DNA repair enzymes as key targets to develop anticancer drugs.ppt
- Количество слайдов: 38
 Human Tyrosyl-DNA Phosphodiesterase 1 : new activities and development of enzyme inhibitors as anticancer drugs Olga Lavrik SB RAS Institute of Chemical Biology and Fundamental Medicine, Department of Physicochemical Biology and Biotechnology of ASU, Novosibirsk, Barnaul, Russia
Human Tyrosyl-DNA Phosphodiesterase 1 : new activities and development of enzyme inhibitors as anticancer drugs Olga Lavrik SB RAS Institute of Chemical Biology and Fundamental Medicine, Department of Physicochemical Biology and Biotechnology of ASU, Novosibirsk, Barnaul, Russia
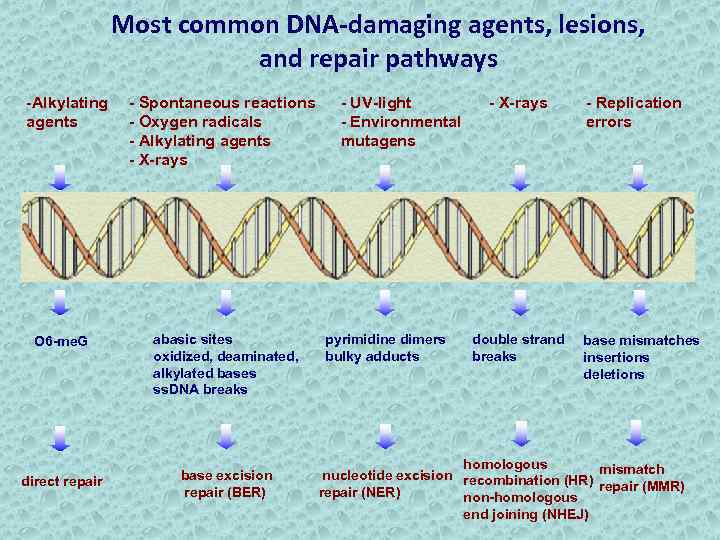 Most common DNA-damaging agents, lesions, and repair pathways -Alkylating agents - Spontaneous reactions - Oxygen radicals - Alkylating agents - X-rays O 6 -me. G abasic sites oxidized, deaminated, alkylated bases ss. DNA breaks direct repair base excision repair (BER) - UV-light - Environmental mutagens pyrimidine dimers bulky adducts - X-rays double strand breaks - Replication errors base mismatches insertions deletions homologous nucleotide excision recombination (HR) mismatch repair (MMR) repair (NER) non-homologous end joining (NHEJ)
Most common DNA-damaging agents, lesions, and repair pathways -Alkylating agents - Spontaneous reactions - Oxygen radicals - Alkylating agents - X-rays O 6 -me. G abasic sites oxidized, deaminated, alkylated bases ss. DNA breaks direct repair base excision repair (BER) - UV-light - Environmental mutagens pyrimidine dimers bulky adducts - X-rays double strand breaks - Replication errors base mismatches insertions deletions homologous nucleotide excision recombination (HR) mismatch repair (MMR) repair (NER) non-homologous end joining (NHEJ)
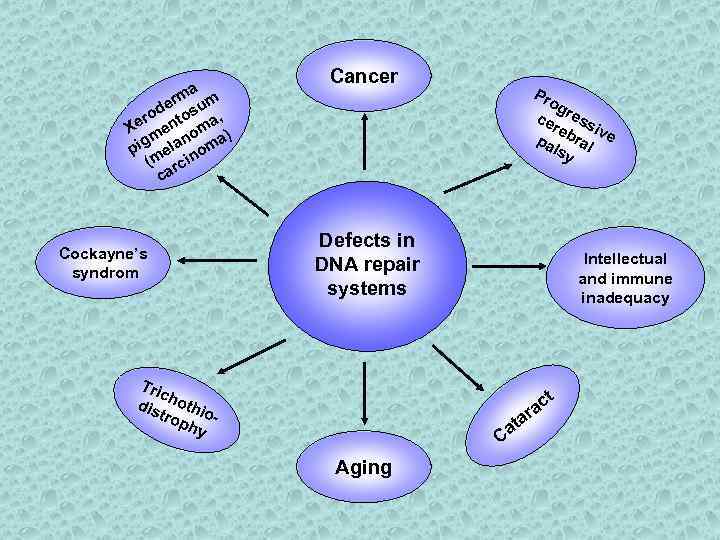 a rm um de ro ntos a, Xe e om ) m n a pig ela nom (m rci ca Cockayne’s syndrom Cancer Pr og ce ress re iv pa bral e lsy Defects in DNA repair systems Tri c dis hothi o tro phy - Intellectual and immune inadequacy t a c ra t Ca Aging
a rm um de ro ntos a, Xe e om ) m n a pig ela nom (m rci ca Cockayne’s syndrom Cancer Pr og ce ress re iv pa bral e lsy Defects in DNA repair systems Tri c dis hothi o tro phy - Intellectual and immune inadequacy t a c ra t Ca Aging
 DNA repair systems are targets for development of anticancer drugs DNA repair systems suppress the efficiency of a number of antitumor drugs that have to reveal their cytotoxic effects by damaging DNA of the cancer cells. Therefore inhibiting of DNA repair can improve the anticancer treatment.
DNA repair systems are targets for development of anticancer drugs DNA repair systems suppress the efficiency of a number of antitumor drugs that have to reveal their cytotoxic effects by damaging DNA of the cancer cells. Therefore inhibiting of DNA repair can improve the anticancer treatment.
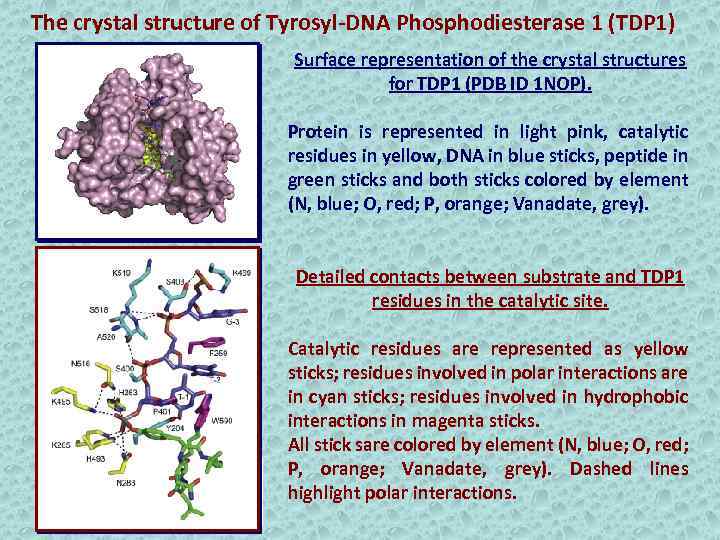 The crystal structure of Tyrosyl-DNA Phosphodiesterase 1 (TDP 1) Surface representation of the crystal structures for TDP 1 (PDB ID 1 NOP). Protein is represented in light pink, catalytic residues in yellow, DNA in blue sticks, peptide in green sticks and both sticks colored by element (N, blue; O, red; P, orange; Vanadate, grey). Detailed contacts between substrate and TDP 1 residues in the catalytic site. Catalytic residues are represented as yellow sticks; residues involved in polar interactions are in cyan sticks; residues involved in hydrophobic interactions in magenta sticks. All stick sare colored by element (N, blue; O, red; P, orange; Vanadate, grey). Dashed lines highlight polar interactions.
The crystal structure of Tyrosyl-DNA Phosphodiesterase 1 (TDP 1) Surface representation of the crystal structures for TDP 1 (PDB ID 1 NOP). Protein is represented in light pink, catalytic residues in yellow, DNA in blue sticks, peptide in green sticks and both sticks colored by element (N, blue; O, red; P, orange; Vanadate, grey). Detailed contacts between substrate and TDP 1 residues in the catalytic site. Catalytic residues are represented as yellow sticks; residues involved in polar interactions are in cyan sticks; residues involved in hydrophobic interactions in magenta sticks. All stick sare colored by element (N, blue; O, red; P, orange; Vanadate, grey). Dashed lines highlight polar interactions.
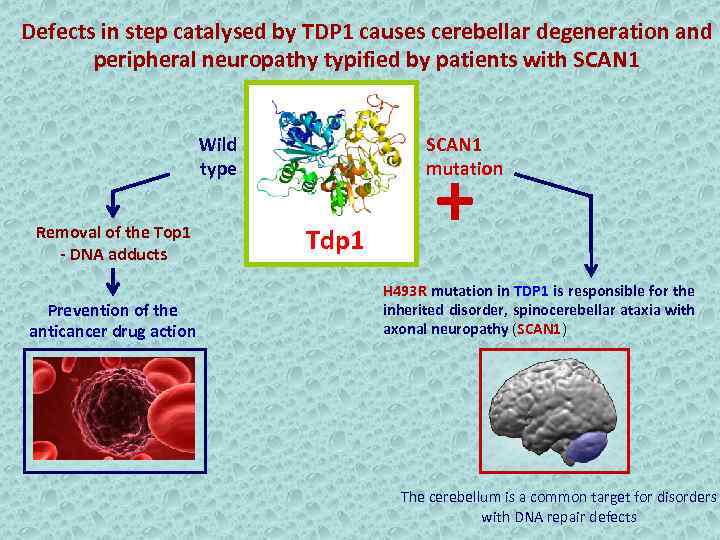 Defects in step catalysed by TDP 1 causes cerebellar degeneration and peripheral neuropathy typified by patients with SCAN 1 Wild type Removal of the Top 1 - DNA adducts Prevention of the anticancer drug action SCAN 1 mutation Tdp 1 + H 493 R mutation in TDP 1 is responsible for the inherited disorder, spinocerebellar ataxia with axonal neuropathy (SCAN 1) The cerebellum is a common target for disorders with DNA repair defects
Defects in step catalysed by TDP 1 causes cerebellar degeneration and peripheral neuropathy typified by patients with SCAN 1 Wild type Removal of the Top 1 - DNA adducts Prevention of the anticancer drug action SCAN 1 mutation Tdp 1 + H 493 R mutation in TDP 1 is responsible for the inherited disorder, spinocerebellar ataxia with axonal neuropathy (SCAN 1) The cerebellum is a common target for disorders with DNA repair defects
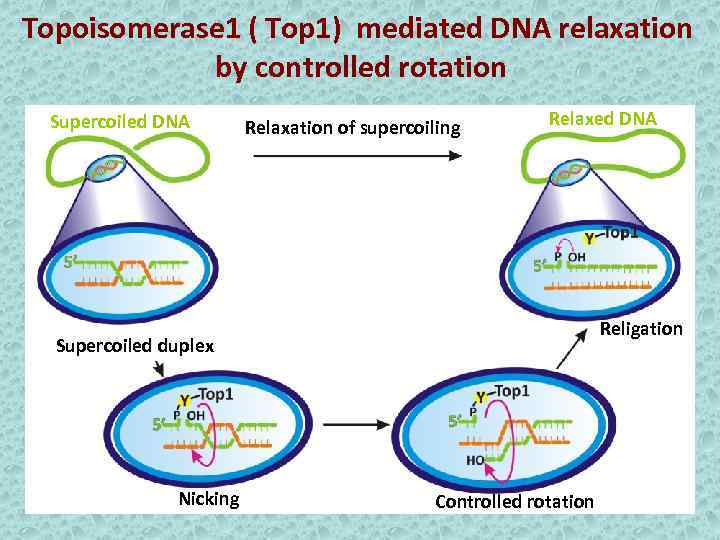 Topoisomerase 1 ( Top 1) mediated DNA relaxation by controlled rotation Supercoiled DNA Relaxation of supercoiling Relaxed DNA Religation Supercoiled duplex Nicking Controlled rotation
Topoisomerase 1 ( Top 1) mediated DNA relaxation by controlled rotation Supercoiled DNA Relaxation of supercoiling Relaxed DNA Religation Supercoiled duplex Nicking Controlled rotation
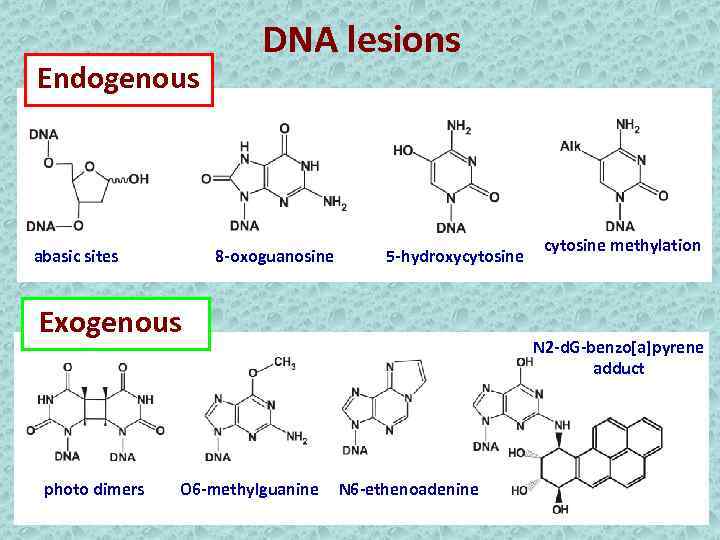 Endogenous abasic sites DNA lesions 8 -oxoguanosine 5 -hydroxycytosine Exogenous photo dimers O 6 -methylguanine cytosine methylation N 2 -d. G-benzo[a]pyrene adduct N 6 -ethenoadenine
Endogenous abasic sites DNA lesions 8 -oxoguanosine 5 -hydroxycytosine Exogenous photo dimers O 6 -methylguanine cytosine methylation N 2 -d. G-benzo[a]pyrene adduct N 6 -ethenoadenine
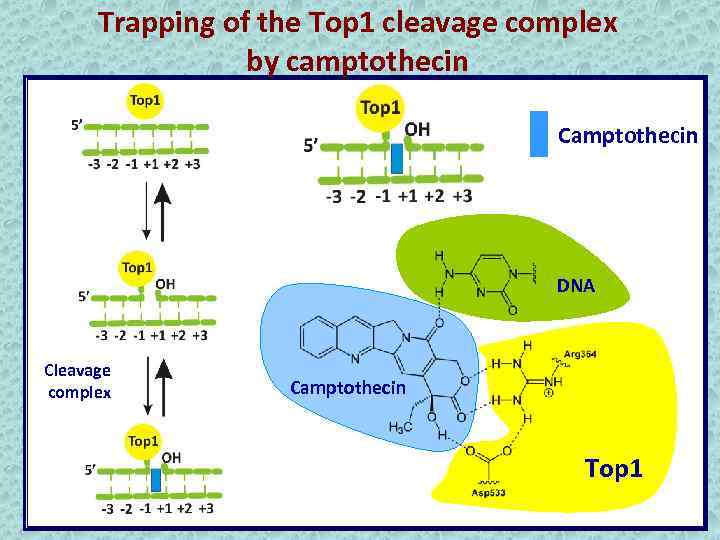 Trapping of the Top 1 cleavage complex by camptothecin Camptothecin DNA Cleavage complex Camptothecin Top 1
Trapping of the Top 1 cleavage complex by camptothecin Camptothecin DNA Cleavage complex Camptothecin Top 1
 Top 1 inhibitors used as anticancer drugs Camptothecin and its clinical derivatives Topotecan Irinotecan Belotecan
Top 1 inhibitors used as anticancer drugs Camptothecin and its clinical derivatives Topotecan Irinotecan Belotecan
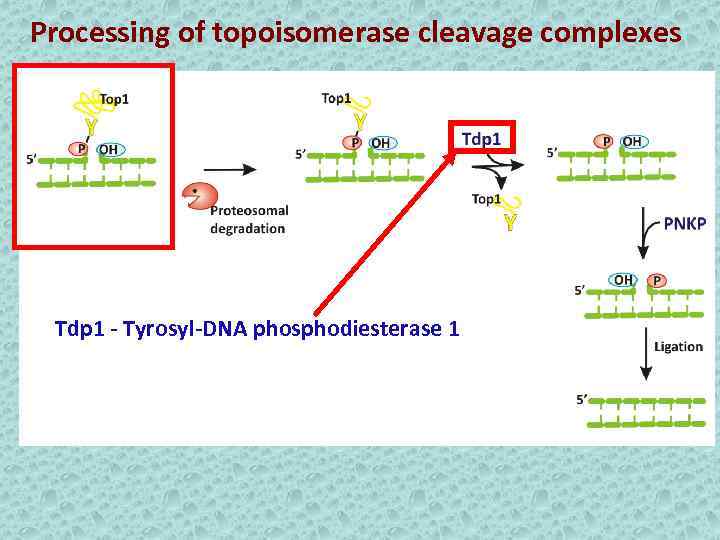 Processing of topoisomerase cleavage complexes Tdp 1 - Tyrosyl-DNA phosphodiesterase 1
Processing of topoisomerase cleavage complexes Tdp 1 - Tyrosyl-DNA phosphodiesterase 1
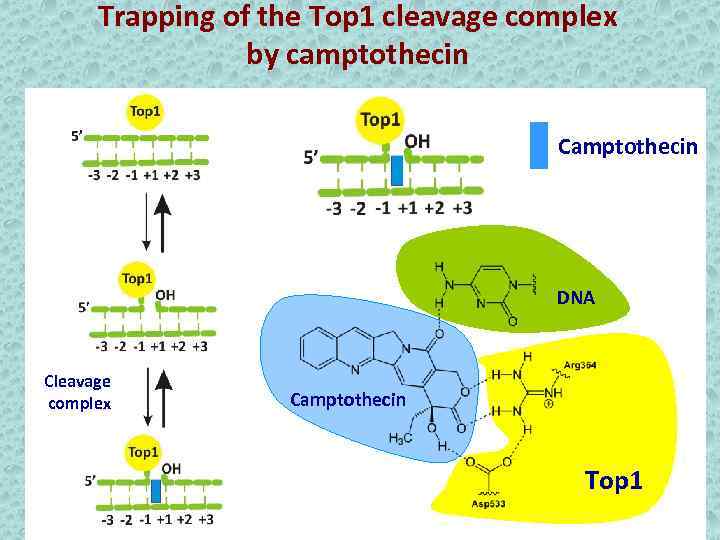 Trapping of the Top 1 cleavage complex by camptothecin Camptothecin DNA Cleavage complex Camptothecin Top 1
Trapping of the Top 1 cleavage complex by camptothecin Camptothecin DNA Cleavage complex Camptothecin Top 1
 Tdp 1 is responsible for the resistance of some types of cancer to anticancer Top 1 inhibitors - Tdp 1 knockout mice and human cell lines, having a mutation of Tdp 1, are hypersensitive to camptothecin - Increased Tdp 1 expression in cells results in less DNA damages induced by camptothecin
Tdp 1 is responsible for the resistance of some types of cancer to anticancer Top 1 inhibitors - Tdp 1 knockout mice and human cell lines, having a mutation of Tdp 1, are hypersensitive to camptothecin - Increased Tdp 1 expression in cells results in less DNA damages induced by camptothecin
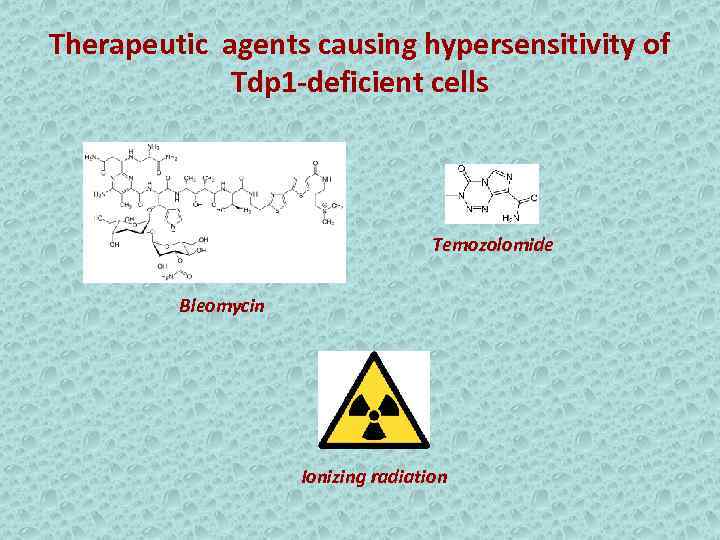 Therapeutic agents causing hypersensitivity of Tdp 1 -deficient cells Temozolomide Bleomycin Ionizing radiation
Therapeutic agents causing hypersensitivity of Tdp 1 -deficient cells Temozolomide Bleomycin Ionizing radiation
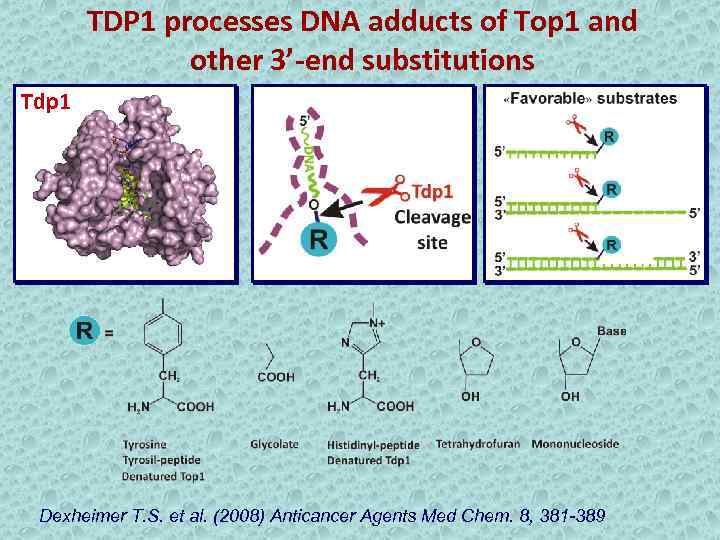 TDP 1 processes DNA adducts of Top 1 and other 3’-end substitutions Tdp 1 Dexheimer T. S. et al. (2008) Anticancer Agents Med Chem. 8, 381 -389
TDP 1 processes DNA adducts of Top 1 and other 3’-end substitutions Tdp 1 Dexheimer T. S. et al. (2008) Anticancer Agents Med Chem. 8, 381 -389
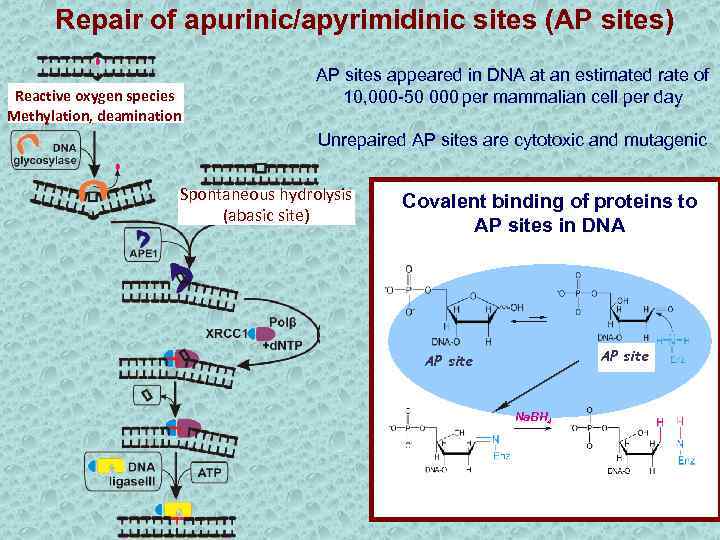 Repair of apurinic/apyrimidinic sites (AP sites) Reactive oxygen species Methylation, deamination АР sites appeared in DNA at an estimated rate of 10, 000 -50 000 per mammalian cell per day Unrepaired AP sites are cytotoxic and mutagenic Spontaneous hydrolysis (abasic site) Covalent binding of proteins to AP sites in DNA АР site Na. BH 4
Repair of apurinic/apyrimidinic sites (AP sites) Reactive oxygen species Methylation, deamination АР sites appeared in DNA at an estimated rate of 10, 000 -50 000 per mammalian cell per day Unrepaired AP sites are cytotoxic and mutagenic Spontaneous hydrolysis (abasic site) Covalent binding of proteins to AP sites in DNA АР site Na. BH 4
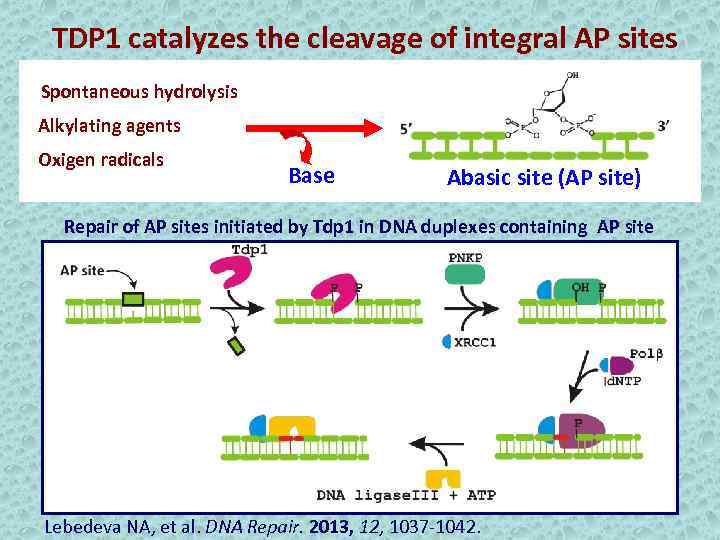 TDP 1 catalyzes the cleavage of integral AP sites Spontaneous hydrolysis Alkylating agents Oxigen radicals Base Abasic site (AP site) Repair of AP sites initiated by Tdp 1 in DNA duplexes containing AP site Lebedeva NA, et al. DNA Repair. 2013, 12, 1037 -1042.
TDP 1 catalyzes the cleavage of integral AP sites Spontaneous hydrolysis Alkylating agents Oxigen radicals Base Abasic site (AP site) Repair of AP sites initiated by Tdp 1 in DNA duplexes containing AP site Lebedeva NA, et al. DNA Repair. 2013, 12, 1037 -1042.
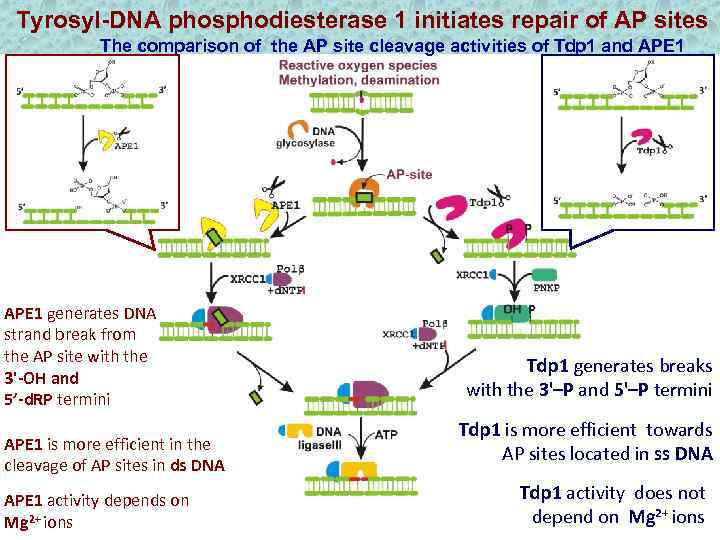 Tyrosyl-DNA phosphodiesterase 1 initiates repair of AP sites The comparison of the AP site cleavage activities of Tdp 1 and APE 1 generates DNA strand break from the AP site with the 3'-OH and 5’-d. RP termini APE 1 is more efficient in the cleavage of AP sites in ds DNA APE 1 activity depends on Mg 2+ ions Tdp 1 generates breaks with the 3'–P and 5'–P termini Tdp 1 is more efficient towards AP sites located in ss DNA Tdp 1 activity does not depend on Mg 2+ ions
Tyrosyl-DNA phosphodiesterase 1 initiates repair of AP sites The comparison of the AP site cleavage activities of Tdp 1 and APE 1 generates DNA strand break from the AP site with the 3'-OH and 5’-d. RP termini APE 1 is more efficient in the cleavage of AP sites in ds DNA APE 1 activity depends on Mg 2+ ions Tdp 1 generates breaks with the 3'–P and 5'–P termini Tdp 1 is more efficient towards AP sites located in ss DNA Tdp 1 activity does not depend on Mg 2+ ions
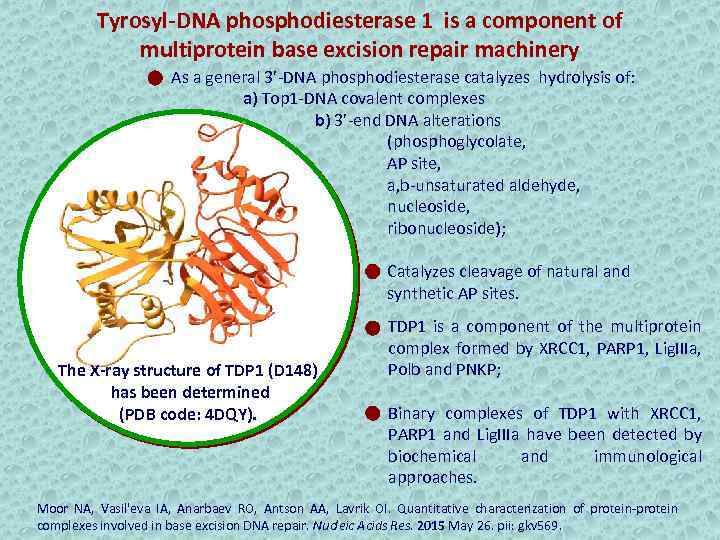 Tyrosyl-DNA phosphodiesterase 1 is a component of multiprotein base excision repair machinery As a general 3’-DNA phosphodiesterase catalyzes hydrolysis of: a) Top 1 -DNA covalent complexes b) 3’-end DNA alterations (phosphoglycolate, AP site, a, b-unsaturated aldehyde, nucleoside, ribonucleoside); Catalyzes cleavage of natural and synthetic AP sites. The X-ray structure of TDP 1 (D 148) has been determined (PDB code: 4 DQY). TDP 1 is a component of the multiprotein complex formed by XRCC 1, PARP 1, Lig. IIIa, Polb and PNKP; Binary complexes of TDP 1 with XRCC 1, PARP 1 and Lig. IIIa have been detected by biochemical and immunological approaches. Moor NA, Vasil'eva IA, Anarbaev RO, Antson AA, Lavrik OI. Quantitative characterization of protein-protein complexes involved in base excision DNA repair. Nucleic Acids Res. 2015 May 26. pii: gkv 569.
Tyrosyl-DNA phosphodiesterase 1 is a component of multiprotein base excision repair machinery As a general 3’-DNA phosphodiesterase catalyzes hydrolysis of: a) Top 1 -DNA covalent complexes b) 3’-end DNA alterations (phosphoglycolate, AP site, a, b-unsaturated aldehyde, nucleoside, ribonucleoside); Catalyzes cleavage of natural and synthetic AP sites. The X-ray structure of TDP 1 (D 148) has been determined (PDB code: 4 DQY). TDP 1 is a component of the multiprotein complex formed by XRCC 1, PARP 1, Lig. IIIa, Polb and PNKP; Binary complexes of TDP 1 with XRCC 1, PARP 1 and Lig. IIIa have been detected by biochemical and immunological approaches. Moor NA, Vasil'eva IA, Anarbaev RO, Antson AA, Lavrik OI. Quantitative characterization of protein-protein complexes involved in base excision DNA repair. Nucleic Acids Res. 2015 May 26. pii: gkv 569.
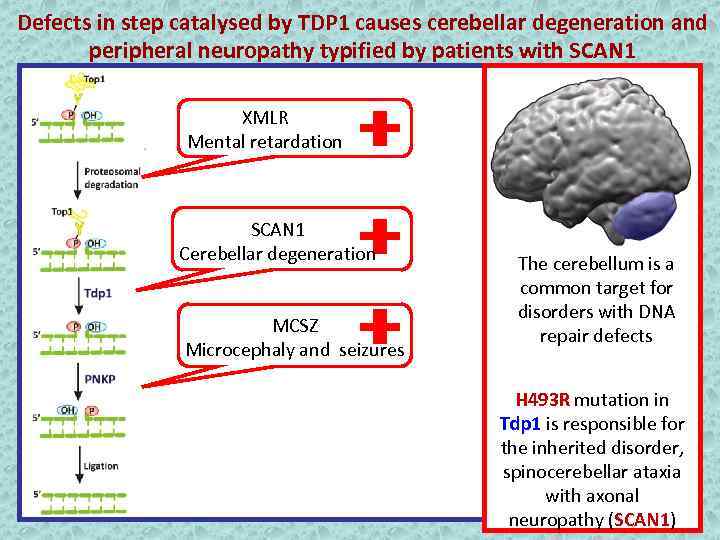 Defects in step catalysed by TDP 1 causes cerebellar degeneration and peripheral neuropathy typified by patients with SCAN 1 XMLR Mental retardation SCAN 1 Cerebellar degeneration MCSZ Microcephaly and seizures The cerebellum is a common target for disorders with DNA repair defects H 493 R mutation in Tdp 1 is responsible for the inherited disorder, spinocerebellar ataxia with axonal neuropathy (SCAN 1)
Defects in step catalysed by TDP 1 causes cerebellar degeneration and peripheral neuropathy typified by patients with SCAN 1 XMLR Mental retardation SCAN 1 Cerebellar degeneration MCSZ Microcephaly and seizures The cerebellum is a common target for disorders with DNA repair defects H 493 R mutation in Tdp 1 is responsible for the inherited disorder, spinocerebellar ataxia with axonal neuropathy (SCAN 1)
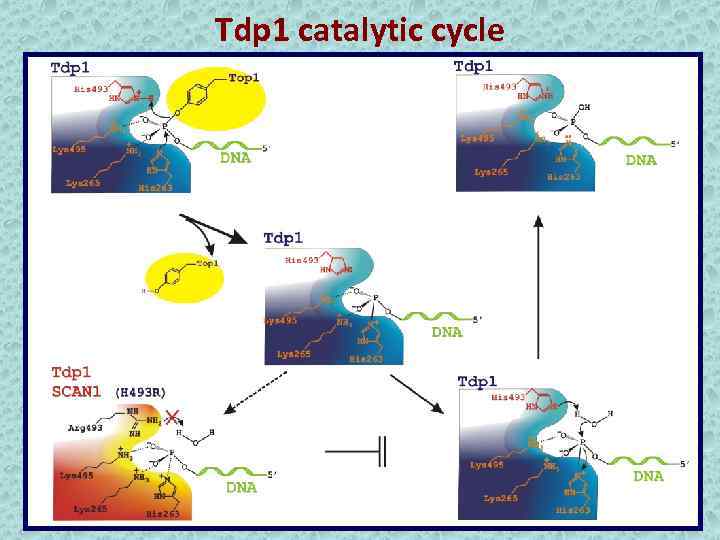 Tdp 1 catalytic cycle
Tdp 1 catalytic cycle
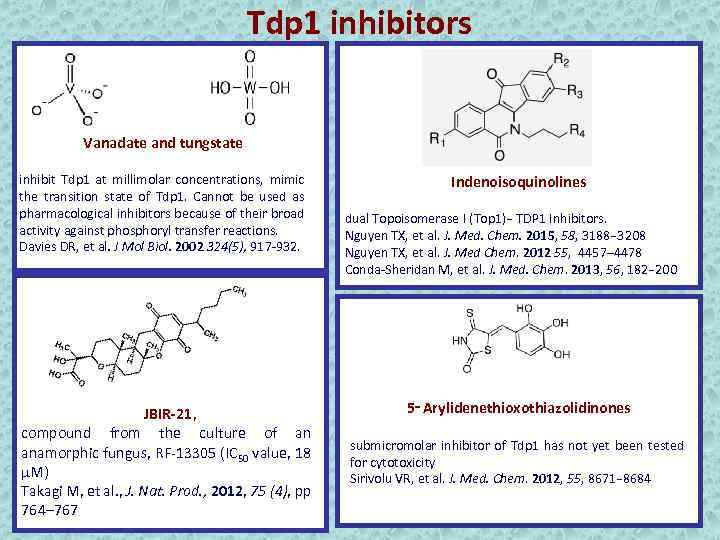 Tdp 1 inhibitors Vanadate and tungstate inhibit Tdp 1 at millimolar concentrations, mimic the transition state of Tdp 1. Cannot be used as pharmacological inhibitors because of their broad activity against phosphoryl transfer reactions. Davies DR, et al. J Mol Biol. 2002 324(5), 917 -932. JBIR-21, compound from the culture of an anamorphic fungus, RF-13305 (IC 50 value, 18 μM) Takagi M, et al. , J. Nat. Prod. , 2012, 75 (4), pp 764– 767 Indenoisoquinolines dual Topoisomerase I (Top 1)− TDP 1 Inhibitors. Nguyen TX, et al. J. Med. Chem. 2015, 58, 3188− 3208 Nguyen TX, et al. J. Med Chem. 2012 55, 4457– 4478 Conda-Sheridan M, et al. J. Med. Chem. 2013, 56, 182− 200 5‑Arylidenethioxothiazolidinones submicromolar inhibitor of Tdp 1 has not yet been tested for cytotoxicity Sirivolu VR, et al. J. Med. Chem. 2012, 55, 8671− 8684
Tdp 1 inhibitors Vanadate and tungstate inhibit Tdp 1 at millimolar concentrations, mimic the transition state of Tdp 1. Cannot be used as pharmacological inhibitors because of their broad activity against phosphoryl transfer reactions. Davies DR, et al. J Mol Biol. 2002 324(5), 917 -932. JBIR-21, compound from the culture of an anamorphic fungus, RF-13305 (IC 50 value, 18 μM) Takagi M, et al. , J. Nat. Prod. , 2012, 75 (4), pp 764– 767 Indenoisoquinolines dual Topoisomerase I (Top 1)− TDP 1 Inhibitors. Nguyen TX, et al. J. Med. Chem. 2015, 58, 3188− 3208 Nguyen TX, et al. J. Med Chem. 2012 55, 4457– 4478 Conda-Sheridan M, et al. J. Med. Chem. 2013, 56, 182− 200 5‑Arylidenethioxothiazolidinones submicromolar inhibitor of Tdp 1 has not yet been tested for cytotoxicity Sirivolu VR, et al. J. Med. Chem. 2012, 55, 8671− 8684
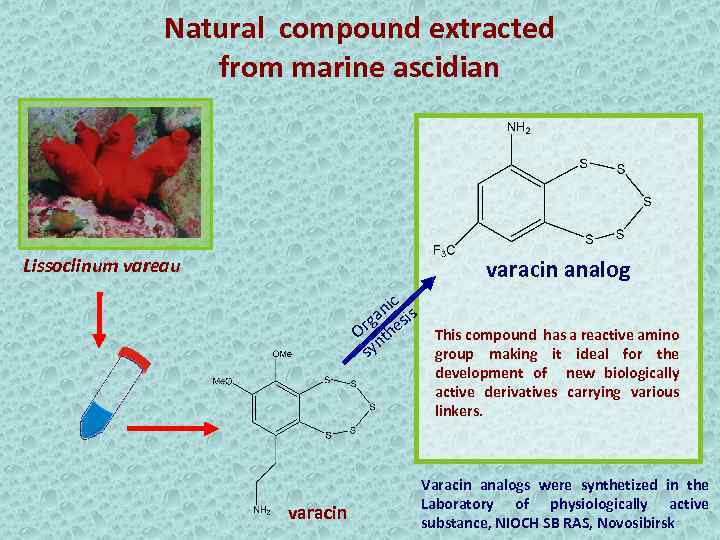 Natural compound extracted from marine ascidian Lissoclinum vareau varacin analog nic is a rg hes This compound has a reactive amino O t n sy group making it ideal for the development of new biologically active derivatives carrying various linkers. varacin Varacin analogs were synthetized in the Laboratory of physiologically active substance, NIOCH SB RAS, Novosibirsk
Natural compound extracted from marine ascidian Lissoclinum vareau varacin analog nic is a rg hes This compound has a reactive amino O t n sy group making it ideal for the development of new biologically active derivatives carrying various linkers. varacin Varacin analogs were synthetized in the Laboratory of physiologically active substance, NIOCH SB RAS, Novosibirsk
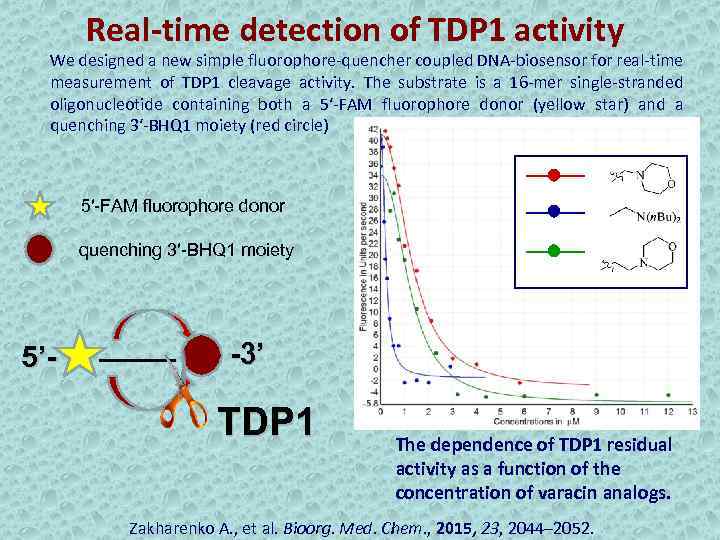 Real-time detection of TDP 1 activity We designed a new simple fluorophore-quencher coupled DNA-biosensor for real-time measurement of TDP 1 cleavage activity. The substrate is a 16 -mer single-stranded oligonucleotide containing both a 5′-FAM fluorophore donor (yellow star) and a quenching 3′-BHQ 1 moiety (red circle) 5′-FAM fluorophore donor quenching 3′-BHQ 1 moiety 5’- -3’ TDP 1 The dependence of TDP 1 residual activity as a function of the concentration of varacin analogs. Zakharenko A. , et al. Bioorg. Med. Chem. , 2015, 23, 2044– 2052.
Real-time detection of TDP 1 activity We designed a new simple fluorophore-quencher coupled DNA-biosensor for real-time measurement of TDP 1 cleavage activity. The substrate is a 16 -mer single-stranded oligonucleotide containing both a 5′-FAM fluorophore donor (yellow star) and a quenching 3′-BHQ 1 moiety (red circle) 5′-FAM fluorophore donor quenching 3′-BHQ 1 moiety 5’- -3’ TDP 1 The dependence of TDP 1 residual activity as a function of the concentration of varacin analogs. Zakharenko A. , et al. Bioorg. Med. Chem. , 2015, 23, 2044– 2052.
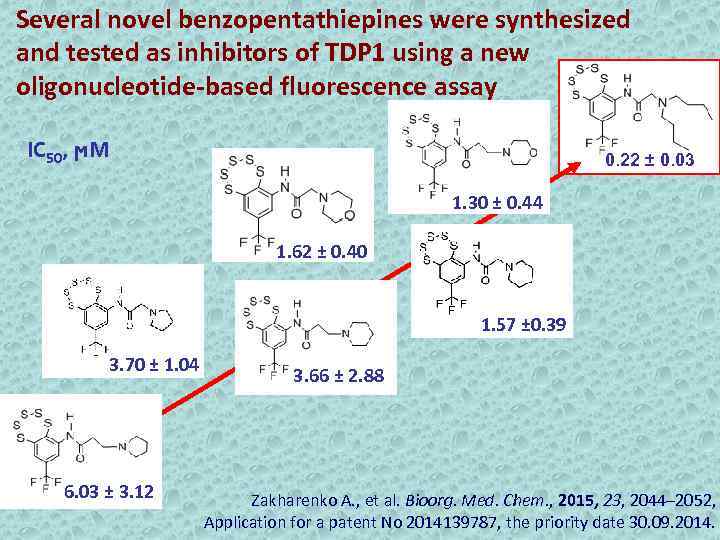 Several novel benzopentathiepines were synthesized and tested as inhibitors of TDP 1 using a new oligonucleotide-based fluorescence assay IC 50, ϻМ 0. 22 ± 0. 03 1. 30 ± 0. 44 1. 62 ± 0. 40 1. 57 ± 0. 39 3. 70 ± 1. 04 6. 03 ± 3. 12 3. 66 ± 2. 88 Zakharenko A. , et al. Bioorg. Med. Chem. , 2015, 23, 2044– 2052, Application for a patent No 2014139787, the priority date 30. 09. 2014.
Several novel benzopentathiepines were synthesized and tested as inhibitors of TDP 1 using a new oligonucleotide-based fluorescence assay IC 50, ϻМ 0. 22 ± 0. 03 1. 30 ± 0. 44 1. 62 ± 0. 40 1. 57 ± 0. 39 3. 70 ± 1. 04 6. 03 ± 3. 12 3. 66 ± 2. 88 Zakharenko A. , et al. Bioorg. Med. Chem. , 2015, 23, 2044– 2052, Application for a patent No 2014139787, the priority date 30. 09. 2014.
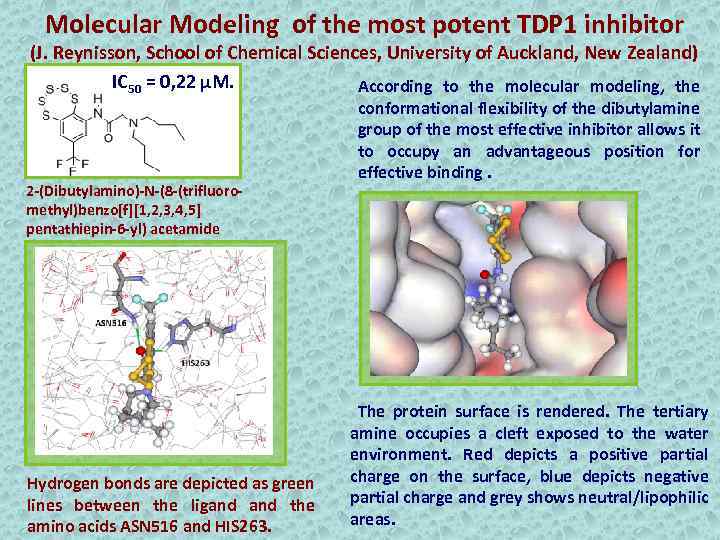 Molecular Modeling of the most potent TDP 1 inhibitor (J. Reynisson, School of Chemical Sciences, University of Auckland, New Zealand) IC 50 = 0, 22 µM. According to the molecular modeling, the 2 -(Dibutylamino)-N-(8 -(trifluoromethyl)benzo[f][1, 2, 3, 4, 5] pentathiepin-6 -yl) acetamide Hydrogen bonds are depicted as green lines between the ligand the amino acids ASN 516 and HIS 263. conformational flexibility of the dibutylamine group of the most effective inhibitor allows it to occupy an advantageous position for effective binding. The protein surface is rendered. The tertiary amine occupies a cleft exposed to the water environment. Red depicts a positive partial charge on the surface, blue depicts negative partial charge and grey shows neutral/lipophilic areas.
Molecular Modeling of the most potent TDP 1 inhibitor (J. Reynisson, School of Chemical Sciences, University of Auckland, New Zealand) IC 50 = 0, 22 µM. According to the molecular modeling, the 2 -(Dibutylamino)-N-(8 -(trifluoromethyl)benzo[f][1, 2, 3, 4, 5] pentathiepin-6 -yl) acetamide Hydrogen bonds are depicted as green lines between the ligand the amino acids ASN 516 and HIS 263. conformational flexibility of the dibutylamine group of the most effective inhibitor allows it to occupy an advantageous position for effective binding. The protein surface is rendered. The tertiary amine occupies a cleft exposed to the water environment. Red depicts a positive partial charge on the surface, blue depicts negative partial charge and grey shows neutral/lipophilic areas.
 Cytotoxicity of varacin analogs - The cytotoxicity study of the compounds showed that they caused apoptotic cell death in human mammary adenocarcinoma cell line MCF-7 and human liver cell carcinoma Hep G 2. - Several apoptotic features were observed with light microscopy: condensation of nuclei, shrinkage of the cytoplasm, convolution of outlines and formation of apoptotic bodies. - The most potent TDP 1 inhibitor (dibutylamine derivative) with 200 μM concentration caused 10. 5% of the cells to enter apoptosis
Cytotoxicity of varacin analogs - The cytotoxicity study of the compounds showed that they caused apoptotic cell death in human mammary adenocarcinoma cell line MCF-7 and human liver cell carcinoma Hep G 2. - Several apoptotic features were observed with light microscopy: condensation of nuclei, shrinkage of the cytoplasm, convolution of outlines and formation of apoptotic bodies. - The most potent TDP 1 inhibitor (dibutylamine derivative) with 200 μM concentration caused 10. 5% of the cells to enter apoptosis
 Conclusions The X-ray structure of TDP 1 (D 148) - Tyrosyl-DNA phosphodiestherase 1 is the beneficial target to design the new classes of anticancer drugs. These drugs are extremely important to improve anticancer therapy by topoisomerase inhibitors. - A new class of TDP 1 benzopentathiepine inhibitors active in the low micromolar or high nanomolar range. The derivative containing dibutylamine substituent demonstrates the highest activity. Its potency can be attributed to its high lipophility and a greater conformational flexibility allowing it to effectively fit to the TDP 1 active center. - Tdp 1 inhibitors caused apoptotic cell death in cancer cell lines - The most potent TDP 1 inhibitor has the greatest potential to be developed further as an anticancer drug in combination with the established Top 1 inhibitors.
Conclusions The X-ray structure of TDP 1 (D 148) - Tyrosyl-DNA phosphodiestherase 1 is the beneficial target to design the new classes of anticancer drugs. These drugs are extremely important to improve anticancer therapy by topoisomerase inhibitors. - A new class of TDP 1 benzopentathiepine inhibitors active in the low micromolar or high nanomolar range. The derivative containing dibutylamine substituent demonstrates the highest activity. Its potency can be attributed to its high lipophility and a greater conformational flexibility allowing it to effectively fit to the TDP 1 active center. - Tdp 1 inhibitors caused apoptotic cell death in cancer cell lines - The most potent TDP 1 inhibitor has the greatest potential to be developed further as an anticancer drug in combination with the established Top 1 inhibitors.
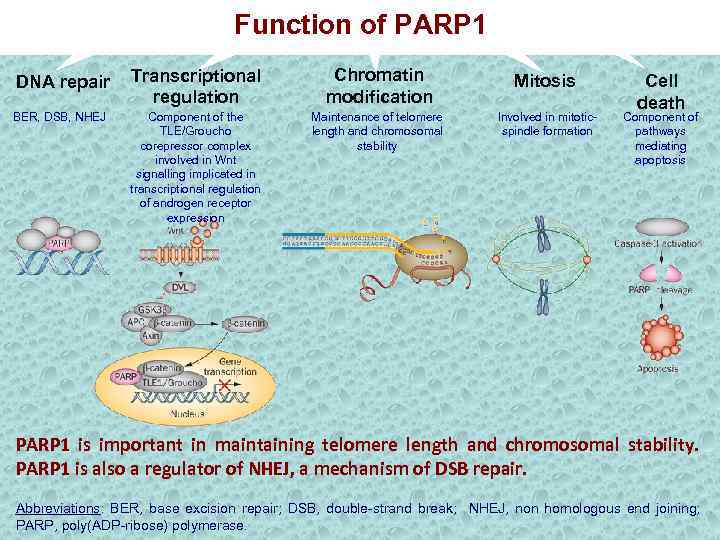 Function of PARP 1 DNA repair Transcriptional regulation Chromatin modification Mitosis BER, DSB, NHEJ Component of the TLE/Groucho corepressor complex involved in Wnt signalling implicated in transcriptional regulation of androgen receptor expression Maintenance of telomere length and chromosomal stability Involved in mitoticspindle formation Cell death Component of pathways mediating apoptosis PARP 1 is important in maintaining telomere length and chromosomal stability. PARP 1 is also a regulator of NHEJ, a mechanism of DSB repair. Abbreviations: BER, base excision repair; DSB, double-strand break; NHEJ, non homologous end joining; PARP, poly(ADP-ribose) polymerase.
Function of PARP 1 DNA repair Transcriptional regulation Chromatin modification Mitosis BER, DSB, NHEJ Component of the TLE/Groucho corepressor complex involved in Wnt signalling implicated in transcriptional regulation of androgen receptor expression Maintenance of telomere length and chromosomal stability Involved in mitoticspindle formation Cell death Component of pathways mediating apoptosis PARP 1 is important in maintaining telomere length and chromosomal stability. PARP 1 is also a regulator of NHEJ, a mechanism of DSB repair. Abbreviations: BER, base excision repair; DSB, double-strand break; NHEJ, non homologous end joining; PARP, poly(ADP-ribose) polymerase.
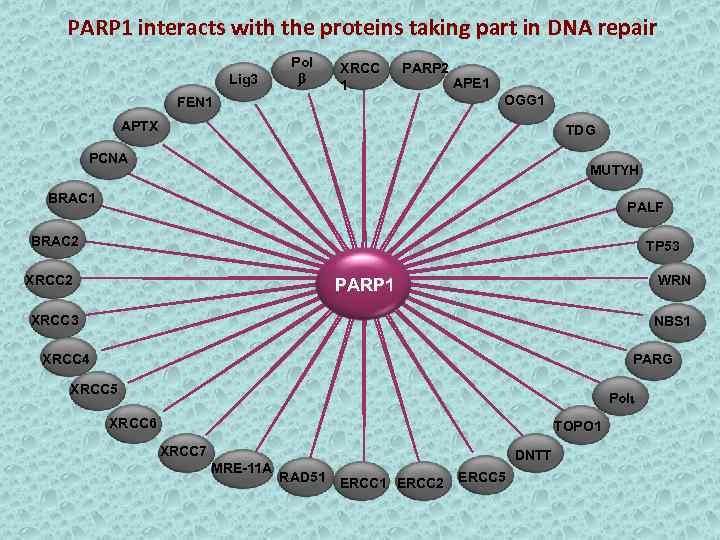 PARP 1 interacts with the proteins taking part in DNA repair Lig 3 Pol b XRCC 1 FEN 1 PARP 2 APE 1 OGG 1 APTX TDG PCNA MUTYH BRAC 1 PALF BRAC 2 TP 53 XRCC 2 WRN PARP 1 XRCC 3 NBS 1 XRCC 4 PARG XRCC 5 Poli XRCC 6 TOPO 1 XRCC 7 MRE-11 A DNTT RAD 51 ERCC 2 ERCC 5
PARP 1 interacts with the proteins taking part in DNA repair Lig 3 Pol b XRCC 1 FEN 1 PARP 2 APE 1 OGG 1 APTX TDG PCNA MUTYH BRAC 1 PALF BRAC 2 TP 53 XRCC 2 WRN PARP 1 XRCC 3 NBS 1 XRCC 4 PARG XRCC 5 Poli XRCC 6 TOPO 1 XRCC 7 MRE-11 A DNTT RAD 51 ERCC 2 ERCC 5
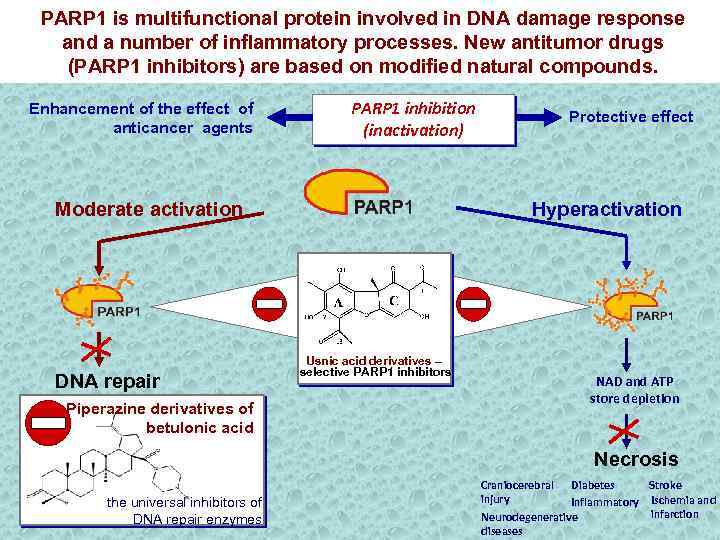 PARP 1 is multifunctional protein involved in DNA damage response and a number of inflammatory processes. New antitumor drugs (PARP 1 inhibitors) are based on modified natural compounds. Enhancement of the effect of anticancer agents PARP 1 inhibition (inactivation) Moderate activation DNA repair Piperazine derivatives of betulonic acid Protective effect Hyperactivation Usnic acid derivatives – selective PARP 1 inhibitors NAD and ATP store depletion Necrosis the universal inhibitors of DNA repair enzymes Craniocerebral Diabetes Stroke injury Inflammatory Ischemia and infarction Neurodegenerative diseases
PARP 1 is multifunctional protein involved in DNA damage response and a number of inflammatory processes. New antitumor drugs (PARP 1 inhibitors) are based on modified natural compounds. Enhancement of the effect of anticancer agents PARP 1 inhibition (inactivation) Moderate activation DNA repair Piperazine derivatives of betulonic acid Protective effect Hyperactivation Usnic acid derivatives – selective PARP 1 inhibitors NAD and ATP store depletion Necrosis the universal inhibitors of DNA repair enzymes Craniocerebral Diabetes Stroke injury Inflammatory Ischemia and infarction Neurodegenerative diseases
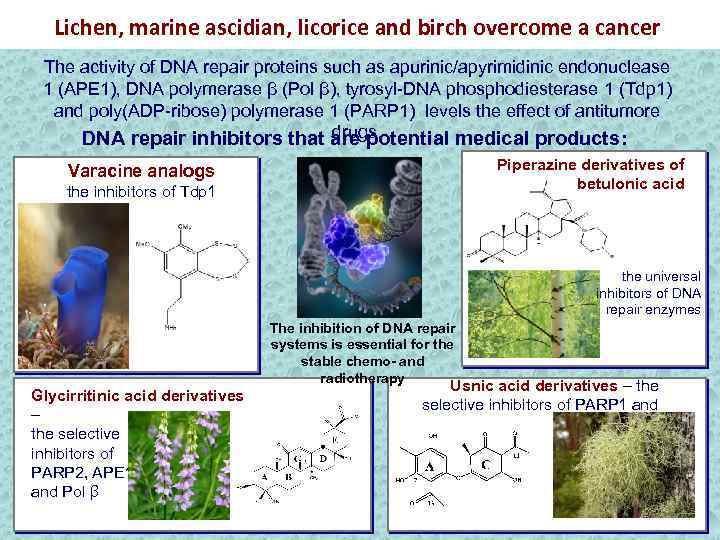 Lichen, marine ascidian, licorice and birch overcome a cancer The activity of DNA repair proteins such as apurinic/apyrimidinic endonuclease 1 (АРЕ 1), DNA polymerase β (Pol β), tyrosyl-DNA phosphodiesterase 1 (Tdp 1) and poly(ADP-ribose) polymerase 1 (PARP 1) levels the effect of antitumore drugs. DNA repair inhibitors that are potential medical products: Piperazine derivatives of betulonic acid Varacine analogs the inhibitors of Tdp 1 the universal inhibitors of DNA repair enzymes The inhibition of DNA repair systems is essential for the stable chemo- and radiotherapy Glycirritinic acid derivatives – the selective inhibitors of PARP 2, APE 1 and Pol β Usnic acid derivatives – the selective inhibitors of PARP 1 and Tdp 1
Lichen, marine ascidian, licorice and birch overcome a cancer The activity of DNA repair proteins such as apurinic/apyrimidinic endonuclease 1 (АРЕ 1), DNA polymerase β (Pol β), tyrosyl-DNA phosphodiesterase 1 (Tdp 1) and poly(ADP-ribose) polymerase 1 (PARP 1) levels the effect of antitumore drugs. DNA repair inhibitors that are potential medical products: Piperazine derivatives of betulonic acid Varacine analogs the inhibitors of Tdp 1 the universal inhibitors of DNA repair enzymes The inhibition of DNA repair systems is essential for the stable chemo- and radiotherapy Glycirritinic acid derivatives – the selective inhibitors of PARP 2, APE 1 and Pol β Usnic acid derivatives – the selective inhibitors of PARP 1 and Tdp 1
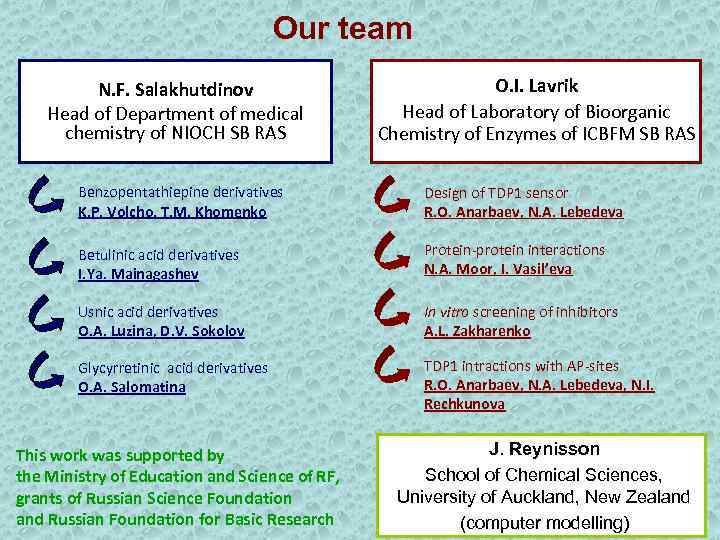 Our team N. F. Salakhutdinov Head of Department of medical chemistry of NIOCH SB RAS O. I. Lavrik Head of Laboratory of Bioorganic Chemistry of Enzymes of ICBFM SB RAS Benzopentathiepine derivatives K. P. Volcho, T. M. Khomenko Design of TDP 1 sensor R. O. Anarbaev, N. A. Lebedeva Betulinic acid derivatives I. Ya. Mainagashev Protein-protein interactions N. A. Moor, I. Vasil’eva Usnic acid derivatives O. A. Luzina, D. V. Sokolov In vitro screening of inhibitors A. L. Zakharenko Glycyrretinic acid derivatives O. A. Salomatina TDP 1 intractions with AP-sites R. O. Anarbaev, N. A. Lebedeva, N. I. Rechkunova This work was supported by the Ministry of Education and Science of RF, grants of Russian Science Foundation and Russian Foundation for Basic Research J. Reynisson School of Chemical Sciences, University of Auckland, New Zealand (computer modelling)
Our team N. F. Salakhutdinov Head of Department of medical chemistry of NIOCH SB RAS O. I. Lavrik Head of Laboratory of Bioorganic Chemistry of Enzymes of ICBFM SB RAS Benzopentathiepine derivatives K. P. Volcho, T. M. Khomenko Design of TDP 1 sensor R. O. Anarbaev, N. A. Lebedeva Betulinic acid derivatives I. Ya. Mainagashev Protein-protein interactions N. A. Moor, I. Vasil’eva Usnic acid derivatives O. A. Luzina, D. V. Sokolov In vitro screening of inhibitors A. L. Zakharenko Glycyrretinic acid derivatives O. A. Salomatina TDP 1 intractions with AP-sites R. O. Anarbaev, N. A. Lebedeva, N. I. Rechkunova This work was supported by the Ministry of Education and Science of RF, grants of Russian Science Foundation and Russian Foundation for Basic Research J. Reynisson School of Chemical Sciences, University of Auckland, New Zealand (computer modelling)
 Thank you for your attention!
Thank you for your attention!
 Therapeutic agents causing hypersensitivity of Tdp 1 -deficient cells Temozolomide Bleomycin Ionizing radiation
Therapeutic agents causing hypersensitivity of Tdp 1 -deficient cells Temozolomide Bleomycin Ionizing radiation
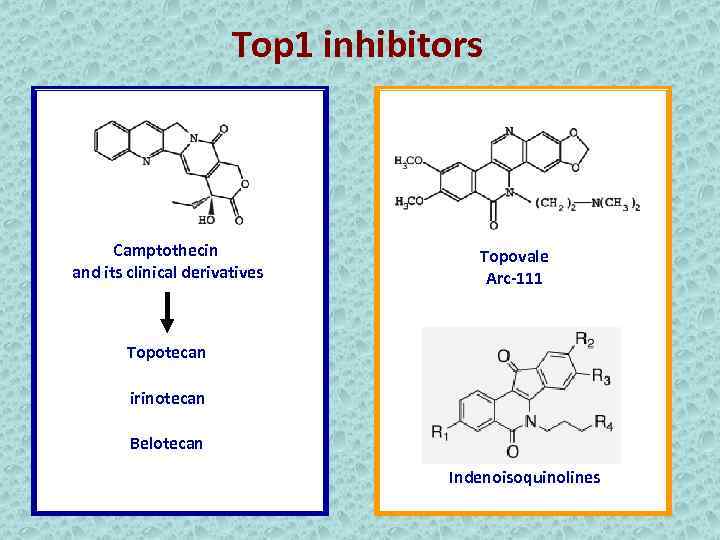 Top 1 inhibitors Camptothecin and its clinical derivatives Topovale Arc-111 Topotecan irinotecan Belotecan Indenoisoquinolines
Top 1 inhibitors Camptothecin and its clinical derivatives Topovale Arc-111 Topotecan irinotecan Belotecan Indenoisoquinolines
 The study of cytotoxicity of these compounds revealed that all compounds cause an apoptotic cell death in human mammary adenocarcinoma cell line MCF-7 or human liver cell carcinoma Hep G 2 24 h Compound 48 h 100 µM 200 µM 100 µM 0 0 nd 0, 8 3 d nd* 1, 5 nd 10, 5 MCF-7 Hep G 2 Varacin analog 14. 0± 2. 7 26. 6 ± 1. 6 3 d 28. 1± 8. 7 >100 200 µM Varacin analog CC 50, µM *not determined The compound 3 d demonstrated concentrationdependence as evaluated by the MTT cytotoxicity assay. The 50% cytotoxic concentration (CC 50) was defined as the concentration required to reduce the cell number by 50% compared to that for the untreated controls. The CC 50 values of Tdp 1 -active compounds after 72 hours treatment applying the MTT cell based assay. The detection of apoptotic DNA fragments after 72 hours treatment of MCF-7 cells with 100 μM of the derivatives. Electrophoretic analysis revealed the appearance of DNA fragments corresponded to mono- (170 -200) and oligonucleosoms, confirming an apoptotic mechanism of cell death for the all pentathiepines tested. The increment of FITC -labeled annexin V and propidium iodide fluorescence in MCF-7 cells treated with the pentathiepines for 24 h and 48 h, relative to control of untreated MCF-7 cells (%). 3000 2000 1000 750 540 -600 360 -400 180 -200 1 - designates DNA markers 2 - control cells; 3 - 1%DMSO; 4 - empty; 5 - 3 d; 6 - varacin analog.
The study of cytotoxicity of these compounds revealed that all compounds cause an apoptotic cell death in human mammary adenocarcinoma cell line MCF-7 or human liver cell carcinoma Hep G 2 24 h Compound 48 h 100 µM 200 µM 100 µM 0 0 nd 0, 8 3 d nd* 1, 5 nd 10, 5 MCF-7 Hep G 2 Varacin analog 14. 0± 2. 7 26. 6 ± 1. 6 3 d 28. 1± 8. 7 >100 200 µM Varacin analog CC 50, µM *not determined The compound 3 d demonstrated concentrationdependence as evaluated by the MTT cytotoxicity assay. The 50% cytotoxic concentration (CC 50) was defined as the concentration required to reduce the cell number by 50% compared to that for the untreated controls. The CC 50 values of Tdp 1 -active compounds after 72 hours treatment applying the MTT cell based assay. The detection of apoptotic DNA fragments after 72 hours treatment of MCF-7 cells with 100 μM of the derivatives. Electrophoretic analysis revealed the appearance of DNA fragments corresponded to mono- (170 -200) and oligonucleosoms, confirming an apoptotic mechanism of cell death for the all pentathiepines tested. The increment of FITC -labeled annexin V and propidium iodide fluorescence in MCF-7 cells treated with the pentathiepines for 24 h and 48 h, relative to control of untreated MCF-7 cells (%). 3000 2000 1000 750 540 -600 360 -400 180 -200 1 - designates DNA markers 2 - control cells; 3 - 1%DMSO; 4 - empty; 5 - 3 d; 6 - varacin analog.
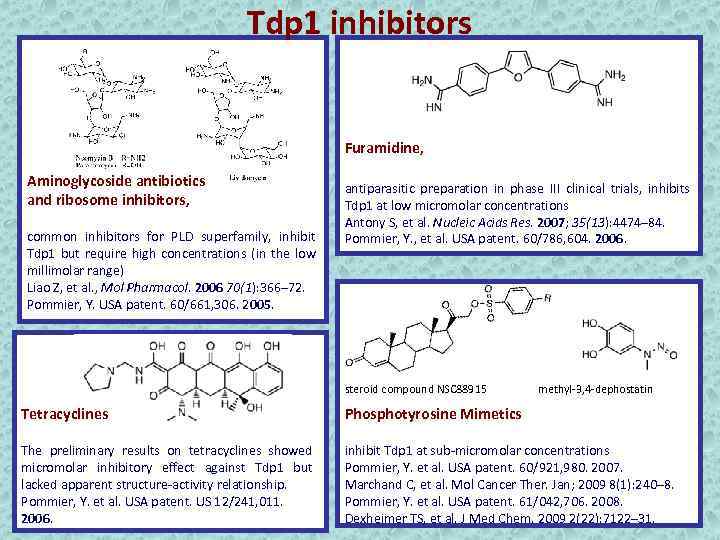 Tdp 1 inhibitors Furamidine, Aminoglycoside antibiotics and ribosome inhibitors, common inhibitors for PLD superfamily, inhibit Tdp 1 but require high concentrations (in the low millimolar range) Liao Z, et al. , Mol Pharmacol. 2006 70(1): 366– 72. Pommier, Y. USA patent. 60/661, 306. 2005. antiparasitic preparation in phase III clinical trials, inhibits Tdp 1 at low micromolar concentrations Antony S, et al. Nucleic Acids Res. 2007; 35(13): 4474– 84. Pommier, Y. , et al. USA patent. 60/786, 604. 2006. steroid compound NSC 88915 methyl-3, 4 -dephostatin Tetracyclines Phosphotyrosine Mimetics The preliminary results on tetracyclines showed micromolar inhibitory effect against Tdp 1 but lacked apparent structure-activity relationship. Pommier, Y. et al. USA patent. US 12/241, 011. 2006. inhibit Tdp 1 at sub-micromolar concentrations Pommier, Y. et al. USA patent. 60/921, 980. 2007. Marchand C, et al. Mol Cancer Ther. Jan; 2009 8(1): 240– 8. Pommier, Y. et al. USA patent. 61/042, 706. 2008. Dexheimer TS, et al. J Med Chem. 2009 2(22): 7122– 31.
Tdp 1 inhibitors Furamidine, Aminoglycoside antibiotics and ribosome inhibitors, common inhibitors for PLD superfamily, inhibit Tdp 1 but require high concentrations (in the low millimolar range) Liao Z, et al. , Mol Pharmacol. 2006 70(1): 366– 72. Pommier, Y. USA patent. 60/661, 306. 2005. antiparasitic preparation in phase III clinical trials, inhibits Tdp 1 at low micromolar concentrations Antony S, et al. Nucleic Acids Res. 2007; 35(13): 4474– 84. Pommier, Y. , et al. USA patent. 60/786, 604. 2006. steroid compound NSC 88915 methyl-3, 4 -dephostatin Tetracyclines Phosphotyrosine Mimetics The preliminary results on tetracyclines showed micromolar inhibitory effect against Tdp 1 but lacked apparent structure-activity relationship. Pommier, Y. et al. USA patent. US 12/241, 011. 2006. inhibit Tdp 1 at sub-micromolar concentrations Pommier, Y. et al. USA patent. 60/921, 980. 2007. Marchand C, et al. Mol Cancer Ther. Jan; 2009 8(1): 240– 8. Pommier, Y. et al. USA patent. 61/042, 706. 2008. Dexheimer TS, et al. J Med Chem. 2009 2(22): 7122– 31.


