2875274a263268063e2f41f63a0c4580.ppt
- Количество слайдов: 36
 http: //www. nearingzero. net (nz 122. jpg)
http: //www. nearingzero. net (nz 122. jpg)
 Announcements ·Soon I’ll be asking you to tell me which Issue you will be handing in. ·Good news: China is about to overtake US in production of electronic waste. ·Grades spreadsheets wil be available here “soon: ” http: //campus. mst. edu/physics/courses/6/grades/.
Announcements ·Soon I’ll be asking you to tell me which Issue you will be handing in. ·Good news: China is about to overtake US in production of electronic waste. ·Grades spreadsheets wil be available here “soon: ” http: //campus. mst. edu/physics/courses/6/grades/.
 Ozone
Ozone
 Hole In The Sky Your comments on this video? Some observations: Where was the science (there’s more than you first think)? Gloom and doom. I almost felt they were happy when North America was affected. uva/uvb/uvc “…mankind has changed the laws of nature…” “…you couldn’t quantify it…” but then he did. Left me unsatisfied and wanting answers.
Hole In The Sky Your comments on this video? Some observations: Where was the science (there’s more than you first think)? Gloom and doom. I almost felt they were happy when North America was affected. uva/uvb/uvc “…mankind has changed the laws of nature…” “…you couldn’t quantify it…” but then he did. Left me unsatisfied and wanting answers.
 We have seen how physicists define work… …let’s talk about energy.
We have seen how physicists define work… …let’s talk about energy.
 Work and Energy: Kinetic Energy A bit of review… Think of work and energy as two sides of the same coin. A very important theory of physics says that work and energy can be converted back and forth into each other. In grade school, you probably learned about two of the main kinds of energy: kinetic energy and potential energy. An object has kinetic energy whenever it is moving.
Work and Energy: Kinetic Energy A bit of review… Think of work and energy as two sides of the same coin. A very important theory of physics says that work and energy can be converted back and forth into each other. In grade school, you probably learned about two of the main kinds of energy: kinetic energy and potential energy. An object has kinetic energy whenever it is moving.
 Work and Energy: Potential Energy In grade school, you probably learned that if an object has potential energy, it has the ability to do work. It has the "potential" to do work; hence the name. Potential energy is stored energy. Potential energy does not involve motion (that would be kinetic energy).
Work and Energy: Potential Energy In grade school, you probably learned that if an object has potential energy, it has the ability to do work. It has the "potential" to do work; hence the name. Potential energy is stored energy. Potential energy does not involve motion (that would be kinetic energy).
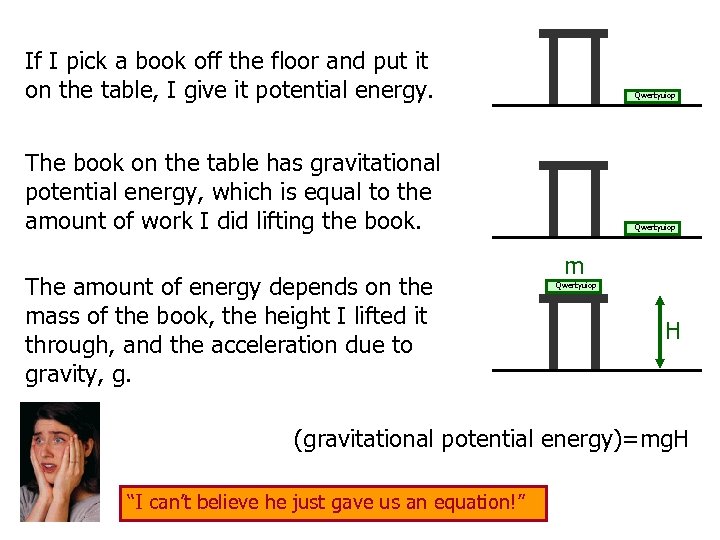 If I pick a book off the floor and put it on the table, I give it potential energy. Qwertyuiop The book on the table has gravitational potential energy, which is equal to the amount of work I did lifting the book. The amount of energy depends on the mass of the book, the height I lifted it through, and the acceleration due to gravity, g. Qwertyuiop m Qwertyuiop H (gravitational potential energy)=mg. H “I can’t believe he just gave us an equation!”
If I pick a book off the floor and put it on the table, I give it potential energy. Qwertyuiop The book on the table has gravitational potential energy, which is equal to the amount of work I did lifting the book. The amount of energy depends on the mass of the book, the height I lifted it through, and the acceleration due to gravity, g. Qwertyuiop m Qwertyuiop H (gravitational potential energy)=mg. H “I can’t believe he just gave us an equation!”
 F Where does PE=mg. H come from? H Remember F=ma? In this case, the weight of the book is equal to mg (F=ma=mg). Qwertyuiop mg The force I have to exert to lift it at a constant rate, by Newton's second law, is also mg. The height I lift it is H, so W=(Force times Distance)=mg. H. That's the amount of work I do on the book, and the amount by which its potential energy is increased. “Noooo! We just did a Physics derivation!”
F Where does PE=mg. H come from? H Remember F=ma? In this case, the weight of the book is equal to mg (F=ma=mg). Qwertyuiop mg The force I have to exert to lift it at a constant rate, by Newton's second law, is also mg. The height I lift it is H, so W=(Force times Distance)=mg. H. That's the amount of work I do on the book, and the amount by which its potential energy is increased. “Noooo! We just did a Physics derivation!”
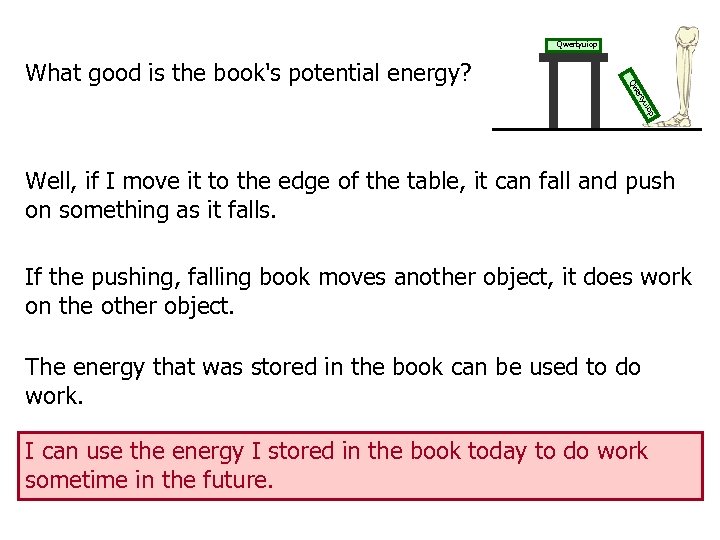 Qwertyuiop yu ert Qw What good is the book's potential energy? Well, if I move it to the edge of the table, it can fall and push on something as it falls. If the pushing, falling book moves another object, it does work on the other object. The energy that was stored in the book can be used to do work. I can use the energy I stored in the book today to do work sometime in the future.
Qwertyuiop yu ert Qw What good is the book's potential energy? Well, if I move it to the edge of the table, it can fall and push on something as it falls. If the pushing, falling book moves another object, it does work on the other object. The energy that was stored in the book can be used to do work. I can use the energy I stored in the book today to do work sometime in the future.
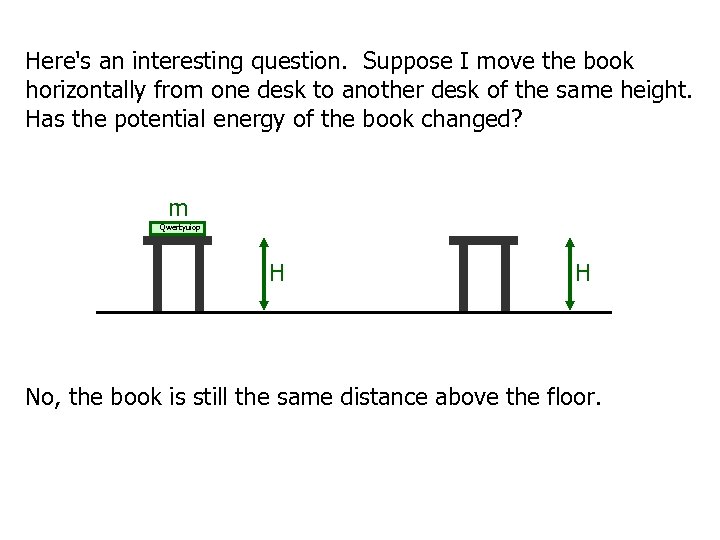 Here's an interesting question. Suppose I move the book horizontally from one desk to another desk of the same height. Has the potential energy of the book changed? m Qwertyuiop H H No, the book is still the same distance above the floor.
Here's an interesting question. Suppose I move the book horizontally from one desk to another desk of the same height. Has the potential energy of the book changed? m Qwertyuiop H H No, the book is still the same distance above the floor.
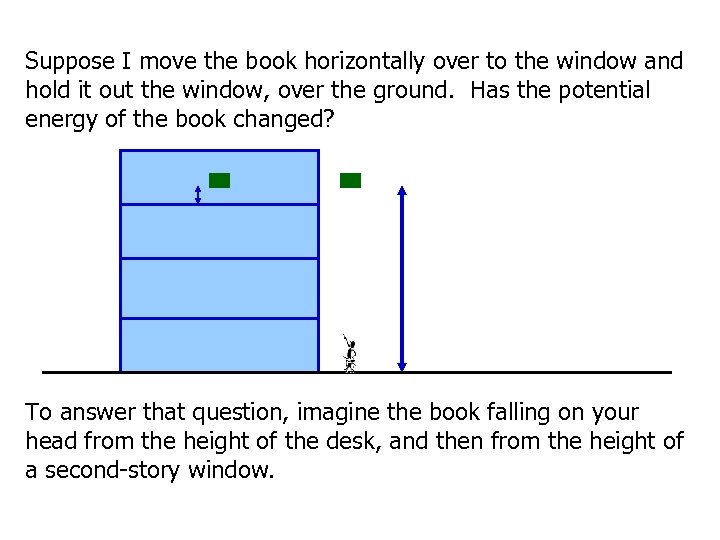 Suppose I move the book horizontally over to the window and hold it out the window, over the ground. Has the potential energy of the book changed? To answer that question, imagine the book falling on your head from the height of the desk, and then from the height of a second-story window.
Suppose I move the book horizontally over to the window and hold it out the window, over the ground. Has the potential energy of the book changed? To answer that question, imagine the book falling on your head from the height of the desk, and then from the height of a second-story window.
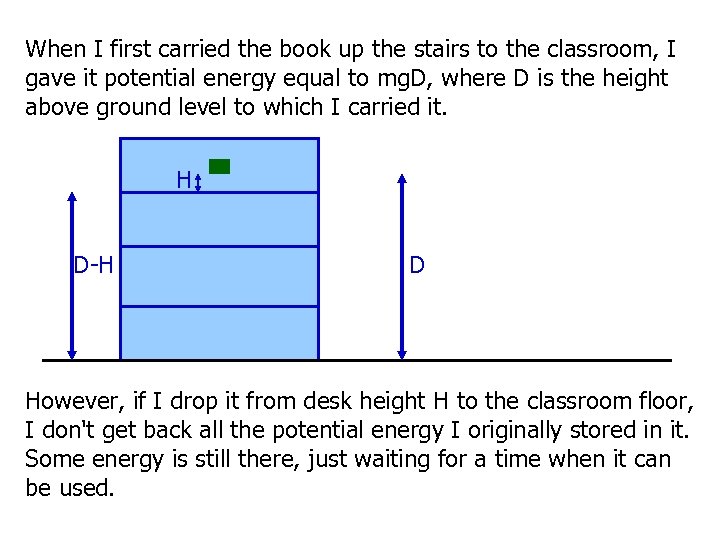 When I first carried the book up the stairs to the classroom, I gave it potential energy equal to mg. D, where D is the height above ground level to which I carried it. H D-H D However, if I drop it from desk height H to the classroom floor, I don't get back all the potential energy I originally stored in it. Some energy is still there, just waiting for a time when it can be used.
When I first carried the book up the stairs to the classroom, I gave it potential energy equal to mg. D, where D is the height above ground level to which I carried it. H D-H D However, if I drop it from desk height H to the classroom floor, I don't get back all the potential energy I originally stored in it. Some energy is still there, just waiting for a time when it can be used.
 There are other kinds of potential energy. One is chemical potential energy. USS Atlanta (CL-104), 610 feet long. (Edward Jones Dome 600 x 747 feet) TNT has the ability to do work. Something has to trigger the release of energy, and when that happens, TNT can move lots of mass around. Chemical potential energy is stored in foods, batteries, explosives, etc.
There are other kinds of potential energy. One is chemical potential energy. USS Atlanta (CL-104), 610 feet long. (Edward Jones Dome 600 x 747 feet) TNT has the ability to do work. Something has to trigger the release of energy, and when that happens, TNT can move lots of mass around. Chemical potential energy is stored in foods, batteries, explosives, etc.
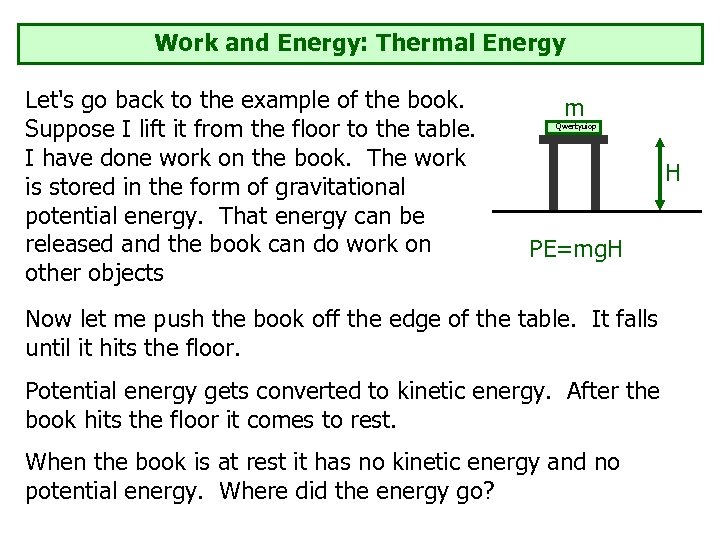 Work and Energy: Thermal Energy Let's go back to the example of the book. Suppose I lift it from the floor to the table. I have done work on the book. The work is stored in the form of gravitational potential energy. That energy can be released and the book can do work on other objects m Qwertyuiop H PE=mg. H Now let me push the book off the edge of the table. It falls until it hits the floor. Potential energy gets converted to kinetic energy. After the book hits the floor it comes to rest. When the book is at rest it has no kinetic energy and no potential energy. Where did the energy go?
Work and Energy: Thermal Energy Let's go back to the example of the book. Suppose I lift it from the floor to the table. I have done work on the book. The work is stored in the form of gravitational potential energy. That energy can be released and the book can do work on other objects m Qwertyuiop H PE=mg. H Now let me push the book off the edge of the table. It falls until it hits the floor. Potential energy gets converted to kinetic energy. After the book hits the floor it comes to rest. When the book is at rest it has no kinetic energy and no potential energy. Where did the energy go?
 Remember conservation of momentum, which I talked about earlier? In every case physicists have observed, total momentum before and after any event are always the same. You have to make sure you find all the moving objects to account for all the momentum, plus any external forces that “remove” momentum, but momentum is always conserved. Because momentum is always conserved, because conservation of momentum has a theoretical basis (remember Emmy Noether), and because it has been used successfully over and over again to predict new phenomena, we call it a "law. "
Remember conservation of momentum, which I talked about earlier? In every case physicists have observed, total momentum before and after any event are always the same. You have to make sure you find all the moving objects to account for all the momentum, plus any external forces that “remove” momentum, but momentum is always conserved. Because momentum is always conserved, because conservation of momentum has a theoretical basis (remember Emmy Noether), and because it has been used successfully over and over again to predict new phenomena, we call it a "law. "
 There is another conservation law—the Law of Conservation of Energy is neither created nor destroyed, but only changed from one form into another. * So let's go back to the falling book. *In a closed system.
There is another conservation law—the Law of Conservation of Energy is neither created nor destroyed, but only changed from one form into another. * So let's go back to the falling book. *In a closed system.
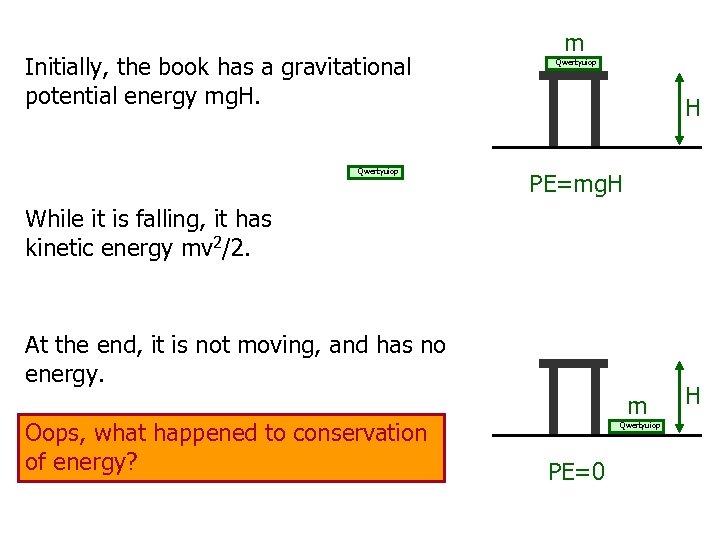 Initially, the book has a gravitational potential energy mg. H. Qwertyuiop m Qwertyuiop H PE=mg. H While it is falling, it has kinetic energy mv 2/2. At the end, it is not moving, and has no energy. Oops, what happened to conservation of energy? m Qwertyuiop PE=0 H
Initially, the book has a gravitational potential energy mg. H. Qwertyuiop m Qwertyuiop H PE=mg. H While it is falling, it has kinetic energy mv 2/2. At the end, it is not moving, and has no energy. Oops, what happened to conservation of energy? m Qwertyuiop PE=0 H
 That’s all time I will spend on energy today. I hope that unanswered question doesn’t hurt anyone’s brain. (http: //www. nearingzero. net/screen_res/nz 233. jpg)
That’s all time I will spend on energy today. I hope that unanswered question doesn’t hurt anyone’s brain. (http: //www. nearingzero. net/screen_res/nz 233. jpg)
 Your attendance assignment for today: should we worry about what is in the atmosphere? Just answer yes or no. If “no, ” you are done. If “yes, ” please give me one reason to justify your answer. Hand in your answer (plus name!) when we take a break. I will not penalize anybody who answers “no” or who chooses to put their name on a blank piece of paper. * *…which is the same as answering “no. ”
Your attendance assignment for today: should we worry about what is in the atmosphere? Just answer yes or no. If “no, ” you are done. If “yes, ” please give me one reason to justify your answer. Hand in your answer (plus name!) when we take a break. I will not penalize anybody who answers “no” or who chooses to put their name on a blank piece of paper. * *…which is the same as answering “no. ”
 Antarctica… Why would anybody want to go there? lots of images in this lecture; click on an image to see the web page where I found it Why would anybody want to do science there?
Antarctica… Why would anybody want to go there? lots of images in this lecture; click on an image to see the web page where I found it Why would anybody want to do science there?
 Why waste resources (money) doing science in Antarctica? A little while ago, I asked if we should worry about what we put in the atmosphere. If your answer is “yes, ” would you like to share the reason you put down?
Why waste resources (money) doing science in Antarctica? A little while ago, I asked if we should worry about what we put in the atmosphere. If your answer is “yes, ” would you like to share the reason you put down?
 There’s not much of it between us and… … nothing.
There’s not much of it between us and… … nothing.
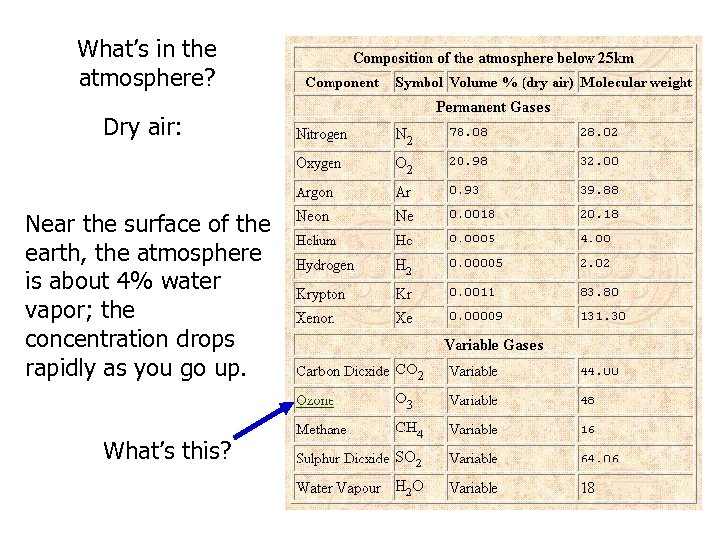 What’s in the atmosphere? Dry air: Near the surface of the earth, the atmosphere is about 4% water vapor; the concentration drops rapidly as you go up. What’s this?
What’s in the atmosphere? Dry air: Near the surface of the earth, the atmosphere is about 4% water vapor; the concentration drops rapidly as you go up. What’s this?
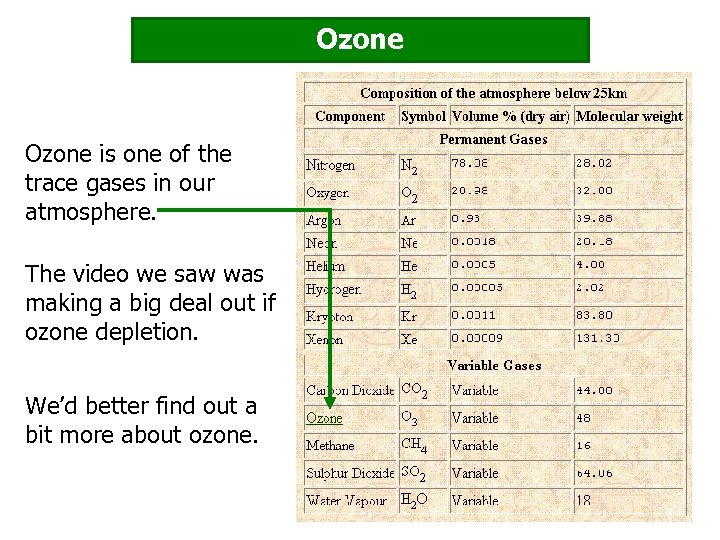 Ozone is one of the trace gases in our atmosphere. The video we saw was making a big deal out if ozone depletion. We’d better find out a bit more about ozone.
Ozone is one of the trace gases in our atmosphere. The video we saw was making a big deal out if ozone depletion. We’d better find out a bit more about ozone.
 Ozone was “discovered” in 1840 by German-born Swiss chemist Christian Freidrich Schonbein was passing electricity through a flask of water. He noticed a strange odor. He attributed the odor to a new substance, which he named ozone after the Greek word “ozein, ” “to smell” (i. e. , “to stink). Schonbein identified ozone as a component of the lower atmosphere. Actually, von Marum synthesized ozone in 1785 using some spectacularly dangerous apparatus, but didn’t identify what he had found.
Ozone was “discovered” in 1840 by German-born Swiss chemist Christian Freidrich Schonbein was passing electricity through a flask of water. He noticed a strange odor. He attributed the odor to a new substance, which he named ozone after the Greek word “ozein, ” “to smell” (i. e. , “to stink). Schonbein identified ozone as a component of the lower atmosphere. Actually, von Marum synthesized ozone in 1785 using some spectacularly dangerous apparatus, but didn’t identify what he had found.
 Schonbein is also noteworthy for his invention of guncotton. According to www. anecdotage. com/index. php? aid=4466: “Though strictly forbidden by his wife from conducting experiments in their home, the German chemist Christian F. Schonbein sometimes disobeyed her when she wasn't around. ” “While experimenting in the kitchen one day in 1845, Schonbein accidentally spilled a mixture of nitric and sulfuric acids. ” “In a panic, he grabbed his wife's cotton apron and mopped up the mess. He then hung it over the stove to dry before its unsuspecting owner came home. ” “Schonbein's deception went unnoticed - until the apron spontaneously ignited and burned so quickly that virtually disappeared in a flash. Fortunately, his wife wasn't wearing it at the time. ” Alas, history did not record Mrs. Schonbein’s response.
Schonbein is also noteworthy for his invention of guncotton. According to www. anecdotage. com/index. php? aid=4466: “Though strictly forbidden by his wife from conducting experiments in their home, the German chemist Christian F. Schonbein sometimes disobeyed her when she wasn't around. ” “While experimenting in the kitchen one day in 1845, Schonbein accidentally spilled a mixture of nitric and sulfuric acids. ” “In a panic, he grabbed his wife's cotton apron and mopped up the mess. He then hung it over the stove to dry before its unsuspecting owner came home. ” “Schonbein's deception went unnoticed - until the apron spontaneously ignited and burned so quickly that virtually disappeared in a flash. Fortunately, his wife wasn't wearing it at the time. ” Alas, history did not record Mrs. Schonbein’s response.
 Anyway, back to ozone. . . Ozone is an unstable form of oxygen. Most of the oxygen in the atmosphere is in the form of (relatively) stable O 2 molecules. Ozone (O 3) is an unstable bluish gas, and forms an explosive blue liquid or dangerous purple crystals if you cool it enough. Ozone is characterized by its unique odor, which is often noticed during electrical storms and around high-voltage electrical equipment, such as photocopiers and laser printers.
Anyway, back to ozone. . . Ozone is an unstable form of oxygen. Most of the oxygen in the atmosphere is in the form of (relatively) stable O 2 molecules. Ozone (O 3) is an unstable bluish gas, and forms an explosive blue liquid or dangerous purple crystals if you cool it enough. Ozone is characterized by its unique odor, which is often noticed during electrical storms and around high-voltage electrical equipment, such as photocopiers and laser printers.
 Ozone both harms and protects life on earth. Ozone is an oxidizing agent. Its excess oxygen atoms, known as free radicals, oxidize other materials and are associated with the aging process. Ozone in the troposphere (the part of the atmosphere we live in) causes damage to lung tissue, plants, rubber, and plastic. Ozone also reacts with chemical from automobile exhausts to produce other dangerous chemicals. Although ozone pollution originates primarily in urban areas, it can be carried for many kilometers into adjoining forest and crop regions. Ozone has been demonstrated to damage crops in the United States, Canada, and Europe. An estimated 90% of crop losses due to pollution is due to ozone.
Ozone both harms and protects life on earth. Ozone is an oxidizing agent. Its excess oxygen atoms, known as free radicals, oxidize other materials and are associated with the aging process. Ozone in the troposphere (the part of the atmosphere we live in) causes damage to lung tissue, plants, rubber, and plastic. Ozone also reacts with chemical from automobile exhausts to produce other dangerous chemicals. Although ozone pollution originates primarily in urban areas, it can be carried for many kilometers into adjoining forest and crop regions. Ozone has been demonstrated to damage crops in the United States, Canada, and Europe. An estimated 90% of crop losses due to pollution is due to ozone.
 Ozone near the surface of the earth is generally “bad ozone. ”* Ozone in the stratosphere is “good ozone. ” The stratosphere is the part of the atmosphere that begins roughly 10 to 15 km above the earth. The stratosphere is closest to the surface of the earth near the poles. How far is 12 kilometers? Ozone in the stratosphere shields the earth’s surface by absorbing harmful ultraviolet radiation. *Nevertheless, you can buy your own personal ozone generator here.
Ozone near the surface of the earth is generally “bad ozone. ”* Ozone in the stratosphere is “good ozone. ” The stratosphere is the part of the atmosphere that begins roughly 10 to 15 km above the earth. The stratosphere is closest to the surface of the earth near the poles. How far is 12 kilometers? Ozone in the stratosphere shields the earth’s surface by absorbing harmful ultraviolet radiation. *Nevertheless, you can buy your own personal ozone generator here.
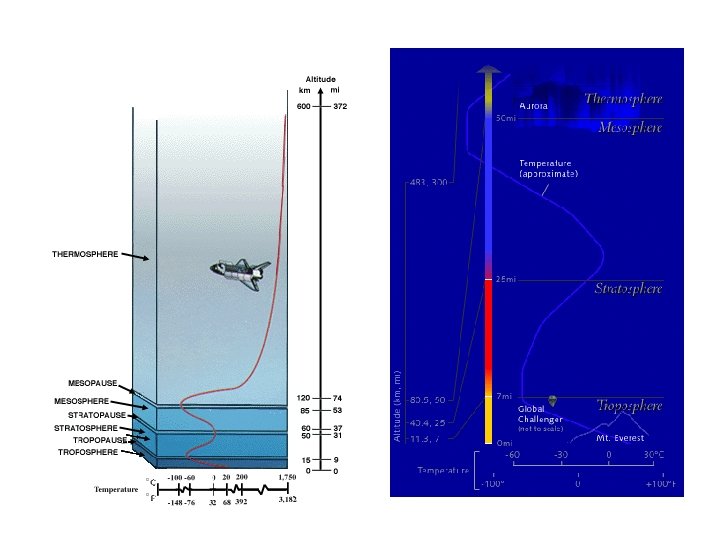
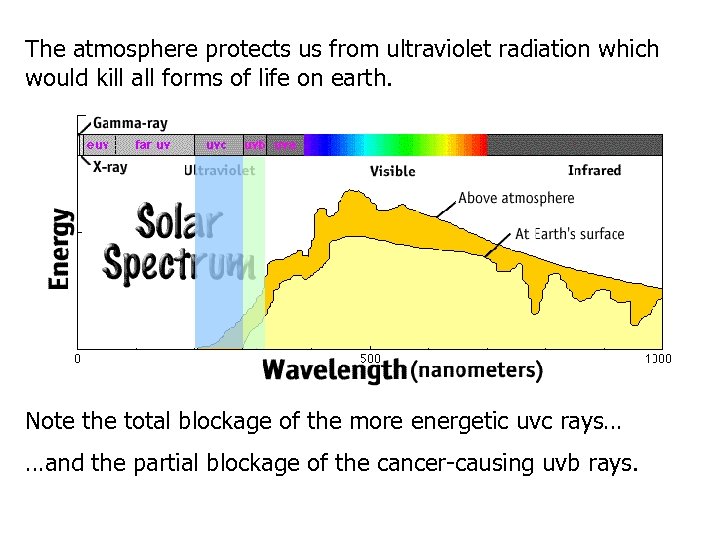 The atmosphere protects us from ultraviolet radiation which would kill all forms of life on earth. Note the total blockage of the more energetic uvc rays… …and the partial blockage of the cancer-causing uvb rays.
The atmosphere protects us from ultraviolet radiation which would kill all forms of life on earth. Note the total blockage of the more energetic uvc rays… …and the partial blockage of the cancer-causing uvb rays.
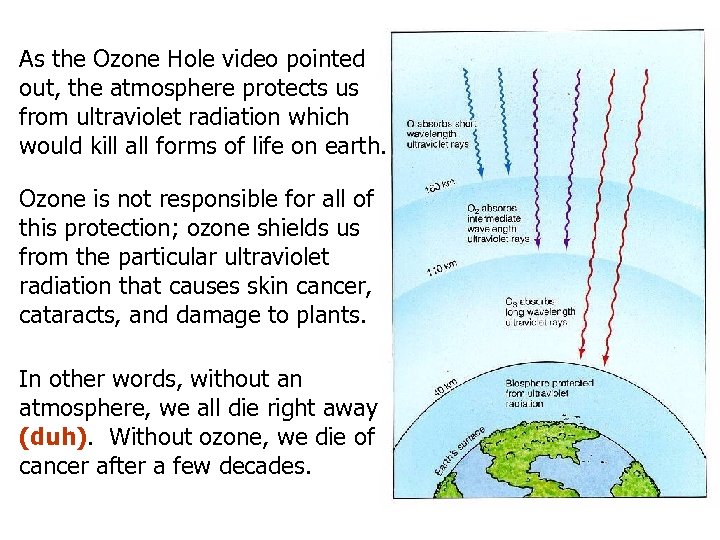 As the Ozone Hole video pointed out, the atmosphere protects us from ultraviolet radiation which would kill all forms of life on earth. Ozone is not responsible for all of this protection; ozone shields us from the particular ultraviolet radiation that causes skin cancer, cataracts, and damage to plants. In other words, without an atmosphere, we all die right away (duh). Without ozone, we die of cancer after a few decades.
As the Ozone Hole video pointed out, the atmosphere protects us from ultraviolet radiation which would kill all forms of life on earth. Ozone is not responsible for all of this protection; ozone shields us from the particular ultraviolet radiation that causes skin cancer, cataracts, and damage to plants. In other words, without an atmosphere, we all die right away (duh). Without ozone, we die of cancer after a few decades.
 UV radiation causes cancer by damaging DNA, which is vital to all living organisms, is damaged by those wavelengths of UV radiation that the ozone shields us from. As the video pointed out, a 60% decrease in the ozone layer resulted in a 120% increase in damaging UV radiation. According to the EPA, a 1% decrease in the ozone layer results in a 2% increase in uv radiation at the earth’s surface, which in turn would be increase skin cancer rates by 8%--or about 100, 000 “extra” people every year in the US. Even relatively small decreases in the ozone layer are worrisome, especially if you are unfortunate enough to be one of those 100, 000.
UV radiation causes cancer by damaging DNA, which is vital to all living organisms, is damaged by those wavelengths of UV radiation that the ozone shields us from. As the video pointed out, a 60% decrease in the ozone layer resulted in a 120% increase in damaging UV radiation. According to the EPA, a 1% decrease in the ozone layer results in a 2% increase in uv radiation at the earth’s surface, which in turn would be increase skin cancer rates by 8%--or about 100, 000 “extra” people every year in the US. Even relatively small decreases in the ozone layer are worrisome, especially if you are unfortunate enough to be one of those 100, 000.
 “If ozone in the troposphere (where we live) is bad, and ozone in the stratosphere is good, why not just take the bad ozone and put it where it will do some good? ” Ozone does have important uses. Good question! Ozone in the air we breathe is increasing, and protective ozone is being destroyed, but because ozone is so fragile (remember, it is highly unstable and quick to react), it doesn’t get from “down here” to “up there. ” 2008: got to here
“If ozone in the troposphere (where we live) is bad, and ozone in the stratosphere is good, why not just take the bad ozone and put it where it will do some good? ” Ozone does have important uses. Good question! Ozone in the air we breathe is increasing, and protective ozone is being destroyed, but because ozone is so fragile (remember, it is highly unstable and quick to react), it doesn’t get from “down here” to “up there. ” 2008: got to here
 Homework Assignment #7 (due in one week). Go to http: //www. archive. org. In the WAYBACK MACHINE box, type http: //www. word. com/machine/dray. Click “Take Me Back. ” Read the October 21, 2000 article. Print out ONLY the last page and turn it in (with your name on it, of course). The pages are very slow to load. A few pictures and animations are missing. Be patient! How do you know when your are on the last page? When you can’t go “deeper!” 2012: got to here
Homework Assignment #7 (due in one week). Go to http: //www. archive. org. In the WAYBACK MACHINE box, type http: //www. word. com/machine/dray. Click “Take Me Back. ” Read the October 21, 2000 article. Print out ONLY the last page and turn it in (with your name on it, of course). The pages are very slow to load. A few pictures and animations are missing. Be patient! How do you know when your are on the last page? When you can’t go “deeper!” 2012: got to here


