9bf5652001d6029c4b46008b874ab997.ppt
- Количество слайдов: 23
 HPV
HPV
 Carcinoma of the Cervix § Many risk factors for development of cervical cancer. • no routinely used positive predictive biological markers, which identify women at risk of developing high-grade lesions and ultimately invasive cancer.
Carcinoma of the Cervix § Many risk factors for development of cervical cancer. • no routinely used positive predictive biological markers, which identify women at risk of developing high-grade lesions and ultimately invasive cancer.
 Human Papillomavirus (HPV) • • • Strong association with development of invasive cancer. >70 types of HPV. Low risk (6, 11). High risk (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68). Exposure to HPV is followed by a serological response to viral capsid proteins (VLPs). • Immune response is assoc. with persistent HPV infection and is type specific.
Human Papillomavirus (HPV) • • • Strong association with development of invasive cancer. >70 types of HPV. Low risk (6, 11). High risk (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, 68). Exposure to HPV is followed by a serological response to viral capsid proteins (VLPs). • Immune response is assoc. with persistent HPV infection and is type specific.
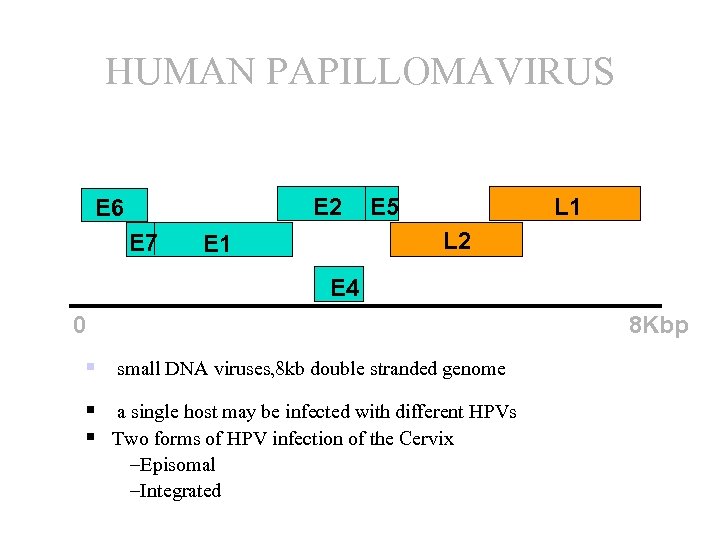 HUMAN PAPILLOMAVIRUS E 2 E 6 E 7 E 5 L 1 L 2 E 1 E 4 8 Kbp 0 § small DNA viruses, 8 kb double stranded genome § a single host may be infected with different HPVs § Two forms of HPV infection of the Cervix –Episomal –Integrated
HUMAN PAPILLOMAVIRUS E 2 E 6 E 7 E 5 L 1 L 2 E 1 E 4 8 Kbp 0 § small DNA viruses, 8 kb double stranded genome § a single host may be infected with different HPVs § Two forms of HPV infection of the Cervix –Episomal –Integrated
 HPV • Integration of HPV DNA into host loss of E 2 orf. • Transcription of E 6 and E 7 is unregulated. • Transformation events within the cell. • Checkpoint for cell proliferation and transcription is lost.
HPV • Integration of HPV DNA into host loss of E 2 orf. • Transcription of E 6 and E 7 is unregulated. • Transformation events within the cell. • Checkpoint for cell proliferation and transcription is lost.
 HPV • Expressed E 6 and E 7 proteins can then interact with other tumour suppressor genes including p 53 and p. RB uncontrolled cellular proliferation and malignant transformation. • 3 splice variants of E 6 HPV 16 recognised: E 6 I, II and III.
HPV • Expressed E 6 and E 7 proteins can then interact with other tumour suppressor genes including p 53 and p. RB uncontrolled cellular proliferation and malignant transformation. • 3 splice variants of E 6 HPV 16 recognised: E 6 I, II and III.
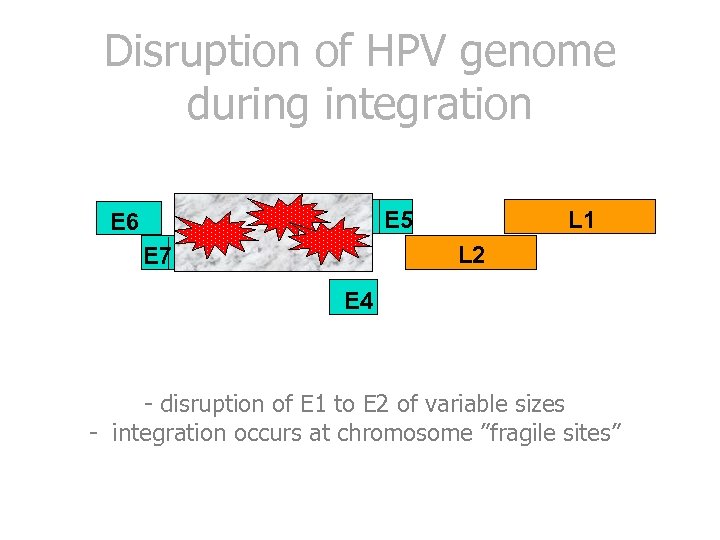 Disruption of HPV genome during integration E 2 E 6 E 7 E 5 L 1 L 2 E 1 E 4 - disruption of E 1 to E 2 of variable sizes - integration occurs at chromosome ”fragile sites”
Disruption of HPV genome during integration E 2 E 6 E 7 E 5 L 1 L 2 E 1 E 4 - disruption of E 1 to E 2 of variable sizes - integration occurs at chromosome ”fragile sites”
 Experimental evidence of HPV transforming capacity RAFT culture experiments with wild type and mutant E 6/E 7 constructs E 6 mutant: in RAFT culture
Experimental evidence of HPV transforming capacity RAFT culture experiments with wild type and mutant E 6/E 7 constructs E 6 mutant: in RAFT culture
 HPV Cells infected with oncogenic HPV types Immortalisation Uncontrolled cell proliferation
HPV Cells infected with oncogenic HPV types Immortalisation Uncontrolled cell proliferation
 Carcinoma of the cervix § MOLECULAR ONCOLOGY § over 95% of cervical SCCs associated with high risk HPV types (16, 18, 31, 33, 45); 40 -70% of adenocarcinomas. § HPVs also found in CIN: § • • • 4 -6% of normal women HPV 6 and 11 positive. CIN 1: 10 - 30% HPV 6 &11 positive. CIN 2 - 3: 75 - 80% HPV 16, 18, 31, 33 positive; 1 - 5% HPV 6, 11 positive. HPV E 6 and E 7 regions can transform epithelial cells and increase cellular levels of cyclins A, B and p 34 -cdc 2 and cyclin E.
Carcinoma of the cervix § MOLECULAR ONCOLOGY § over 95% of cervical SCCs associated with high risk HPV types (16, 18, 31, 33, 45); 40 -70% of adenocarcinomas. § HPVs also found in CIN: § • • • 4 -6% of normal women HPV 6 and 11 positive. CIN 1: 10 - 30% HPV 6 &11 positive. CIN 2 - 3: 75 - 80% HPV 16, 18, 31, 33 positive; 1 - 5% HPV 6, 11 positive. HPV E 6 and E 7 regions can transform epithelial cells and increase cellular levels of cyclins A, B and p 34 -cdc 2 and cyclin E.
 HPV analysis • Who do we screen? – All Women? – HPV as a triage? • How do we screen? • Does HPV analysis give prognostic information? • HPV and other novel biomarkers of disease
HPV analysis • Who do we screen? – All Women? – HPV as a triage? • How do we screen? • Does HPV analysis give prognostic information? • HPV and other novel biomarkers of disease
 Future role for HPV screening • Post introduction of HPV vaccine § vaccines being produced to target HPV 16 and 18 E 6/E 7 regions. § requirement to monitor HPV status pre and post -vaccination. § possibility of using recombinant § anti-sense PNAs to specifically § target HPV E 6 and E 6 splice variants.
Future role for HPV screening • Post introduction of HPV vaccine § vaccines being produced to target HPV 16 and 18 E 6/E 7 regions. § requirement to monitor HPV status pre and post -vaccination. § possibility of using recombinant § anti-sense PNAs to specifically § target HPV E 6 and E 6 splice variants.
 How do we screen? • HPV analysis – Type – Load – Viral integration
How do we screen? • HPV analysis – Type – Load – Viral integration
 HPV analysis • Technologies available – Hybrid Capture II – PCR generic, (incl. PGYM, GP 5 and 6, green) – Type specific DNA PCR • • • Solution phase PCR Taq. Man PCR NASBA (HPV proofer) In-situ hybridisation (ISH) Sequence genotyping In-cell PCR – ICC SYBR
HPV analysis • Technologies available – Hybrid Capture II – PCR generic, (incl. PGYM, GP 5 and 6, green) – Type specific DNA PCR • • • Solution phase PCR Taq. Man PCR NASBA (HPV proofer) In-situ hybridisation (ISH) Sequence genotyping In-cell PCR – ICC SYBR
 HPV analysis • Hybrid Capture II – Liquid based system. – Low and high risk type analysis. – No information in relation to integration. – Indirect load information but NOT quantitative.
HPV analysis • Hybrid Capture II – Liquid based system. – Low and high risk type analysis. – No information in relation to integration. – Indirect load information but NOT quantitative.
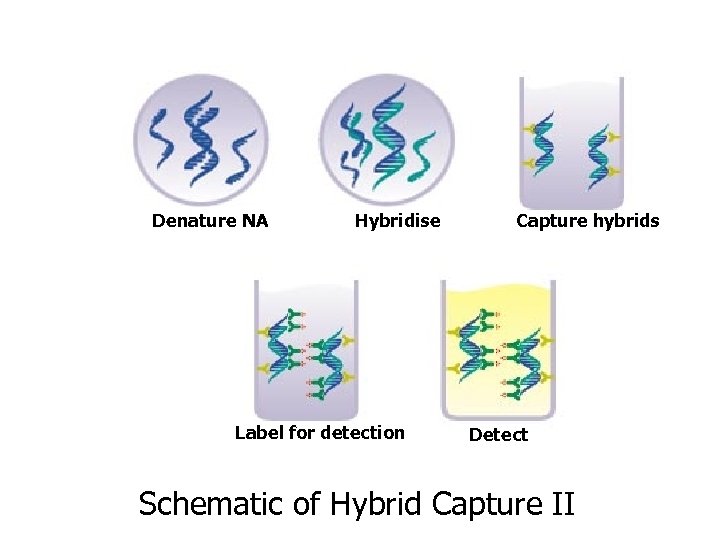 Denature NA Hybridise Label for detection Capture hybrids Detect Schematic of Hybrid Capture II
Denature NA Hybridise Label for detection Capture hybrids Detect Schematic of Hybrid Capture II
 HPV analysis Hybrid Capture II §Recommended cut-off for the HC-II test is 1 pg viral DNA per ml of buffer, equivalent to about 5000 viral genomes. §This cut-off value has been reduced to 0. 2 pg/ml but with the introduction of false positives (Peyton et al). §Data comparing PCR with HC-II found PCR identified HPV in 24. 5% of samples, while HC-II detected HPV in 13% using the recommended cut-off of 1 pg/ml, and in 22. 1% using a cut-off of 0. 2 pg/ml.
HPV analysis Hybrid Capture II §Recommended cut-off for the HC-II test is 1 pg viral DNA per ml of buffer, equivalent to about 5000 viral genomes. §This cut-off value has been reduced to 0. 2 pg/ml but with the introduction of false positives (Peyton et al). §Data comparing PCR with HC-II found PCR identified HPV in 24. 5% of samples, while HC-II detected HPV in 13% using the recommended cut-off of 1 pg/ml, and in 22. 1% using a cut-off of 0. 2 pg/ml.
 HPV analysis -PCR • PCR generic / consensus – GP 5 and 6 – PGYM – MY 09/11 – SPF 10 – GP 5 and 6 + SYBR green
HPV analysis -PCR • PCR generic / consensus – GP 5 and 6 – PGYM – MY 09/11 – SPF 10 – GP 5 and 6 + SYBR green
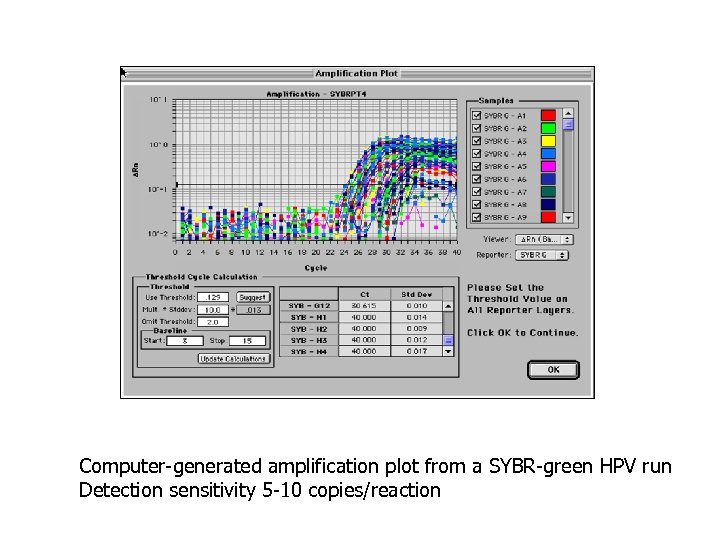 Computer-generated amplification plot from a SYBR-green HPV run Detection sensitivity 5 -10 copies/reaction
Computer-generated amplification plot from a SYBR-green HPV run Detection sensitivity 5 -10 copies/reaction
 HPV analysis • Type specific PCR – Solution phase PCR – Taq Man q(PCR) – NASBA (HPV proofer) – In-situ hybridisation – HPV genotyping
HPV analysis • Type specific PCR – Solution phase PCR – Taq Man q(PCR) – NASBA (HPV proofer) – In-situ hybridisation – HPV genotyping
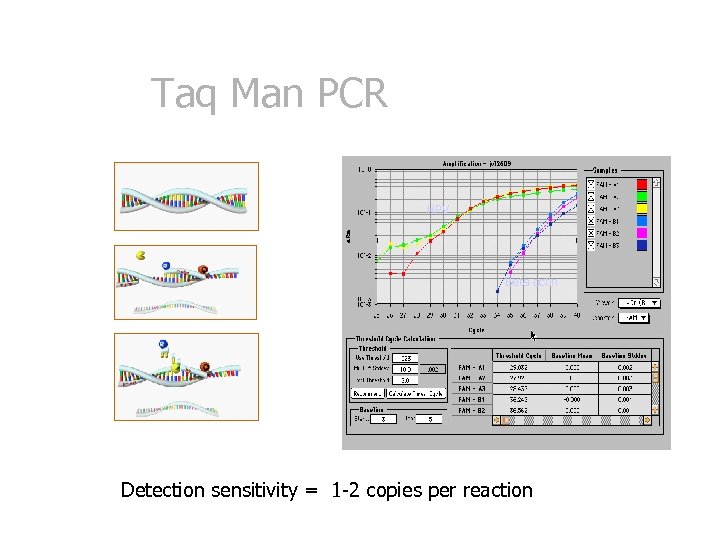 Taq Man PCR HPV Beta actin Detection sensitivity = 1 -2 copies per reaction
Taq Man PCR HPV Beta actin Detection sensitivity = 1 -2 copies per reaction
 HPV analysis • In-situ hybridisation – Cloned HPV subtypes (Zur Hausen) – Automated platforms available. – Commercial probes: • DAKO, Digene, Ventana, etc. Detection sensitivity = 1 -5 copies per biopsy
HPV analysis • In-situ hybridisation – Cloned HPV subtypes (Zur Hausen) – Automated platforms available. – Commercial probes: • DAKO, Digene, Ventana, etc. Detection sensitivity = 1 -5 copies per biopsy
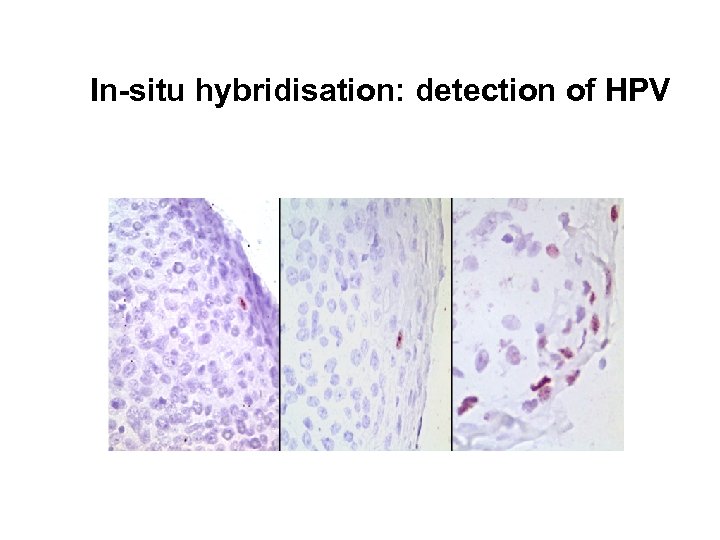 In-situ hybridisation: detection of HPV
In-situ hybridisation: detection of HPV


