52cffff68b67c033e1e16624cbe0d5df.ppt
- Количество слайдов: 48
 Hope and Hype of Hypertension: Renal Denervation… Julie Logan, RN, BS La Jolla, California
Hope and Hype of Hypertension: Renal Denervation… Julie Logan, RN, BS La Jolla, California
 Julie Logan, RN I/we have no real or apparent conflicts of interest to report. Off-Label: Will discuss devices that are not FDA approved but will state accordingly
Julie Logan, RN I/we have no real or apparent conflicts of interest to report. Off-Label: Will discuss devices that are not FDA approved but will state accordingly
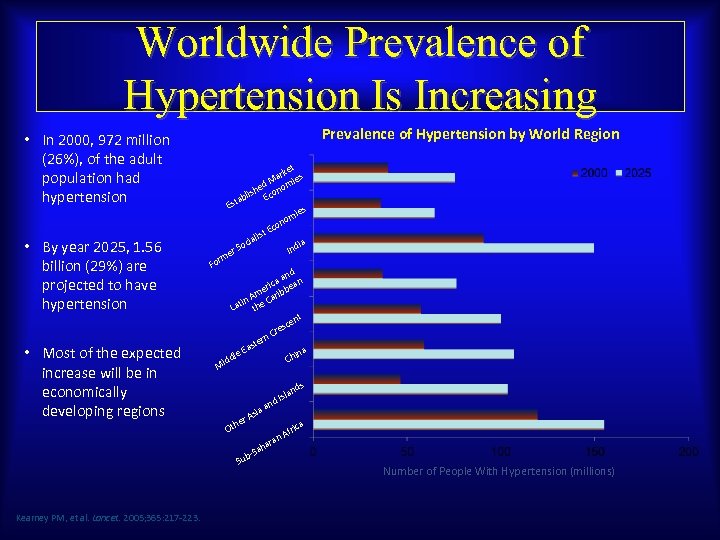 Worldwide Prevalence of Hypertension Is Increasing Prevalence of Hypertension by World Region • In 2000, 972 million (26%), of the adult population had hypertension • By year 2025, 1. 56 billion (29%) are projected to have hypertension • Most of the expected increase will be in economically developing regions F et ark ies M ed om lish Econ ab Est es mi no co t. E alis ci ia So Ind er orm nd aa ric bean e Am arib tin he C La t t cen res C ern ast ina le E Ch idd M n a sia r. A he Ot fr n. A ica ara ah b-S Su Kearney PM, et al. Lancet. 2005; 365: 217 -223. s nd sla d. I Number of People With Hypertension (millions)
Worldwide Prevalence of Hypertension Is Increasing Prevalence of Hypertension by World Region • In 2000, 972 million (26%), of the adult population had hypertension • By year 2025, 1. 56 billion (29%) are projected to have hypertension • Most of the expected increase will be in economically developing regions F et ark ies M ed om lish Econ ab Est es mi no co t. E alis ci ia So Ind er orm nd aa ric bean e Am arib tin he C La t t cen res C ern ast ina le E Ch idd M n a sia r. A he Ot fr n. A ica ara ah b-S Su Kearney PM, et al. Lancet. 2005; 365: 217 -223. s nd sla d. I Number of People With Hypertension (millions)
 A Major Global Health Burden v Most commonly diagnosed condition in the US v Astonishing prevalence v 74 Million Americans (1 in 3 -4 adults) v. Shared prevalence among men and women v Single largest contributor to death worldwide v Dramatically increases the risk of heart attack, stroke, heart failure & kidney failure v Estimated cost this year in US = $73. 4 B
A Major Global Health Burden v Most commonly diagnosed condition in the US v Astonishing prevalence v 74 Million Americans (1 in 3 -4 adults) v. Shared prevalence among men and women v Single largest contributor to death worldwide v Dramatically increases the risk of heart attack, stroke, heart failure & kidney failure v Estimated cost this year in US = $73. 4 B
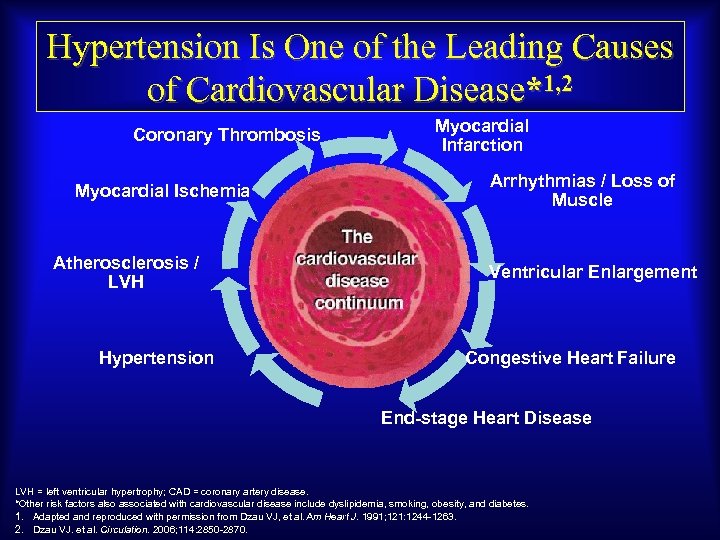 Hypertension Is One of the Leading Causes of Cardiovascular Disease*1, 2 Coronary Thrombosis Myocardial Ischemia Atherosclerosis / LVH Hypertension Myocardial Infarction Arrhythmias / Loss of Muscle Ventricular Enlargement Congestive Heart Failure End-stage Heart Disease LVH = left ventricular hypertrophy; CAD = coronary artery disease. *Other risk factors also associated with cardiovascular disease include dyslipidemia, smoking, obesity, and diabetes. 1. Adapted and reproduced with permission from Dzau VJ, et al. Am Heart J. 1991; 121: 1244 -1263. 2. Dzau VJ. et al. Circulation. 2006; 114: 2850 -2870.
Hypertension Is One of the Leading Causes of Cardiovascular Disease*1, 2 Coronary Thrombosis Myocardial Ischemia Atherosclerosis / LVH Hypertension Myocardial Infarction Arrhythmias / Loss of Muscle Ventricular Enlargement Congestive Heart Failure End-stage Heart Disease LVH = left ventricular hypertrophy; CAD = coronary artery disease. *Other risk factors also associated with cardiovascular disease include dyslipidemia, smoking, obesity, and diabetes. 1. Adapted and reproduced with permission from Dzau VJ, et al. Am Heart J. 1991; 121: 1244 -1263. 2. Dzau VJ. et al. Circulation. 2006; 114: 2850 -2870.
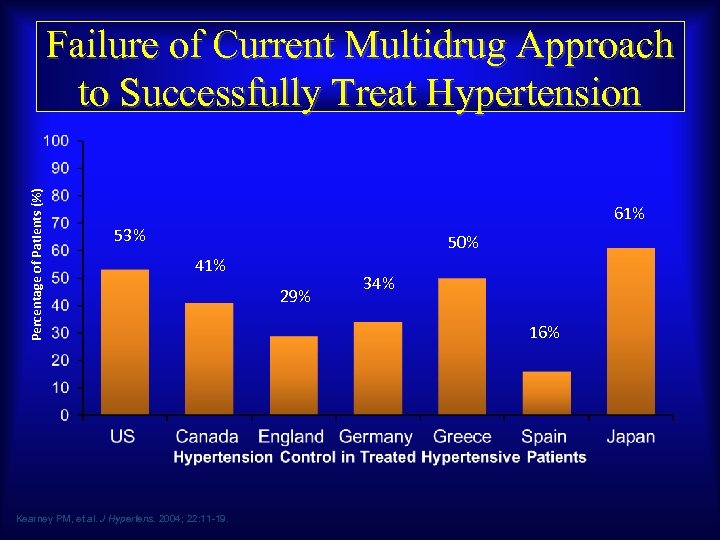 Percentage of Patients (%) Failure of Current Multidrug Approach to Successfully Treat Hypertension 61% 53% 50% 41% Kearney PM, et al. J Hypertens. 2004; 22: 11 -19. 29% 34% 16%
Percentage of Patients (%) Failure of Current Multidrug Approach to Successfully Treat Hypertension 61% 53% 50% 41% Kearney PM, et al. J Hypertens. 2004; 22: 11 -19. 29% 34% 16%
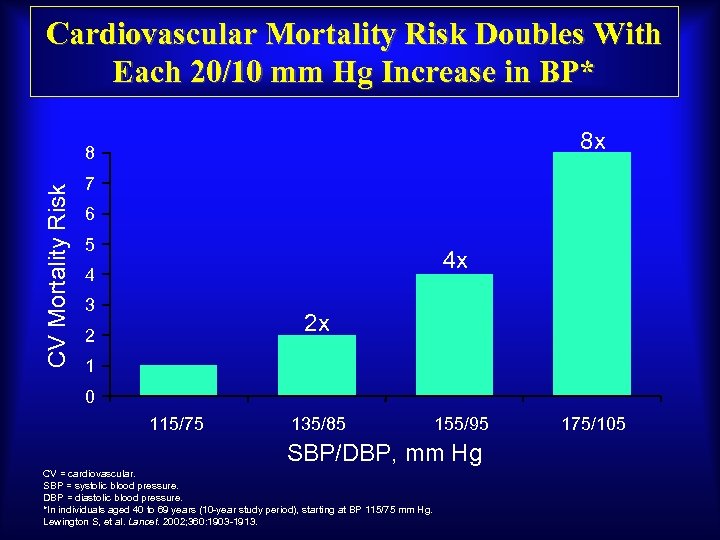 Cardiovascular Mortality Risk Doubles With Each 20/10 mm Hg Increase in BP* 8 x CV Mortality Risk 8 7 6 5 4 x 4 3 2 x 2 1 0 115/75 135/85 155/95 SBP/DBP, mm Hg CV = cardiovascular. SBP = systolic blood pressure. DBP = diastolic blood pressure. *In individuals aged 40 to 69 years (10 -year study period), starting at BP 115/75 mm Hg. Lewington S, et al. Lancet. 2002; 360: 1903 -1913. 175/105
Cardiovascular Mortality Risk Doubles With Each 20/10 mm Hg Increase in BP* 8 x CV Mortality Risk 8 7 6 5 4 x 4 3 2 x 2 1 0 115/75 135/85 155/95 SBP/DBP, mm Hg CV = cardiovascular. SBP = systolic blood pressure. DBP = diastolic blood pressure. *In individuals aged 40 to 69 years (10 -year study period), starting at BP 115/75 mm Hg. Lewington S, et al. Lancet. 2002; 360: 1903 -1913. 175/105
 Consequences of Hypertension Symptomatic Polysymptomatic or end-stage disease Adapted from Messerli FH, et al. Lancet. 2007; 370: 591 -603. Ventricular tachycardia/ fibrillation Congestive heart failure Coronary artery disease Angina Oligosymptomatic End-stage renal disease Systolic/ diastolic dysfunction Asymptomatic Chronic Renal Failure Atrial fibrillation Ventricular arrhythmias Prehypertension Myocardial infarction Dementia Transient ischemic attack Stroke
Consequences of Hypertension Symptomatic Polysymptomatic or end-stage disease Adapted from Messerli FH, et al. Lancet. 2007; 370: 591 -603. Ventricular tachycardia/ fibrillation Congestive heart failure Coronary artery disease Angina Oligosymptomatic End-stage renal disease Systolic/ diastolic dysfunction Asymptomatic Chronic Renal Failure Atrial fibrillation Ventricular arrhythmias Prehypertension Myocardial infarction Dementia Transient ischemic attack Stroke
 Definition of Resistant Hypertension Uncontrolled Hypertension • Includes all patients who lack BP control on treatment, including those on inadequate treatment regimens, those with poor adherence, those with undetected secondary hypertension, as well as those with true treatment resistance 1 Resistant Hypertension • BP that remains above goal in spite of compliance with full doses of ≥ 3 antihypertensive medications of different classes; ideally, 1 of the 3 agents should be a diuretic 1 ─ The treatment plan must include attention to lifestyle measures 2 • Includes those patients who achieve BP control but require ≥ 4 antihypertensive agents to do so 1 1. Calhoun DA, et al. Circulation. 2008; 117: e 510 -e 526. 2. Mancia G, et al. Eur Heart J. 2007; 28: 1462 -1536.
Definition of Resistant Hypertension Uncontrolled Hypertension • Includes all patients who lack BP control on treatment, including those on inadequate treatment regimens, those with poor adherence, those with undetected secondary hypertension, as well as those with true treatment resistance 1 Resistant Hypertension • BP that remains above goal in spite of compliance with full doses of ≥ 3 antihypertensive medications of different classes; ideally, 1 of the 3 agents should be a diuretic 1 ─ The treatment plan must include attention to lifestyle measures 2 • Includes those patients who achieve BP control but require ≥ 4 antihypertensive agents to do so 1 1. Calhoun DA, et al. Circulation. 2008; 117: e 510 -e 526. 2. Mancia G, et al. Eur Heart J. 2007; 28: 1462 -1536.
 BP Regulation Is Complex and Involves Many Components Mosaic Theory of Hypertension, published in 1967 Chemical Factors Neurosympathetic Activity Vascular Wall Elasticity Reactivity of Blood Vessels to Hormones Tissue Perfusion Pressure/Resistance Cardiac Output Vascular Caliber 10 Viscosity of Blood Page IH. Perspect Biol Med. 1967; 10(3): 325 -333. Volume Status
BP Regulation Is Complex and Involves Many Components Mosaic Theory of Hypertension, published in 1967 Chemical Factors Neurosympathetic Activity Vascular Wall Elasticity Reactivity of Blood Vessels to Hormones Tissue Perfusion Pressure/Resistance Cardiac Output Vascular Caliber 10 Viscosity of Blood Page IH. Perspect Biol Med. 1967; 10(3): 325 -333. Volume Status
 Renal Denervation
Renal Denervation
 Rational for Renal Sympathectomy • Pts with essential hypertension exhibit increased renal NA spillover into plasma • Increased renal and cardiac NA spillover is consistent with the hemodynamic profile of essential hypertension – Increased heart rate – Increased cardiac output – Renovascular resistence • Essential hypertension has been recognized as a NEUROGENIC phenomenon
Rational for Renal Sympathectomy • Pts with essential hypertension exhibit increased renal NA spillover into plasma • Increased renal and cardiac NA spillover is consistent with the hemodynamic profile of essential hypertension – Increased heart rate – Increased cardiac output – Renovascular resistence • Essential hypertension has been recognized as a NEUROGENIC phenomenon
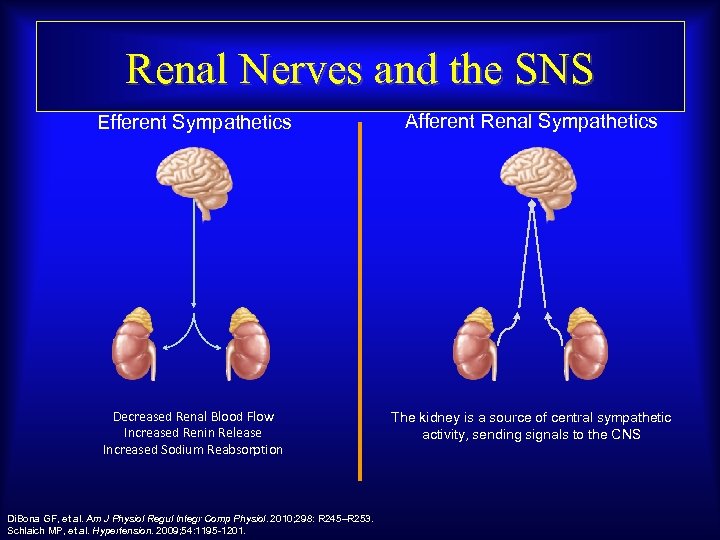 Renal Nerves and the SNS Efferent Sympathetics Afferent Renal Sympathetics Decreased Renal Blood Flow Increased Renin Release Increased Sodium Reabsorption The kidney is a source of central sympathetic activity, sending signals to the CNS Di. Bona GF, et al. Am J Physiol Regul Integr Comp Physiol. 2010; 298: R 245–R 253. Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201.
Renal Nerves and the SNS Efferent Sympathetics Afferent Renal Sympathetics Decreased Renal Blood Flow Increased Renin Release Increased Sodium Reabsorption The kidney is a source of central sympathetic activity, sending signals to the CNS Di. Bona GF, et al. Am J Physiol Regul Integr Comp Physiol. 2010; 298: R 245–R 253. Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201.
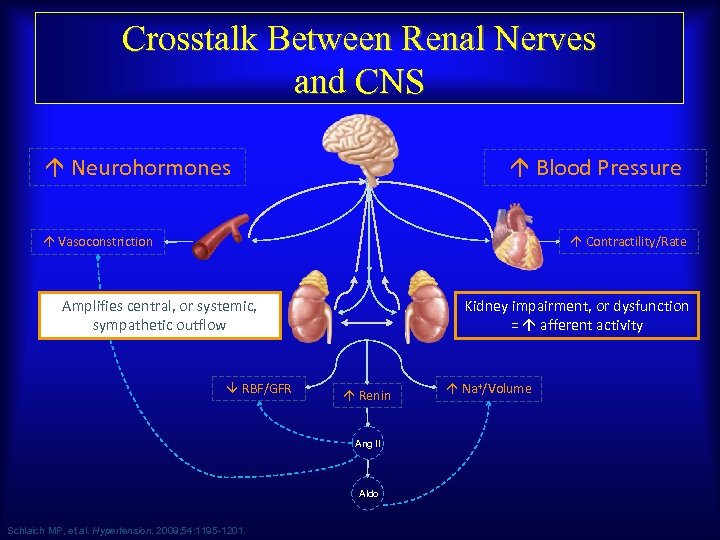 Crosstalk Between Renal Nerves and CNS Neurohormones Blood Pressure Contractility/Rate Vasoconstriction Kidney impairment, or dysfunction = afferent activity Amplifies central, or systemic, sympathetic outflow RBF/GFR Renin Ang II Aldo Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201. Na+/Volume
Crosstalk Between Renal Nerves and CNS Neurohormones Blood Pressure Contractility/Rate Vasoconstriction Kidney impairment, or dysfunction = afferent activity Amplifies central, or systemic, sympathetic outflow RBF/GFR Renin Ang II Aldo Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201. Na+/Volume
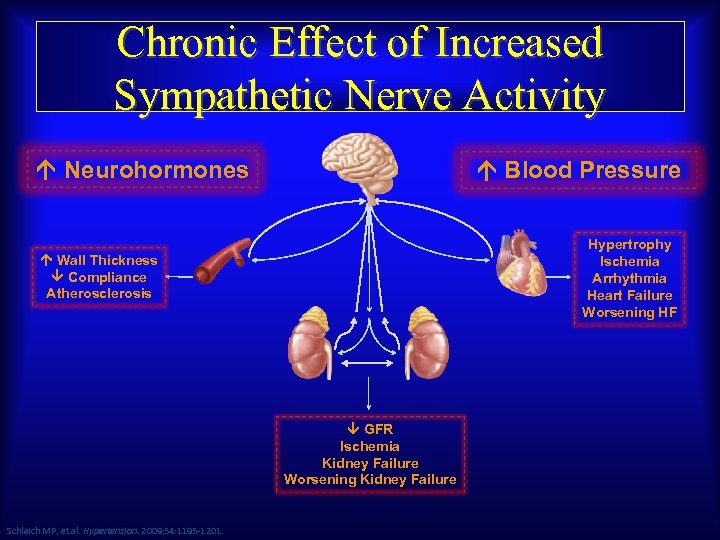 Chronic Effect of Increased Sympathetic Nerve Activity Neurohormones Blood Pressure Hypertrophy Ischemia Arrhythmia Heart Failure Worsening HF Wall Thickness Compliance Atherosclerosis GFR Ischemia Kidney Failure Worsening Kidney Failure Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201.
Chronic Effect of Increased Sympathetic Nerve Activity Neurohormones Blood Pressure Hypertrophy Ischemia Arrhythmia Heart Failure Worsening HF Wall Thickness Compliance Atherosclerosis GFR Ischemia Kidney Failure Worsening Kidney Failure Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201.
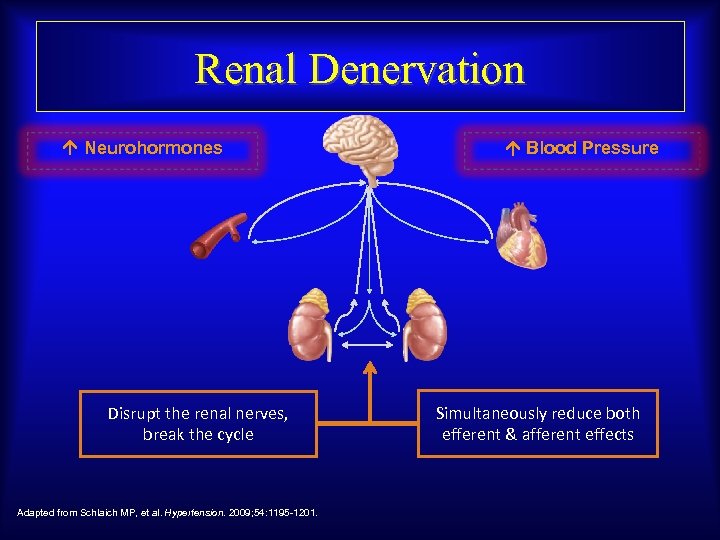 Renal Denervation Neurohormones Disrupt the renal nerves, break the cycle Adapted from Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201. Blood Pressure Simultaneously reduce both efferent & afferent effects
Renal Denervation Neurohormones Disrupt the renal nerves, break the cycle Adapted from Schlaich MP, et al. Hypertension. 2009; 54: 1195 -1201. Blood Pressure Simultaneously reduce both efferent & afferent effects
 HYPE? To an Interventionalist with a hammer, everything looks like a nail
HYPE? To an Interventionalist with a hammer, everything looks like a nail
 . 130 -micron diameter needle
. 130 -micron diameter needle
 New Devices • Radiofrequency catheters – – – St. Jude Medical Cordis Medtronic • Radiofrequency balloons – Covidien – Maya – Vessix Vascular • Nano particles – Apex Nano • Drugs - Mercator - Kipprokration Hospital, Athens • Radiation - Best Medical Int. • Ultrasound - Recor Medical Cardio. Sonic Sound Interventions Kona • Other • Rox. Medical • CVRX
New Devices • Radiofrequency catheters – – – St. Jude Medical Cordis Medtronic • Radiofrequency balloons – Covidien – Maya – Vessix Vascular • Nano particles – Apex Nano • Drugs - Mercator - Kipprokration Hospital, Athens • Radiation - Best Medical Int. • Ultrasound - Recor Medical Cardio. Sonic Sound Interventions Kona • Other • Rox. Medical • CVRX
 New Devices Approved in Europe • Radiofrequency catheters – St. Jude Medical- Enlig. HTN – Medtronic- Symplicity • Radiofrequency balloons – Covidien- One. Shot – BSC (Vessix)- V 2 • Ultrasound - Recor Medical- Paradise • Other • Rox. Medical- Rox. Flow 2 W • CVRx- Barostim Neo
New Devices Approved in Europe • Radiofrequency catheters – St. Jude Medical- Enlig. HTN – Medtronic- Symplicity • Radiofrequency balloons – Covidien- One. Shot – BSC (Vessix)- V 2 • Ultrasound - Recor Medical- Paradise • Other • Rox. Medical- Rox. Flow 2 W • CVRx- Barostim Neo
 Show me the “DATA”…
Show me the “DATA”…
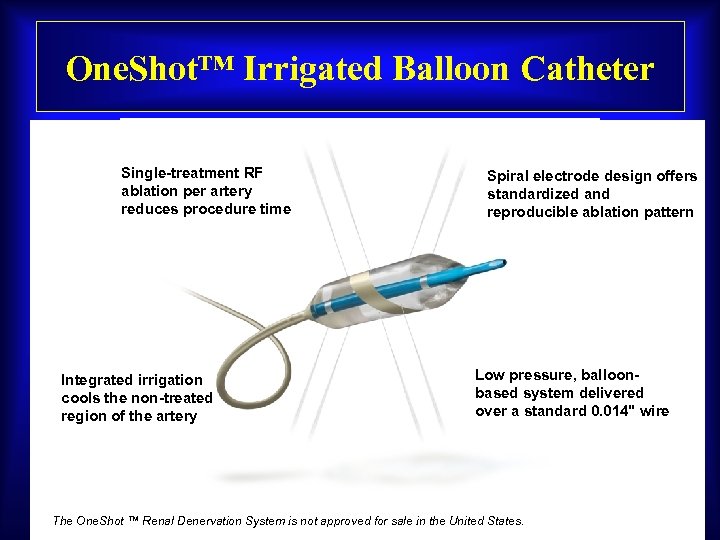 One. Shot™ Irrigated Balloon Catheter Single-treatment RF ablation per artery reduces procedure time Integrated irrigation cools the non-treated region of the artery Spiral electrode design offers standardized and reproducible ablation pattern Low pressure, balloonbased system delivered over a standard 0. 014" wire The One. Shot ™ Renal Denervation System is not approved for sale in the United States.
One. Shot™ Irrigated Balloon Catheter Single-treatment RF ablation per artery reduces procedure time Integrated irrigation cools the non-treated region of the artery Spiral electrode design offers standardized and reproducible ablation pattern Low pressure, balloonbased system delivered over a standard 0. 014" wire The One. Shot ™ Renal Denervation System is not approved for sale in the United States.
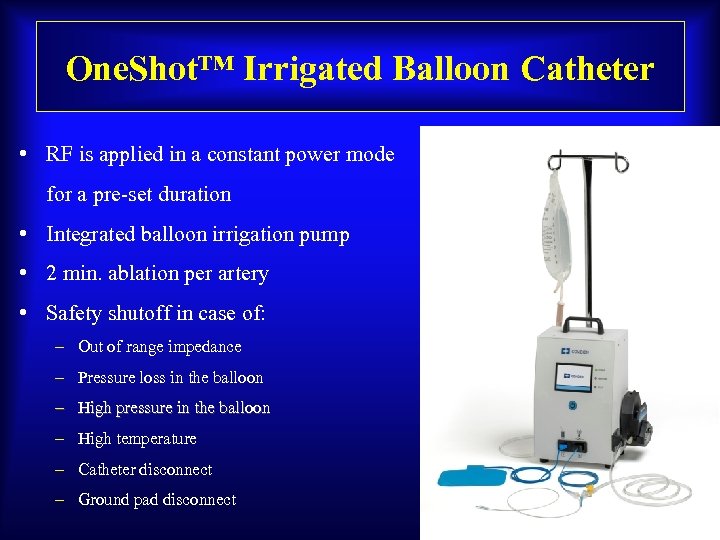 One. Shot™ Irrigated Balloon Catheter • RF is applied in a constant power mode for a pre-set duration • Integrated balloon irrigation pump • 2 min. ablation per artery • Safety shutoff in case of: – Out of range impedance – Pressure loss in the balloon – High pressure in the balloon – High temperature – Catheter disconnect – Ground pad disconnect MKT 3032. A / Effective Date: 12/05/2012
One. Shot™ Irrigated Balloon Catheter • RF is applied in a constant power mode for a pre-set duration • Integrated balloon irrigation pump • 2 min. ablation per artery • Safety shutoff in case of: – Out of range impedance – Pressure loss in the balloon – High pressure in the balloon – High temperature – Catheter disconnect – Ground pad disconnect MKT 3032. A / Effective Date: 12/05/2012
 RHAS First-in-Man Study (Renal Hypertension Ablation System) Study Baseline Variables n=9 patients enrolled Mean ± SD or % (n/N) Age (years) 59. 5 ± 15. 6 Male gender 55. 6% (5/9) Weight (kg) 83. 6 ± 17. 1 # anti-hypertensive meds 2. 9 ± 0. 6 Office SBP at baseline (mm. Hg) 185. 9 ± 17. 5 Office DBP at baseline (mm. Hg) 91. 6 ± 13. 6
RHAS First-in-Man Study (Renal Hypertension Ablation System) Study Baseline Variables n=9 patients enrolled Mean ± SD or % (n/N) Age (years) 59. 5 ± 15. 6 Male gender 55. 6% (5/9) Weight (kg) 83. 6 ± 17. 1 # anti-hypertensive meds 2. 9 ± 0. 6 Office SBP at baseline (mm. Hg) 185. 9 ± 17. 5 Office DBP at baseline (mm. Hg) 91. 6 ± 13. 6
 RHAS First-in-Man Study (Renal Hypertension Ablation System) Study • Defined as the ability to insert the RHAS balloon in each renal artery and deliver low level radiofrequency energy - 8/9 technical success • Only failure was with first patient: RF generator high-impedance safety shut-off threshold set too low for humans minor re-programming led to no further issue Ø Median total procedure time: 35 min Ø Median fluoro time: 7. 7 min Ø Median contrast volume: Ø No serious adverse events 124 ml
RHAS First-in-Man Study (Renal Hypertension Ablation System) Study • Defined as the ability to insert the RHAS balloon in each renal artery and deliver low level radiofrequency energy - 8/9 technical success • Only failure was with first patient: RF generator high-impedance safety shut-off threshold set too low for humans minor re-programming led to no further issue Ø Median total procedure time: 35 min Ø Median fluoro time: 7. 7 min Ø Median contrast volume: Ø No serious adverse events 124 ml
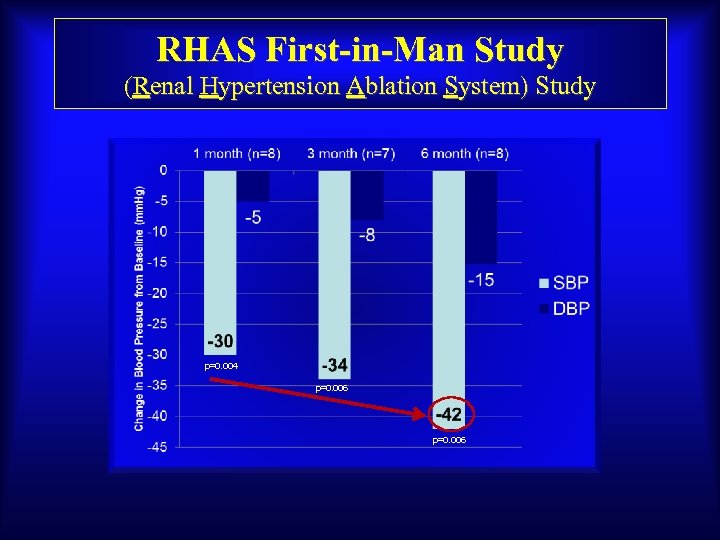 RHAS First-in-Man Study (Renal Hypertension Ablation System) Study p=0. 004 p=0. 006
RHAS First-in-Man Study (Renal Hypertension Ablation System) Study p=0. 004 p=0. 006
 Enlig. HTN™ Multi-Electrode Renal Denervation System
Enlig. HTN™ Multi-Electrode Renal Denervation System
 Enlig. HTN™ Ablation Catheter Key Features • • • Multi-electrode 8 F compatible Deflectable , atraumatic tip Common femoral access Durable electrodes Easy insertion with guiding catheters – Hemostatic hub and Tuohy Borst • Compatible with St. Jude Medical RF generator
Enlig. HTN™ Ablation Catheter Key Features • • • Multi-electrode 8 F compatible Deflectable , atraumatic tip Common femoral access Durable electrodes Easy insertion with guiding catheters – Hemostatic hub and Tuohy Borst • Compatible with St. Jude Medical RF generator
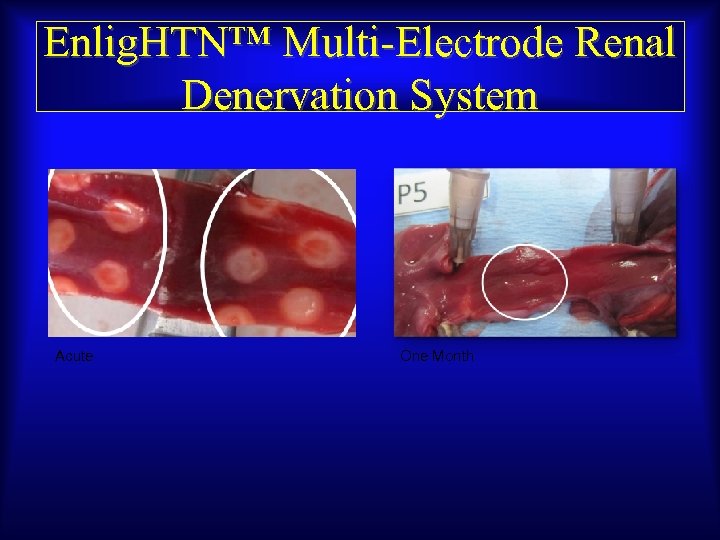 Enlig. HTN™ Multi-Electrode Renal Denervation System Acute One Month
Enlig. HTN™ Multi-Electrode Renal Denervation System Acute One Month
 Enlig. HTN I § The objective of this first-in-human study is to evaluate the safety and efficacy of the St. Jude Medical Enlig. HTN multielectrode renal denervation system in the treatment of patients with resistant hypertension Safety Marker All adverse events during the study period Efficacy Marker Office BP during the study: according to Standard Joint National Committee VII Guidelines / ESC and ESH Guidelines
Enlig. HTN I § The objective of this first-in-human study is to evaluate the safety and efficacy of the St. Jude Medical Enlig. HTN multielectrode renal denervation system in the treatment of patients with resistant hypertension Safety Marker All adverse events during the study period Efficacy Marker Office BP during the study: according to Standard Joint National Committee VII Guidelines / ESC and ESH Guidelines
 Inclusion Criteria § Office systolic blood pressure that remains ≥ 160 mm. Hg (≥ 150 mm. Hg for patient with type 2 diabetes) § Stable use of ≥ 3 antihypertensive medications concurrently at maximally tolerated doses for a minimum of 14 days prior to enrollment § of which one is a diuretic, or § patient was on diuretic previously but documented to be diuretic intolerant § ≥ 18 and ≤ 80 years old
Inclusion Criteria § Office systolic blood pressure that remains ≥ 160 mm. Hg (≥ 150 mm. Hg for patient with type 2 diabetes) § Stable use of ≥ 3 antihypertensive medications concurrently at maximally tolerated doses for a minimum of 14 days prior to enrollment § of which one is a diuretic, or § patient was on diuretic previously but documented to be diuretic intolerant § ≥ 18 and ≤ 80 years old
 Exclusion Criteria § Prior renal artery intervention § Evidence of renal artery atherosclerosis (defined as a stenotic severity of >30%) in either renal artery § Multiple main renal arteries in either kidney § Main renal arteries are <4 mm in diameter or <20 mm in length § e. GFR of <45 m. L/min/1. 73 m 2 using Modification of Diet in Renal Disease (MDRD) formula § Type 1 Diabetes Mellitus § Identified secondary cause of hypertension § Hemodynamically significant valvular heart disease
Exclusion Criteria § Prior renal artery intervention § Evidence of renal artery atherosclerosis (defined as a stenotic severity of >30%) in either renal artery § Multiple main renal arteries in either kidney § Main renal arteries are <4 mm in diameter or <20 mm in length § e. GFR of <45 m. L/min/1. 73 m 2 using Modification of Diet in Renal Disease (MDRD) formula § Type 1 Diabetes Mellitus § Identified secondary cause of hypertension § Hemodynamically significant valvular heart disease
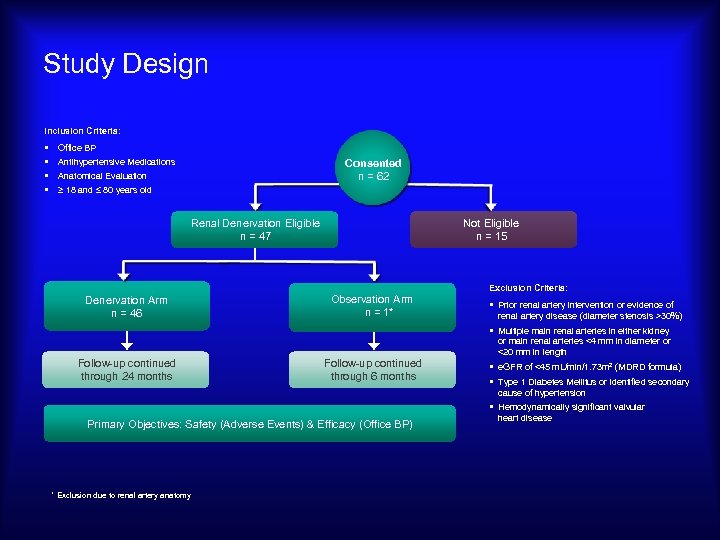 Study Design Inclusion Criteria: § § Office BP Antihypertensive Medications Consented n = 62 Anatomical Evaluation ≥ 18 and ≤ 80 years old Renal Denervation Eligible n = 47 Not Eligible n = 15 Exclusion Criteria: Denervation Arm n = 46 Observation Arm n = 1* § Prior renal artery intervention or evidence of renal artery disease (diameter stenosis >30%) § Multiple main renal arteries in either kidney or main renal arteries <4 mm in diameter or <20 mm in length Follow-up continued through 24 months Follow-up continued through 6 months § e. GFR of <45 m. L/min/1. 73 m 2 (MDRD formula) § Type 1 Diabetes Mellitus or identified secondary cause of hypertension § Hemodynamically significant valvular Primary Objectives: Safety (Adverse Events) & Efficacy (Office BP) * Exclusion due to renal artery anatomy heart disease
Study Design Inclusion Criteria: § § Office BP Antihypertensive Medications Consented n = 62 Anatomical Evaluation ≥ 18 and ≤ 80 years old Renal Denervation Eligible n = 47 Not Eligible n = 15 Exclusion Criteria: Denervation Arm n = 46 Observation Arm n = 1* § Prior renal artery intervention or evidence of renal artery disease (diameter stenosis >30%) § Multiple main renal arteries in either kidney or main renal arteries <4 mm in diameter or <20 mm in length Follow-up continued through 24 months Follow-up continued through 6 months § e. GFR of <45 m. L/min/1. 73 m 2 (MDRD formula) § Type 1 Diabetes Mellitus or identified secondary cause of hypertension § Hemodynamically significant valvular Primary Objectives: Safety (Adverse Events) & Efficacy (Office BP) * Exclusion due to renal artery anatomy heart disease
 Safety Results: Procedural & Device § Treatment delivered with: § § § No renal artery dissections, aneurysms or new stenoses No flow-limiting renal artery vasospasms No major vascular access complications § The primary safety outcome was assessment of all Adverse Events § At the three month time point, one serious event (worsening of pre-existing proteinuria) was classified as related to the device/procedure § There have been other minor procedural-related events such as hematoma (5), vasovagal episodes (2), hypotensive episodes (2) and bradycardia (2)
Safety Results: Procedural & Device § Treatment delivered with: § § § No renal artery dissections, aneurysms or new stenoses No flow-limiting renal artery vasospasms No major vascular access complications § The primary safety outcome was assessment of all Adverse Events § At the three month time point, one serious event (worsening of pre-existing proteinuria) was classified as related to the device/procedure § There have been other minor procedural-related events such as hematoma (5), vasovagal episodes (2), hypotensive episodes (2) and bradycardia (2)
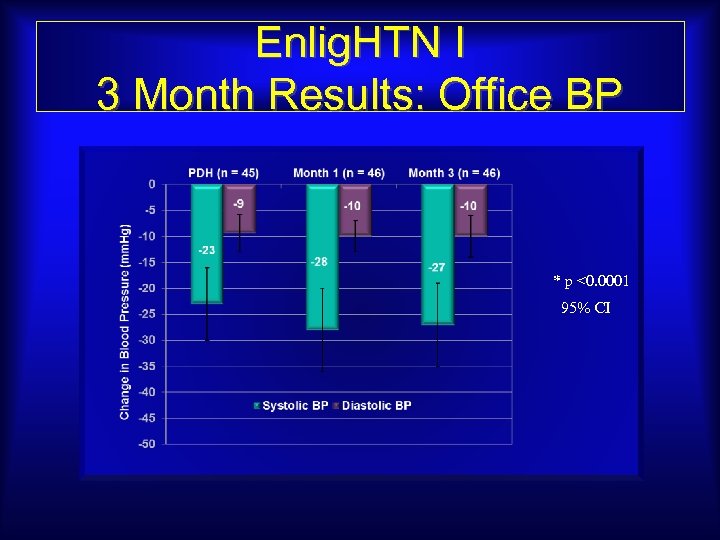 Enlig. HTN I 3 Month Results: Office BP * p <0. 0001 95% CI
Enlig. HTN I 3 Month Results: Office BP * p <0. 0001 95% CI
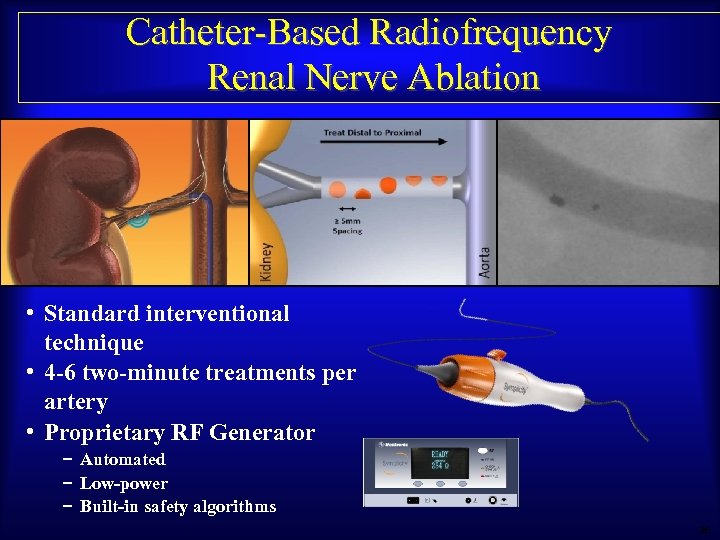 Catheter-Based Radiofrequency Renal Nerve Ablation • Standard interventional technique • 4 -6 two-minute treatments per artery • Proprietary RF Generator − Automated − Low-power − Built-in safety algorithms 36
Catheter-Based Radiofrequency Renal Nerve Ablation • Standard interventional technique • 4 -6 two-minute treatments per artery • Proprietary RF Generator − Automated − Low-power − Built-in safety algorithms 36
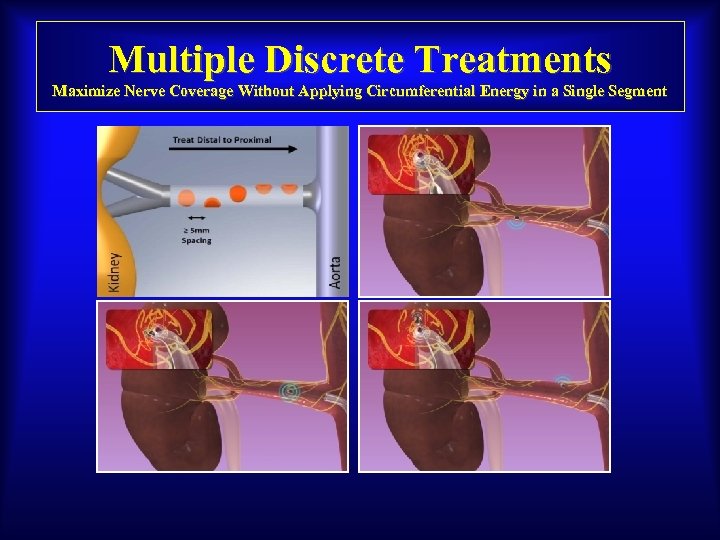 Multiple Discrete Treatments Maximize Nerve Coverage Without Applying Circumferential Energy in a Single Segment
Multiple Discrete Treatments Maximize Nerve Coverage Without Applying Circumferential Energy in a Single Segment
 The Symplicity HTN Clinical Trial Program Symplicity HTN-1 First-in-Man, and Expanded Cohort (N=153)1, 2 Symplicity HTN-2 Randomized, Controlled Trial (N=106)3 lairdezem 03 tno. R , T d i , ldor 5~N 4 ) o dna. B l ednil. C ( Symplicity HTN-3 = Primary endpoint = Planned follow up = Partial cohort reports 2006 2007 2008 2009 2010 2011 2012 2013 2014 Shading on bars indicates clinical trial enrollment periods. Enrollment period for HTN-3 is estimated. 1. 2. 3. 4. Krum H, et al. Lancet. 2009; 373: 1275 -1281. Symplicity HTN-1 Investigators. Hypertension. 2011; 57: 911 -917. Esler et al. Lancet. 2010; 376: 1903 -1909. Data on file, Medtronic. 2015 2016
The Symplicity HTN Clinical Trial Program Symplicity HTN-1 First-in-Man, and Expanded Cohort (N=153)1, 2 Symplicity HTN-2 Randomized, Controlled Trial (N=106)3 lairdezem 03 tno. R , T d i , ldor 5~N 4 ) o dna. B l ednil. C ( Symplicity HTN-3 = Primary endpoint = Planned follow up = Partial cohort reports 2006 2007 2008 2009 2010 2011 2012 2013 2014 Shading on bars indicates clinical trial enrollment periods. Enrollment period for HTN-3 is estimated. 1. 2. 3. 4. Krum H, et al. Lancet. 2009; 373: 1275 -1281. Symplicity HTN-1 Investigators. Hypertension. 2011; 57: 911 -917. Esler et al. Lancet. 2010; 376: 1903 -1909. Data on file, Medtronic. 2015 2016
 Symplicity HTN-2 Design • Purpose: To demonstrate the effectiveness of catheter-based renal denervation (RDN) for reducing blood pressure in patients with uncontrolled hypertension in a prospective, randomized, controlled, clinical trial • Patients: 106 patients with drug-resistant hypertension randomized 1: 1 to treatment with RDN vs. control • Clinical Sites: 24 centers in Europe, Australia, & New Zealand • 67% were designated hypertension centers of excellence • Primary Endpoint: Office systolic BP change from baseline at 6 months Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909.
Symplicity HTN-2 Design • Purpose: To demonstrate the effectiveness of catheter-based renal denervation (RDN) for reducing blood pressure in patients with uncontrolled hypertension in a prospective, randomized, controlled, clinical trial • Patients: 106 patients with drug-resistant hypertension randomized 1: 1 to treatment with RDN vs. control • Clinical Sites: 24 centers in Europe, Australia, & New Zealand • 67% were designated hypertension centers of excellence • Primary Endpoint: Office systolic BP change from baseline at 6 months Symplicity HTN-2 Investigators. Lancet. 2010; 376: 1903 -1909.
 Patient Population Inclusion Criteria: – – – Office SBP ≥ 160 mm. Hg (≥ 150 mm. Hg with type II diabetes mellitus) Stable drug regimen of 3+ more anti-HTN medications Age 18 -85 years Exclusion Criteria: – Hemodynamically or anatomically significant renal artery abnormalities or prior renal artery intervention – e. GFR <45 m. L/min/1. 73 m 2 (MDRD formula) – Type 1 diabetes mellitus – Stenotic valvular heart disease for which reduction of BP would be hazardous – MI, unstable angina, or CVA in the prior 6 months
Patient Population Inclusion Criteria: – – – Office SBP ≥ 160 mm. Hg (≥ 150 mm. Hg with type II diabetes mellitus) Stable drug regimen of 3+ more anti-HTN medications Age 18 -85 years Exclusion Criteria: – Hemodynamically or anatomically significant renal artery abnormalities or prior renal artery intervention – e. GFR <45 m. L/min/1. 73 m 2 (MDRD formula) – Type 1 diabetes mellitus – Stenotic valvular heart disease for which reduction of BP would be hazardous – MI, unstable angina, or CVA in the prior 6 months
 Procedure Detail & Safety • 38 minute median procedure time – Average of 4 ablations per artery • Intravenous narcotics & sedatives used to manage pain during delivery of RF energy • No catheter or generator malfunctions • No major complications • Minor complications 4/153: – 1 renal artery dissection during catheter delivery (prior to RF energy), no sequelae – 3 access site complications, treated without further sequelae
Procedure Detail & Safety • 38 minute median procedure time – Average of 4 ablations per artery • Intravenous narcotics & sedatives used to manage pain during delivery of RF energy • No catheter or generator malfunctions • No major complications • Minor complications 4/153: – 1 renal artery dissection during catheter delivery (prior to RF energy), no sequelae – 3 access site complications, treated without further sequelae
 Primary Endpoint: 6 -Month Office BP ∆ from Baseline to 6 Months (mm. Hg) Systolic Diastolic Systolic • • 33/11 mm. Hg difference between RDN and Control (p<0. 0001) 84% of RDN patients had ≥ 10 mm. Hg reduction in SBP 10% of RDN patients had no reduction in SBP Symplicity HTN-2 Investigators. The Lancet. 2010. 42
Primary Endpoint: 6 -Month Office BP ∆ from Baseline to 6 Months (mm. Hg) Systolic Diastolic Systolic • • 33/11 mm. Hg difference between RDN and Control (p<0. 0001) 84% of RDN patients had ≥ 10 mm. Hg reduction in SBP 10% of RDN patients had no reduction in SBP Symplicity HTN-2 Investigators. The Lancet. 2010. 42
 Office Systolic BP Distribution Symplicity HTN-2 Investigators. The Lancet. 2010. 43
Office Systolic BP Distribution Symplicity HTN-2 Investigators. The Lancet. 2010. 43
 Time Course of Office BP Change †† RDN ∆ from Baseline (mm. Hg) ††† † † p<0. 0001 for between-group comparisons p=0. 002 for between-group comparisons ††† p=0. 005 for between-group comparisons Two-way repeated measures ANOVA, p=0. 001 † †† Control ∆ from Baseline (mm. Hg) Symplicity HTN-2 Investigators. The Lancet. 2010. 44
Time Course of Office BP Change †† RDN ∆ from Baseline (mm. Hg) ††† † † p<0. 0001 for between-group comparisons p=0. 002 for between-group comparisons ††† p=0. 005 for between-group comparisons Two-way repeated measures ANOVA, p=0. 001 † †† Control ∆ from Baseline (mm. Hg) Symplicity HTN-2 Investigators. The Lancet. 2010. 44
 Office BP in RDN & Crossover Groups Diastolic BP 2 week compliance period Diastolic BP 178. 3 ± 18. 2 96. 1 ± 15. 5 6 month BP Change (p-value) 12 months BP Change (p-value) 146. 7 ± 23. 3 -31. 7 ± 23. 1 p<0. 001 150. 7 ± 21. 9 -28. 1 ± 24. 9 p<0. 001 84. 4 ± 17. 0 -11. 7 ± 11. 2 p<0. 001 87. 0 ± 16. 1 -9. 7 ± 10. 6 P<0. 001 Treated with RDN at 6 -mo Follow-up Crossover N=35 Systolic BP Baseline 2 week compliance period Systolic BP Treated with RDN 2 week compliance period RDN N=47 Pre-RDN* 6 month BP Change (p-value) 190. 0 ± 19. 6 166. 3 ± 24. 7 -23. 7 ± 27. 5 p<0. 001 99. 9 ± 15. 1 91. 5 ± 14. 6 -8. 4 ± 12. 1 p<0. 001 * At 6 months post-randomization No compliance period for Crossover patients prior to 6 month post-RDN BP measurements
Office BP in RDN & Crossover Groups Diastolic BP 2 week compliance period Diastolic BP 178. 3 ± 18. 2 96. 1 ± 15. 5 6 month BP Change (p-value) 12 months BP Change (p-value) 146. 7 ± 23. 3 -31. 7 ± 23. 1 p<0. 001 150. 7 ± 21. 9 -28. 1 ± 24. 9 p<0. 001 84. 4 ± 17. 0 -11. 7 ± 11. 2 p<0. 001 87. 0 ± 16. 1 -9. 7 ± 10. 6 P<0. 001 Treated with RDN at 6 -mo Follow-up Crossover N=35 Systolic BP Baseline 2 week compliance period Systolic BP Treated with RDN 2 week compliance period RDN N=47 Pre-RDN* 6 month BP Change (p-value) 190. 0 ± 19. 6 166. 3 ± 24. 7 -23. 7 ± 27. 5 p<0. 001 99. 9 ± 15. 1 91. 5 ± 14. 6 -8. 4 ± 12. 1 p<0. 001 * At 6 months post-randomization No compliance period for Crossover patients prior to 6 month post-RDN BP measurements
 Procedure Safety v. No vascular abnormalities at any site of RF delivery v One progression of a pre-existing stenosis unrelated to RF treatment (stented without further sequelae) v Two deaths within the follow-up period; both unrelated to the device or therapy v No orthostatic or electrolyte disturbances v No change in renal function (∆ e. GFR)
Procedure Safety v. No vascular abnormalities at any site of RF delivery v One progression of a pre-existing stenosis unrelated to RF treatment (stented without further sequelae) v Two deaths within the follow-up period; both unrelated to the device or therapy v No orthostatic or electrolyte disturbances v No change in renal function (∆ e. GFR)
 Conclusions v BP reduction can be achieved with catheter based renal denervation. v The treatment effect of renal denervation appears to be significant, sustained, and consistent across subgroups v No major adverse effects of renal denervation v Affirms the crucial relevance of the renal sympathetic system in the maintenance of hypertension.
Conclusions v BP reduction can be achieved with catheter based renal denervation. v The treatment effect of renal denervation appears to be significant, sustained, and consistent across subgroups v No major adverse effects of renal denervation v Affirms the crucial relevance of the renal sympathetic system in the maintenance of hypertension.



