 Stroke 2. 7 Heart failure 1. 5 Peripheral vascular disease Myocardial infarction* =1. 6 Coronary artery disease 1. 5 1. 8 ESRD = end-stage renal disease; SBP 165 mm Hg. *Men only. Adapted from Kannel WB. Am J Hypertens. 2000; 13: 3 S-10 S; Perry HM Jr et al. Hypertension. 1995; 25(part 1): 587 -594; Klag MJ et al. N Engl J Med. 1996; 334: 13 -18; Nielsen WB et al. Ugeskr Laeger. 1996; 158: 3779 -3783; Neaton JD et al. Arch Intern Med. 1992; 152: 56 -64.
Elevated SBP Alone Is Associated With Increased Risk of Cardiovascular and Renal Disease Relative Risk Kidney failure (ESRD) 2. 8 Stroke 2. 7 Heart failure 1. 5 Peripheral vascular disease Myocardial infarction* =1. 6 Coronary artery disease 1. 5 1. 8 ESRD = end-stage renal disease; SBP 165 mm Hg. *Men only. Adapted from Kannel WB. Am J Hypertens. 2000; 13: 3 S-10 S; Perry HM Jr et al. Hypertension. 1995; 25(part 1): 587 -594; Klag MJ et al. N Engl J Med. 1996; 334: 13 -18; Nielsen WB et al. Ugeskr Laeger. 1996; 158: 3779 -3783; Neaton JD et al. Arch Intern Med. 1992; 152: 56 -64.
 Lowering SBP Benefits Older Patients Clinical trials document importance of controlling elevated SBP to prevent cardiovascular disease – SHEP (Systolic Hypertension in the Elderly Program) – Syst-Eur (Systolic Hypertension in Europe) Adapted from SHEP Cooperative Research Group. JAMA. 1991; 265: 3255 -3264; Staessen JA et al. Lancet. 1997; 350: 757 -764.
Lowering SBP Benefits Older Patients Clinical trials document importance of controlling elevated SBP to prevent cardiovascular disease – SHEP (Systolic Hypertension in the Elderly Program) – Syst-Eur (Systolic Hypertension in Europe) Adapted from SHEP Cooperative Research Group. JAMA. 1991; 265: 3255 -3264; Staessen JA et al. Lancet. 1997; 350: 757 -764.
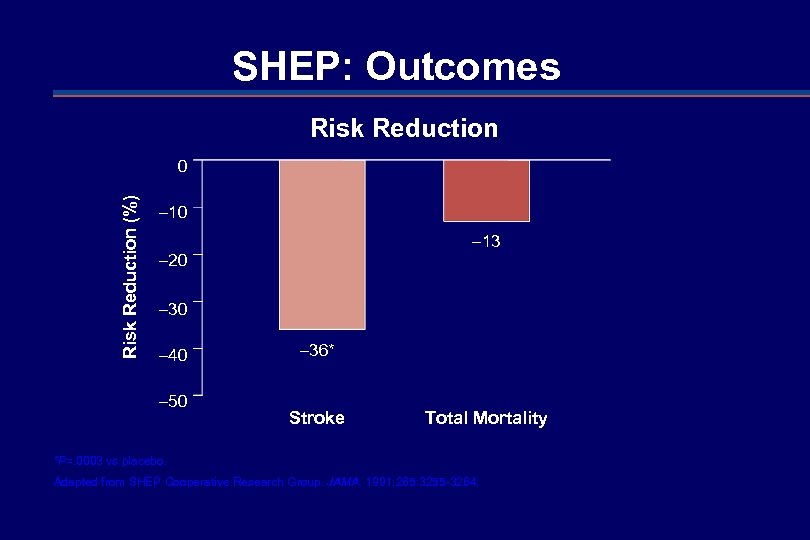 SHEP: Outcomes Risk Reduction (%) 0 – 13 – 20 – 30 – 40 – 50 – 36* Stroke Total Mortality *P=. 0003 vs placebo. Adapted from SHEP Cooperative Research Group. JAMA. 1991; 265: 3255 -3264.
SHEP: Outcomes Risk Reduction (%) 0 – 13 – 20 – 30 – 40 – 50 – 36* Stroke Total Mortality *P=. 0003 vs placebo. Adapted from SHEP Cooperative Research Group. JAMA. 1991; 265: 3255 -3264.
 Systolic Hypertension in Europe (Syst-Eur) Objective: To determine whether antihypertensive treatment reduces cardiovascular complications in older patients with elevated SBP Patients: 4695 patients, 60 years of age, with SBP 160– 219 mm Hg and DBP <95 mm Hg Treatments: Nitrendipine (10– 40 mg/day) with possible addition or substitution of: – Enalapril (5– 20 mg/day) – Hydrochlorothiazide (12. 5– 25 mg/day) Placebo Follow-up: 2 years (median) Endpoint: Total stroke Myocardial infarction Adapted from Staessen JA et al. Lancet. 1997; 350: 757 -764.
Systolic Hypertension in Europe (Syst-Eur) Objective: To determine whether antihypertensive treatment reduces cardiovascular complications in older patients with elevated SBP Patients: 4695 patients, 60 years of age, with SBP 160– 219 mm Hg and DBP <95 mm Hg Treatments: Nitrendipine (10– 40 mg/day) with possible addition or substitution of: – Enalapril (5– 20 mg/day) – Hydrochlorothiazide (12. 5– 25 mg/day) Placebo Follow-up: 2 years (median) Endpoint: Total stroke Myocardial infarction Adapted from Staessen JA et al. Lancet. 1997; 350: 757 -764.
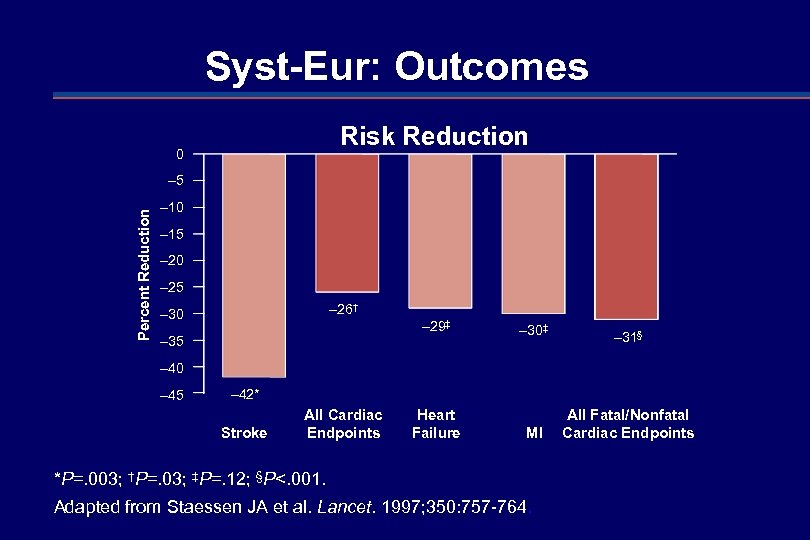 Syst-Eur: Outcomes Risk Reduction 0 Percent Reduction – 5 – 10 – 15 – 20 – 25 – 26† – 30 – 29‡ – 35 – 30‡ – 31§ MI All Fatal/Nonfatal Cardiac Endpoints – 40 – 45 – 42* Stroke All Cardiac Endpoints Heart Failure *P=. 003; †P=. 03; ‡P=. 12; §P<. 001. Adapted from Staessen JA et al. Lancet. 1997; 350: 757 -764.
Syst-Eur: Outcomes Risk Reduction 0 Percent Reduction – 5 – 10 – 15 – 20 – 25 – 26† – 30 – 29‡ – 35 – 30‡ – 31§ MI All Fatal/Nonfatal Cardiac Endpoints – 40 – 45 – 42* Stroke All Cardiac Endpoints Heart Failure *P=. 003; †P=. 03; ‡P=. 12; §P<. 001. Adapted from Staessen JA et al. Lancet. 1997; 350: 757 -764.
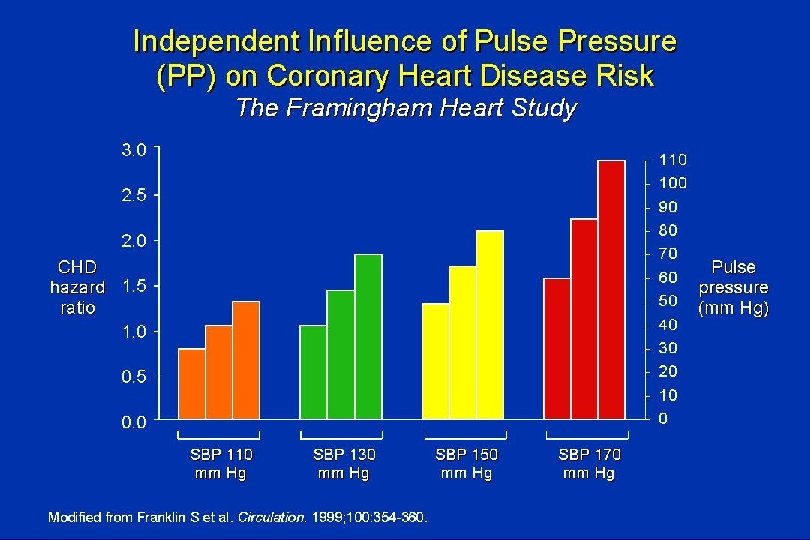
 Pulse Pressure PP = SBP – DBP • Increase in pulse pressure (PP) indicates greater stiffness in large conduit arteries, primarily the thoracic aorta. • PP, therefore, is a surrogate measure of dynamic, cyclic stress during systole. • PP may be a better marker of increased CV risk than either systolic BP or diastolic BP alone in older persons.
Pulse Pressure PP = SBP – DBP • Increase in pulse pressure (PP) indicates greater stiffness in large conduit arteries, primarily the thoracic aorta. • PP, therefore, is a surrogate measure of dynamic, cyclic stress during systole. • PP may be a better marker of increased CV risk than either systolic BP or diastolic BP alone in older persons.
 ATP III: The Metabolic Syndrome* Risk Factor Abdominal obesity† (Waist circumference‡) Men Women TG HDL-C Men Women Blood pressure Fasting glucose Defining Level >102 cm (>40 in) >88 cm (>35 in) 150 mg/d. L <40 mg/d. L <50 mg/d. L 130/ 85 mm Hg 110 mg/d. L *Diagnosis is established when 3 of these risk factors are present. †Abdominal obesity is more highly correlated with metabolic risk factors than is BMI. ‡Some men develop metabolic risk factors when circumference is only marginally increased. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. JAMA. 2001; 285: 2486 -2497. © 2001, Professional Postgraduate Services® www. lipidhealth. org
ATP III: The Metabolic Syndrome* Risk Factor Abdominal obesity† (Waist circumference‡) Men Women TG HDL-C Men Women Blood pressure Fasting glucose Defining Level >102 cm (>40 in) >88 cm (>35 in) 150 mg/d. L <40 mg/d. L <50 mg/d. L 130/ 85 mm Hg 110 mg/d. L *Diagnosis is established when 3 of these risk factors are present. †Abdominal obesity is more highly correlated with metabolic risk factors than is BMI. ‡Some men develop metabolic risk factors when circumference is only marginally increased. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. JAMA. 2001; 285: 2486 -2497. © 2001, Professional Postgraduate Services® www. lipidhealth. org
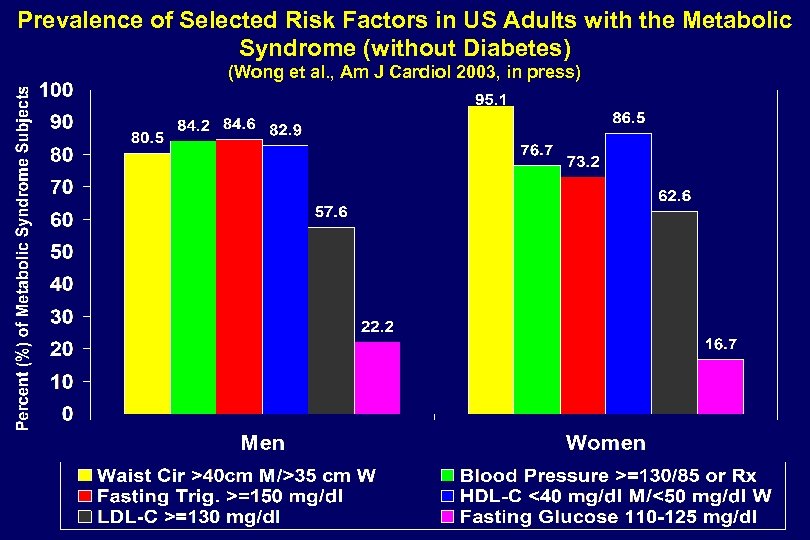 Prevalence of Selected Risk Factors in US Adults with the Metabolic Syndrome (without Diabetes) (Wong et al. , Am J Cardiol 2003, in press)
Prevalence of Selected Risk Factors in US Adults with the Metabolic Syndrome (without Diabetes) (Wong et al. , Am J Cardiol 2003, in press)
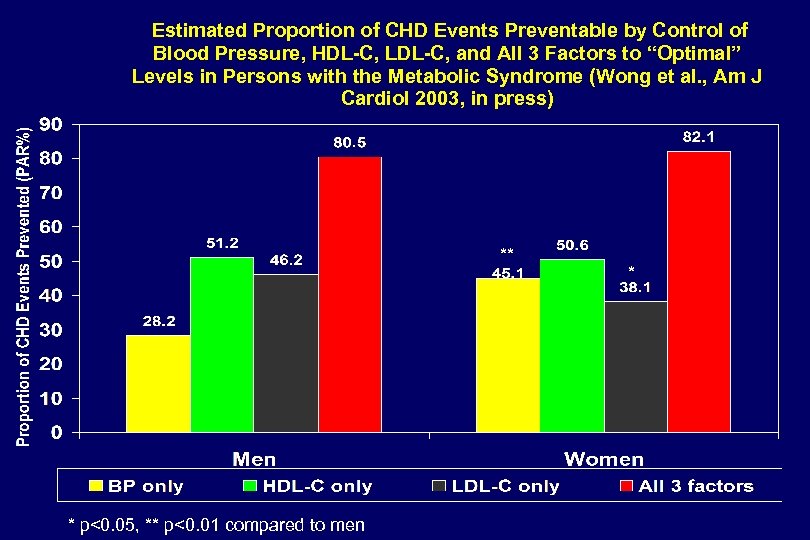 Estimated Proportion of CHD Events Preventable by Control of Blood Pressure, HDL-C, LDL-C, and All 3 Factors to “Optimal” Levels in Persons with the Metabolic Syndrome (Wong et al. , Am J Cardiol 2003, in press) ** * p<0. 05, ** p<0. 01 compared to men *
Estimated Proportion of CHD Events Preventable by Control of Blood Pressure, HDL-C, LDL-C, and All 3 Factors to “Optimal” Levels in Persons with the Metabolic Syndrome (Wong et al. , Am J Cardiol 2003, in press) ** * p<0. 05, ** p<0. 01 compared to men *
 ALLHAT Antihypertensive Trial Design • Randomized, double-blind, multi-center clinical trial • Determine whether occurrence of fatal CHD or nonfatal MI is lower for high-risk hypertensive patients treated with newer agents (CCB, ACEI, alpha-blocker) compared with a diuretic • 42, 418 high-risk hypertensive patients ≥ 55 years
ALLHAT Antihypertensive Trial Design • Randomized, double-blind, multi-center clinical trial • Determine whether occurrence of fatal CHD or nonfatal MI is lower for high-risk hypertensive patients treated with newer agents (CCB, ACEI, alpha-blocker) compared with a diuretic • 42, 418 high-risk hypertensive patients ≥ 55 years
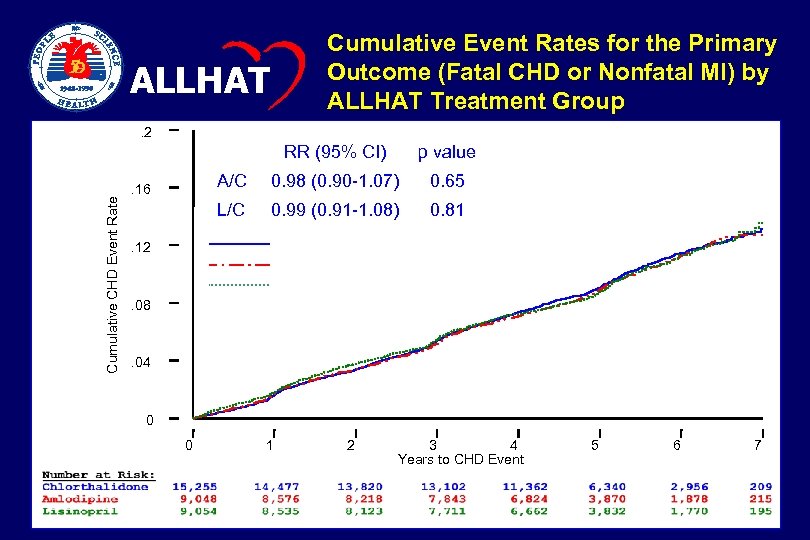 Cumulative Event Rates for the Primary Outcome (Fatal CHD or Nonfatal MI) by ALLHAT Treatment Group ALLHAT . 2 Cumulative CHD Event Rate RR (95% CI) A/C 0. 98 (0. 90 -1. 07) 0. 65 L/C . 16 p value 0. 99 (0. 91 -1. 08) 0. 81 Chlorthalidone Amlodipine Lisinopril . 12 . 08 . 04 0 0 1 2 3 4 Years to CHD Event 5 6 7
Cumulative Event Rates for the Primary Outcome (Fatal CHD or Nonfatal MI) by ALLHAT Treatment Group ALLHAT . 2 Cumulative CHD Event Rate RR (95% CI) A/C 0. 98 (0. 90 -1. 07) 0. 65 L/C . 16 p value 0. 99 (0. 91 -1. 08) 0. 81 Chlorthalidone Amlodipine Lisinopril . 12 . 08 . 04 0 0 1 2 3 4 Years to CHD Event 5 6 7
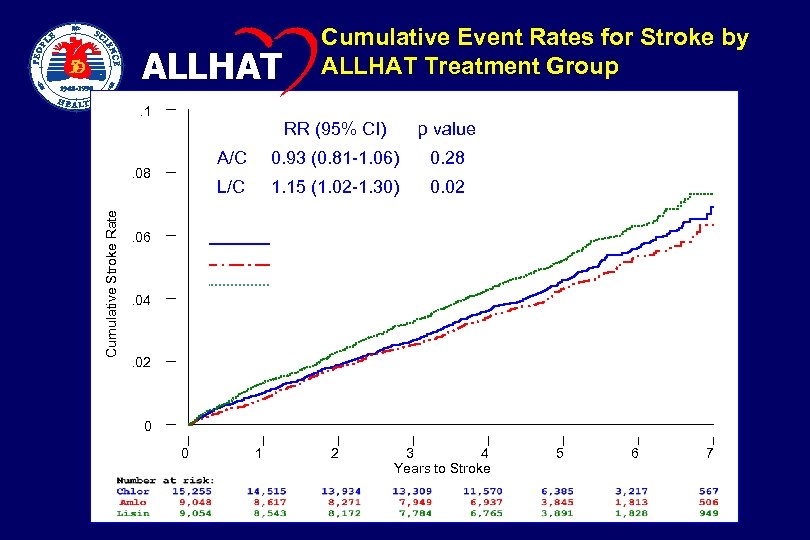 ALLHAT Cumulative Event Rates for Stroke by ALLHAT Treatment Group . 1 RR (95% CI) A/C Cumulative Stroke Rate 0. 93 (0. 81 -1. 06) 0. 28 L/C . 08 p value 1. 15 (1. 02 -1. 30) 0. 02 . 06 Chlorthalidone Amlodipine Lisinopril . 04 . 02 0 0 1 2 3 4 Years to Stroke 5 6 7
ALLHAT Cumulative Event Rates for Stroke by ALLHAT Treatment Group . 1 RR (95% CI) A/C Cumulative Stroke Rate 0. 93 (0. 81 -1. 06) 0. 28 L/C . 08 p value 1. 15 (1. 02 -1. 30) 0. 02 . 06 Chlorthalidone Amlodipine Lisinopril . 04 . 02 0 0 1 2 3 4 Years to Stroke 5 6 7
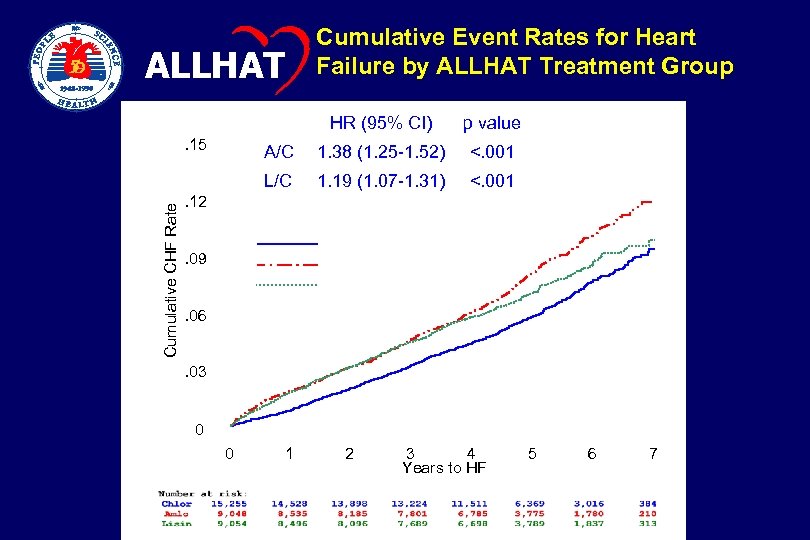 ALLHAT HR (95% CI) p value A/C 1. 38 (1. 25 -1. 52) <. 001 L/C . 15 Cumulative CHF Rate Cumulative Event Rates for Heart Failure by ALLHAT Treatment Group 1. 19 (1. 07 -1. 31) <. 001 . 12 Chlorthalidone Amlodipine Lisinopril . 09. 06. 03 0 0 1 2 3 4 Years to HF 5 6 7
ALLHAT HR (95% CI) p value A/C 1. 38 (1. 25 -1. 52) <. 001 L/C . 15 Cumulative CHF Rate Cumulative Event Rates for Heart Failure by ALLHAT Treatment Group 1. 19 (1. 07 -1. 31) <. 001 . 12 Chlorthalidone Amlodipine Lisinopril . 09. 06. 03 0 0 1 2 3 4 Years to HF 5 6 7
 ALLHAT Overall Conclusions Because of the superiority of thiazide-type diuretics in preventing one or more major forms of CVD and their lower cost, they should be the drugs of choice for first-step antihypertensive drug therapy.
ALLHAT Overall Conclusions Because of the superiority of thiazide-type diuretics in preventing one or more major forms of CVD and their lower cost, they should be the drugs of choice for first-step antihypertensive drug therapy.
 JNC-VII New Features and Key Messages (Continued) § Thiazide-type diuretics should be initial drug therapy for most, either alone or combined with other drug classes. § Certain high-risk conditions are compelling indications for other drug classes. § Most patients will require two or more antihypertensive drugs to achieve goal BP. § If BP is >20/10 mm. Hg above goal, initiate therapy with two agents, one usually should be a thiazide-type diuretic.
JNC-VII New Features and Key Messages (Continued) § Thiazide-type diuretics should be initial drug therapy for most, either alone or combined with other drug classes. § Certain high-risk conditions are compelling indications for other drug classes. § Most patients will require two or more antihypertensive drugs to achieve goal BP. § If BP is >20/10 mm. Hg above goal, initiate therapy with two agents, one usually should be a thiazide-type diuretic.
 JNC-VII New Features and Key Messages (Continued) § The most effective therapy prescribed by the careful clinician will control HTN only if patients are motivated. § Motivation improves when patients have positive experiences with, and trust in, the clinician. § Empathy builds trust and is a potent motivator. § The responsible physician’s judgment remains
JNC-VII New Features and Key Messages (Continued) § The most effective therapy prescribed by the careful clinician will control HTN only if patients are motivated. § Motivation improves when patients have positive experiences with, and trust in, the clinician. § Empathy builds trust and is a potent motivator. § The responsible physician’s judgment remains
 Patient Evaluation of patients with documented HTN has three objectives: 1. Assess lifestyle and identify other CV risk factors or concomitant disorders that affects prognosis and guides treatment. 2. Reveal identifiable causes of high BP. 3. Assess the presence or absence of target organ damage and CVD.
Patient Evaluation of patients with documented HTN has three objectives: 1. Assess lifestyle and identify other CV risk factors or concomitant disorders that affects prognosis and guides treatment. 2. Reveal identifiable causes of high BP. 3. Assess the presence or absence of target organ damage and CVD.
 BP Measurement Techniques Method In-office Ambulatory BP monitoring Self-measurement Brief Description Two readings, 5 minutes apart, sitting in chair. Confirm elevated reading in contralateral arm. Indicated for evaluation of “white-coat” HTN. Absence of 10– 20% BP decrease during sleep may indicate increased CVD risk. Provides information on response to therapy. May help improve adherence to therapy and evaluate “white-coat” HTN.
BP Measurement Techniques Method In-office Ambulatory BP monitoring Self-measurement Brief Description Two readings, 5 minutes apart, sitting in chair. Confirm elevated reading in contralateral arm. Indicated for evaluation of “white-coat” HTN. Absence of 10– 20% BP decrease during sleep may indicate increased CVD risk. Provides information on response to therapy. May help improve adherence to therapy and evaluate “white-coat” HTN.
 CVD Risk Factors § Hypertension* § Cigarette smoking § Obesity* (BMI >30 kg/m 2) § Physical inactivity § Dyslipidemia* § Diabetes mellitus* § Microalbuminuria or estimated GFR <60 ml/min § Age (older than 55 for men, 65 for women) § Family history of premature CVD (men under age 55 or women under age 65) *Components of the metabolic syndrome.
CVD Risk Factors § Hypertension* § Cigarette smoking § Obesity* (BMI >30 kg/m 2) § Physical inactivity § Dyslipidemia* § Diabetes mellitus* § Microalbuminuria or estimated GFR <60 ml/min § Age (older than 55 for men, 65 for women) § Family history of premature CVD (men under age 55 or women under age 65) *Components of the metabolic syndrome.
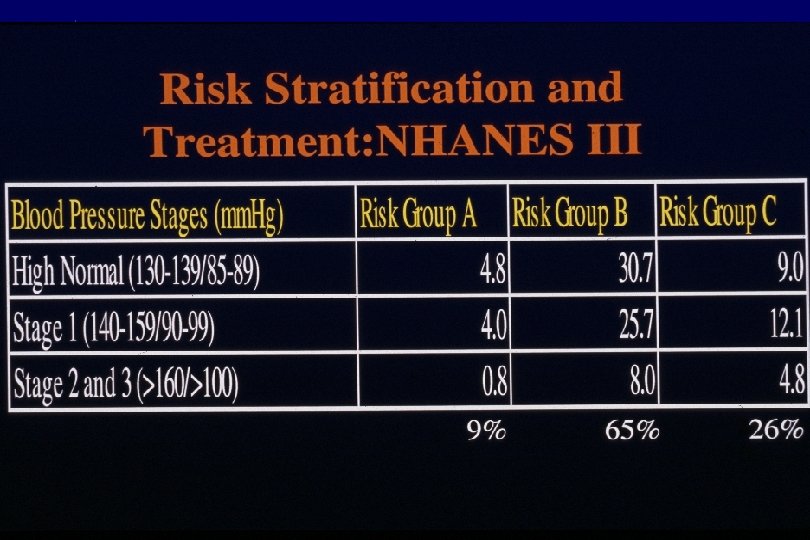
 JNC VI: BP Risk Stratification • Risk Group A – No CV risk factors – No diabetes, target-organ damage, or clinical CVD • Risk Group B – At least one other risk factor: age >60, male gender or postmenopausal status, dyslipidemia, smoking, +FH – (No diabetes, target-organ damage, or clinical CVD) • Risk Group C – Diabetes or target-organ damage or clinical CVD with or without other risk factors JNC VI. Arch Intern Med 1997; 157: 2413.
JNC VI: BP Risk Stratification • Risk Group A – No CV risk factors – No diabetes, target-organ damage, or clinical CVD • Risk Group B – At least one other risk factor: age >60, male gender or postmenopausal status, dyslipidemia, smoking, +FH – (No diabetes, target-organ damage, or clinical CVD) • Risk Group C – Diabetes or target-organ damage or clinical CVD with or without other risk factors JNC VI. Arch Intern Med 1997; 157: 2413.
 Target Organ Damage § Heart • Left ventricular hypertrophy • Angina or prior myocardial infarction • Prior coronary revascularization • Heart failure § Brain • Stroke or transient ischemic attack § Chronic kidney disease § Peripheral arterial disease § Retinopathy
Target Organ Damage § Heart • Left ventricular hypertrophy • Angina or prior myocardial infarction • Prior coronary revascularization • Heart failure § Brain • Stroke or transient ischemic attack § Chronic kidney disease § Peripheral arterial disease § Retinopathy
 Laboratory Tests § Routine Tests • Electrocardiogram • Urinalysis • Blood glucose, and hematocrit • Serum potassium, creatinine, or the corresponding estimated GFR, and calcium • Lipid profile, after 9 - to 12 -hour fast, that includes high-density and low-density lipoprotein cholesterol, and triglycerides § Optional tests • Measurement of urinary albumin excretion or albumin/creatinine ratio § More extensive testing for identifiable causes is not generally
Laboratory Tests § Routine Tests • Electrocardiogram • Urinalysis • Blood glucose, and hematocrit • Serum potassium, creatinine, or the corresponding estimated GFR, and calcium • Lipid profile, after 9 - to 12 -hour fast, that includes high-density and low-density lipoprotein cholesterol, and triglycerides § Optional tests • Measurement of urinary albumin excretion or albumin/creatinine ratio § More extensive testing for identifiable causes is not generally
 Lifestyle Modification Weight reduction Adopt DASH eating plan Dietary sodium reduction Physical activity Moderation of alcohol consumption Approximate SBP reduction (range) 5– 20 mm. Hg/10 kg weight loss 8– 14 mm. Hg 2– 8 mm. Hg 4– 9 mm. Hg 2– 4 mm. Hg
Lifestyle Modification Weight reduction Adopt DASH eating plan Dietary sodium reduction Physical activity Moderation of alcohol consumption Approximate SBP reduction (range) 5– 20 mm. Hg/10 kg weight loss 8– 14 mm. Hg 2– 8 mm. Hg 4– 9 mm. Hg 2– 4 mm. Hg
 Lifestyle Modifications For Prevention and Management For Overall and Cardiovascular Health • Lose weight if overweight • Limit alcohol intake • Increase aerobic physical activity • Reduce sodium intake • Maintain adequate intake of potassium • Maintain adequate intake of calcium and magnesium • Stop smoking • Reduce dietary saturated fat and cholesterol
Lifestyle Modifications For Prevention and Management For Overall and Cardiovascular Health • Lose weight if overweight • Limit alcohol intake • Increase aerobic physical activity • Reduce sodium intake • Maintain adequate intake of potassium • Maintain adequate intake of calcium and magnesium • Stop smoking • Reduce dietary saturated fat and cholesterol
 Dietary Approaches to Stop Hypertension (DASH) • Diet high in fruits and vegetables and low-fat dairy products lowers blood pressure (11 mm. Hg SBP/ 5 mm. Hg DBP lower than traditional US diet), including more than a sodium-restricted diet • Recommends 7 -8 servings/day of grain/grain products, 4 -5 vegetable, 4 -5 fruit, 2 -3 low- or non-fat dairy products, 2 or less meat, poultry, and fish. • NEJM 1997; 366: 1117 -24.
Dietary Approaches to Stop Hypertension (DASH) • Diet high in fruits and vegetables and low-fat dairy products lowers blood pressure (11 mm. Hg SBP/ 5 mm. Hg DBP lower than traditional US diet), including more than a sodium-restricted diet • Recommends 7 -8 servings/day of grain/grain products, 4 -5 vegetable, 4 -5 fruit, 2 -3 low- or non-fat dairy products, 2 or less meat, poultry, and fish. • NEJM 1997; 366: 1117 -24.
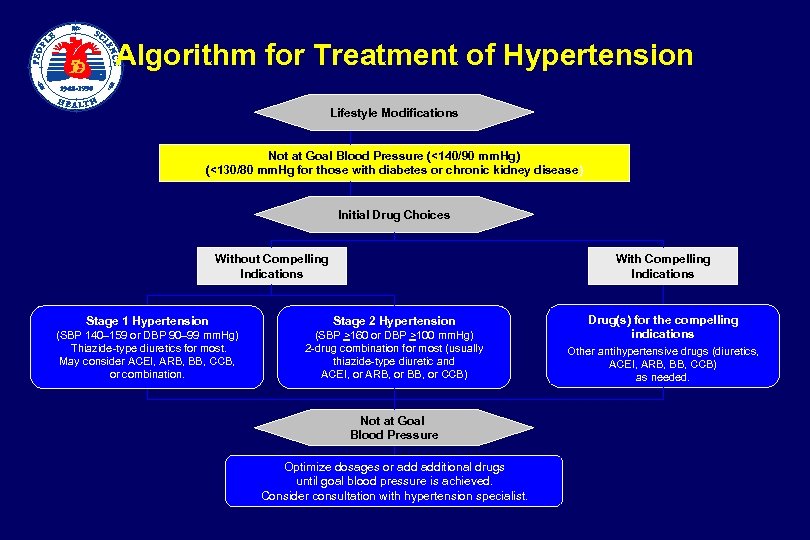 Algorithm for Treatment of Hypertension Lifestyle Modifications Not at Goal Blood Pressure (<140/90 mm. Hg) (<130/80 mm. Hg for those with diabetes or chronic kidney disease) Initial Drug Choices Without Compelling Indications With Compelling Indications Stage 1 Hypertension Stage 2 Hypertension (SBP 140– 159 or DBP 90– 99 mm. Hg) Thiazide-type diuretics for most. May consider ACEI, ARB, BB, CCB, or combination. (SBP >160 or DBP >100 mm. Hg) 2 -drug combination for most (usually thiazide-type diuretic and ACEI, or ARB, or BB, or CCB) Not at Goal Blood Pressure Optimize dosages or additional drugs until goal blood pressure is achieved. Consider consultation with hypertension specialist. Drug(s) for the compelling indications Other antihypertensive drugs (diuretics, ACEI, ARB, BB, CCB) as needed.
Algorithm for Treatment of Hypertension Lifestyle Modifications Not at Goal Blood Pressure (<140/90 mm. Hg) (<130/80 mm. Hg for those with diabetes or chronic kidney disease) Initial Drug Choices Without Compelling Indications With Compelling Indications Stage 1 Hypertension Stage 2 Hypertension (SBP 140– 159 or DBP 90– 99 mm. Hg) Thiazide-type diuretics for most. May consider ACEI, ARB, BB, CCB, or combination. (SBP >160 or DBP >100 mm. Hg) 2 -drug combination for most (usually thiazide-type diuretic and ACEI, or ARB, or BB, or CCB) Not at Goal Blood Pressure Optimize dosages or additional drugs until goal blood pressure is achieved. Consider consultation with hypertension specialist. Drug(s) for the compelling indications Other antihypertensive drugs (diuretics, ACEI, ARB, BB, CCB) as needed.
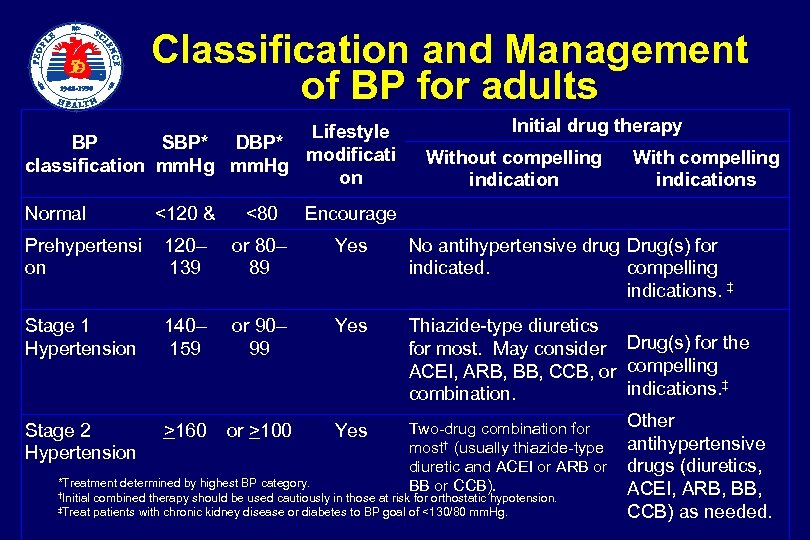 Classification and Management of BP for adults Lifestyle BP SBP* DBP* modificati classification mm. Hg on Normal <120 & <80 120– 139 or 80– 89 Yes Stage 1 Hypertension 140– 159 or 90– 99 Yes Without compelling indication With compelling indications Encourage Prehypertensi on Initial drug therapy No antihypertensive drug Drug(s) for indicated. compelling indications. ‡ Thiazide-type diuretics for most. May consider Drug(s) for the ACEI, ARB, BB, CCB, or compelling indications. ‡ combination. Other Two-drug combination for Stage 2 >160 or >100 Yes most† (usually thiazide-type antihypertensive Hypertension diuretic and ACEI or ARB or drugs (diuretics, *Treatment determined by highest BP category. BB or CCB). ACEI, ARB, BB, †Initial combined therapy should be used cautiously in those at risk for orthostatic hypotension. ‡Treat patients with chronic kidney disease or diabetes to BP goal of <130/80 mm. Hg. CCB) as needed.
Classification and Management of BP for adults Lifestyle BP SBP* DBP* modificati classification mm. Hg on Normal <120 & <80 120– 139 or 80– 89 Yes Stage 1 Hypertension 140– 159 or 90– 99 Yes Without compelling indication With compelling indications Encourage Prehypertensi on Initial drug therapy No antihypertensive drug Drug(s) for indicated. compelling indications. ‡ Thiazide-type diuretics for most. May consider Drug(s) for the ACEI, ARB, BB, CCB, or compelling indications. ‡ combination. Other Two-drug combination for Stage 2 >160 or >100 Yes most† (usually thiazide-type antihypertensive Hypertension diuretic and ACEI or ARB or drugs (diuretics, *Treatment determined by highest BP category. BB or CCB). ACEI, ARB, BB, †Initial combined therapy should be used cautiously in those at risk for orthostatic hypotension. ‡Treat patients with chronic kidney disease or diabetes to BP goal of <130/80 mm. Hg. CCB) as needed.
 Followup and Monitoring § Patients should return for followup and adjustment of medications until the BP goal is reached. § More frequent visits for stage 2 HTN or with complicating comorbid conditions. § Serum potassium and creatinine monitored 1– 2 times per year.
Followup and Monitoring § Patients should return for followup and adjustment of medications until the BP goal is reached. § More frequent visits for stage 2 HTN or with complicating comorbid conditions. § Serum potassium and creatinine monitored 1– 2 times per year.
 Followup and Monitoring (continued) § After BP at goal and stable, followup visits at 3 - to 6 month intervals. § Comorbidities, such as heart failure, associated diseases, such as diabetes, and the need for laboratory tests influence the frequency of visits.
Followup and Monitoring (continued) § After BP at goal and stable, followup visits at 3 - to 6 month intervals. § Comorbidities, such as heart failure, associated diseases, such as diabetes, and the need for laboratory tests influence the frequency of visits.
 Special Considerations § Compelling Indications § Other Special Situations • Minority populations • Obesity and the metabolic syndrome • Left ventricular hypertrophy • Peripheral arterial disease • Hypertension in older persons • Postural hypotension • Dementia • Hypertension in women • Hypertension in children and adolescents • Hypertension urgencies and emergencies
Special Considerations § Compelling Indications § Other Special Situations • Minority populations • Obesity and the metabolic syndrome • Left ventricular hypertrophy • Peripheral arterial disease • Hypertension in older persons • Postural hypotension • Dementia • Hypertension in women • Hypertension in children and adolescents • Hypertension urgencies and emergencies
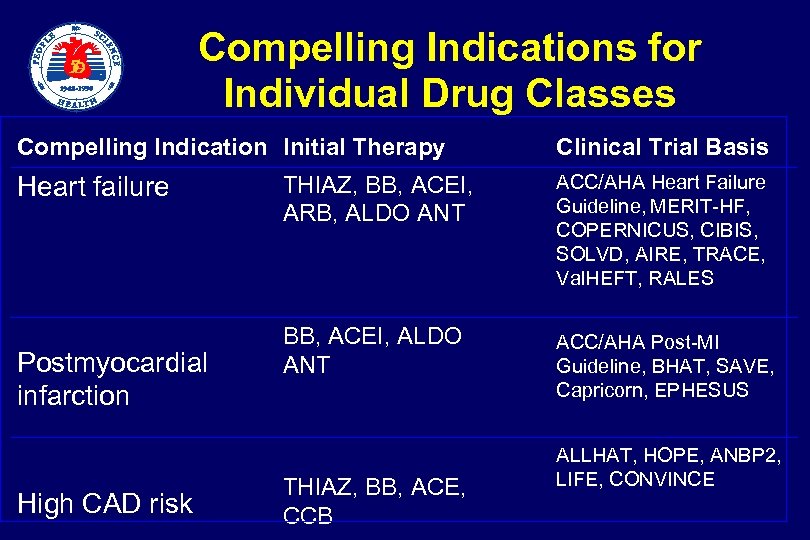 Compelling Indications for Individual Drug Classes Compelling Indication Initial Therapy Heart failure Postmyocardial infarction High CAD risk Clinical Trial Basis THIAZ, BB, ACEI, ARB, ALDO ANT ACC/AHA Heart Failure Guideline, MERIT-HF, COPERNICUS, CIBIS, SOLVD, AIRE, TRACE, Val. HEFT, RALES BB, ACEI, ALDO ANT ACC/AHA Post-MI Guideline, BHAT, SAVE, Capricorn, EPHESUS THIAZ, BB, ACE, CCB ALLHAT, HOPE, ANBP 2, LIFE, CONVINCE
Compelling Indications for Individual Drug Classes Compelling Indication Initial Therapy Heart failure Postmyocardial infarction High CAD risk Clinical Trial Basis THIAZ, BB, ACEI, ARB, ALDO ANT ACC/AHA Heart Failure Guideline, MERIT-HF, COPERNICUS, CIBIS, SOLVD, AIRE, TRACE, Val. HEFT, RALES BB, ACEI, ALDO ANT ACC/AHA Post-MI Guideline, BHAT, SAVE, Capricorn, EPHESUS THIAZ, BB, ACE, CCB ALLHAT, HOPE, ANBP 2, LIFE, CONVINCE
 Compelling Indications for Individual Drug Classes Compelling Indication Initial Therapy Options Clinical Trial Basis Diabetes Chronic kidney disease Recurrent stroke prevention THIAZ, BB, ACE, ARB, CCB ACEI, ARB THIAZ, ACEI NKF-ADA Guideline, UKPDS, ALLHAT NKF Guideline, Captopril Trial, RENAAL, IDNT, REIN, AASK PROGRESS
Compelling Indications for Individual Drug Classes Compelling Indication Initial Therapy Options Clinical Trial Basis Diabetes Chronic kidney disease Recurrent stroke prevention THIAZ, BB, ACE, ARB, CCB ACEI, ARB THIAZ, ACEI NKF-ADA Guideline, UKPDS, ALLHAT NKF Guideline, Captopril Trial, RENAAL, IDNT, REIN, AASK PROGRESS
 Cardiovascular Diseases • Cerebrovascular disease – Indication for treatment, except immediately after ischemic cerebral infarction. • Coronary artery disease – Benefits of therapy well established. • Left ventricular hypertrophy – Antihypertensive agents (except direct vasodilators) indicated. – Reduced weight and decreased sodium intake beneficial.
Cardiovascular Diseases • Cerebrovascular disease – Indication for treatment, except immediately after ischemic cerebral infarction. • Coronary artery disease – Benefits of therapy well established. • Left ventricular hypertrophy – Antihypertensive agents (except direct vasodilators) indicated. – Reduced weight and decreased sodium intake beneficial.
 Cardiovascular Diseases (continued) • Cardiac failure – ACE inhibitors, especially with digoxin or diuretics, shown to prevent subsequent heart failure. • Peripheral arterial disease – Limited or no data available.
Cardiovascular Diseases (continued) • Cardiac failure – ACE inhibitors, especially with digoxin or diuretics, shown to prevent subsequent heart failure. • Peripheral arterial disease – Limited or no data available.
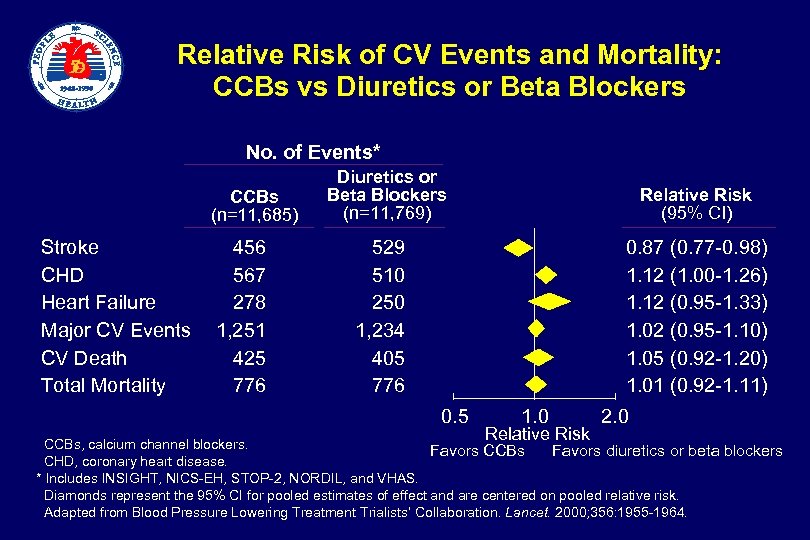 Relative Risk of CV Events and Mortality: CCBs vs Diuretics or Beta Blockers No. of Events* CCBs (n=11, 685) Stroke CHD Heart Failure Major CV Events CV Death Total Mortality 456 567 278 1, 251 425 776 Diuretics or Beta Blockers (n=11, 769) Relative Risk (95% CI) 529 510 250 1, 234 405 776 0. 87 (0. 77 -0. 98) 1. 12 (1. 00 -1. 26) 1. 12 (0. 95 -1. 33) 1. 02 (0. 95 -1. 10) 1. 05 (0. 92 -1. 20) 1. 01 (0. 92 -1. 11) 0. 5 1. 0 Relative Risk 2. 0 CCBs, calcium channel blockers. Favors CCBs Favors diuretics or beta blockers CHD, coronary heart disease. * Includes INSIGHT, NICS-EH, STOP-2, NORDIL, and VHAS. Diamonds represent the 95% CI for pooled estimates of effect and are centered on pooled relative risk. Adapted from Blood Pressure Lowering Treatment Trialists’ Collaboration. Lancet. 2000; 356: 1955 -1964.
Relative Risk of CV Events and Mortality: CCBs vs Diuretics or Beta Blockers No. of Events* CCBs (n=11, 685) Stroke CHD Heart Failure Major CV Events CV Death Total Mortality 456 567 278 1, 251 425 776 Diuretics or Beta Blockers (n=11, 769) Relative Risk (95% CI) 529 510 250 1, 234 405 776 0. 87 (0. 77 -0. 98) 1. 12 (1. 00 -1. 26) 1. 12 (0. 95 -1. 33) 1. 02 (0. 95 -1. 10) 1. 05 (0. 92 -1. 20) 1. 01 (0. 92 -1. 11) 0. 5 1. 0 Relative Risk 2. 0 CCBs, calcium channel blockers. Favors CCBs Favors diuretics or beta blockers CHD, coronary heart disease. * Includes INSIGHT, NICS-EH, STOP-2, NORDIL, and VHAS. Diamonds represent the 95% CI for pooled estimates of effect and are centered on pooled relative risk. Adapted from Blood Pressure Lowering Treatment Trialists’ Collaboration. Lancet. 2000; 356: 1955 -1964.
 HOPE: Risk Reduction of CV Events Associated with ACEI (RAS Inhibition) Treatment 0 MI, Stroke, CV Death (primary end point) CV Death MI Stroke All-cause Death Risk Reduction (%) -5 -10 -15 -20 -25 -30 -35 -22 (P<. 001) -16 (P=. 005) -20 (P<. 001) -26 (P<. 001) -32 (P<. 001) Adapted from The Heart Outcomes Prevention Evaluation Study Investigators. Effects of an angiotensin-converting enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N Engl J Med. 2000; 342: 145 -153.
HOPE: Risk Reduction of CV Events Associated with ACEI (RAS Inhibition) Treatment 0 MI, Stroke, CV Death (primary end point) CV Death MI Stroke All-cause Death Risk Reduction (%) -5 -10 -15 -20 -25 -30 -35 -22 (P<. 001) -16 (P=. 005) -20 (P<. 001) -26 (P<. 001) -32 (P<. 001) Adapted from The Heart Outcomes Prevention Evaluation Study Investigators. Effects of an angiotensin-converting enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. N Engl J Med. 2000; 342: 145 -153.
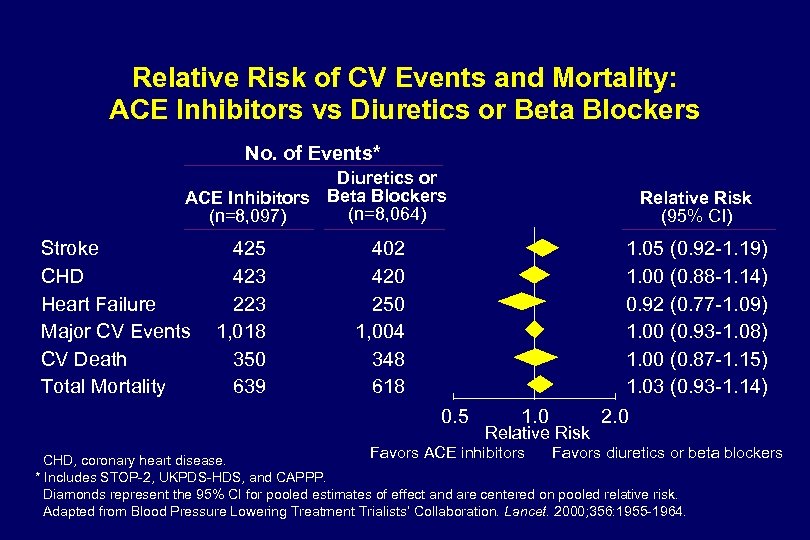 Relative Risk of CV Events and Mortality: ACE Inhibitors vs Diuretics or Beta Blockers No. of Events* Diuretics or ACE Inhibitors Beta Blockers (n=8, 064) (n=8, 097) Stroke CHD Heart Failure Major CV Events CV Death Total Mortality 425 423 223 1, 018 350 639 Relative Risk (95% CI) 402 420 250 1, 004 348 618 0. 5 1. 0 Relative Risk Favors ACE inhibitors 1. 05 (0. 92 -1. 19) 1. 00 (0. 88 -1. 14) 0. 92 (0. 77 -1. 09) 1. 00 (0. 93 -1. 08) 1. 00 (0. 87 -1. 15) 1. 03 (0. 93 -1. 14) 2. 0 Favors diuretics or beta blockers CHD, coronary heart disease. * Includes STOP-2, UKPDS-HDS, and CAPPP. Diamonds represent the 95% CI for pooled estimates of effect and are centered on pooled relative risk. Adapted from Blood Pressure Lowering Treatment Trialists’ Collaboration. Lancet. 2000; 356: 1955 -1964.
Relative Risk of CV Events and Mortality: ACE Inhibitors vs Diuretics or Beta Blockers No. of Events* Diuretics or ACE Inhibitors Beta Blockers (n=8, 064) (n=8, 097) Stroke CHD Heart Failure Major CV Events CV Death Total Mortality 425 423 223 1, 018 350 639 Relative Risk (95% CI) 402 420 250 1, 004 348 618 0. 5 1. 0 Relative Risk Favors ACE inhibitors 1. 05 (0. 92 -1. 19) 1. 00 (0. 88 -1. 14) 0. 92 (0. 77 -1. 09) 1. 00 (0. 93 -1. 08) 1. 00 (0. 87 -1. 15) 1. 03 (0. 93 -1. 14) 2. 0 Favors diuretics or beta blockers CHD, coronary heart disease. * Includes STOP-2, UKPDS-HDS, and CAPPP. Diamonds represent the 95% CI for pooled estimates of effect and are centered on pooled relative risk. Adapted from Blood Pressure Lowering Treatment Trialists’ Collaboration. Lancet. 2000; 356: 1955 -1964.
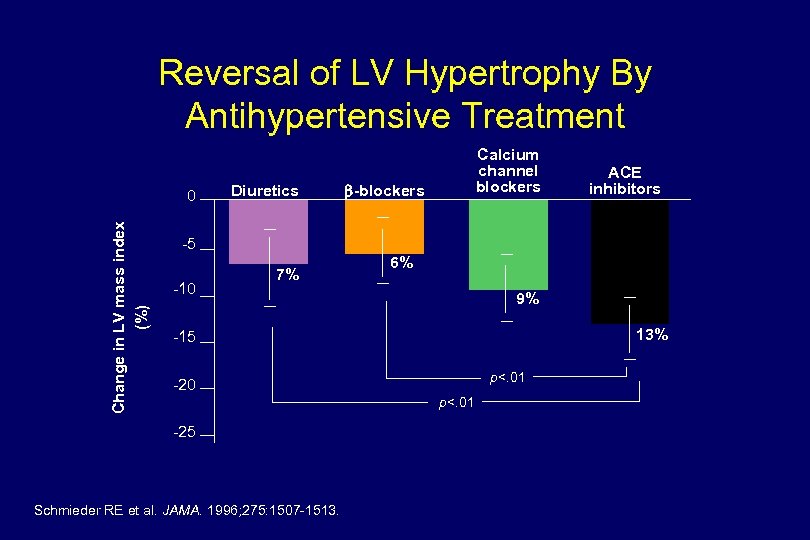 Reversal of LV Hypertrophy By Antihypertensive Treatment Change in LV mass index (%) 0 Diuretics Calcium channel blockers -blockers ACE inhibitors -5 -10 7% 6% 9% 13% -15 p<. 01 -20 p<. 01 -25 Schmieder RE et al. JAMA. 1996; 275: 1507 -1513.
Reversal of LV Hypertrophy By Antihypertensive Treatment Change in LV mass index (%) 0 Diuretics Calcium channel blockers -blockers ACE inhibitors -5 -10 7% 6% 9% 13% -15 p<. 01 -20 p<. 01 -25 Schmieder RE et al. JAMA. 1996; 275: 1507 -1513.
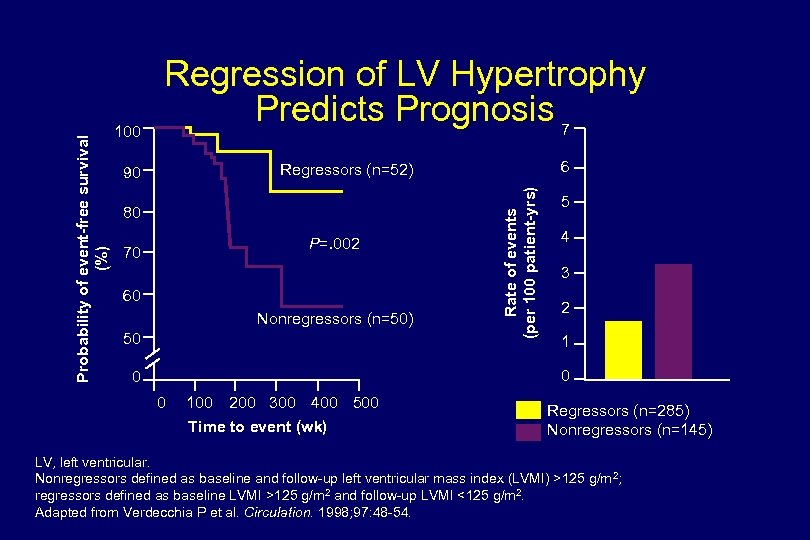 6 Regressors (n=52) 90 80 P=. 002 70 60 Nonregressors (n=50) 50 Rate of events (per 100 patient-yrs) Probability of event-free survival (%) 100 Regression of LV Hypertrophy Predicts Prognosis 7 5 4 3 2 1 0 0 0 100 200 300 400 500 Time to event (wk) Regressors (n=285) Nonregressors (n=145) LV, left ventricular. Nonregressors defined as baseline and follow-up left ventricular mass index (LVMI) >125 g/m 2; regressors defined as baseline LVMI >125 g/m 2 and follow-up LVMI <125 g/m 2. Adapted from Verdecchia P et al. Circulation. 1998; 97: 48 -54.
6 Regressors (n=52) 90 80 P=. 002 70 60 Nonregressors (n=50) 50 Rate of events (per 100 patient-yrs) Probability of event-free survival (%) 100 Regression of LV Hypertrophy Predicts Prognosis 7 5 4 3 2 1 0 0 0 100 200 300 400 500 Time to event (wk) Regressors (n=285) Nonregressors (n=145) LV, left ventricular. Nonregressors defined as baseline and follow-up left ventricular mass index (LVMI) >125 g/m 2; regressors defined as baseline LVMI >125 g/m 2 and follow-up LVMI <125 g/m 2. Adapted from Verdecchia P et al. Circulation. 1998; 97: 48 -54.
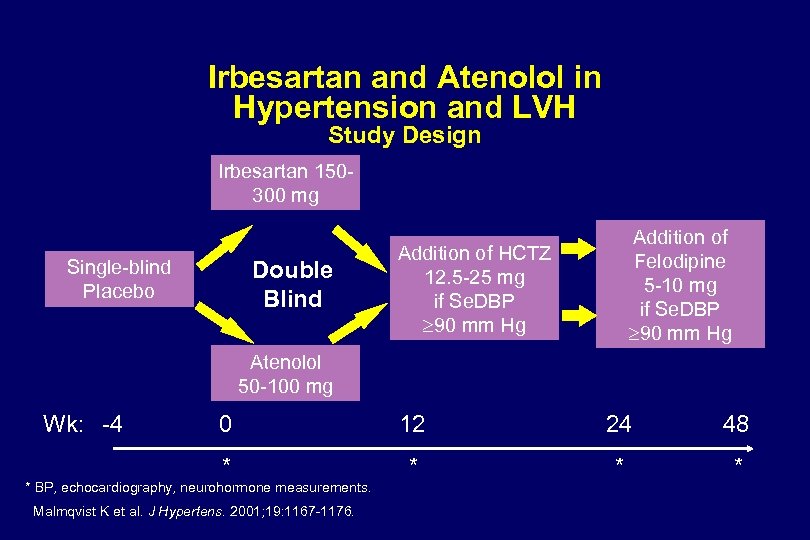 Irbesartan and Atenolol in Hypertension and LVH Study Design Irbesartan 150300 mg Single-blind Placebo Double Blind Addition of Felodipine 5 -10 mg if Se. DBP 90 mm Hg Addition of HCTZ 12. 5 -25 mg if Se. DBP 90 mm Hg Atenolol 50 -100 mg Wk: -4 0 12 24 48 * * * BP, echocardiography, neurohormone measurements. Malmqvist K et al. J Hypertens. 2001; 19: 1167 -1176.
Irbesartan and Atenolol in Hypertension and LVH Study Design Irbesartan 150300 mg Single-blind Placebo Double Blind Addition of Felodipine 5 -10 mg if Se. DBP 90 mm Hg Addition of HCTZ 12. 5 -25 mg if Se. DBP 90 mm Hg Atenolol 50 -100 mg Wk: -4 0 12 24 48 * * * BP, echocardiography, neurohormone measurements. Malmqvist K et al. J Hypertens. 2001; 19: 1167 -1176.
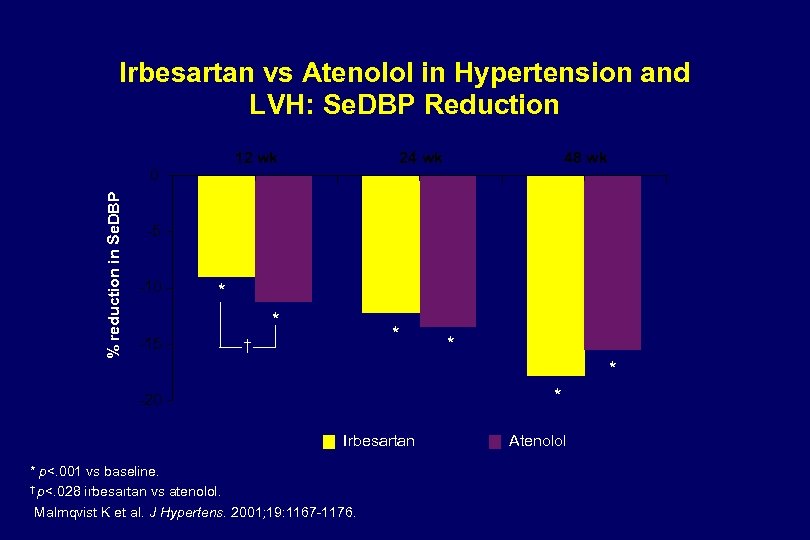 Irbesartan vs Atenolol in Hypertension and LVH: Se. DBP Reduction 12 wk % reduction in Se. DBP 0 24 wk 48 wk -5 -10 * * -15 * † * * * -20 Irbesartan * p<. 001 vs baseline. † p<. 028 irbesartan vs atenolol. Malmqvist K et al. J Hypertens. 2001; 19: 1167 -1176. Atenolol
Irbesartan vs Atenolol in Hypertension and LVH: Se. DBP Reduction 12 wk % reduction in Se. DBP 0 24 wk 48 wk -5 -10 * * -15 * † * * * -20 Irbesartan * p<. 001 vs baseline. † p<. 028 irbesartan vs atenolol. Malmqvist K et al. J Hypertens. 2001; 19: 1167 -1176. Atenolol
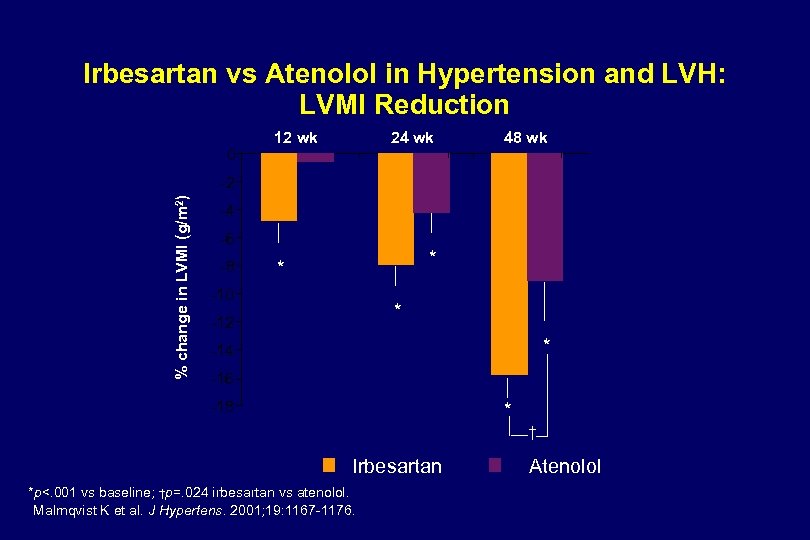 Irbesartan vs Atenolol in Hypertension and LVH: LVMI Reduction 0 12 wk 24 wk 48 wk % change in LVMI (g/m 2) -2 -4 -6 -8 * * -10 * -12 * -14 -16 -18 * † Irbesartan *p<. 001 vs baseline; †p=. 024 irbesartan vs atenolol. Malmqvist K et al. J Hypertens. 2001; 19: 1167 -1176. Atenolol
Irbesartan vs Atenolol in Hypertension and LVH: LVMI Reduction 0 12 wk 24 wk 48 wk % change in LVMI (g/m 2) -2 -4 -6 -8 * * -10 * -12 * -14 -16 -18 * † Irbesartan *p<. 001 vs baseline; †p=. 024 irbesartan vs atenolol. Malmqvist K et al. J Hypertens. 2001; 19: 1167 -1176. Atenolol
 LIFE: Inclusion Criteria • Age 55 -80 years • Previously treated or untreated hypertension • Systolic BP 160 -200 mm. Hg or Diastolic BP 95 -115 mm. Hg • ECG LVH Adapted from Dahlöf B et al. Am J Hypertens. 1997; 10: 705 -13.
LIFE: Inclusion Criteria • Age 55 -80 years • Previously treated or untreated hypertension • Systolic BP 160 -200 mm. Hg or Diastolic BP 95 -115 mm. Hg • ECG LVH Adapted from Dahlöf B et al. Am J Hypertens. 1997; 10: 705 -13.
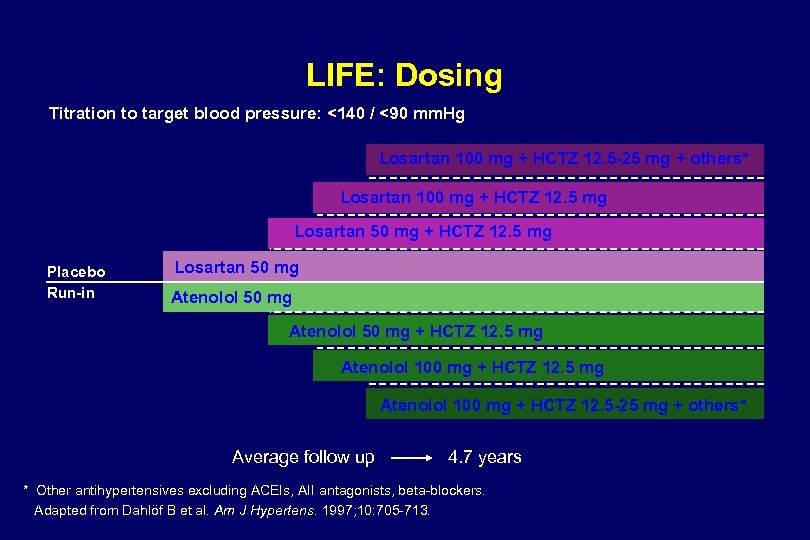 LIFE: Dosing Titration to target blood pressure: <140 / <90 mm. Hg Losartan 100 mg + HCTZ 12. 5 -25 mg + others* Losartan 100 mg + HCTZ 12. 5 mg Losartan 50 mg + HCTZ 12. 5 mg Placebo Run-in Losartan 50 mg Atenolol 50 mg + HCTZ 12. 5 mg Atenolol 100 mg + HCTZ 12. 5 -25 mg + others* Average follow up 4. 7 years * Other antihypertensives excluding ACEIs, AII antagonists, beta-blockers. Adapted from Dahlöf B et al. Am J Hypertens. 1997; 10: 705 -713.
LIFE: Dosing Titration to target blood pressure: <140 / <90 mm. Hg Losartan 100 mg + HCTZ 12. 5 -25 mg + others* Losartan 100 mg + HCTZ 12. 5 mg Losartan 50 mg + HCTZ 12. 5 mg Placebo Run-in Losartan 50 mg Atenolol 50 mg + HCTZ 12. 5 mg Atenolol 100 mg + HCTZ 12. 5 -25 mg + others* Average follow up 4. 7 years * Other antihypertensives excluding ACEIs, AII antagonists, beta-blockers. Adapted from Dahlöf B et al. Am J Hypertens. 1997; 10: 705 -713.
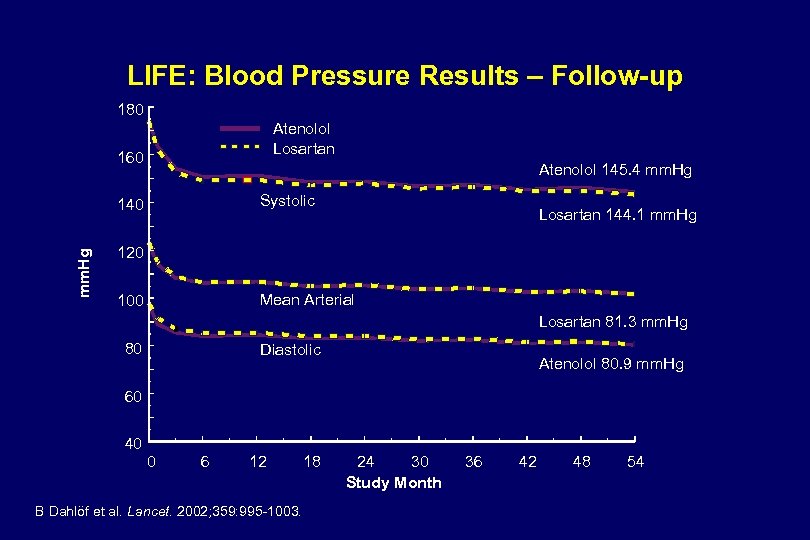 LIFE: Blood Pressure Results – Follow-up 180 Atenolol Losartan 160 Atenolol 145. 4 mm. Hg Systolic mm. Hg 140 Losartan 144. 1 mm. Hg 120 Mean Arterial 100 Losartan 81. 3 mm. Hg 80 Diastolic Atenolol 80. 9 mm. Hg 60 40 0 6 12 B Dahlöf et al. Lancet. 2002; 359: 995 -1003. 18 24 30 Study Month 36 42 48 54
LIFE: Blood Pressure Results – Follow-up 180 Atenolol Losartan 160 Atenolol 145. 4 mm. Hg Systolic mm. Hg 140 Losartan 144. 1 mm. Hg 120 Mean Arterial 100 Losartan 81. 3 mm. Hg 80 Diastolic Atenolol 80. 9 mm. Hg 60 40 0 6 12 B Dahlöf et al. Lancet. 2002; 359: 995 -1003. 18 24 30 Study Month 36 42 48 54
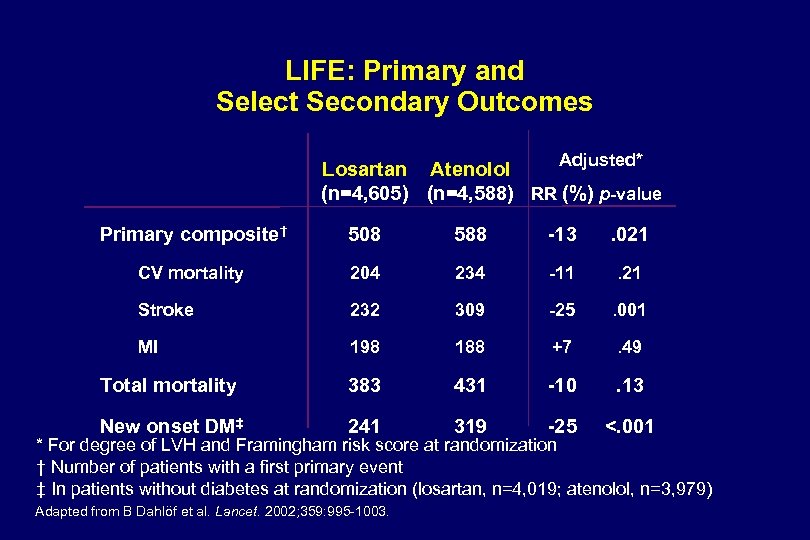 LIFE: Primary and Select Secondary Outcomes Adjusted* Losartan Atenolol (n=4, 605) (n=4, 588) RR (%) p-value Primary composite† 508 588 -13 . 021 CV mortality 204 234 -11 . 21 Stroke 232 309 -25 . 001 MI 198 188 +7 . 49 Total mortality 383 431 -10 . 13 New onset DM‡ 241 319 -25 <. 001 * For degree of LVH and Framingham risk score at randomization † Number of patients with a first primary event ‡ In patients without diabetes at randomization (losartan, n=4, 019; atenolol, n=3, 979) Adapted from B Dahlöf et al. Lancet. 2002; 359: 995 -1003.
LIFE: Primary and Select Secondary Outcomes Adjusted* Losartan Atenolol (n=4, 605) (n=4, 588) RR (%) p-value Primary composite† 508 588 -13 . 021 CV mortality 204 234 -11 . 21 Stroke 232 309 -25 . 001 MI 198 188 +7 . 49 Total mortality 383 431 -10 . 13 New onset DM‡ 241 319 -25 <. 001 * For degree of LVH and Framingham risk score at randomization † Number of patients with a first primary event ‡ In patients without diabetes at randomization (losartan, n=4, 019; atenolol, n=3, 979) Adapted from B Dahlöf et al. Lancet. 2002; 359: 995 -1003.
 Valsartan Heart Failure Trial (Val-He. FT) Study Characteristics: • 5, 010 total patients randomized with NYHA class II, III, or IV HF • Two groups: valsartan (target dose 160 mg BID) plus standard therapy vs placebo plus standard therapy • Mean duration of follow-up: 23 months (range 0 -38) • Two primary end points: – Mortality – Combined mortality and morbidity (morbidity defined as cardiac arrest with resuscitation, hospitalization for HF, or administration of IV inotropic or vasodilator drugs for > 4 hours without hospitalization)
Valsartan Heart Failure Trial (Val-He. FT) Study Characteristics: • 5, 010 total patients randomized with NYHA class II, III, or IV HF • Two groups: valsartan (target dose 160 mg BID) plus standard therapy vs placebo plus standard therapy • Mean duration of follow-up: 23 months (range 0 -38) • Two primary end points: – Mortality – Combined mortality and morbidity (morbidity defined as cardiac arrest with resuscitation, hospitalization for HF, or administration of IV inotropic or vasodilator drugs for > 4 hours without hospitalization)
 Val-He. FT Results • Overall mortality was similar in the two groups • 13% RRR (p=. 009) in combined end point • Predominantly because of a 27% decrease in hospitalization for HF in the valsartan group • Subgroup analyses: – Valsartan had a favorable effect in patients receiving neither an ACE inhibitor nor a beta-blocker – Valsartan had a favorable effect in patients receiving an ACE inhibitor or a beta blocker – Valsartan demonstrated a statistically non-significant trend towards an adverse outcome in patients receiving an ACE inhibitor and a beta blocker
Val-He. FT Results • Overall mortality was similar in the two groups • 13% RRR (p=. 009) in combined end point • Predominantly because of a 27% decrease in hospitalization for HF in the valsartan group • Subgroup analyses: – Valsartan had a favorable effect in patients receiving neither an ACE inhibitor nor a beta-blocker – Valsartan had a favorable effect in patients receiving an ACE inhibitor or a beta blocker – Valsartan demonstrated a statistically non-significant trend towards an adverse outcome in patients receiving an ACE inhibitor and a beta blocker
 Web site www. nhlbi. nih. gov/
Web site www. nhlbi. nih. gov/
 DASH Fact Sheet
DASH Fact Sheet
 Your Guide to Lowering Blood Pressure
Your Guide to Lowering Blood Pressure
 Reference Card
Reference Card
 Diabetes Mellitus • Drug therapy should begin along with lifestyle modifications to reduce blood pressure to < 130/85 mm Hg. • ACE inhibitors, -blockers, calcium antagonists, and low-dose diuretics are preferred. • Insulin resistance or high peripheral insulin levels may cause hypertension, which can be treated with lifestyle changes, insulin-sensitizing agents, vasodilating antihypertensive drugs, and lipidlowering agents.
Diabetes Mellitus • Drug therapy should begin along with lifestyle modifications to reduce blood pressure to < 130/85 mm Hg. • ACE inhibitors, -blockers, calcium antagonists, and low-dose diuretics are preferred. • Insulin resistance or high peripheral insulin levels may cause hypertension, which can be treated with lifestyle changes, insulin-sensitizing agents, vasodilating antihypertensive drugs, and lipidlowering agents.
 Renal Disease • Hypertension may result from renal disease that reduces functioning nephrons. • Evidence shows a clear relationship between high blood pressure and end-stage renal disease. • Blood pressure should be controlled to < 130/85 mm Hg or lower (< 125/75 mm Hg) in patients with proteinuria in excess of 1 gram per 24 hours. • ACE inhibitors work well to control blood pressure and slow progression of renal failure.
Renal Disease • Hypertension may result from renal disease that reduces functioning nephrons. • Evidence shows a clear relationship between high blood pressure and end-stage renal disease. • Blood pressure should be controlled to < 130/85 mm Hg or lower (< 125/75 mm Hg) in patients with proteinuria in excess of 1 gram per 24 hours. • ACE inhibitors work well to control blood pressure and slow progression of renal failure.
 ADA Guidelines on Management of Diabetic Nephropathy Hypertensive Type 2 Diabetic Patients* ARBs are the initial agents of choice Type 1 Diabetics with or without hypertension* ACEIs are the initial agents of choice If one class is not tolerated the other should be substituted * With microalbuminuria and clinical proteinuria. Adapted from American Diabetes Association. Diabetes Care. 2002; 25: S 85 -S 89.
ADA Guidelines on Management of Diabetic Nephropathy Hypertensive Type 2 Diabetic Patients* ARBs are the initial agents of choice Type 1 Diabetics with or without hypertension* ACEIs are the initial agents of choice If one class is not tolerated the other should be substituted * With microalbuminuria and clinical proteinuria. Adapted from American Diabetes Association. Diabetes Care. 2002; 25: S 85 -S 89.
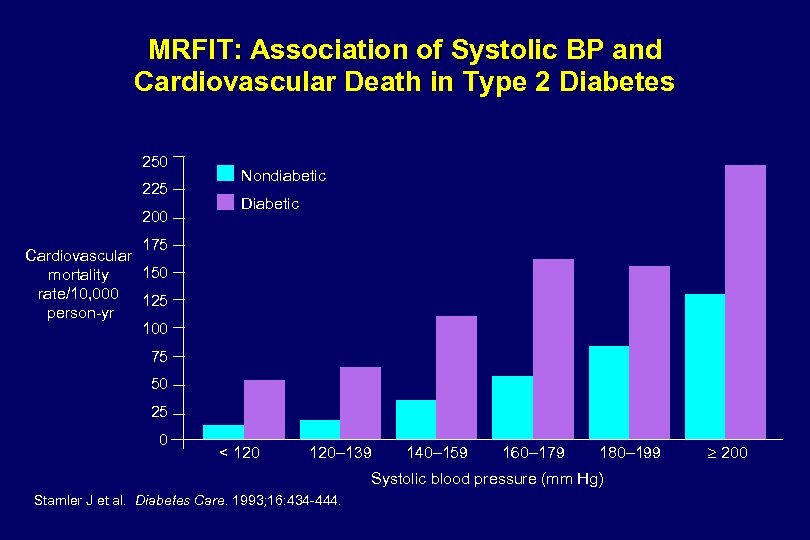 MRFIT: Association of Systolic BP and Cardiovascular Death in Type 2 Diabetes 250 225 200 Nondiabetic Diabetic 175 Cardiovascular 150 mortality rate/10, 000 125 person-yr 100 75 50 25 0 < 120– 139 140– 159 160– 179 180– 199 Systolic blood pressure (mm Hg) Stamler J et al. Diabetes Care. 1993; 16: 434 -444. 200
MRFIT: Association of Systolic BP and Cardiovascular Death in Type 2 Diabetes 250 225 200 Nondiabetic Diabetic 175 Cardiovascular 150 mortality rate/10, 000 125 person-yr 100 75 50 25 0 < 120– 139 140– 159 160– 179 180– 199 Systolic blood pressure (mm Hg) Stamler J et al. Diabetes Care. 1993; 16: 434 -444. 200
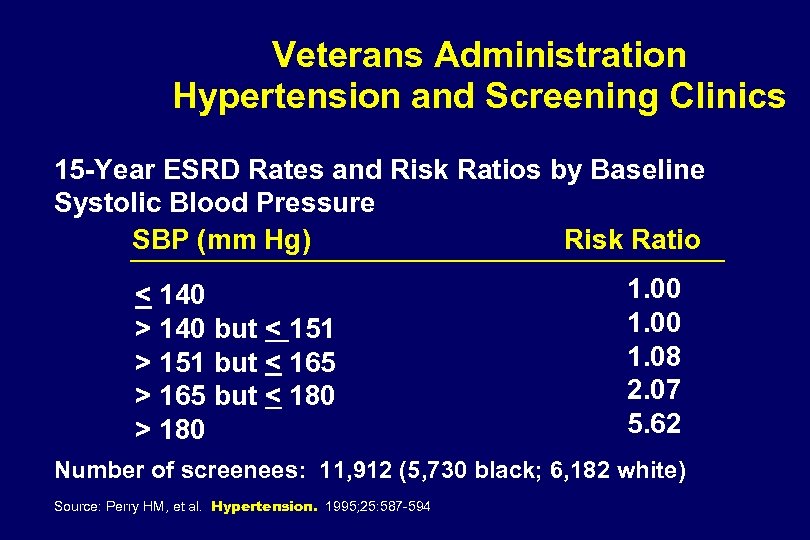 Veterans Administration Hypertension and Screening Clinics 15 -Year ESRD Rates and Risk Ratios by Baseline Systolic Blood Pressure SBP (mm Hg) Risk Ratio < 140 > 140 but < 151 > 151 but < 165 > 165 but < 180 > 180 1. 00 1. 08 2. 07 5. 62 Number of screenees: 11, 912 (5, 730 black; 6, 182 white) Source: Perry HM, et al. Hypertension. 1995; 25: 587 -594
Veterans Administration Hypertension and Screening Clinics 15 -Year ESRD Rates and Risk Ratios by Baseline Systolic Blood Pressure SBP (mm Hg) Risk Ratio < 140 > 140 but < 151 > 151 but < 165 > 165 but < 180 > 180 1. 00 1. 08 2. 07 5. 62 Number of screenees: 11, 912 (5, 730 black; 6, 182 white) Source: Perry HM, et al. Hypertension. 1995; 25: 587 -594
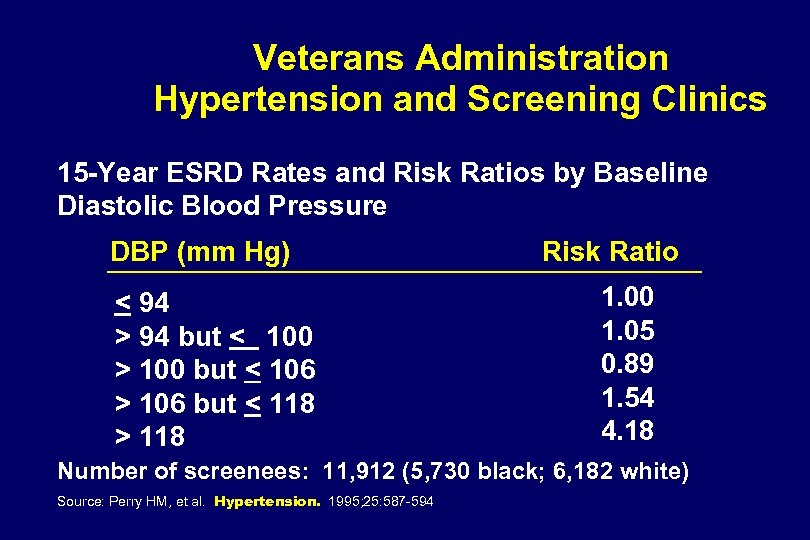 Veterans Administration Hypertension and Screening Clinics 15 -Year ESRD Rates and Risk Ratios by Baseline Diastolic Blood Pressure DBP (mm Hg) < 94 > 94 but < 100 > 100 but < 106 > 106 but < 118 > 118 Risk Ratio 1. 00 1. 05 0. 89 1. 54 4. 18 Number of screenees: 11, 912 (5, 730 black; 6, 182 white) Source: Perry HM, et al. Hypertension. 1995; 25: 587 -594
Veterans Administration Hypertension and Screening Clinics 15 -Year ESRD Rates and Risk Ratios by Baseline Diastolic Blood Pressure DBP (mm Hg) < 94 > 94 but < 100 > 100 but < 106 > 106 but < 118 > 118 Risk Ratio 1. 00 1. 05 0. 89 1. 54 4. 18 Number of screenees: 11, 912 (5, 730 black; 6, 182 white) Source: Perry HM, et al. Hypertension. 1995; 25: 587 -594
 United Kingdom Prospective Diabetes Study (UKPDS): Results • Tight blood pressure control* with captopril- or atenolol-based therapy reduces risk of Risk reduction – Any diabetes-related endpoints 24% – Diabetes-related deaths 32% 0. 019 37% 0. 009 – Stroke 44% 0. 013 – Microvascular endpoints * Mean blood pressure achieved: 144/82 vs 154/87 mm Hg. UK Prospective Diabetes Study Group 38. BMJ. 1998; 317: 703 -713. UK Prospective Diabetes Study Group 33. Lancet. 1998; 352: 837 -853. 0. 005 p-value
United Kingdom Prospective Diabetes Study (UKPDS): Results • Tight blood pressure control* with captopril- or atenolol-based therapy reduces risk of Risk reduction – Any diabetes-related endpoints 24% – Diabetes-related deaths 32% 0. 019 37% 0. 009 – Stroke 44% 0. 013 – Microvascular endpoints * Mean blood pressure achieved: 144/82 vs 154/87 mm Hg. UK Prospective Diabetes Study Group 38. BMJ. 1998; 317: 703 -713. UK Prospective Diabetes Study Group 33. Lancet. 1998; 352: 837 -853. 0. 005 p-value
 Diabetic Nephropathy Burden of Illness Incidence Approximately 40% of all new cases of ESRD in the U. S. are due to diabetes 1 Type 2 diabetes accounts for most cases of diabetic nephropathy 2, 3 Prevalence of nephropathy 57% after 25 years of type 2 diabetes 4 Cost In U. S. alone, total annual spending for ESRD > $15 billion 1 Cost/patient-year higher for diabetic ESRD ($51, 000) than nondiabetic ESRD ($39, 000)5 1. USRDS Coordinating Center. USRDS 1999 Annual Data Report. The Kidney Epidemiology and Cost Center of the University of Michigan; 1999. NIH Contract no. NO 1 -DK-3 -2202. 2. American Diabetes Association. Diabetes Care. 2001; 24 (supp 1): S 69 -72. 3. Ritz E, et al. Am J Kidney Dis. 1996; 27: 167 -194. 4. Bakris GL et al. Am J Kidney Dis. 2000; 36: 646 -661. 5. Ruggenenti P et al. J Am Soc Nephrol. 1998; 9: 2336 -2343.
Diabetic Nephropathy Burden of Illness Incidence Approximately 40% of all new cases of ESRD in the U. S. are due to diabetes 1 Type 2 diabetes accounts for most cases of diabetic nephropathy 2, 3 Prevalence of nephropathy 57% after 25 years of type 2 diabetes 4 Cost In U. S. alone, total annual spending for ESRD > $15 billion 1 Cost/patient-year higher for diabetic ESRD ($51, 000) than nondiabetic ESRD ($39, 000)5 1. USRDS Coordinating Center. USRDS 1999 Annual Data Report. The Kidney Epidemiology and Cost Center of the University of Michigan; 1999. NIH Contract no. NO 1 -DK-3 -2202. 2. American Diabetes Association. Diabetes Care. 2001; 24 (supp 1): S 69 -72. 3. Ritz E, et al. Am J Kidney Dis. 1996; 27: 167 -194. 4. Bakris GL et al. Am J Kidney Dis. 2000; 36: 646 -661. 5. Ruggenenti P et al. J Am Soc Nephrol. 1998; 9: 2336 -2343.
 Diabetic Nephropathy Burden of Illness (continued) Mortality 1. 5 -2. 5 x greater mortality among diabetics with ESRD than nondiabetics 1 < 20% of diabetics with ESRD survive 5 years after initiation of dialysis 1 Cardiovascular complications the most common cause of death 2, 3 1. Koch M et al. Diabetologia. 1993; 36: 1113 -1117. 2. Bakris GL. Diabetes Res Clin Pract. 1998; 39: S 35 -S 42. 3. Grundy SM et al. Circulation. 1999; 100: 1134 -1146.
Diabetic Nephropathy Burden of Illness (continued) Mortality 1. 5 -2. 5 x greater mortality among diabetics with ESRD than nondiabetics 1 < 20% of diabetics with ESRD survive 5 years after initiation of dialysis 1 Cardiovascular complications the most common cause of death 2, 3 1. Koch M et al. Diabetologia. 1993; 36: 1113 -1117. 2. Bakris GL. Diabetes Res Clin Pract. 1998; 39: S 35 -S 42. 3. Grundy SM et al. Circulation. 1999; 100: 1134 -1146.
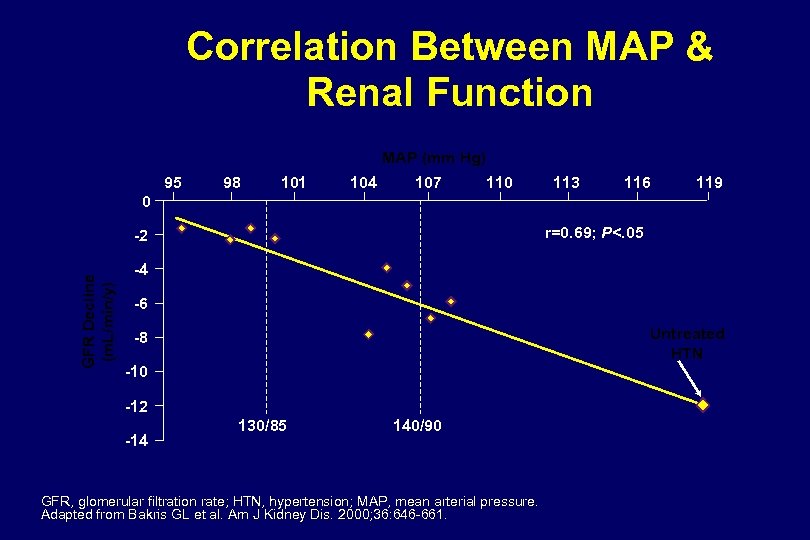 Correlation Between MAP & Renal Function MAP (mm Hg) 95 98 101 104 107 110 113 116 119 0 r=0. 69; P<. 05 GFR Decline (m. L/min/y) -2 -4 -6 Untreated HTN -8 -10 -12 -14 130/85 140/90 GFR, glomerular filtration rate; HTN, hypertension; MAP, mean arterial pressure. Adapted from Bakris GL et al. Am J Kidney Dis. 2000; 36: 646 -661.
Correlation Between MAP & Renal Function MAP (mm Hg) 95 98 101 104 107 110 113 116 119 0 r=0. 69; P<. 05 GFR Decline (m. L/min/y) -2 -4 -6 Untreated HTN -8 -10 -12 -14 130/85 140/90 GFR, glomerular filtration rate; HTN, hypertension; MAP, mean arterial pressure. Adapted from Bakris GL et al. Am J Kidney Dis. 2000; 36: 646 -661.
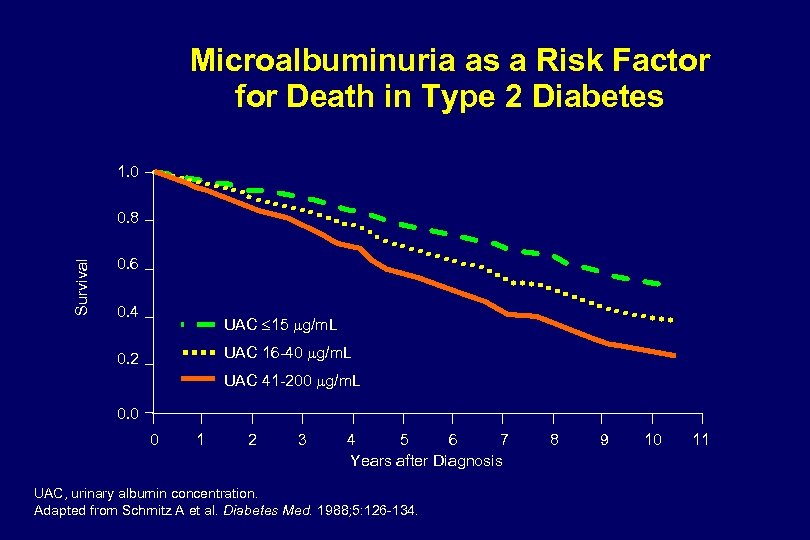 Microalbuminuria as a Risk Factor for Death in Type 2 Diabetes 1. 0 Survival 0. 8 0. 6 0. 4 UAC 15 g/m. L UAC 16 -40 g/m. L 0. 2 UAC 41 -200 g/m. L 0. 0 0 1 2 3 4 5 6 7 Years after Diagnosis UAC, urinary albumin concentration. Adapted from Schmitz A et al. Diabetes Med. 1988; 5: 126 -134. 8 9 10 11
Microalbuminuria as a Risk Factor for Death in Type 2 Diabetes 1. 0 Survival 0. 8 0. 6 0. 4 UAC 15 g/m. L UAC 16 -40 g/m. L 0. 2 UAC 41 -200 g/m. L 0. 0 0 1 2 3 4 5 6 7 Years after Diagnosis UAC, urinary albumin concentration. Adapted from Schmitz A et al. Diabetes Med. 1988; 5: 126 -134. 8 9 10 11
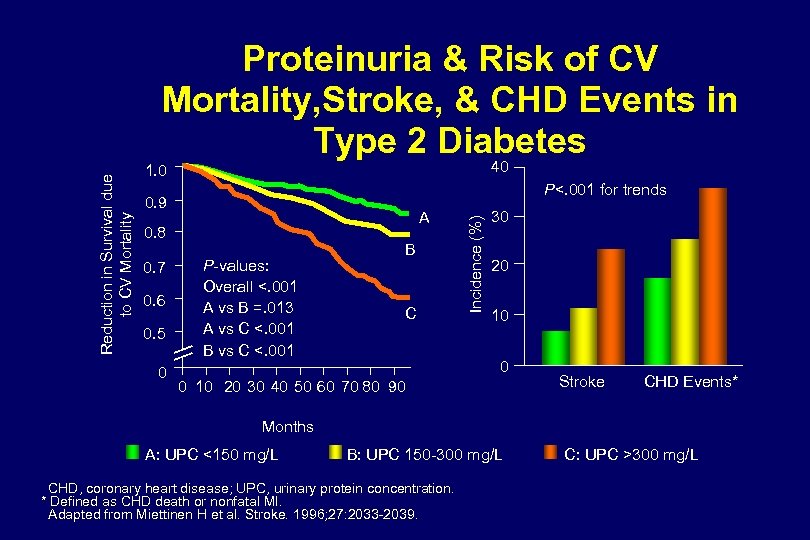 40 1. 0 P<. 001 for trends 0. 9 A 0. 8 0. 7 0. 6 0. 5 0 P-values: Overall <. 001 A vs B =. 013 A vs C <. 001 B C Incidence (%) Reduction in Survival due to CV Mortality Proteinuria & Risk of CV Mortality, Stroke, & CHD Events in Type 2 Diabetes 30 20 10 0 0 10 20 30 40 50 60 70 80 90 Stroke CHD Events* Months A: UPC <150 mg/L B: UPC 150 -300 mg/L CHD, coronary heart disease; UPC, urinary protein concentration. * Defined as CHD death or nonfatal MI. Adapted from Miettinen H et al. Stroke. 1996; 27: 2033 -2039. C: UPC >300 mg/L
40 1. 0 P<. 001 for trends 0. 9 A 0. 8 0. 7 0. 6 0. 5 0 P-values: Overall <. 001 A vs B =. 013 A vs C <. 001 B C Incidence (%) Reduction in Survival due to CV Mortality Proteinuria & Risk of CV Mortality, Stroke, & CHD Events in Type 2 Diabetes 30 20 10 0 0 10 20 30 40 50 60 70 80 90 Stroke CHD Events* Months A: UPC <150 mg/L B: UPC 150 -300 mg/L CHD, coronary heart disease; UPC, urinary protein concentration. * Defined as CHD death or nonfatal MI. Adapted from Miettinen H et al. Stroke. 1996; 27: 2033 -2039. C: UPC >300 mg/L
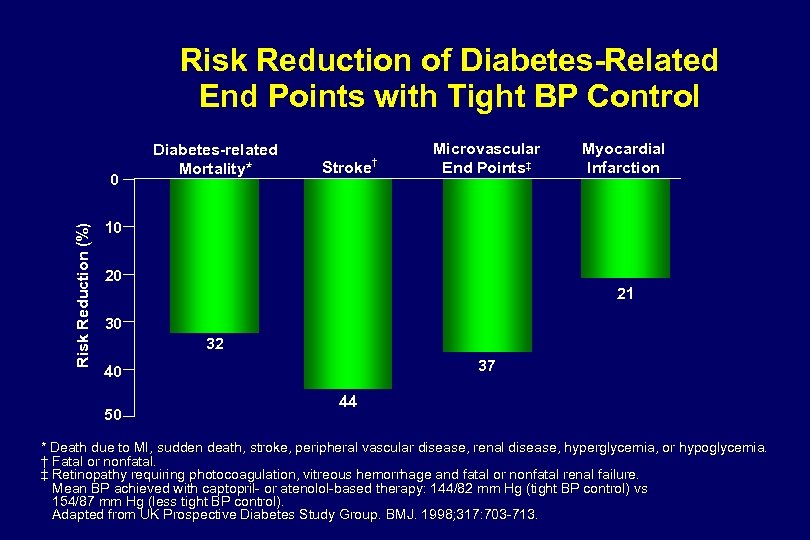 Risk Reduction of Diabetes-Related End Points with Tight BP Control Risk Reduction (%) 0 Diabetes-related Mortality* Stroke † Microvascular End Points‡ Myocardial Infarction 10 20 21 30 32 37 40 50 44 * Death due to MI, sudden death, stroke, peripheral vascular disease, renal disease, hyperglycemia, or hypoglycemia. † Fatal or nonfatal. ‡ Retinopathy requiring photocoagulation, vitreous hemorrhage and fatal or nonfatal renal failure. Mean BP achieved with captopril- or atenolol-based therapy: 144/82 mm Hg (tight BP control) vs 154/87 mm Hg (less tight BP control). Adapted from UK Prospective Diabetes Study Group. BMJ. 1998; 317: 703 -713.
Risk Reduction of Diabetes-Related End Points with Tight BP Control Risk Reduction (%) 0 Diabetes-related Mortality* Stroke † Microvascular End Points‡ Myocardial Infarction 10 20 21 30 32 37 40 50 44 * Death due to MI, sudden death, stroke, peripheral vascular disease, renal disease, hyperglycemia, or hypoglycemia. † Fatal or nonfatal. ‡ Retinopathy requiring photocoagulation, vitreous hemorrhage and fatal or nonfatal renal failure. Mean BP achieved with captopril- or atenolol-based therapy: 144/82 mm Hg (tight BP control) vs 154/87 mm Hg (less tight BP control). Adapted from UK Prospective Diabetes Study Group. BMJ. 1998; 317: 703 -713.
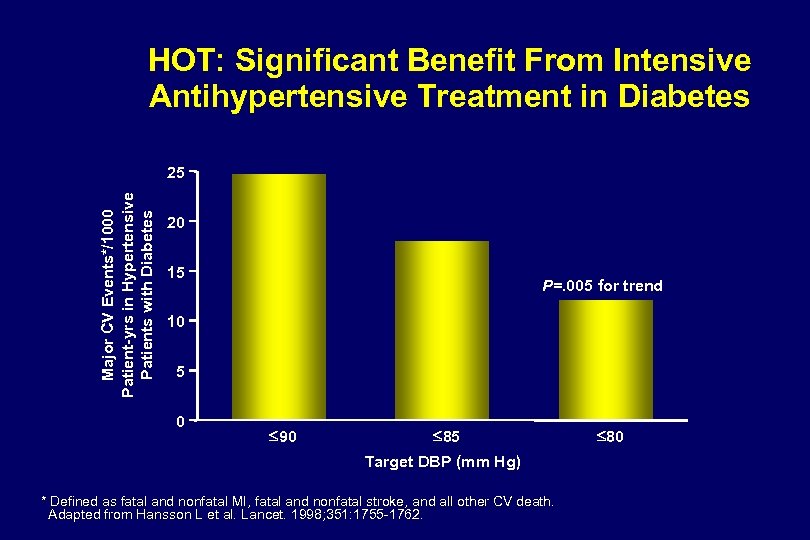 HOT: Significant Benefit From Intensive Antihypertensive Treatment in Diabetes Major CV Events*/1000 Patient-yrs in Hypertensive Patients with Diabetes 25 20 15 P=. 005 for trend 10 5 0 £ 90 £ 85 Target DBP (mm Hg) * Defined as fatal and nonfatal MI, fatal and nonfatal stroke, and all other CV death. Adapted from Hansson L et al. Lancet. 1998; 351: 1755 -1762. £ 80
HOT: Significant Benefit From Intensive Antihypertensive Treatment in Diabetes Major CV Events*/1000 Patient-yrs in Hypertensive Patients with Diabetes 25 20 15 P=. 005 for trend 10 5 0 £ 90 £ 85 Target DBP (mm Hg) * Defined as fatal and nonfatal MI, fatal and nonfatal stroke, and all other CV death. Adapted from Hansson L et al. Lancet. 1998; 351: 1755 -1762. £ 80
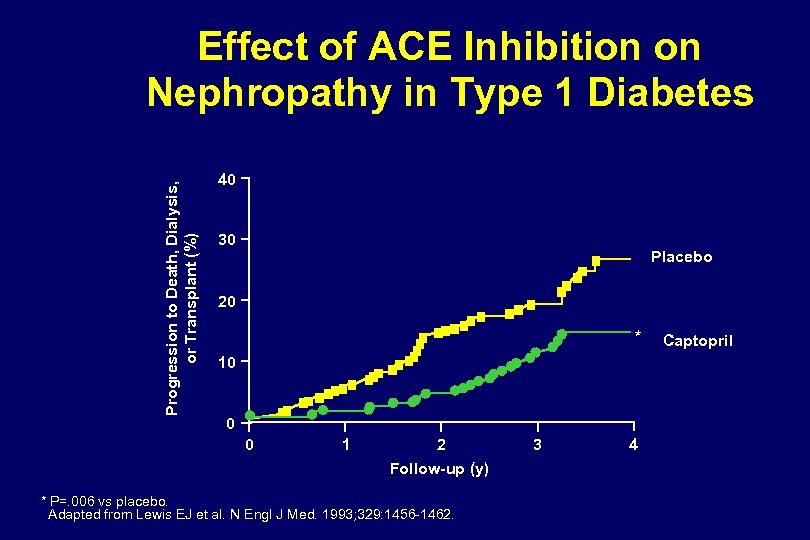 Progression to Death, Dialysis, or Transplant (%) Effect of ACE Inhibition on Nephropathy in Type 1 Diabetes 40 30 Placebo 20 * 10 0 0 1 2 Follow-up (y) * P=. 006 vs placebo. Adapted from Lewis EJ et al. N Engl J Med. 1993; 329: 1456 -1462. 3 4 Captopril
Progression to Death, Dialysis, or Transplant (%) Effect of ACE Inhibition on Nephropathy in Type 1 Diabetes 40 30 Placebo 20 * 10 0 0 1 2 Follow-up (y) * P=. 006 vs placebo. Adapted from Lewis EJ et al. N Engl J Med. 1993; 329: 1456 -1462. 3 4 Captopril
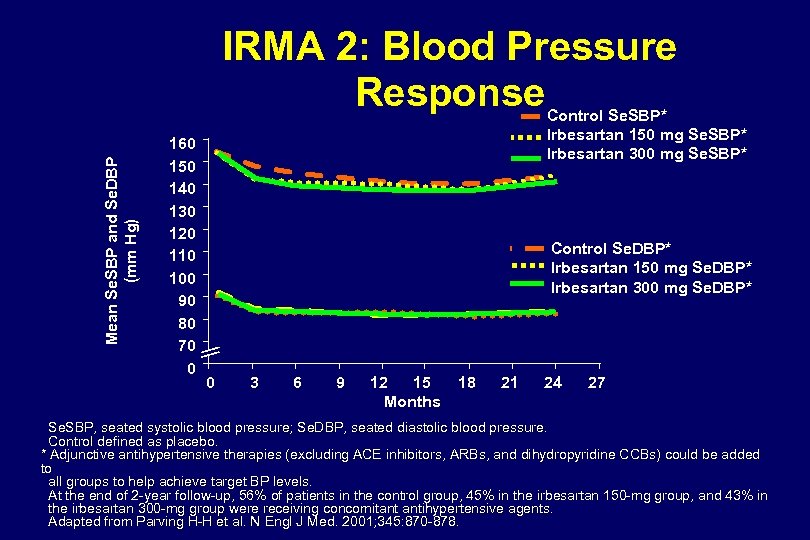 Mean Se. SBP and Se. DBP (mm Hg) IRMA 2: Blood Pressure Response 160 150 140 130 120 110 100 90 80 70 0 Control Se. SBP* Irbesartan 150 mg Se. SBP* Irbesartan 300 mg Se. SBP* Control Se. DBP* Irbesartan 150 mg Se. DBP* Irbesartan 300 mg Se. DBP* 0 3 6 9 12 15 Months 18 21 24 27 Se. SBP, seated systolic blood pressure; Se. DBP, seated diastolic blood pressure. Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. At the end of 2 -year follow-up, 56% of patients in the control group, 45% in the irbesartan 150 -mg group, and 43% in the irbesartan 300 -mg group were receiving concomitant antihypertensive agents. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
Mean Se. SBP and Se. DBP (mm Hg) IRMA 2: Blood Pressure Response 160 150 140 130 120 110 100 90 80 70 0 Control Se. SBP* Irbesartan 150 mg Se. SBP* Irbesartan 300 mg Se. SBP* Control Se. DBP* Irbesartan 150 mg Se. DBP* Irbesartan 300 mg Se. DBP* 0 3 6 9 12 15 Months 18 21 24 27 Se. SBP, seated systolic blood pressure; Se. DBP, seated diastolic blood pressure. Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. At the end of 2 -year follow-up, 56% of patients in the control group, 45% in the irbesartan 150 -mg group, and 43% in the irbesartan 300 -mg group were receiving concomitant antihypertensive agents. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
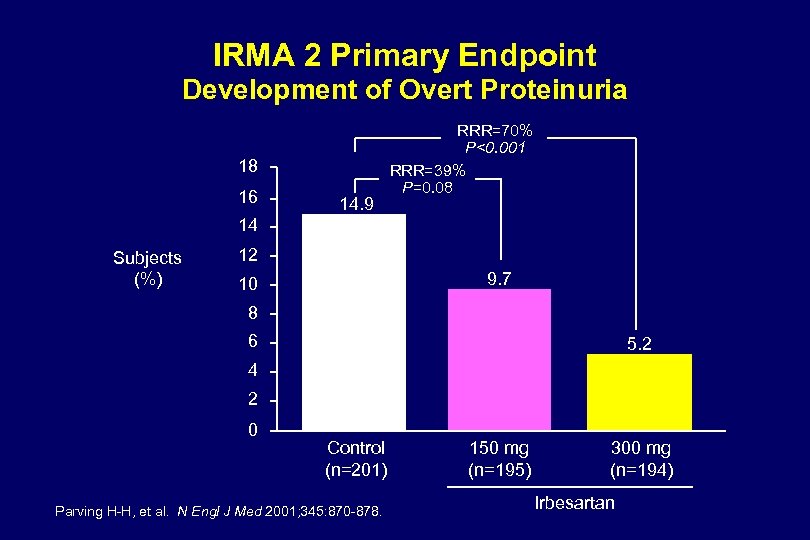 IRMA 2 Primary Endpoint Development of Overt Proteinuria 18 16 14. 9 RRR=70% P<0. 001 RRR=39% P=0. 08 14 Subjects (%) 12 9. 7 10 8 6 5. 2 4 2 0 Control (n=201) Parving H-H, et al. N Engl J Med 2001; 345: 870 -878. 150 mg (n=195) 300 mg (n=194) Irbesartan
IRMA 2 Primary Endpoint Development of Overt Proteinuria 18 16 14. 9 RRR=70% P<0. 001 RRR=39% P=0. 08 14 Subjects (%) 12 9. 7 10 8 6 5. 2 4 2 0 Control (n=201) Parving H-H, et al. N Engl J Med 2001; 345: 870 -878. 150 mg (n=195) 300 mg (n=194) Irbesartan
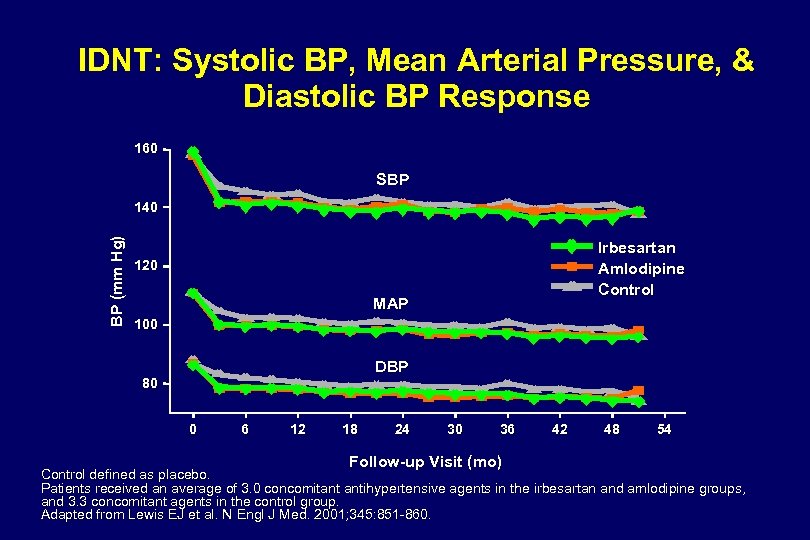 IDNT: Systolic BP, Mean Arterial Pressure, & Diastolic BP Response 160 SBP BP (mm Hg) 140 Irbesartan Amlodipine Control 120 MAP 100 DBP 80 0 6 12 18 24 30 36 Follow-up Visit (mo) 42 48 54 Control defined as placebo. Patients received an average of 3. 0 concomitant antihypertensive agents in the irbesartan and amlodipine groups, and 3. 3 concomitant agents in the control group. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
IDNT: Systolic BP, Mean Arterial Pressure, & Diastolic BP Response 160 SBP BP (mm Hg) 140 Irbesartan Amlodipine Control 120 MAP 100 DBP 80 0 6 12 18 24 30 36 Follow-up Visit (mo) 42 48 54 Control defined as placebo. Patients received an average of 3. 0 concomitant antihypertensive agents in the irbesartan and amlodipine groups, and 3. 3 concomitant agents in the control group. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
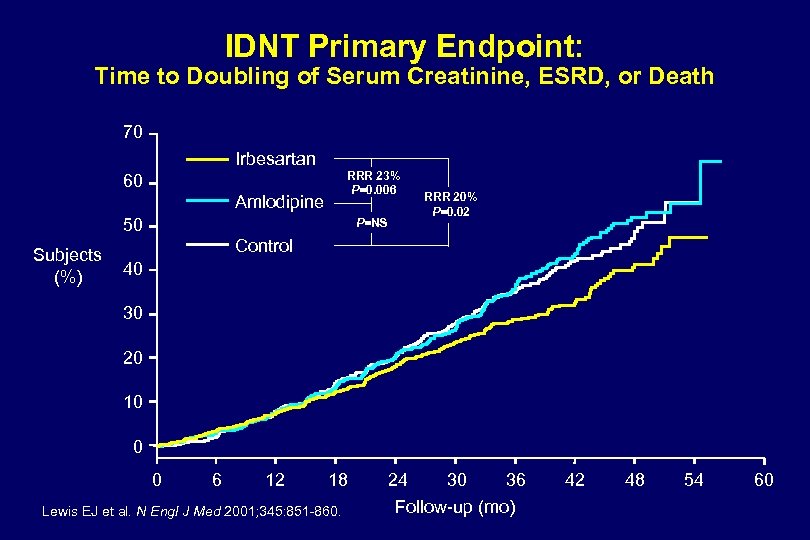 IDNT Primary Endpoint: Time to Doubling of Serum Creatinine, ESRD, or Death 70 Irbesartan RRR 23% P=0. 006 60 Amlodipine 50 P=NS RRR 20% P=0. 02 Control Subjects 40 (%) 30 20 10 0 0 6 12 18 Lewis EJ et al. N Engl J Med 2001; 345: 851 -860. 24 30 36 Follow-up (mo) 42 48 54 60
IDNT Primary Endpoint: Time to Doubling of Serum Creatinine, ESRD, or Death 70 Irbesartan RRR 23% P=0. 006 60 Amlodipine 50 P=NS RRR 20% P=0. 02 Control Subjects 40 (%) 30 20 10 0 0 6 12 18 Lewis EJ et al. N Engl J Med 2001; 345: 851 -860. 24 30 36 Follow-up (mo) 42 48 54 60
 IDNT & RENAAL: Study Design RENAAL‡ IDNT† Patients: & nephropathy Treatment arms: 1, 715 HTN patients with type 2 diabetes & irbesartan, amlodipine, Target BP: Adjunctive therapy: inhibitors, or CCBs Primary outcome: ESRD, or death Secondary outcomes: placebo 135/85 mm Hg Permitted except ARBs, CCBs, except ARBs or Composite of doubling of Se. Cr, ESRD, or death CV events Mean Follow-up: 2. 6 years Se. Cr, serum creatinine; ESRD, end-stage renal disease. † Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. ‡ Brenner BM et al. N Engl J Med. 2001; 345: 861 -869. 1, 513 HTN patients with nephropathy losartan, placebo 140/90 mm Hg Permitted including ACE inhibitors Composite of doubling of CV events 3. 4 years diabetes ACE Se. Cr,
IDNT & RENAAL: Study Design RENAAL‡ IDNT† Patients: & nephropathy Treatment arms: 1, 715 HTN patients with type 2 diabetes & irbesartan, amlodipine, Target BP: Adjunctive therapy: inhibitors, or CCBs Primary outcome: ESRD, or death Secondary outcomes: placebo 135/85 mm Hg Permitted except ARBs, CCBs, except ARBs or Composite of doubling of Se. Cr, ESRD, or death CV events Mean Follow-up: 2. 6 years Se. Cr, serum creatinine; ESRD, end-stage renal disease. † Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. ‡ Brenner BM et al. N Engl J Med. 2001; 345: 861 -869. 1, 513 HTN patients with nephropathy losartan, placebo 140/90 mm Hg Permitted including ACE inhibitors Composite of doubling of CV events 3. 4 years diabetes ACE Se. Cr,
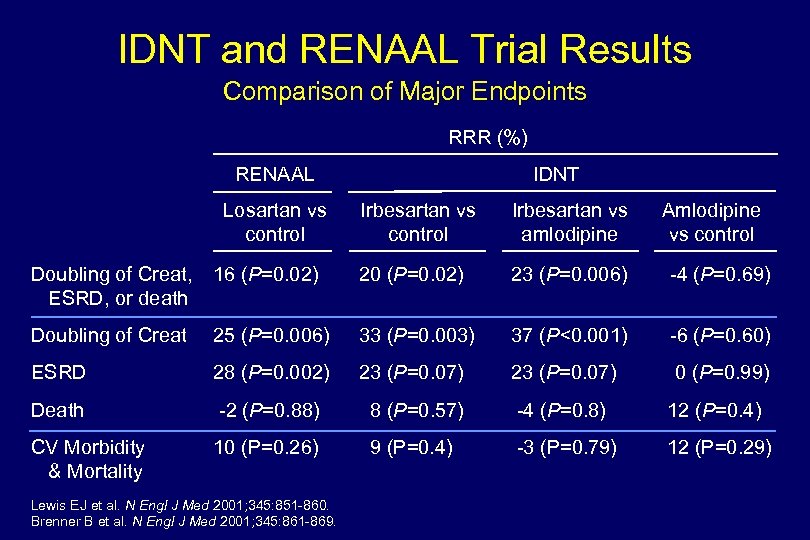 IDNT and RENAAL Trial Results Comparison of Major Endpoints RRR (%) RENAAL Losartan vs control IDNT Irbesartan vs control Doubling of Creat, 16 (P=0. 02) 20 (P=0. 02) ESRD, or death Irbesartan vs amlodipine Amlodipine vs control 23 (P=0. 006) -4 (P=0. 69) Doubling of Creat 25 (P=0. 006) 33 (P=0. 003) 37 (P<0. 001) -6 (P=0. 60) ESRD 28 (P=0. 002) 23 (P=0. 07) 0 (P=0. 99) Death -2 (P=0. 88) 8 (P=0. 57) -4 (P=0. 8) 12 (P=0. 4) CV Morbidity & Mortality 10 (P=0. 26) 9 (P=0. 4) -3 (P=0. 79) 12 (P=0. 29) Lewis EJ et al. N Engl J Med 2001; 345: 851 -860. Brenner B et al. N Engl J Med 2001; 345: 861 -869.
IDNT and RENAAL Trial Results Comparison of Major Endpoints RRR (%) RENAAL Losartan vs control IDNT Irbesartan vs control Doubling of Creat, 16 (P=0. 02) 20 (P=0. 02) ESRD, or death Irbesartan vs amlodipine Amlodipine vs control 23 (P=0. 006) -4 (P=0. 69) Doubling of Creat 25 (P=0. 006) 33 (P=0. 003) 37 (P<0. 001) -6 (P=0. 60) ESRD 28 (P=0. 002) 23 (P=0. 07) 0 (P=0. 99) Death -2 (P=0. 88) 8 (P=0. 57) -4 (P=0. 8) 12 (P=0. 4) CV Morbidity & Mortality 10 (P=0. 26) 9 (P=0. 4) -3 (P=0. 79) 12 (P=0. 29) Lewis EJ et al. N Engl J Med 2001; 345: 851 -860. Brenner B et al. N Engl J Med 2001; 345: 861 -869.
 Minority Populations § In general, treatment similar for all demographic groups. § Socioeconomic factors and lifestyle important barriers to BP control. § Prevalence, severity of HTN increased in African Americans. § African Americans demonstrate somewhat reduced BP responses to monotherapy with BBs, ACEIs, or ARBs compared to diuretics or CCBs.
Minority Populations § In general, treatment similar for all demographic groups. § Socioeconomic factors and lifestyle important barriers to BP control. § Prevalence, severity of HTN increased in African Americans. § African Americans demonstrate somewhat reduced BP responses to monotherapy with BBs, ACEIs, or ARBs compared to diuretics or CCBs.
 Left Ventricular Hypertrophy § LVH is an independent risk factor that increases the risk of CVD. § Regression of LVH occurs with aggressive BP management: weight loss, sodium restriction, and treatment with all classes of drugs except the direct vasodilators hydralazine and minoxidil.
Left Ventricular Hypertrophy § LVH is an independent risk factor that increases the risk of CVD. § Regression of LVH occurs with aggressive BP management: weight loss, sodium restriction, and treatment with all classes of drugs except the direct vasodilators hydralazine and minoxidil.
 Peripheral Arterial Disease (PAD) § PAD is equivalent in risk to ischemic heart disease. § Any class of drugs can be used in most PAD patients. § Other risk factors should be managed aggressively. § Aspirin should be used.
Peripheral Arterial Disease (PAD) § PAD is equivalent in risk to ischemic heart disease. § Any class of drugs can be used in most PAD patients. § Other risk factors should be managed aggressively. § Aspirin should be used.
 Hypertension in Older Persons § More than two-thirds of people over 65 have HTN. § This population has the lowest rates of BP control. § Treatment, including those who with isolated systolic HTN, should follow same principles outlined for general care of HTN. § Lower initial drug doses may be indicated to avoid symptoms; standard doses and multiple drugs will be needed to reach BP targets.
Hypertension in Older Persons § More than two-thirds of people over 65 have HTN. § This population has the lowest rates of BP control. § Treatment, including those who with isolated systolic HTN, should follow same principles outlined for general care of HTN. § Lower initial drug doses may be indicated to avoid symptoms; standard doses and multiple drugs will be needed to reach BP targets.
 Postural Hypotension § Decrease in standing SBP >10 mm. Hg, when associated with dizziness/fainting, more frequent in older SBP patients with diabetes, taking diuretics, venodilators, and some psychotropic drugs. § BP in these individuals should be monitored in the upright position. § Avoid volume depletion and excessively rapid dose titration of drugs.
Postural Hypotension § Decrease in standing SBP >10 mm. Hg, when associated with dizziness/fainting, more frequent in older SBP patients with diabetes, taking diuretics, venodilators, and some psychotropic drugs. § BP in these individuals should be monitored in the upright position. § Avoid volume depletion and excessively rapid dose titration of drugs.
 Dementia § Dementia and cognitive impairment occur more commonly in people with HTN. § Reduced progression of cognitive impairment occurs with effective antihypertensive therapy.
Dementia § Dementia and cognitive impairment occur more commonly in people with HTN. § Reduced progression of cognitive impairment occurs with effective antihypertensive therapy.
 Hypertension in Women § Oral contraceptives may increase BP, and BP should be checked regularly. In contrast, HRT does not raise BP. § Development of HTN—consider other forms of contraception. § Pregnant women with HTN should be followed carefully. Methyldopa, BBs, and vasodilators, preferred for the safety of the fetus. ACEI and ARBs contraindicated in pregnancy.
Hypertension in Women § Oral contraceptives may increase BP, and BP should be checked regularly. In contrast, HRT does not raise BP. § Development of HTN—consider other forms of contraception. § Pregnant women with HTN should be followed carefully. Methyldopa, BBs, and vasodilators, preferred for the safety of the fetus. ACEI and ARBs contraindicated in pregnancy.
 Strategies for Improving Adherence to Regimens § Clinician empathy increases patient trust, motivation, and adherence to therapy. § Physicians should consider their patients’ cultural beliefs and individual attitudes in formulating therapy.
Strategies for Improving Adherence to Regimens § Clinician empathy increases patient trust, motivation, and adherence to therapy. § Physicians should consider their patients’ cultural beliefs and individual attitudes in formulating therapy.
 Causes of Resistant Hypertension § Improper BP measurement § Excess sodium intake § Inadequate diuretic therapy § Medication • Inadequate doses • Drug actions and interactions (e. g. , nonsteroidal antiinflammatory drugs (NSAIDs), illicit drugs, sympathomimetics, oral contraceptives) • Over-the-counter (OTC) drugs and herbal supplements § Excess alcohol intake § Identifiable causes of HTN
Causes of Resistant Hypertension § Improper BP measurement § Excess sodium intake § Inadequate diuretic therapy § Medication • Inadequate doses • Drug actions and interactions (e. g. , nonsteroidal antiinflammatory drugs (NSAIDs), illicit drugs, sympathomimetics, oral contraceptives) • Over-the-counter (OTC) drugs and herbal supplements § Excess alcohol intake § Identifiable causes of HTN
 Public Health Challenges and Community Programs § Public health approaches (e. g. reducing calories, saturated fat, and salt in processed foods and increasing community/school opportunities for physical activity) can achieve a downward shift in the distribution of a population’s BP, thus potentially reducing morbidity, mortality, and the lifetime risk of an individual’s becoming hypertensive. § These public health approaches can provide an attractive opportunity to interrupt and prevent the continuing costly cycle of managing HTN and its complications.
Public Health Challenges and Community Programs § Public health approaches (e. g. reducing calories, saturated fat, and salt in processed foods and increasing community/school opportunities for physical activity) can achieve a downward shift in the distribution of a population’s BP, thus potentially reducing morbidity, mortality, and the lifetime risk of an individual’s becoming hypertensive. § These public health approaches can provide an attractive opportunity to interrupt and prevent the continuing costly cycle of managing HTN and its complications.
 Supporting Materials § Web site www. nhlbi. nih. gov/ § For patients and the general public • “Facts About the DASH Eating Plan” (Revised May 2003) • “Your Guide to Lowering Blood Pressure” § For health professionals • Reference Card • Slide Show
Supporting Materials § Web site www. nhlbi. nih. gov/ § For patients and the general public • “Facts About the DASH Eating Plan” (Revised May 2003) • “Your Guide to Lowering Blood Pressure” § For health professionals • Reference Card • Slide Show
 Back-up Slides: Special Populations • Racial and ethnic groups • Children and adolescents • Women • Older persons
Back-up Slides: Special Populations • Racial and ethnic groups • Children and adolescents • Women • Older persons
 Potential Pathogenic Properties of Angiotensin II § Heart – Myocardial hypertrophy – Interstitial fibrosis § Coronary Arteries – Endothelial dysfunction with decreased release of nitric oxide – Coronary constriction via release of norepinephrine – Formation of oxygen-derived free radicals via NADH (nicotinamide adenine dinucleotide) oxidase – Promotion of inflammatory response and plaque instability – Promotion of low-density lipoprotein cholesterol uptake Adapted from Opie and Gersh. Drugs for the Heart, 2001.
Potential Pathogenic Properties of Angiotensin II § Heart – Myocardial hypertrophy – Interstitial fibrosis § Coronary Arteries – Endothelial dysfunction with decreased release of nitric oxide – Coronary constriction via release of norepinephrine – Formation of oxygen-derived free radicals via NADH (nicotinamide adenine dinucleotide) oxidase – Promotion of inflammatory response and plaque instability – Promotion of low-density lipoprotein cholesterol uptake Adapted from Opie and Gersh. Drugs for the Heart, 2001.
 Potential Pathogenic Properties of Angiotensin II (continued) § Kidneys – – – Increased intraglomerular pressure Increased protein leak Glomerular growth and fibrosis Increased sodium reabsorption Decreased renal blood flow § Adrenal Glands – Increased formation of aldosterone § Coagulation System – Increased fibrinogen – Increased PAI-1 (plasminogen activator inhibitor-1) relative to tissue plasminogen factor Adapted from Opie and Gersh. Drugs for the Heart, 2001.
Potential Pathogenic Properties of Angiotensin II (continued) § Kidneys – – – Increased intraglomerular pressure Increased protein leak Glomerular growth and fibrosis Increased sodium reabsorption Decreased renal blood flow § Adrenal Glands – Increased formation of aldosterone § Coagulation System – Increased fibrinogen – Increased PAI-1 (plasminogen activator inhibitor-1) relative to tissue plasminogen factor Adapted from Opie and Gersh. Drugs for the Heart, 2001.
 Summary of Chapter 3 (continued) • Management strategies can improve adherence through the use of multidisciplinary teams. • The reductions in cardiovascular events demonstrated in randomized controlled trials have important implications for managed care organizations. • Management of hypertensive emergencies requires immediate action, whereas urgencies benefit from reducing blood pressure within a few hours.
Summary of Chapter 3 (continued) • Management strategies can improve adherence through the use of multidisciplinary teams. • The reductions in cardiovascular events demonstrated in randomized controlled trials have important implications for managed care organizations. • Management of hypertensive emergencies requires immediate action, whereas urgencies benefit from reducing blood pressure within a few hours.
 Drug Therapy • A low dose of initial drug should be used, slowly titrating upward. • Optimal formulation should provide 24 -hour efficacy with once-daily dose with at least 50% of peak effect remaining at end of 24 hours. • Combination therapies may provide additional efficacy with fewer adverse effects.
Drug Therapy • A low dose of initial drug should be used, slowly titrating upward. • Optimal formulation should provide 24 -hour efficacy with once-daily dose with at least 50% of peak effect remaining at end of 24 hours. • Combination therapies may provide additional efficacy with fewer adverse effects.
 Classes of Antihypertensive Drugs • ACE inhibitors • Adrenergic inhibitors • Angiotensin II receptor blockers • Calcium antagonists • Direct vasodilators • Diuretics
Classes of Antihypertensive Drugs • ACE inhibitors • Adrenergic inhibitors • Angiotensin II receptor blockers • Calcium antagonists • Direct vasodilators • Diuretics
 Combination Therapies • -adrenergic blockers and diuretics • ACE inhibitors and diuretics • Angiotensin II receptor antagonists and diuretics • Calcium antagonists and ACE inhibitors • Other combinations
Combination Therapies • -adrenergic blockers and diuretics • ACE inhibitors and diuretics • Angiotensin II receptor antagonists and diuretics • Calcium antagonists and ACE inhibitors • Other combinations
 Followup • Followup within 1 to 2 months after initiating therapy. • Recognize that high-risk patients often require high dose or combination therapies and shorter intervals between changes in medications. • Consider reasons for lack of responsiveness if blood pressure is uncontrolled after reaching full dose. • Consider reducing dose and number of agents after 1 year at or below goal.
Followup • Followup within 1 to 2 months after initiating therapy. • Recognize that high-risk patients often require high dose or combination therapies and shorter intervals between changes in medications. • Consider reasons for lack of responsiveness if blood pressure is uncontrolled after reaching full dose. • Consider reducing dose and number of agents after 1 year at or below goal.
 Guidelines for Improving Adherence to Therapy • Be aware of signs of nonadherence. • Establish goal of therapy. • Encourage a positive attitude about achieving goals. • Educate patients about the disease and therapy. • Maintain contact with patients. • Encourage lifestyle modifications. • Keep care inexpensive and simple.
Guidelines for Improving Adherence to Therapy • Be aware of signs of nonadherence. • Establish goal of therapy. • Encourage a positive attitude about achieving goals. • Educate patients about the disease and therapy. • Maintain contact with patients. • Encourage lifestyle modifications. • Keep care inexpensive and simple.
 Guidelines for Improving Adherence to Therapy (continued) • Integrate therapy into daily routine. • Prescribe long-acting drugs. • Adjust therapy to minimize adverse effects. • Continue to add drugs systematically to meet goal. • Consider using nurse case management. • Utilize other health professionals. • Try a new approach if current regime is inadequate.
Guidelines for Improving Adherence to Therapy (continued) • Integrate therapy into daily routine. • Prescribe long-acting drugs. • Adjust therapy to minimize adverse effects. • Continue to add drugs systematically to meet goal. • Consider using nurse case management. • Utilize other health professionals. • Try a new approach if current regime is inadequate.
 Racial and Ethnic Groups African Americans • Among the highest prevalence • Early onset • Delayed treatment Hispanics • Generally low prevalence • Lowest control rate in Mexican Americans Asian and Pacific Islanders • May be more responsive to treatment than other groups American Indians • Similar prevalence to general population • High prevalence of diabetes and obesity
Racial and Ethnic Groups African Americans • Among the highest prevalence • Early onset • Delayed treatment Hispanics • Generally low prevalence • Lowest control rate in Mexican Americans Asian and Pacific Islanders • May be more responsive to treatment than other groups American Indians • Similar prevalence to general population • High prevalence of diabetes and obesity
 Women • Clinical trials have not demonstrated significant differences between men and women in treatment response and outcomes. • Some women using oral contraceptives may have significant increases in blood pressure. • High blood pressure is not a contraindication to hormone replacement therapy.
Women • Clinical trials have not demonstrated significant differences between men and women in treatment response and outcomes. • Some women using oral contraceptives may have significant increases in blood pressure. • High blood pressure is not a contraindication to hormone replacement therapy.
 Pregnant Women • Chronic hypertension is high blood pressure present before pregnancy or diagnosed before the 20 th week of gestation. • Preeclampsia is increased blood pressure that occurs in pregnancy (generally after the 20 th week) and is accompanied by edema, proteinuria, or both. • ACE inhibitors and angiotensin II receptor blockers are contraindicated for pregnant women. • Methyldopa is recommended for women diagnosed during pregnancy.
Pregnant Women • Chronic hypertension is high blood pressure present before pregnancy or diagnosed before the 20 th week of gestation. • Preeclampsia is increased blood pressure that occurs in pregnancy (generally after the 20 th week) and is accompanied by edema, proteinuria, or both. • ACE inhibitors and angiotensin II receptor blockers are contraindicated for pregnant women. • Methyldopa is recommended for women diagnosed during pregnancy.
 Antihypertensive Drugs Used in Pregnancy These agents* may be used with chronic hypertension (DBP > 100 mm Hg) or acute hypertension (DBP > 105 mm Hg). Central -agonists Methyldopa is the drug of choice. -blockers and - -blockers Atenolol, metoprolol, and labetalol appear safe and effective in late pregnancy. Calcium antagonists Potential synergism with magnesium sulfate may lead to precipitous hypotension. *Limited or no controlled trials in pregnant women.
Antihypertensive Drugs Used in Pregnancy These agents* may be used with chronic hypertension (DBP > 100 mm Hg) or acute hypertension (DBP > 105 mm Hg). Central -agonists Methyldopa is the drug of choice. -blockers and - -blockers Atenolol, metoprolol, and labetalol appear safe and effective in late pregnancy. Calcium antagonists Potential synergism with magnesium sulfate may lead to precipitous hypotension. *Limited or no controlled trials in pregnant women.
 Antihypertensive Drugs Used in Pregnancy (continued) These agents* may be used with chronic hypertension (DBP > 100 mm Hg) or acute hypertension (DBP > 105). Diuretics are recommended for chronic hypertension if prescribed before gestation, but they are not recommended for preeclampsia. Direct vasodilators Hydralazine is the parenteral drug of choice based on its long history of safety and efficacy. *Limited or no controlled trials in pregnant women. ACE inhibitors and angiotensin II receptor blockers are contraindicated.
Antihypertensive Drugs Used in Pregnancy (continued) These agents* may be used with chronic hypertension (DBP > 100 mm Hg) or acute hypertension (DBP > 105). Diuretics are recommended for chronic hypertension if prescribed before gestation, but they are not recommended for preeclampsia. Direct vasodilators Hydralazine is the parenteral drug of choice based on its long history of safety and efficacy. *Limited or no controlled trials in pregnant women. ACE inhibitors and angiotensin II receptor blockers are contraindicated.
 Older Persons • Hypertension is common. • SBP is a better predictor of events than DBP. • Pseudohypertension and “white-coat hypertension” may indicate a need for readings outside the office. • Primary hypertension is the most common cause, but common identifiable causes (e. g. , renovascular hypertension) should be considered.
Older Persons • Hypertension is common. • SBP is a better predictor of events than DBP. • Pseudohypertension and “white-coat hypertension” may indicate a need for readings outside the office. • Primary hypertension is the most common cause, but common identifiable causes (e. g. , renovascular hypertension) should be considered.
 Older Persons (continued) • Therapy should begin with lifestyle modifications. • Starting doses for drug therapy should be lower than those used in younger adults. • Goal of therapy is the same (< 140/90 mm Hg), although an interim goal of SBP < 160 mm Hg may be necessary.
Older Persons (continued) • Therapy should begin with lifestyle modifications. • Starting doses for drug therapy should be lower than those used in younger adults. • Goal of therapy is the same (< 140/90 mm Hg), although an interim goal of SBP < 160 mm Hg may be necessary.
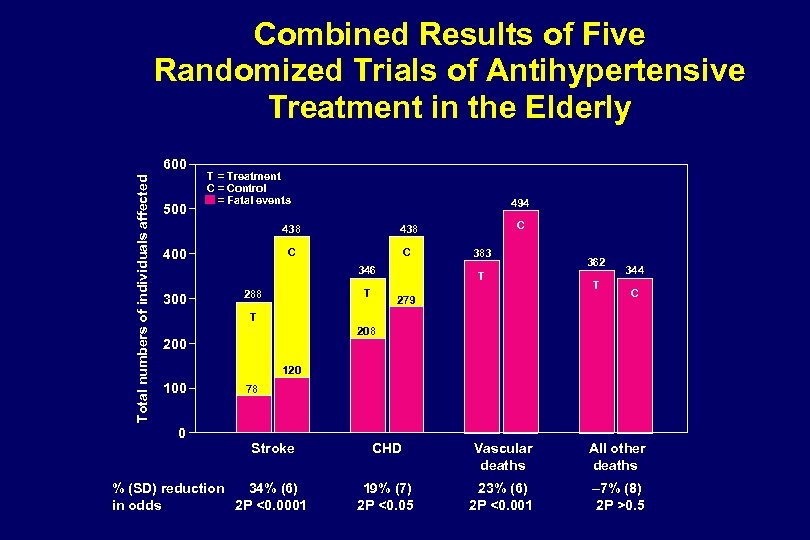 Combined Results of Five Randomized Trials of Antihypertensive Treatment in the Elderly Total numbers of individuals affected 600 500 T = Treatment C = Control = Fatal events 494 438 C 400 C 346 300 C 438 T 288 383 T 279 362 T 344 C T 208 200 120 100 0 78 Stroke % (SD) reduction 34% (6) in odds 2 P <0. 0001 CHD Vascular deaths All other deaths 19% (7) 2 P <0. 05 23% (6) 2 P <0. 001 – 7% (8) 2 P >0. 5
Combined Results of Five Randomized Trials of Antihypertensive Treatment in the Elderly Total numbers of individuals affected 600 500 T = Treatment C = Control = Fatal events 494 438 C 400 C 346 300 C 438 T 288 383 T 279 362 T 344 C T 208 200 120 100 0 78 Stroke % (SD) reduction 34% (6) in odds 2 P <0. 0001 CHD Vascular deaths All other deaths 19% (7) 2 P <0. 05 23% (6) 2 P <0. 001 – 7% (8) 2 P >0. 5
 Effects of Therapy in Elderly Hypertensive Patients SHEP (1991) Mean BP at entry (mm Hg) STOP-HTN (Dahlöf, 1991) MRC (1992) SYST-EUR (1997) 170/77 195/102 185/91 174/85
Effects of Therapy in Elderly Hypertensive Patients SHEP (1991) Mean BP at entry (mm Hg) STOP-HTN (Dahlöf, 1991) MRC (1992) SYST-EUR (1997) 170/77 195/102 185/91 174/85
 IRMA 2: Study Design • 590 patients with hypertension, type 2 diabetes, microalbuminuria (albumin excretion rate 20– 200 µg/min), and normal renal function Double-blind Treatment Placebo/Control group* Screening/Enrollment Up to 5 weeks Irbesartan 150 mg* Irbesartan 300 mg* Follow-up: 2 years * Adjunctive antihypertensive therapies (excluding ACE inhibitors, angiotensin II receptor antagonists, and dihydropyridine calcium channel blockers) could be added to all groups to help achieve equal blood pressure levels. Parving H-H, et al. N Engl J Med 2001; 345: 870 -878.
IRMA 2: Study Design • 590 patients with hypertension, type 2 diabetes, microalbuminuria (albumin excretion rate 20– 200 µg/min), and normal renal function Double-blind Treatment Placebo/Control group* Screening/Enrollment Up to 5 weeks Irbesartan 150 mg* Irbesartan 300 mg* Follow-up: 2 years * Adjunctive antihypertensive therapies (excluding ACE inhibitors, angiotensin II receptor antagonists, and dihydropyridine calcium channel blockers) could be added to all groups to help achieve equal blood pressure levels. Parving H-H, et al. N Engl J Med 2001; 345: 870 -878.
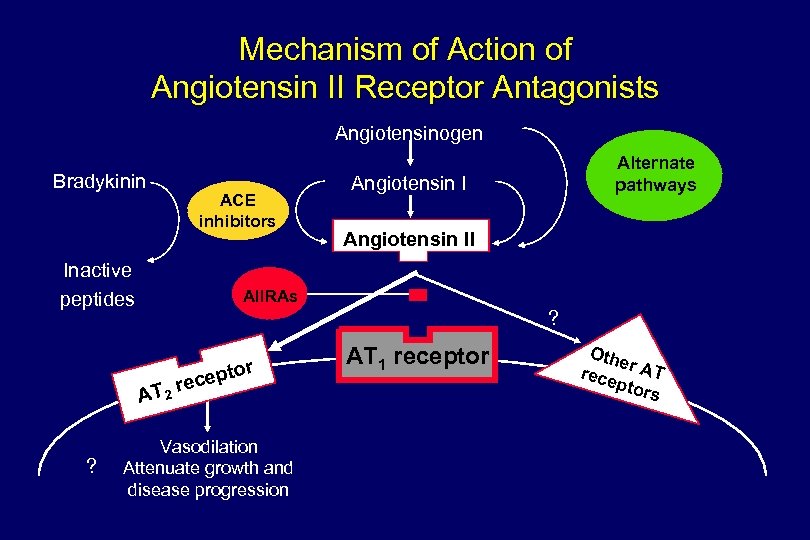 Mechanism of Action of Angiotensin II Receptor Antagonists Angiotensinogen Bradykinin Inactive peptides ACE inhibitors Angiotensin II AIIRAs ? or cept re AT 2 ? Alternate pathways Vasodilation Attenuate growth and disease progression AT 1 receptor Oth e rece r AT ptor s
Mechanism of Action of Angiotensin II Receptor Antagonists Angiotensinogen Bradykinin Inactive peptides ACE inhibitors Angiotensin II AIIRAs ? or cept re AT 2 ? Alternate pathways Vasodilation Attenuate growth and disease progression AT 1 receptor Oth e rece r AT ptor s
 IRMA 2: Clinical Outcome Measures • Primary outcome: – Time to occurrence of overt proteinuria (AER > 200 g/min) • Secondary outcomes: – Change in AER – Regression to normoalbuminuria (AER < 20 g/min) – Change in creatinine clearance – Clotting factors and lipid profile Parving H-H, et al. N Engl J Med 2001; 345: 870 -878.
IRMA 2: Clinical Outcome Measures • Primary outcome: – Time to occurrence of overt proteinuria (AER > 200 g/min) • Secondary outcomes: – Change in AER – Regression to normoalbuminuria (AER < 20 g/min) – Change in creatinine clearance – Clotting factors and lipid profile Parving H-H, et al. N Engl J Med 2001; 345: 870 -878.
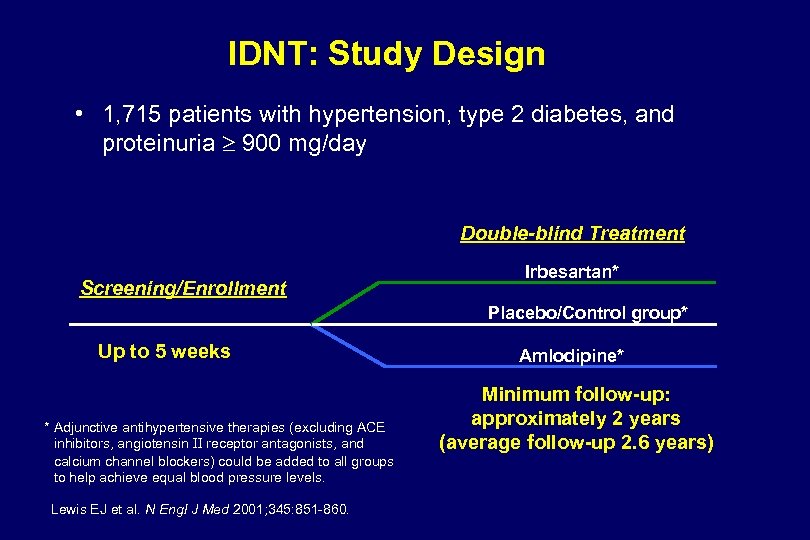 IDNT: Study Design • 1, 715 patients with hypertension, type 2 diabetes, and proteinuria 900 mg/day Double-blind Treatment Screening/Enrollment Irbesartan* Placebo/Control group* Up to 5 weeks * Adjunctive antihypertensive therapies (excluding ACE inhibitors, angiotensin II receptor antagonists, and calcium channel blockers) could be added to all groups to help achieve equal blood pressure levels. Lewis EJ et al. N Engl J Med 2001; 345: 851 -860. Amlodipine* Minimum follow-up: approximately 2 years (average follow-up 2. 6 years)
IDNT: Study Design • 1, 715 patients with hypertension, type 2 diabetes, and proteinuria 900 mg/day Double-blind Treatment Screening/Enrollment Irbesartan* Placebo/Control group* Up to 5 weeks * Adjunctive antihypertensive therapies (excluding ACE inhibitors, angiotensin II receptor antagonists, and calcium channel blockers) could be added to all groups to help achieve equal blood pressure levels. Lewis EJ et al. N Engl J Med 2001; 345: 851 -860. Amlodipine* Minimum follow-up: approximately 2 years (average follow-up 2. 6 years)
 IDNT Clinical Outcome Measures • Primary outcome is time to a composite endpoint consisting of: – Doubling of baseline serum creatinine – End-stage renal disease (dialysis, renal transplant, or serum creatinine 6 mg/d. L) – Death (all-cause mortality) • Secondary outcome is time to a composite endpoint of fatal or nonfatal cardiovascular events Lewis EJ et al. N Engl J Med 2001; 345: 851 -860.
IDNT Clinical Outcome Measures • Primary outcome is time to a composite endpoint consisting of: – Doubling of baseline serum creatinine – End-stage renal disease (dialysis, renal transplant, or serum creatinine 6 mg/d. L) – Death (all-cause mortality) • Secondary outcome is time to a composite endpoint of fatal or nonfatal cardiovascular events Lewis EJ et al. N Engl J Med 2001; 345: 851 -860.
 Potential Pathogenic Properties of Angiotensin II Kidneys Increased intraglomerular pressure Increased protein leak Glomerular growth and fibrosis Increased sodium reabsorption Decreased renal blood flow Adapted from Opie and Gersh. Drugs for the Heart, 2001.
Potential Pathogenic Properties of Angiotensin II Kidneys Increased intraglomerular pressure Increased protein leak Glomerular growth and fibrosis Increased sodium reabsorption Decreased renal blood flow Adapted from Opie and Gersh. Drugs for the Heart, 2001.
 IRMA 2: Study Design § 590 patients with hypertension, type 2 diabetes, microalbuminuria, * and normal renal function Double-blind treatment Screening/enrollment Control† Irbesartan 150 mg/d† Irbesartan 300 mg/d† 3 weeks Follow-up: 2 years Control defined as placebo. * Defined as albumin excretion rate 20 -200 µg/min. † Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
IRMA 2: Study Design § 590 patients with hypertension, type 2 diabetes, microalbuminuria, * and normal renal function Double-blind treatment Screening/enrollment Control† Irbesartan 150 mg/d† Irbesartan 300 mg/d† 3 weeks Follow-up: 2 years Control defined as placebo. * Defined as albumin excretion rate 20 -200 µg/min. † Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
 IRMA 2: Clinical Outcome Measures Primary outcome: Time to occurrence of overt proteinuria (UAER >200 mg/min) Secondary outcomes: Change in UAER Regression to normoalbuminuria (UAER <20 mg/min) Change in creatinine clearance UAER, urinary albumin excretion rate. Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
IRMA 2: Clinical Outcome Measures Primary outcome: Time to occurrence of overt proteinuria (UAER >200 mg/min) Secondary outcomes: Change in UAER Regression to normoalbuminuria (UAER <20 mg/min) Change in creatinine clearance UAER, urinary albumin excretion rate. Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
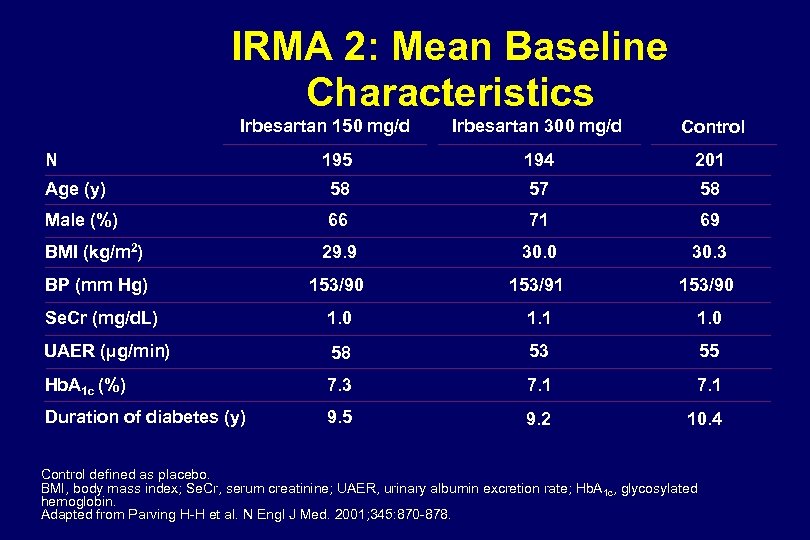 IRMA 2: Mean Baseline Characteristics Irbesartan 150 mg/d Irbesartan 300 mg/d Control 195 194 201 Age (y) 58 57 58 Male (%) 66 71 69 BMI (kg/m 2) 29. 9 30. 0 30. 3 BP (mm Hg) 153/90 153/91 153/90 Se. Cr (mg/d. L) 1. 0 1. 1 1. 0 UAER (µg/min) 58 53 55 Hb. A 1 c (%) 7. 3 7. 1 Duration of diabetes (y) 9. 5 9. 2 10. 4 N Control defined as placebo. BMI, body mass index; Se. Cr, serum creatinine; UAER, urinary albumin excretion rate; Hb. A 1 c, glycosylated hemoglobin. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
IRMA 2: Mean Baseline Characteristics Irbesartan 150 mg/d Irbesartan 300 mg/d Control 195 194 201 Age (y) 58 57 58 Male (%) 66 71 69 BMI (kg/m 2) 29. 9 30. 0 30. 3 BP (mm Hg) 153/90 153/91 153/90 Se. Cr (mg/d. L) 1. 0 1. 1 1. 0 UAER (µg/min) 58 53 55 Hb. A 1 c (%) 7. 3 7. 1 Duration of diabetes (y) 9. 5 9. 2 10. 4 N Control defined as placebo. BMI, body mass index; Se. Cr, serum creatinine; UAER, urinary albumin excretion rate; Hb. A 1 c, glycosylated hemoglobin. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
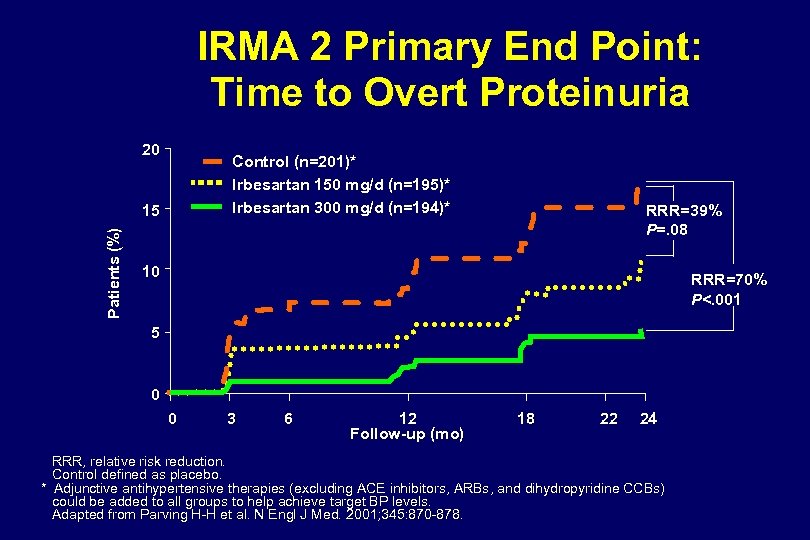 IRMA 2 Primary End Point: Time to Overt Proteinuria 20 Control (n=201)* Irbesartan 150 mg/d (n=195)* Irbesartan 300 mg/d (n=194)* Patients (%) 15 RRR=39% P=. 08 10 RRR=70% P<. 001 5 0 0 3 6 12 Follow-up (mo) 18 22 24 RRR, relative risk reduction. Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
IRMA 2 Primary End Point: Time to Overt Proteinuria 20 Control (n=201)* Irbesartan 150 mg/d (n=195)* Irbesartan 300 mg/d (n=194)* Patients (%) 15 RRR=39% P=. 08 10 RRR=70% P<. 001 5 0 0 3 6 12 Follow-up (mo) 18 22 24 RRR, relative risk reduction. Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. Adapted from Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
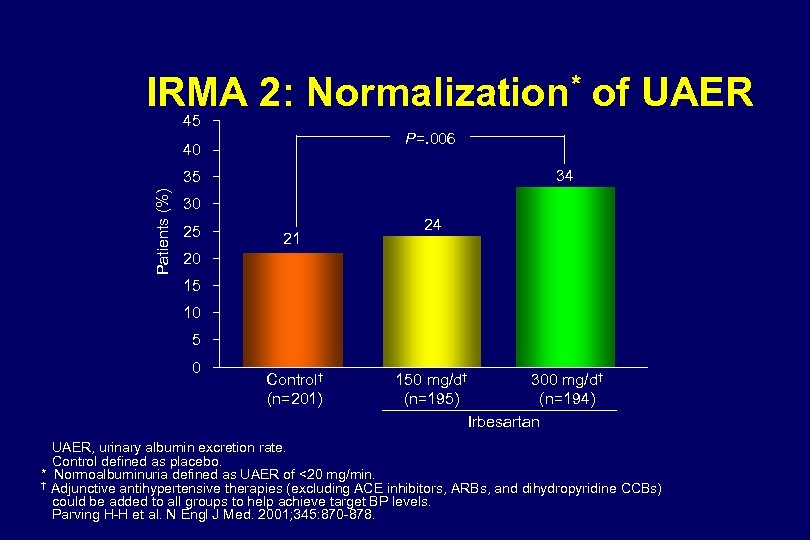 IRMA 2: Normalization* of UAER 45 P=. 006 40 34 Patients (%) 35 30 25 21 24 20 15 10 5 0 Control† (n=201) 150 mg/d† 300 mg/d† (n=195) (n=194) Irbesartan UAER, urinary albumin excretion rate. Control defined as placebo. * Normoalbuminuria defined as UAER of <20 mg/min. † Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
IRMA 2: Normalization* of UAER 45 P=. 006 40 34 Patients (%) 35 30 25 21 24 20 15 10 5 0 Control† (n=201) 150 mg/d† 300 mg/d† (n=195) (n=194) Irbesartan UAER, urinary albumin excretion rate. Control defined as placebo. * Normoalbuminuria defined as UAER of <20 mg/min. † Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. Parving H-H et al. N Engl J Med. 2001; 345: 870 -878.
 IRMA 2: Adverse Events No. of Adverse Events (%) Control group* (n=201) Irbesartan 150 mg* (n=195) 300 mg* (n=194) Cardiovascular events 18 (8. 7)1 14 (6. 9)2 9 (4. 5)1 Serious AE 47 (22. 8)1 32 (15. 8)2 30 (15. 0)2 18 (8. 9)2 11 (5. 5)2 Discontinuations due to AE 19 (9. 2)2 Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. 1. Parving H-H, et al. N Engl J Med. 2001; 345: 870 -878. 2. Data on file, Bristol-Myers Squibb and Sanofi-Synthelabo, Inc.
IRMA 2: Adverse Events No. of Adverse Events (%) Control group* (n=201) Irbesartan 150 mg* (n=195) 300 mg* (n=194) Cardiovascular events 18 (8. 7)1 14 (6. 9)2 9 (4. 5)1 Serious AE 47 (22. 8)1 32 (15. 8)2 30 (15. 0)2 18 (8. 9)2 11 (5. 5)2 Discontinuations due to AE 19 (9. 2)2 Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and dihydropyridine CCBs) could be added to all groups to help achieve target BP levels. 1. Parving H-H, et al. N Engl J Med. 2001; 345: 870 -878. 2. Data on file, Bristol-Myers Squibb and Sanofi-Synthelabo, Inc.
 IRMA 2: Summary The renal benefits of irbesartan are independent of its BP-lowering effects 1 70% risk reduction in the progression from microalbuminuria to overt diabetic nephropathy with irbesartan 300 mg/d vs control (P<. 001)1 More frequent restoration of normoalbuminuria with irbesartan 300 mg/d vs control (P=. 006)1 Irbesartan is safe & well tolerated 2 Fewer nonfatal CV events, serious AEs, and AE, adverse event. 1. Parving H-H et al. N Engl J Med. 2001; 345: 870 -878. discontinuations due to AEs in the irbesartan groups 2 2. Data on file, Bristol-Myers Squibb and Sanofi-Synthelabo, Inc.
IRMA 2: Summary The renal benefits of irbesartan are independent of its BP-lowering effects 1 70% risk reduction in the progression from microalbuminuria to overt diabetic nephropathy with irbesartan 300 mg/d vs control (P<. 001)1 More frequent restoration of normoalbuminuria with irbesartan 300 mg/d vs control (P=. 006)1 Irbesartan is safe & well tolerated 2 Fewer nonfatal CV events, serious AEs, and AE, adverse event. 1. Parving H-H et al. N Engl J Med. 2001; 345: 870 -878. discontinuations due to AEs in the irbesartan groups 2 2. Data on file, Bristol-Myers Squibb and Sanofi-Synthelabo, Inc.
 IDNT: Study Design 1, 715 patients with hypertension, type 2 diabetes, & proteinuria ³ 900 mg/d Double-blind treatment Screening/enrollment Irbesartan* Control* Amlodipine* Up to 5 weeks Minimum follow-up: approximately 2 years (average follow-up, 2. 6 years) Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and CCBs) could be added to all groups to help achieve target BP. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
IDNT: Study Design 1, 715 patients with hypertension, type 2 diabetes, & proteinuria ³ 900 mg/d Double-blind treatment Screening/enrollment Irbesartan* Control* Amlodipine* Up to 5 weeks Minimum follow-up: approximately 2 years (average follow-up, 2. 6 years) Control defined as placebo. * Adjunctive antihypertensive therapies (excluding ACE inhibitors, ARBs, and CCBs) could be added to all groups to help achieve target BP. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
 IDNT: Clinical Outcome Measures Primary outcome: time to composite end point of Doubling of baseline Se. Cr ESRD (dialysis, renal transplant, or Se. Cr ³ 6 mg/d. L) Death (all-cause mortality) Secondary outcome: time to composite end point of Se. Cr, serum creatinine; ESRD, end-stage renal disease. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. fatal or nonfatal CV events
IDNT: Clinical Outcome Measures Primary outcome: time to composite end point of Doubling of baseline Se. Cr ESRD (dialysis, renal transplant, or Se. Cr ³ 6 mg/d. L) Death (all-cause mortality) Secondary outcome: time to composite end point of Se. Cr, serum creatinine; ESRD, end-stage renal disease. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. fatal or nonfatal CV events
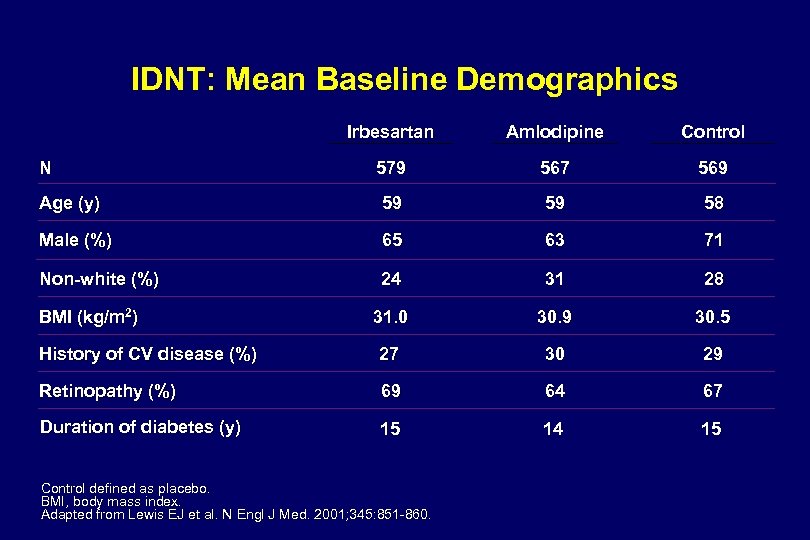 IDNT: Mean Baseline Demographics Irbesartan Amlodipine Control 579 567 569 Age (y) 59 59 58 Male (%) 65 63 71 Non-white (%) 24 31 28 31. 0 30. 9 30. 5 History of CV disease (%) 27 30 29 Retinopathy (%) 69 64 67 Duration of diabetes (y) 15 14 15 N BMI (kg/m 2) Control defined as placebo. BMI, body mass index. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
IDNT: Mean Baseline Demographics Irbesartan Amlodipine Control 579 567 569 Age (y) 59 59 58 Male (%) 65 63 71 Non-white (%) 24 31 28 31. 0 30. 9 30. 5 History of CV disease (%) 27 30 29 Retinopathy (%) 69 64 67 Duration of diabetes (y) 15 14 15 N BMI (kg/m 2) Control defined as placebo. BMI, body mass index. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
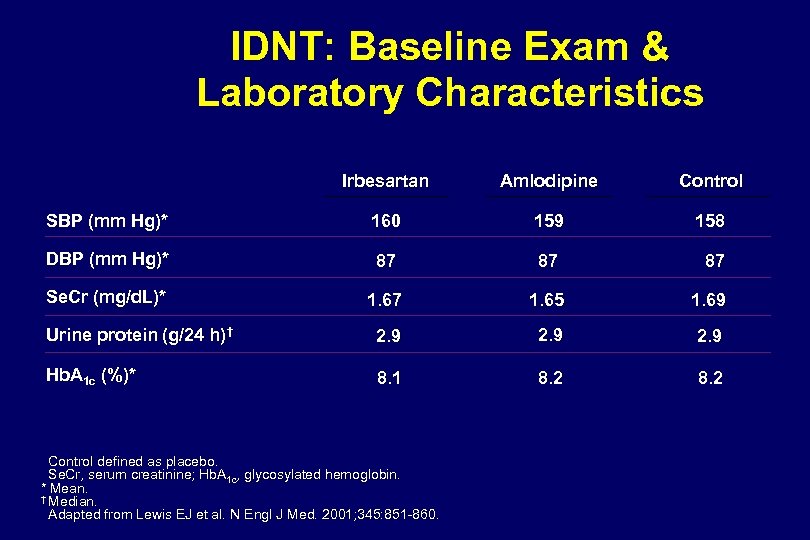 IDNT: Baseline Exam & Laboratory Characteristics Irbesartan Amlodipine Control SBP (mm Hg)* 160 159 158 DBP (mm Hg)* 87 87 Se. Cr (mg/d. L)* 1. 67 1. 65 1. 69 Urine protein (g/24 h)† 2. 9 Hb. A 1 c (%)* 8. 1 8. 2 Control defined as placebo. Se. Cr, serum creatinine; Hb. A 1 c, glycosylated hemoglobin. * Mean. † Median. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 87
IDNT: Baseline Exam & Laboratory Characteristics Irbesartan Amlodipine Control SBP (mm Hg)* 160 159 158 DBP (mm Hg)* 87 87 Se. Cr (mg/d. L)* 1. 67 1. 65 1. 69 Urine protein (g/24 h)† 2. 9 Hb. A 1 c (%)* 8. 1 8. 2 Control defined as placebo. Se. Cr, serum creatinine; Hb. A 1 c, glycosylated hemoglobin. * Mean. † Median. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 87
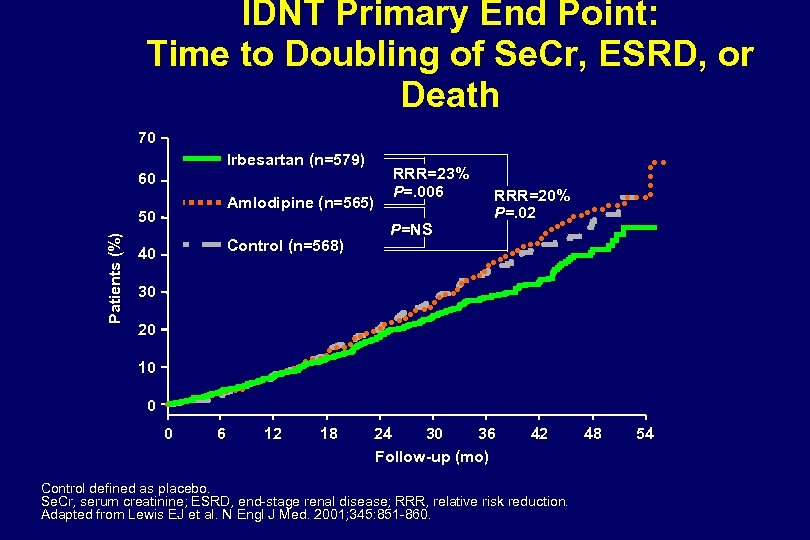 IDNT Primary End Point: Time to Doubling of Se. Cr, ESRD, or Death 70 Irbesartan (n=579) 60 Amlodipine (n=565) Patients (%) 50 Control (n=568) 40 RRR=23% P=. 006 P=NS RRR=20% P=. 02 30 20 10 0 0 6 12 18 24 30 36 Follow-up (mo) 42 Control defined as placebo. Se. Cr, serum creatinine; ESRD, end-stage renal disease; RRR, relative risk reduction. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 48 54
IDNT Primary End Point: Time to Doubling of Se. Cr, ESRD, or Death 70 Irbesartan (n=579) 60 Amlodipine (n=565) Patients (%) 50 Control (n=568) 40 RRR=23% P=. 006 P=NS RRR=20% P=. 02 30 20 10 0 0 6 12 18 24 30 36 Follow-up (mo) 42 Control defined as placebo. Se. Cr, serum creatinine; ESRD, end-stage renal disease; RRR, relative risk reduction. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 48 54
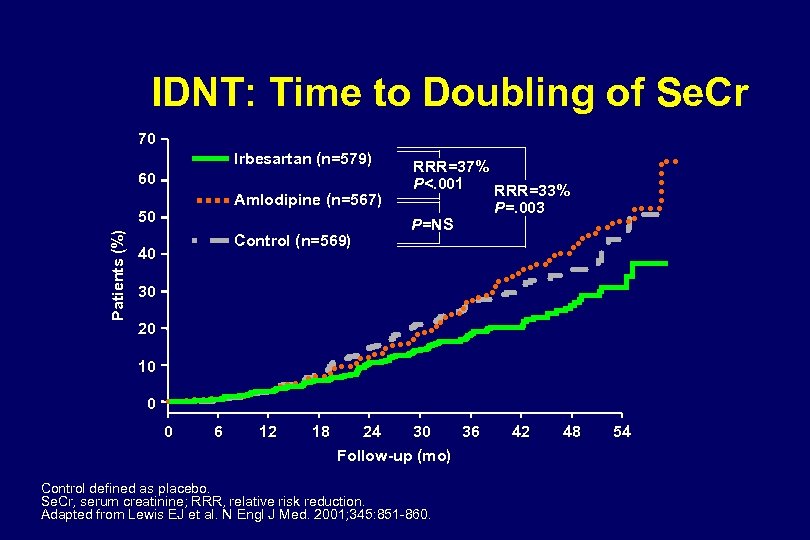 IDNT: Time to Doubling of Se. Cr 70 Irbesartan (n=579) 60 Amlodipine (n=567) Patients (%) 50 Control (n=569) 40 RRR=37% P<. 001 RRR=33% P=. 003 P=NS 30 20 10 0 0 6 12 18 24 30 36 Follow-up (mo) Control defined as placebo. Se. Cr, serum creatinine; RRR, relative risk reduction. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 42 48 54
IDNT: Time to Doubling of Se. Cr 70 Irbesartan (n=579) 60 Amlodipine (n=567) Patients (%) 50 Control (n=569) 40 RRR=37% P<. 001 RRR=33% P=. 003 P=NS 30 20 10 0 0 6 12 18 24 30 36 Follow-up (mo) Control defined as placebo. Se. Cr, serum creatinine; RRR, relative risk reduction. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 42 48 54
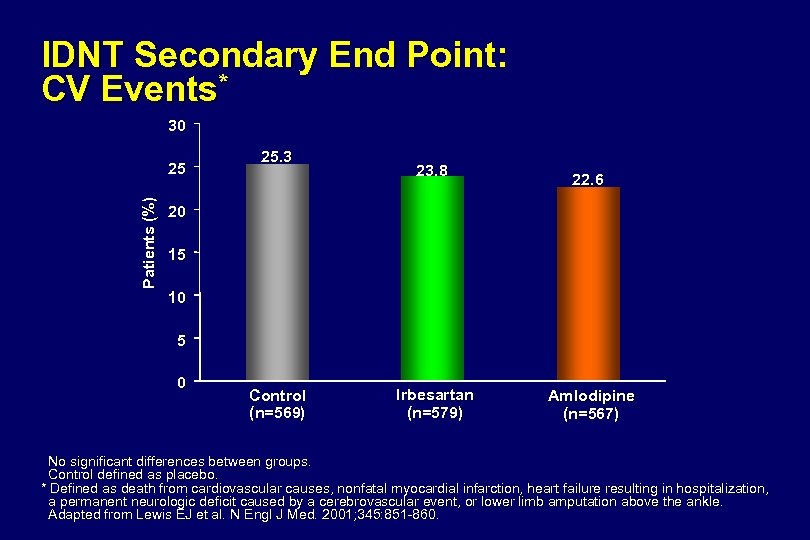 IDNT Secondary End Point: CV Events* 30 Patients (%) 25 25. 3 23. 8 22. 6 20 15 10 5 0 Control (n=569) Irbesartan (n=579) Amlodipine (n=567) No significant differences between groups. Control defined as placebo. * Defined as death from cardiovascular causes, nonfatal myocardial infarction, heart failure resulting in hospitalization, a permanent neurologic deficit caused by a cerebrovascular event, or lower limb amputation above the ankle. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
IDNT Secondary End Point: CV Events* 30 Patients (%) 25 25. 3 23. 8 22. 6 20 15 10 5 0 Control (n=569) Irbesartan (n=579) Amlodipine (n=567) No significant differences between groups. Control defined as placebo. * Defined as death from cardiovascular causes, nonfatal myocardial infarction, heart failure resulting in hospitalization, a permanent neurologic deficit caused by a cerebrovascular event, or lower limb amputation above the ankle. Adapted from Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
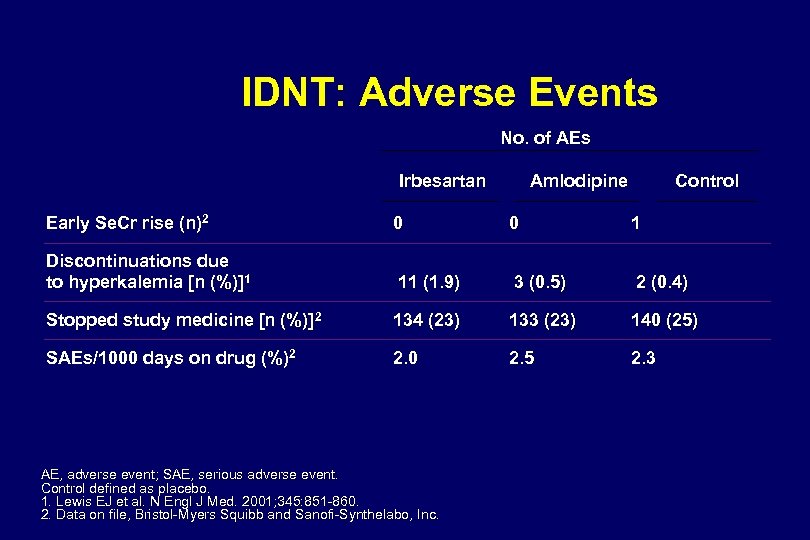 IDNT: Adverse Events No. of AEs Irbesartan Amlodipine Control Early Se. Cr rise (n)2 0 0 1 Discontinuations due to hyperkalemia [n (%)]1 11 (1. 9) 3 (0. 5) 2 (0. 4) Stopped study medicine [n (%)]2 134 (23) 133 (23) 140 (25) SAEs/1000 days on drug (%)2 2. 0 2. 5 2. 3 AE, adverse event; SAE, serious adverse event. Control defined as placebo. 1. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 2. Data on file, Bristol-Myers Squibb and Sanofi-Synthelabo, Inc.
IDNT: Adverse Events No. of AEs Irbesartan Amlodipine Control Early Se. Cr rise (n)2 0 0 1 Discontinuations due to hyperkalemia [n (%)]1 11 (1. 9) 3 (0. 5) 2 (0. 4) Stopped study medicine [n (%)]2 134 (23) 133 (23) 140 (25) SAEs/1000 days on drug (%)2 2. 0 2. 5 2. 3 AE, adverse event; SAE, serious adverse event. Control defined as placebo. 1. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860. 2. Data on file, Bristol-Myers Squibb and Sanofi-Synthelabo, Inc.
 IDNT: Summary Irbesartan reduced the composite risk of progression of renal disease or total mortality, independent of its BP-lowering effects 20% RRR vs control (P=. 02) 23% RRR vs amlodipine (P=. 006) No significant differences among groups for CV outcomes Size and duration of study was insufficient to detect any differences Irbesartan was generally safe and well tolerated Lower rate of SAEs in the irbesartan group SAE, serious adverse event; RRR, relative risk reduction. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.
IDNT: Summary Irbesartan reduced the composite risk of progression of renal disease or total mortality, independent of its BP-lowering effects 20% RRR vs control (P=. 02) 23% RRR vs amlodipine (P=. 006) No significant differences among groups for CV outcomes Size and duration of study was insufficient to detect any differences Irbesartan was generally safe and well tolerated Lower rate of SAEs in the irbesartan group SAE, serious adverse event; RRR, relative risk reduction. Lewis EJ et al. N Engl J Med. 2001; 345: 851 -860.


