46291b4e0687a1be0b1531d91d22071a.ppt
- Количество слайдов: 98
 Homocysteine and Creatine in Schizophrenia Prof. J. Levine Beer Sheva Mental Health Center, Ben Gurion University, Beer Sheva, Israel
Homocysteine and Creatine in Schizophrenia Prof. J. Levine Beer Sheva Mental Health Center, Ben Gurion University, Beer Sheva, Israel
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
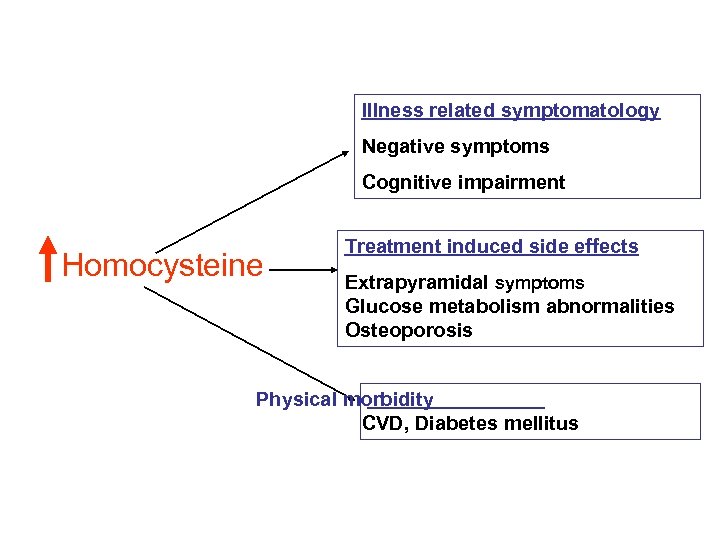
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
 HOMOCYSTINURIA • Rare autosomal disease: 1: 200, 000 • High blood and urine homocysteine levels • Mental retardation, skeletal abnormalities, premature arteriosclerosis
HOMOCYSTINURIA • Rare autosomal disease: 1: 200, 000 • High blood and urine homocysteine levels • Mental retardation, skeletal abnormalities, premature arteriosclerosis
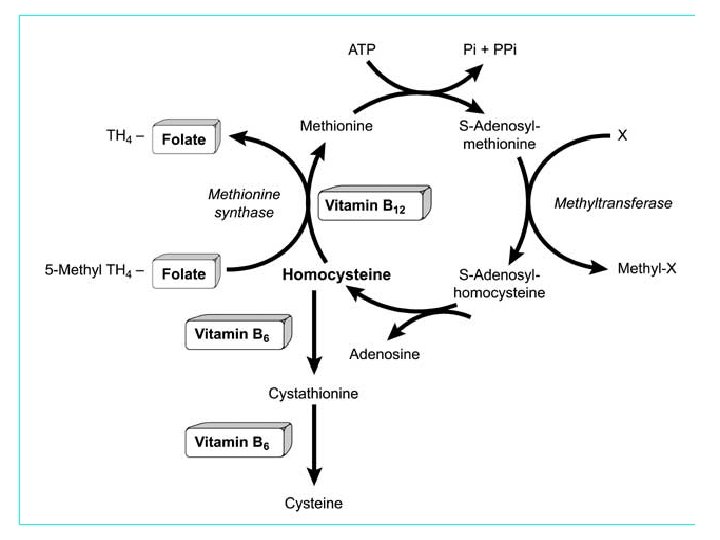
 Homocysteine blood level “Normal” values: 5 -15 microgram/liter (µg/L) Moderate elevation: 16 -30 µg/L Intermediate elevation: 31 -100 µg/L Severe elevation: >100 µg/L
Homocysteine blood level “Normal” values: 5 -15 microgram/liter (µg/L) Moderate elevation: 16 -30 µg/L Intermediate elevation: 31 -100 µg/L Severe elevation: >100 µg/L
![Vitamin status Enzyme Deficiency [B-12, folate, B-6] Life style habits (smoking, CVD Renal failure Vitamin status Enzyme Deficiency [B-12, folate, B-6] Life style habits (smoking, CVD Renal failure](https://present5.com/presentation/46291b4e0687a1be0b1531d91d22071a/image-6.jpg) Vitamin status Enzyme Deficiency [B-12, folate, B-6] Life style habits (smoking, CVD Renal failure Diabetes Thyroid disease Cancer obesity, coffee consumption, decreased physical activity) Age Gender Genetics (MTHFR) Homocysteine Level Drugs FACTORS EFFECTING HOMOCYSTEINE LEVEL
Vitamin status Enzyme Deficiency [B-12, folate, B-6] Life style habits (smoking, CVD Renal failure Diabetes Thyroid disease Cancer obesity, coffee consumption, decreased physical activity) Age Gender Genetics (MTHFR) Homocysteine Level Drugs FACTORS EFFECTING HOMOCYSTEINE LEVEL
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
 Homocysteine as a risk factor for cognitive deterioration and Alzheimer Disease
Homocysteine as a risk factor for cognitive deterioration and Alzheimer Disease
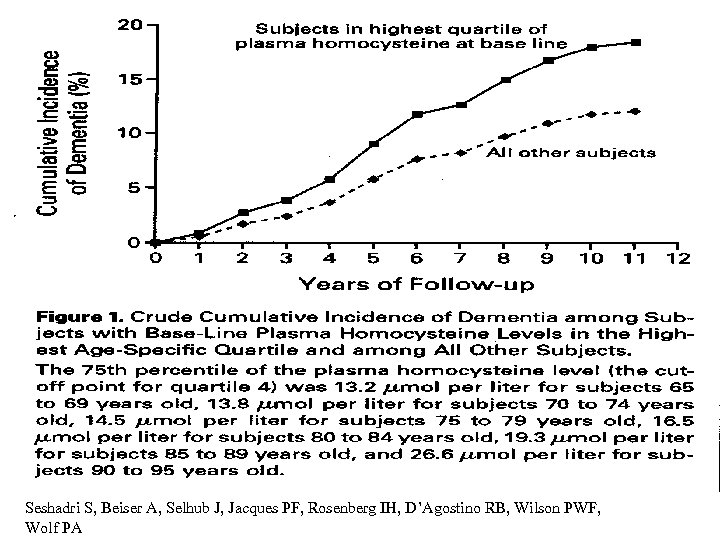 Seshadri S, Beiser A, Selhub J, Jacques PF, Rosenberg IH, D’Agostino RB, Wilson PWF, Wolf PA
Seshadri S, Beiser A, Selhub J, Jacques PF, Rosenberg IH, D’Agostino RB, Wilson PWF, Wolf PA
 Homocysteine may be a risk factor for several CNS disorders Elevated plasma homocysteine has been found to be a risk factor for Alzheimer disease as well as for cerebral vascular disease, suggesting that some risk factors can accelerate or increase the severity of several CNS disease processes.
Homocysteine may be a risk factor for several CNS disorders Elevated plasma homocysteine has been found to be a risk factor for Alzheimer disease as well as for cerebral vascular disease, suggesting that some risk factors can accelerate or increase the severity of several CNS disease processes.
 Elevated Homocysteine in Mental Disorders Schizophrenia Depression Bipolar Disorder Anxiety Disorders (OCD, PTSD) Eating Disorders
Elevated Homocysteine in Mental Disorders Schizophrenia Depression Bipolar Disorder Anxiety Disorders (OCD, PTSD) Eating Disorders
 Proc. Natl. Acad. Sci. USA. 1997 May 27; 94 (11): 5923– 5928 Neurobiology Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor Lipton et al n With physiological levels of glycine, homocysteine acts as a partial antagonist at the glycine coagonist site of the N-methyl-D-aspartate receptor. Homocysteine acts as an agonist at the glutamate binding site of the N-methyl-D-aspartate receptor, under pathological conditions in which glycine levels in the nervous system are elevated, such as stroke and head trauma. In this case, homocysteine neurotoxicity (agonist effect) at 10– 100 μM level outweighs its neuroprotective antagonist activity. n
Proc. Natl. Acad. Sci. USA. 1997 May 27; 94 (11): 5923– 5928 Neurobiology Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor Lipton et al n With physiological levels of glycine, homocysteine acts as a partial antagonist at the glycine coagonist site of the N-methyl-D-aspartate receptor. Homocysteine acts as an agonist at the glutamate binding site of the N-methyl-D-aspartate receptor, under pathological conditions in which glycine levels in the nervous system are elevated, such as stroke and head trauma. In this case, homocysteine neurotoxicity (agonist effect) at 10– 100 μM level outweighs its neuroprotective antagonist activity. n
 Proc. Natl. Acad. Sci. USA. 1997 May 27; 94 (11): 5923– 5928 Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor Stuart A. Lipton et al • Under these conditions neuronal damage derives from excessive Ca++ influx and reactive oxygen generation. • Accordingly, homocysteine neurotoxicity through overstimulation of N-methyl-Daspartate receptors may contribute to the pathogenesis of both homocystinuria and modest hyperhomocysteinemia.
Proc. Natl. Acad. Sci. USA. 1997 May 27; 94 (11): 5923– 5928 Neurotoxicity associated with dual actions of homocysteine at the N-methyl-D-aspartate receptor Stuart A. Lipton et al • Under these conditions neuronal damage derives from excessive Ca++ influx and reactive oxygen generation. • Accordingly, homocysteine neurotoxicity through overstimulation of N-methyl-Daspartate receptors may contribute to the pathogenesis of both homocystinuria and modest hyperhomocysteinemia.
 J Neurosci 2000 Sep 15; 20(18): 6920 -6 Homocysteine elicits a DNA damage response in neurons that promotes apoptosis and hypersensitivity to excitotoxicity. Kruman et al • Kruman et al (2000) reported that homocysteine induces apoptosis in rat hippocampal neurons. • DNA strand breaks occur rapidly after exposure to homocysteine and precede mitochondrial dysfunction, oxidative stress, and caspase activation. • Homocysteine markedly increases the vulnerability of hippocampal neurons to excitotoxic and oxidative injury in cell culture and in vivo, suggesting a mechanism by which homocysteine may contribute to the pathogenesis of neurodegenerative disorders.
J Neurosci 2000 Sep 15; 20(18): 6920 -6 Homocysteine elicits a DNA damage response in neurons that promotes apoptosis and hypersensitivity to excitotoxicity. Kruman et al • Kruman et al (2000) reported that homocysteine induces apoptosis in rat hippocampal neurons. • DNA strand breaks occur rapidly after exposure to homocysteine and precede mitochondrial dysfunction, oxidative stress, and caspase activation. • Homocysteine markedly increases the vulnerability of hippocampal neurons to excitotoxic and oxidative injury in cell culture and in vivo, suggesting a mechanism by which homocysteine may contribute to the pathogenesis of neurodegenerative disorders.
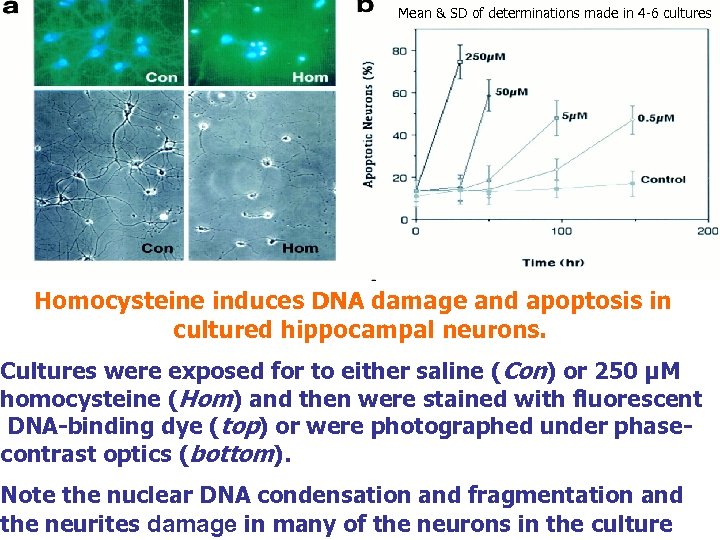 Mean & SD of determinations made in 4 -6 cultures Homocysteine induces DNA damage and apoptosis in cultured hippocampal neurons. Cultures were exposed for to either saline (Con) or 250 µM homocysteine (Hom) and then were stained with fluorescent DNA-binding dye (top) or were photographed under phasecontrast optics (bottom). Note the nuclear DNA condensation and fragmentation and the neurites damage in many of the neurons in the culture
Mean & SD of determinations made in 4 -6 cultures Homocysteine induces DNA damage and apoptosis in cultured hippocampal neurons. Cultures were exposed for to either saline (Con) or 250 µM homocysteine (Hom) and then were stained with fluorescent DNA-binding dye (top) or were photographed under phasecontrast optics (bottom). Note the nuclear DNA condensation and fragmentation and the neurites damage in many of the neurons in the culture
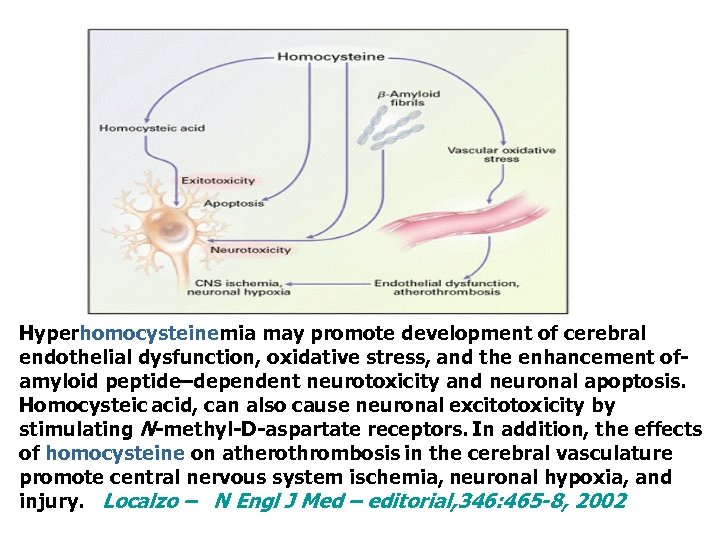 Hyperhomocysteinemia may promote development of cerebral endothelial dysfunction, oxidative stress, and the enhancement ofamyloid peptide–dependent neurotoxicity and neuronal apoptosis. Homocysteic acid, can also cause neuronal excitotoxicity by stimulating N-methyl-D-aspartate receptors. In addition, the effects of homocysteine on atherothrombosis in the cerebral vasculature promote central nervous system ischemia, neuronal hypoxia, and injury. Localzo – N Engl J Med – editorial, 346: 465 -8, 2002
Hyperhomocysteinemia may promote development of cerebral endothelial dysfunction, oxidative stress, and the enhancement ofamyloid peptide–dependent neurotoxicity and neuronal apoptosis. Homocysteic acid, can also cause neuronal excitotoxicity by stimulating N-methyl-D-aspartate receptors. In addition, the effects of homocysteine on atherothrombosis in the cerebral vasculature promote central nervous system ischemia, neuronal hypoxia, and injury. Localzo – N Engl J Med – editorial, 346: 465 -8, 2002
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
 Does Homocysteine Play a Role in Schizophrenia ?
Does Homocysteine Play a Role in Schizophrenia ?
 • An oral methionine load has classically and consistently been reported to exacerbate schizophrenia and is of course converted to homocysteine. • Several authors including Regland (1997) and Susser (1998) suggested that high homocysteine levels may consist of a risk factor for schizophrenia. • In order to find whether elevated homocysteine levels may be associated with schizophrenia we screened schizophrenic patients in our catchment area for plasma homocysteine levels.
• An oral methionine load has classically and consistently been reported to exacerbate schizophrenia and is of course converted to homocysteine. • Several authors including Regland (1997) and Susser (1998) suggested that high homocysteine levels may consist of a risk factor for schizophrenia. • In order to find whether elevated homocysteine levels may be associated with schizophrenia we screened schizophrenic patients in our catchment area for plasma homocysteine levels.
 Elevated Homocysteine Levels in Young Male Schizophrenic Inatients Joseph Levine, Ziva Stahl, Ben Ami Sela, Slava Gavendo Vladimir Ruderman, RH Belmaker Ben Gurion University of the Negev, Beer Sheva, Israel Am J Psychiat 159: 1790 -1792, 2002
Elevated Homocysteine Levels in Young Male Schizophrenic Inatients Joseph Levine, Ziva Stahl, Ben Ami Sela, Slava Gavendo Vladimir Ruderman, RH Belmaker Ben Gurion University of the Negev, Beer Sheva, Israel Am J Psychiat 159: 1790 -1792, 2002
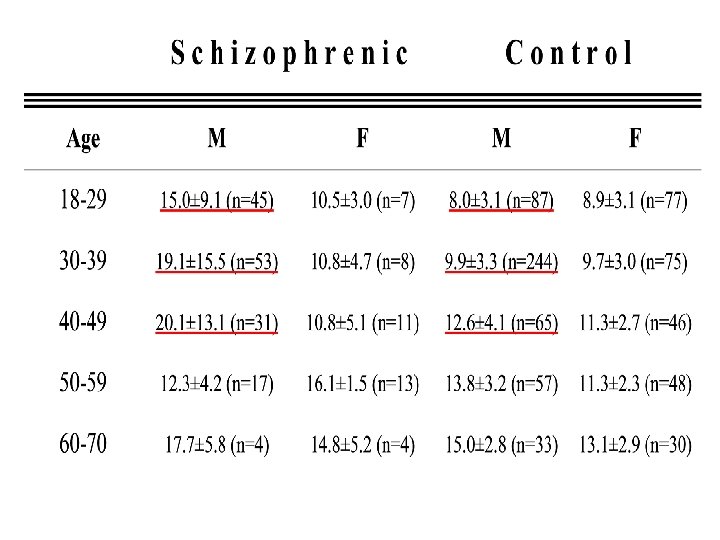
 Total plasma homocysteine levels were screened in: 193 schizophrenic patients compared to 762 controls subjects (evaluated in a screening program for employee health).
Total plasma homocysteine levels were screened in: 193 schizophrenic patients compared to 762 controls subjects (evaluated in a screening program for employee health).
 Results Homoysteine levels were significantly higher in schizophrenia patients compared with control subjects mean homocysteine level was: 16. 3 ± 11. 8 (SD) m. M in schizophrenic patients versus 10. 6 ± 3. 6 (SD) m. M in healthy controls. [One-way ANCOVA with age and sex as covariants showed a marked effect of diagnosis on homocysteine levels (F=135. 7, df= 1; 951, p<0. 0001)] The increase was almost entirely in young male schizophrenic patients
Results Homoysteine levels were significantly higher in schizophrenia patients compared with control subjects mean homocysteine level was: 16. 3 ± 11. 8 (SD) m. M in schizophrenic patients versus 10. 6 ± 3. 6 (SD) m. M in healthy controls. [One-way ANCOVA with age and sex as covariants showed a marked effect of diagnosis on homocysteine levels (F=135. 7, df= 1; 951, p<0. 0001)] The increase was almost entirely in young male schizophrenic patients
 Next step • Next, we turned to explore whether the finding is related to poor hospital nutrition or to other yet unknown factors associated with hospitalization ? • One way to examine it, is to study homocysteine levels in newly admitted schizophrenic patients.
Next step • Next, we turned to explore whether the finding is related to poor hospital nutrition or to other yet unknown factors associated with hospitalization ? • One way to examine it, is to study homocysteine levels in newly admitted schizophrenic patients.
 Plasma Homocysteine Levels in Newly Admitted Schizophrenic Patients J Applebaum, Hady Shimon, B-A Sela, RH Belmaker and J Levine 1 Ben Gurion University of the Negev, Beersheva, Israel, J Psychiatric Research. 3: 413 -416, 2004
Plasma Homocysteine Levels in Newly Admitted Schizophrenic Patients J Applebaum, Hady Shimon, B-A Sela, RH Belmaker and J Levine 1 Ben Gurion University of the Negev, Beersheva, Israel, J Psychiatric Research. 3: 413 -416, 2004
 Total plasma total homocysteine levels were screened in: 184 Newly admitted schizophrenic patients versus 305 controls subjects (evaluated in a screening program for employee health).
Total plasma total homocysteine levels were screened in: 184 Newly admitted schizophrenic patients versus 305 controls subjects (evaluated in a screening program for employee health).
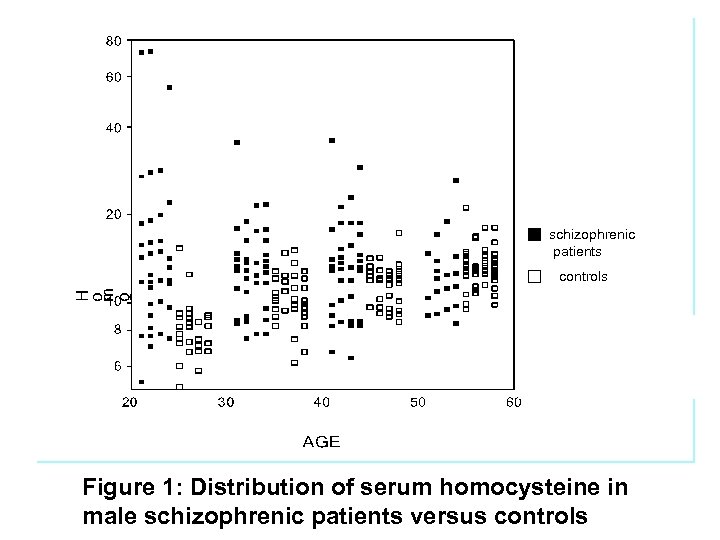 schizophrenic patients controls Figure 1: Distribution of serum homocysteine in male schizophrenic patients versus controls
schizophrenic patients controls Figure 1: Distribution of serum homocysteine in male schizophrenic patients versus controls
 Homocysteine blood levels are mainly elevated in a sub-group of YOUNG MALE SCHIZOPHRENIA PATIENTS
Homocysteine blood levels are mainly elevated in a sub-group of YOUNG MALE SCHIZOPHRENIA PATIENTS
 Homocysteine, methylenetetrahydrofolate reductase and risk of schizophrenia: a meta-analysis: Muntjewerff et al A meta-analysis of eight retrospective studies (812 cases and 2113 control subjects) was carried out to examine the association between homocysteine and schizophrenia. A 5 mol/l higher homocysteine level was associated with a 70% higher risk of schizophrenia. Molecular Psychiatry (2006) 11, 143– 149.
Homocysteine, methylenetetrahydrofolate reductase and risk of schizophrenia: a meta-analysis: Muntjewerff et al A meta-analysis of eight retrospective studies (812 cases and 2113 control subjects) was carried out to examine the association between homocysteine and schizophrenia. A 5 mol/l higher homocysteine level was associated with a 70% higher risk of schizophrenia. Molecular Psychiatry (2006) 11, 143– 149.
 What next • Can anything be done to lower homocysteine levels in schizophrenia? Well, elevated homocysteine can be lowered by oral administration of folic acid, B-12 and pyridoxine. • If so, will such homocysteine lowering strategy be associated with clinical improvement or improved cognitive functioning in schizophrenia?
What next • Can anything be done to lower homocysteine levels in schizophrenia? Well, elevated homocysteine can be lowered by oral administration of folic acid, B-12 and pyridoxine. • If so, will such homocysteine lowering strategy be associated with clinical improvement or improved cognitive functioning in schizophrenia?
 Homocysteine Reducing Strategy in Schizophrenia
Homocysteine Reducing Strategy in Schizophrenia
 Homocysteine Reducing Strategies Improve Symptoms in Chronic Schizophrenic Patients with Hyperhomocysteinemia Joseph Levine, MD 1, Ziva Stahl, MSc 1, Ben-Ami Sela, Ph. D 2, Vladimir Ruderman MD 1, Oleg Shumaico MD 1, RH Belmaker MD 1 1 Stanley Research Center & Beersheva Mental Health Center Ben Gurion University of the Negev, Beersheva, Israel, 2 The Institute of Chemical Pathology, Sheba Medical Center, Tel-Hashomer, Sackler Faculty of Medicine, Tel Aviv University Biol Psychiatry. 2006 1; 60(3): 265 -9
Homocysteine Reducing Strategies Improve Symptoms in Chronic Schizophrenic Patients with Hyperhomocysteinemia Joseph Levine, MD 1, Ziva Stahl, MSc 1, Ben-Ami Sela, Ph. D 2, Vladimir Ruderman MD 1, Oleg Shumaico MD 1, RH Belmaker MD 1 1 Stanley Research Center & Beersheva Mental Health Center Ben Gurion University of the Negev, Beersheva, Israel, 2 The Institute of Chemical Pathology, Sheba Medical Center, Tel-Hashomer, Sackler Faculty of Medicine, Tel Aviv University Biol Psychiatry. 2006 1; 60(3): 265 -9
 Homocysteine lowering strategy in schizophrenia Inclusion criteria: Schizophrenic patients with baseline homocysteine plasma levels >15 micro. M/L Exclusion Criteria: Patients with any physical illness or abnormality in blood chemistry; patients with alcohol or drug abuse in the last 6 months The design was a double-blind crossover with one capsule a day containing 2 mg folic acid, 25 mg pyridoxine and 400 mg B-12. After 3 months patients were crossed over for another 3 months from active vitamin to placebo or vice versa. Positive and Negative Symptom Scale (PANSS) was used to measure severity of symptoms Fifty five patients entered the study. All patients entering the study were highly symptomatic but had shown no major clinical changes for at least one month
Homocysteine lowering strategy in schizophrenia Inclusion criteria: Schizophrenic patients with baseline homocysteine plasma levels >15 micro. M/L Exclusion Criteria: Patients with any physical illness or abnormality in blood chemistry; patients with alcohol or drug abuse in the last 6 months The design was a double-blind crossover with one capsule a day containing 2 mg folic acid, 25 mg pyridoxine and 400 mg B-12. After 3 months patients were crossed over for another 3 months from active vitamin to placebo or vice versa. Positive and Negative Symptom Scale (PANSS) was used to measure severity of symptoms Fifty five patients entered the study. All patients entering the study were highly symptomatic but had shown no major clinical changes for at least one month
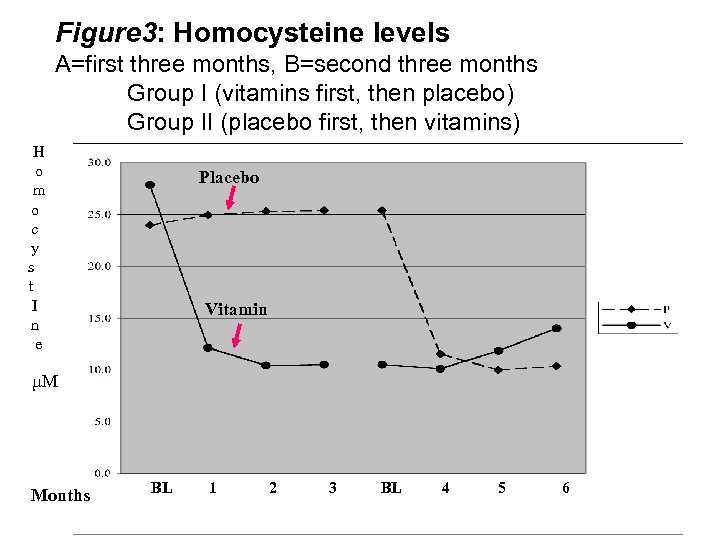 Figure 3: Homocysteine levels A=first three months, B=second three months Group I (vitamins first, then placebo) Group II (placebo first, then vitamins) H o m o c y s t I n e Placebo Vitamin µM Months BL 1 2 3 BL 4 5 6
Figure 3: Homocysteine levels A=first three months, B=second three months Group I (vitamins first, then placebo) Group II (placebo first, then vitamins) H o m o c y s t I n e Placebo Vitamin µM Months BL 1 2 3 BL 4 5 6
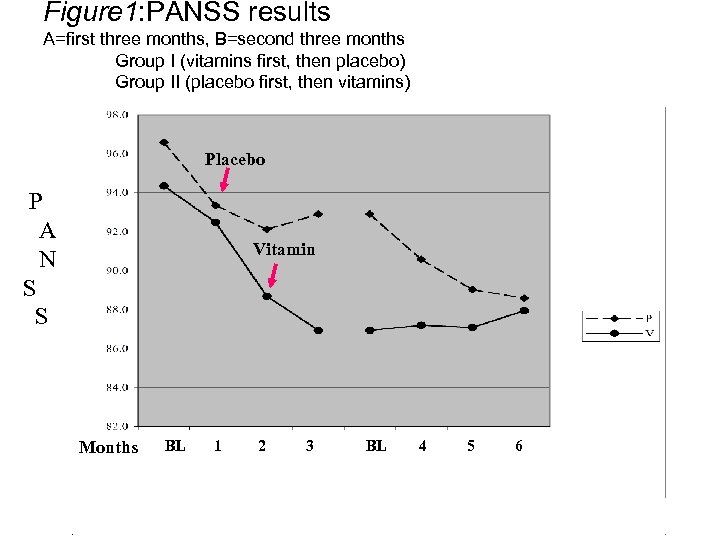 Figure 1: PANSS results A=first three months, B=second three months Group I (vitamins first, then placebo) Group II (placebo first, then vitamins) Placebo P A N S S Vitamin Months BL 1 2 3 BL 4 5 6
Figure 1: PANSS results A=first three months, B=second three months Group I (vitamins first, then placebo) Group II (placebo first, then vitamins) Placebo P A N S S Vitamin Months BL 1 2 3 BL 4 5 6
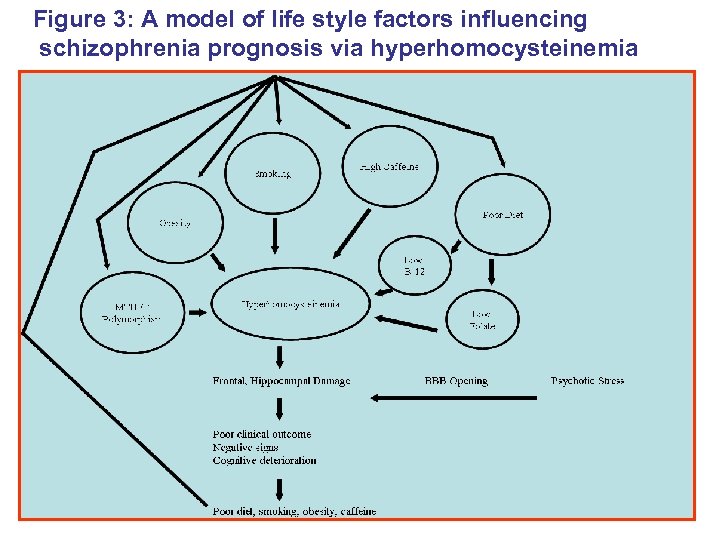 Figure 3: A model of life style factors influencing schizophrenia prognosis via hyperhomocysteinemia
Figure 3: A model of life style factors influencing schizophrenia prognosis via hyperhomocysteinemia
 Arch Gen Psychiatry. 2007 Jan; 64(1): 31 -9. Elevated prenatal homocysteine levels as a risk factor for schizophrenia. Brown AS, Bottiglieri T, Schaefer CA, Quesenberry CP Jr, Liu L, Bresnahan M, Susser ES. DESIGN: Nested case-control study of a large birth cohort, born from 1959 through 1967 and followed up for schizophrenia from 1981 through 1997. PARTICIPANTS: Cases (n = 63) were diagnosed with schizophrenia and other schizophrenia spectrum disorders. Controls (n = 122) belonged to the birth cohort and were matched to cases on date of birth, sex, length of time in the cohort, and availability of maternal serum samples. . RESULTS: In a model that tested for a threshold effect of third-trimester homocysteine levels, an elevated homocysteine level was associated with a greater than 2 -fold statistically significant increase in schizophrenia risk (odds ratio, 2. 39; 95% confidence interval, 1. 18 -4. 81; P =. 02). CONCLUSIONS: These findings indicate that elevated third-trimester homocysteine levels may be a risk factor for schizophrenia. as a strategy for prevention of schizophrenia in offspring.
Arch Gen Psychiatry. 2007 Jan; 64(1): 31 -9. Elevated prenatal homocysteine levels as a risk factor for schizophrenia. Brown AS, Bottiglieri T, Schaefer CA, Quesenberry CP Jr, Liu L, Bresnahan M, Susser ES. DESIGN: Nested case-control study of a large birth cohort, born from 1959 through 1967 and followed up for schizophrenia from 1981 through 1997. PARTICIPANTS: Cases (n = 63) were diagnosed with schizophrenia and other schizophrenia spectrum disorders. Controls (n = 122) belonged to the birth cohort and were matched to cases on date of birth, sex, length of time in the cohort, and availability of maternal serum samples. . RESULTS: In a model that tested for a threshold effect of third-trimester homocysteine levels, an elevated homocysteine level was associated with a greater than 2 -fold statistically significant increase in schizophrenia risk (odds ratio, 2. 39; 95% confidence interval, 1. 18 -4. 81; P =. 02). CONCLUSIONS: These findings indicate that elevated third-trimester homocysteine levels may be a risk factor for schizophrenia. as a strategy for prevention of schizophrenia in offspring.
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
 Does Homocysteine Play a Role in Neuroleptic induced ? Drugs side effects ?
Does Homocysteine Play a Role in Neuroleptic induced ? Drugs side effects ?
 Extrapyramidal Side Effects
Extrapyramidal Side Effects
 J Clin Psychiatry. 2005 ; 66: 1558 -63. High serum homocysteine levels in young male schizophrenic and schizoaffective patients with tardive parkinsonism and/or tardive dyskinesia. Lerner V, Miodownik C, Kaptsan A, Vishne T, Sela BA and Levine J. An elevated serum level of total homocysteine has been implicated as a risk factor for various neuropathologic states and some movement disorders. The aim of our study was to determine whethere is an association between serum total homocysteine level and the presence of tardive movement disorders [TMD] among schizophrenic and schizoaffective patients.
J Clin Psychiatry. 2005 ; 66: 1558 -63. High serum homocysteine levels in young male schizophrenic and schizoaffective patients with tardive parkinsonism and/or tardive dyskinesia. Lerner V, Miodownik C, Kaptsan A, Vishne T, Sela BA and Levine J. An elevated serum level of total homocysteine has been implicated as a risk factor for various neuropathologic states and some movement disorders. The aim of our study was to determine whethere is an association between serum total homocysteine level and the presence of tardive movement disorders [TMD] among schizophrenic and schizoaffective patients.
 METHOD: 58 patients with schizophrenia or – schizoaffective disorder (DSM-IV) and TMD for at least 1 year (38 men, 20 women; age range, 28 -73 years) were compared to a control group of 188 patients with DSM-IV -diagnosed schizophrenia or schizoaffective disorder without TMD (123 men, 65 women; age range, 19 -66 years) regarding serum total homocysteine levels. RESULTS: Men with TMD (demonstrating tardive – parkinsonism and/or TD) had significantly higher mean serum total homocysteine levels compared to sex- and age group-matched controls. The difference between groups was almost entirely attributable to the homocysteine levels of young male patients (age group, 19 -40 years old) with TMD. CONCLUSION: High serum total homocysteine level may – constitute a risk factor for certain variants of TMD, especially in young schizophrenic or schizo-affective male patients. Further prospective studies are needed to clarify these findings.
METHOD: 58 patients with schizophrenia or – schizoaffective disorder (DSM-IV) and TMD for at least 1 year (38 men, 20 women; age range, 28 -73 years) were compared to a control group of 188 patients with DSM-IV -diagnosed schizophrenia or schizoaffective disorder without TMD (123 men, 65 women; age range, 19 -66 years) regarding serum total homocysteine levels. RESULTS: Men with TMD (demonstrating tardive – parkinsonism and/or TD) had significantly higher mean serum total homocysteine levels compared to sex- and age group-matched controls. The difference between groups was almost entirely attributable to the homocysteine levels of young male patients (age group, 19 -40 years old) with TMD. CONCLUSION: High serum total homocysteine level may – constitute a risk factor for certain variants of TMD, especially in young schizophrenic or schizo-affective male patients. Further prospective studies are needed to clarify these findings.
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, diabetes mellitus
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, diabetes mellitus
 Osteoporosis
Osteoporosis
 • Am J Psychiatry 163: 549 -a-550, March 2006 Letter to the Editor Osteoporosis and Schizophrenia JOSEPH LEVINE, and ROBERT H. BELMAKER • Martina Hummer, M. D. , et al. (2005) reported the occurrence of low bone mineral density in a group of young male subjects with schizophrenia. Levine et al. (2002) and Applebaum et al (2004) reported elevated plasma homocysteine levels in young male schizophrenic patients. • Elevated homocysteine plasma levels were recently reported to be associated with osteoporotic bone fractures in the elderly in two large follow-up studies. • Mc. Lean et al. (2004) analyzed blood samples obtained and stored from 1, 999 men and women as part of the long-term Framingham Study. These researchers found that men and women in the upper quartile of homocysteine concentrations were nearly four and two times, respectively, as likely to later have a hip fracture in comparison to the lower quartile of homocysteine concentrations.
• Am J Psychiatry 163: 549 -a-550, March 2006 Letter to the Editor Osteoporosis and Schizophrenia JOSEPH LEVINE, and ROBERT H. BELMAKER • Martina Hummer, M. D. , et al. (2005) reported the occurrence of low bone mineral density in a group of young male subjects with schizophrenia. Levine et al. (2002) and Applebaum et al (2004) reported elevated plasma homocysteine levels in young male schizophrenic patients. • Elevated homocysteine plasma levels were recently reported to be associated with osteoporotic bone fractures in the elderly in two large follow-up studies. • Mc. Lean et al. (2004) analyzed blood samples obtained and stored from 1, 999 men and women as part of the long-term Framingham Study. These researchers found that men and women in the upper quartile of homocysteine concentrations were nearly four and two times, respectively, as likely to later have a hip fracture in comparison to the lower quartile of homocysteine concentrations.
 • The mechanism underlying homocysteine’s effect on bone metabolism is not yet clear. However, several mechanisms were suggested, including that elevated homocysteine disturbs the cross-linking of collagen in bone and disturbs osteoblast formation. • Thus, it is suggested that elevated homocysteine levels may be a mechanism of the low bone mineral density reported by Dr. Hummer et al. (2005) among young male subjects suffering from schizophrenia. • References • Hummer M, Malik P, Gasser RW, Hofer A, Kemmler G, Naveda RCM, Rettenbacher MA, Fleischhacker WW: Osteoporosis in patients with schizophrenia. Am J Psychiatry 2005; 162: 162– 167 • Mc. Lean RR, Jacques PF, Selhub J, Tucker KL, Samelson EJ, Broe KE, Hannan MT, Cupples LA, Kiel DP: Homocysteine as a predictive factor for hip fracture in older persons. N Engl J Med 2004; 350: 2042– 2049
• The mechanism underlying homocysteine’s effect on bone metabolism is not yet clear. However, several mechanisms were suggested, including that elevated homocysteine disturbs the cross-linking of collagen in bone and disturbs osteoblast formation. • Thus, it is suggested that elevated homocysteine levels may be a mechanism of the low bone mineral density reported by Dr. Hummer et al. (2005) among young male subjects suffering from schizophrenia. • References • Hummer M, Malik P, Gasser RW, Hofer A, Kemmler G, Naveda RCM, Rettenbacher MA, Fleischhacker WW: Osteoporosis in patients with schizophrenia. Am J Psychiatry 2005; 162: 162– 167 • Mc. Lean RR, Jacques PF, Selhub J, Tucker KL, Samelson EJ, Broe KE, Hannan MT, Cupples LA, Kiel DP: Homocysteine as a predictive factor for hip fracture in older persons. N Engl J Med 2004; 350: 2042– 2049
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
 Glucose Metabolism
Glucose Metabolism
 Homocysteine levels and glucose metabolism in non -obese, non-diabetic chronic schizophrenia Henderson DC, Copeland PM, Nguyen DD, Borba CP, Cather C, Eden Evins A, Freudenreich O, Baer L, Goff DC. METHOD: Subjects underwent a nutritional assessment and fasting plasma, serum insulin and homocysteine tests. RESULTS: Males had a significantly higher homocysteine levels than females. Subjects with impaired fasting glucose had significantly higher homocysteine levels than those with normal fasting glucose CONCLUSION: The group with impaired fasting glucose had higher fasting serum homocysteine concentrations than those with normal fasting glucose which supports a connection to elevated homocysteine: an important cardiovascular risk factor.
Homocysteine levels and glucose metabolism in non -obese, non-diabetic chronic schizophrenia Henderson DC, Copeland PM, Nguyen DD, Borba CP, Cather C, Eden Evins A, Freudenreich O, Baer L, Goff DC. METHOD: Subjects underwent a nutritional assessment and fasting plasma, serum insulin and homocysteine tests. RESULTS: Males had a significantly higher homocysteine levels than females. Subjects with impaired fasting glucose had significantly higher homocysteine levels than those with normal fasting glucose CONCLUSION: The group with impaired fasting glucose had higher fasting serum homocysteine concentrations than those with normal fasting glucose which supports a connection to elevated homocysteine: an important cardiovascular risk factor.
 Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
Illness related symptomatology Negative symptoms Cognitive impairment Homocysteine Treatment induced side effects Extrapyramidal symptoms Glucose metabolism abnormalities Osteoporosis Physical morbidity CVD, Diabetes mellitus
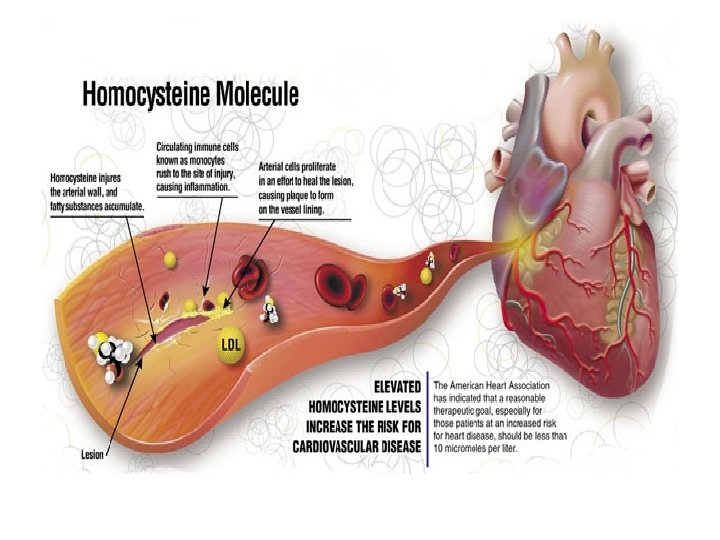
 Does Homocysteine Play a Role in Cardiovascular Morbidity Associated with Schizophrenia ?
Does Homocysteine Play a Role in Cardiovascular Morbidity Associated with Schizophrenia ?
 Acknowledgement: Supported by a Stanley Medical Research Institute Grant (RHB & JL). The Stanley Medical Research Institute had no role in study design, data collection, analysis or interpretation of data or in writing the report or in the decision to submit for publication.
Acknowledgement: Supported by a Stanley Medical Research Institute Grant (RHB & JL). The Stanley Medical Research Institute had no role in study design, data collection, analysis or interpretation of data or in writing the report or in the decision to submit for publication.
 Collaborators: Belmaker RH Ruderman Vladimir Shumeiko Oleg Agam Galila Babushkin I Shimon Hady Ben-Ami Sela Bromberg Anna Lerner Vladimir Bersudsky Yuly Stahl Ziva Appelbaum Julie Beer Sheva Mental Health Center, Israel
Collaborators: Belmaker RH Ruderman Vladimir Shumeiko Oleg Agam Galila Babushkin I Shimon Hady Ben-Ami Sela Bromberg Anna Lerner Vladimir Bersudsky Yuly Stahl Ziva Appelbaum Julie Beer Sheva Mental Health Center, Israel
 Creatine in Psychiatric Disorders
Creatine in Psychiatric Disorders

 Creatine
Creatine
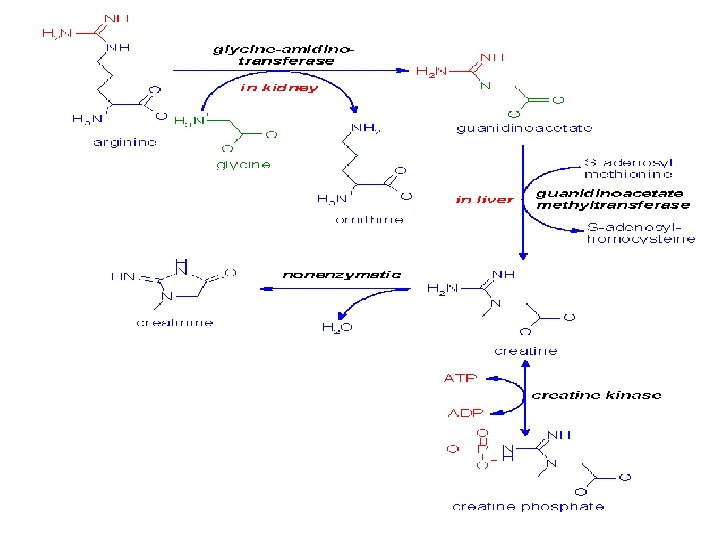
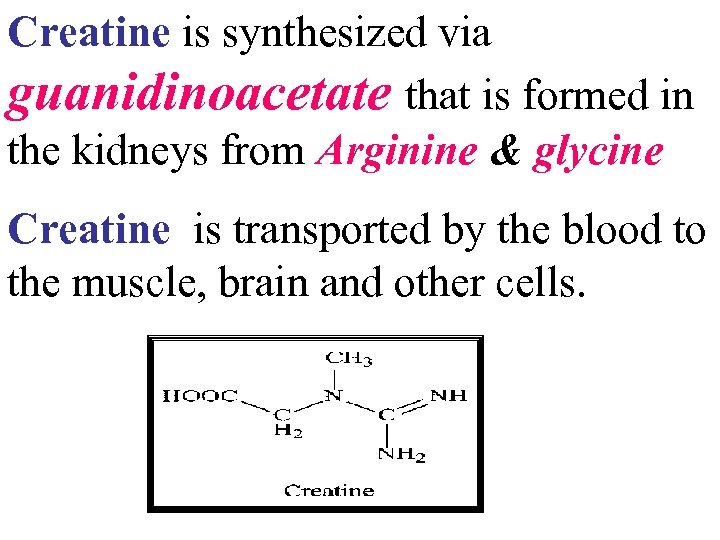 Creatine is synthesized via guanidinoacetate that is formed in the kidneys from Arginine & glycine Creatine is transported by the blood to the muscle, brain and other cells.
Creatine is synthesized via guanidinoacetate that is formed in the kidneys from Arginine & glycine Creatine is transported by the blood to the muscle, brain and other cells.
 Creatine is degraded by non-enzymatic cyclization to creatinine that is renally excreted. This process leads to the loss of about 2 – 4 grams of the total creatine pool (about 140 grams) per day that have to be replaced by creatine synthesized by the liver or taken in with the diet.
Creatine is degraded by non-enzymatic cyclization to creatinine that is renally excreted. This process leads to the loss of about 2 – 4 grams of the total creatine pool (about 140 grams) per day that have to be replaced by creatine synthesized by the liver or taken in with the diet.
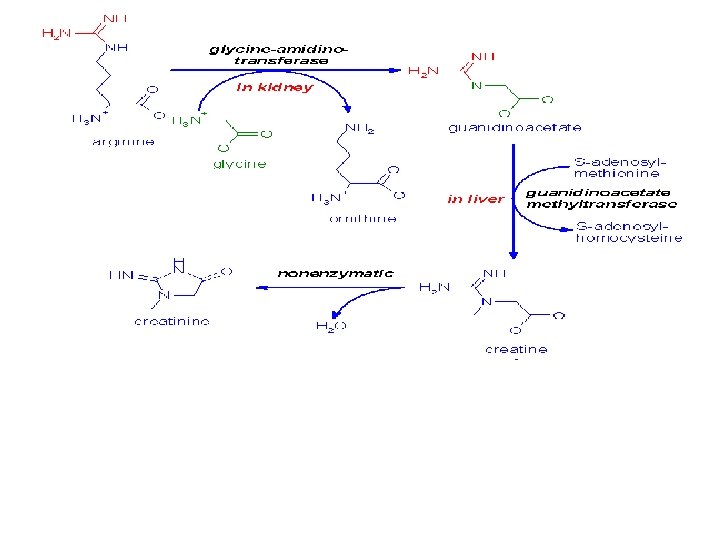
 • Creatine is used as a storage form of high energy phosphate. The phosphate of ATP is transferred to creatine, generating creatine phosphate, through the action of creatine phosphokinase. • The reaction is reversible such that when energy demand is high creatine phosphate donates its phosphate to ADP to yield ATP. Both creatine and creatine phosphate are found in muscle, brain and blood.
• Creatine is used as a storage form of high energy phosphate. The phosphate of ATP is transferred to creatine, generating creatine phosphate, through the action of creatine phosphokinase. • The reaction is reversible such that when energy demand is high creatine phosphate donates its phosphate to ADP to yield ATP. Both creatine and creatine phosphate are found in muscle, brain and blood.
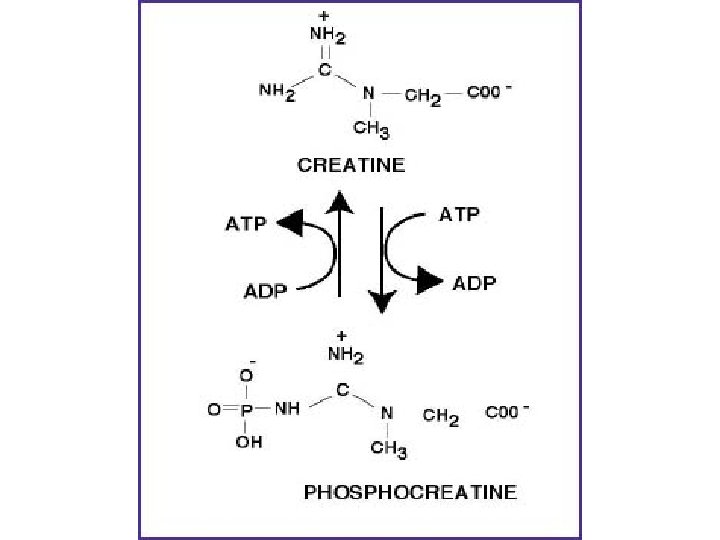
 Creatine plays a pivotal role in brain energy homeostasis, being a temporal and spatial buffer for cytosolic and mitochondrial pools of the cellular energy currency adenosinetriphosphate (Wyss & Kaddurah-Daouk, 2000).
Creatine plays a pivotal role in brain energy homeostasis, being a temporal and spatial buffer for cytosolic and mitochondrial pools of the cellular energy currency adenosinetriphosphate (Wyss & Kaddurah-Daouk, 2000).
 Creatine supplementation is widely used in enhancing sports performance, and has been tried in the treatment of neurological, neuromuscular and atherosclerotic disease with a paucity of side effects (Persky & Brazeua, 2001).
Creatine supplementation is widely used in enhancing sports performance, and has been tried in the treatment of neurological, neuromuscular and atherosclerotic disease with a paucity of side effects (Persky & Brazeua, 2001).
 Creatine in Huntington disease is safe, tolerable, bioavailable in brain and reduces serum 8 OH 2'd. G. Hersch et al In a randomized, double-blind, placebo-controlled study in 64 subjects with Huntington disease (HD), 8 g/day of creatine administered for 16 weeks was well tolerated and safe. Serum and brain creatine concentrations increased in the creatine-treated group and returned to baseline after washout. Serum 8 -hydroxy-2'-deoxyguanosine (8 OH 2'd. G) levels, an indicator of oxidative injury to DNA, were markedly elevated in HD and reduced by creatine treatment. Neurology. 2006 Jan 24; 66(2): 250 -2.
Creatine in Huntington disease is safe, tolerable, bioavailable in brain and reduces serum 8 OH 2'd. G. Hersch et al In a randomized, double-blind, placebo-controlled study in 64 subjects with Huntington disease (HD), 8 g/day of creatine administered for 16 weeks was well tolerated and safe. Serum and brain creatine concentrations increased in the creatine-treated group and returned to baseline after washout. Serum 8 -hydroxy-2'-deoxyguanosine (8 OH 2'd. G) levels, an indicator of oxidative injury to DNA, were markedly elevated in HD and reduced by creatine treatment. Neurology. 2006 Jan 24; 66(2): 250 -2.
 A pilot clinical trial of creatine and minocycline in early Parkinson disease: 18 -month results. NINDS NET-PD Investigators. The NET-PD FS-1 futility study on creatine and minocycline found neither agent futile in slowing down the progression of disability in Parkinson disease (PD) at 12 months using the prespecified futility threshold. Additional 6 months of follow-up in randomized, blinded phase II trial of creatine (dosage, 10 g/d) and minocycline (dosage, 200 mg/d) in subjects with early PD. Data from this small, 18 -month phase II trial of creatine and minocycline do not demonstrate safety concerns that would preclude a large, phase III efficacy trial, although the decreased tolerability of minocycline is a concern. Clin Neuropharmacol. 2008 May-Jun; 31(3): 141 -50
A pilot clinical trial of creatine and minocycline in early Parkinson disease: 18 -month results. NINDS NET-PD Investigators. The NET-PD FS-1 futility study on creatine and minocycline found neither agent futile in slowing down the progression of disability in Parkinson disease (PD) at 12 months using the prespecified futility threshold. Additional 6 months of follow-up in randomized, blinded phase II trial of creatine (dosage, 10 g/d) and minocycline (dosage, 200 mg/d) in subjects with early PD. Data from this small, 18 -month phase II trial of creatine and minocycline do not demonstrate safety concerns that would preclude a large, phase III efficacy trial, although the decreased tolerability of minocycline is a concern. Clin Neuropharmacol. 2008 May-Jun; 31(3): 141 -50
 Creatine enters the brain via a specialized sodium dependent transporter. Dechent et al (1999) studied the effect of oral creatine supplementation of 20 g/day for 4 wk demonstrating a significant increase of mean concentration of total creatine across brain regions (8. 7% corresponding to 0. 6 m. M, P < 0. 001). Lyoo et al (2003) studied magnetic resonance spectroscopy of high-energy phosphate metabolites in human brain following oral supplementation of creatine reporting that creatine (0. 3 g/kg/day for the first 7 days and 0. 03 g/kg/day for the next 7 days) significantly increased brain creatine levels.
Creatine enters the brain via a specialized sodium dependent transporter. Dechent et al (1999) studied the effect of oral creatine supplementation of 20 g/day for 4 wk demonstrating a significant increase of mean concentration of total creatine across brain regions (8. 7% corresponding to 0. 6 m. M, P < 0. 001). Lyoo et al (2003) studied magnetic resonance spectroscopy of high-energy phosphate metabolites in human brain following oral supplementation of creatine reporting that creatine (0. 3 g/kg/day for the first 7 days and 0. 03 g/kg/day for the next 7 days) significantly increased brain creatine levels.
 These findings suggest the possibility of using oral creatine supplementation to modify brain high-energy phosphate metabolism in subjects with various brain disorders, including; schizophrenia major depression and bipolar disorder where alterations in brain highenergy phosphate metabolism have been reported.
These findings suggest the possibility of using oral creatine supplementation to modify brain high-energy phosphate metabolism in subjects with various brain disorders, including; schizophrenia major depression and bipolar disorder where alterations in brain highenergy phosphate metabolism have been reported.

 Rae et al (2003) reported that creatine supplementation (5 grams per day for 6 weeks) had a significant positive effect on both working memory (backward digit span) and Raven's Advanced Progressive Matrices. These findings suggest a role of brain energy capacity in influencing brain cognitive performance and that creatine via its effects on brain energy metabolism may exert beneficial effects on cognition.
Rae et al (2003) reported that creatine supplementation (5 grams per day for 6 weeks) had a significant positive effect on both working memory (backward digit span) and Raven's Advanced Progressive Matrices. These findings suggest a role of brain energy capacity in influencing brain cognitive performance and that creatine via its effects on brain energy metabolism may exert beneficial effects on cognition.
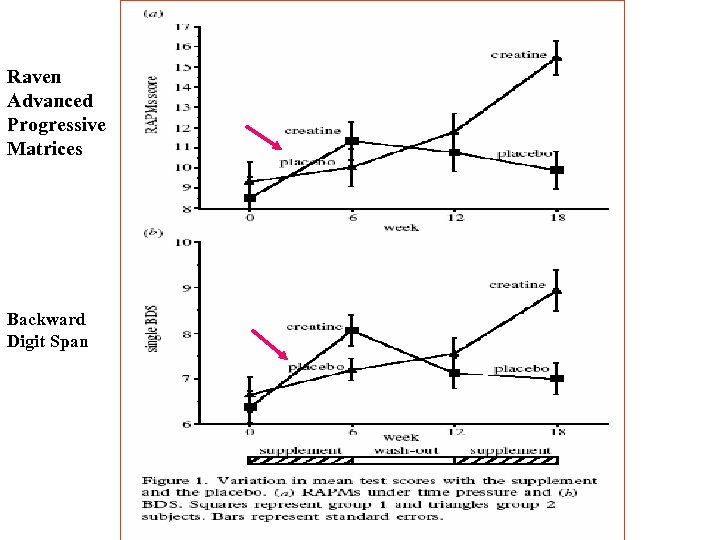 Raven Advanced Progressive Matrices Backward Digit Span
Raven Advanced Progressive Matrices Backward Digit Span
 Several independent lines of evidence suggest the possible involvement of altered cerebral energy metabolism in the pathophysiology of schizophrenia and affective disorders.
Several independent lines of evidence suggest the possible involvement of altered cerebral energy metabolism in the pathophysiology of schizophrenia and affective disorders.
 Several studies also observed alterations in brain metabolic rates in other brain regions including the temporal lobes, the thalamus and the basal ganglia in schizophrenia. This led to the suggestion of an impairment in the fronto-striatal-thalamic circuitry in schizophrenia rather than in a specific brain region (Andreasen et al. 1997).
Several studies also observed alterations in brain metabolic rates in other brain regions including the temporal lobes, the thalamus and the basal ganglia in schizophrenia. This led to the suggestion of an impairment in the fronto-striatal-thalamic circuitry in schizophrenia rather than in a specific brain region (Andreasen et al. 1997).
 A direct link to phosphocreatine and ATP energy systems came from studies using 31 P-MRS with or without chemical shift imaging, which enabled the measurement of ATP, phosphocreatine and inorganic phosphate. These studies showed reduced ATP in the frontal lobe and in left temporal lobe of schizophrenic patients as compared to controls (Volz et al. 2000). Altered brain energy metabolism could be due to impairment of mitochondria and a variety of studies reviewed recently by Ben Shachar (2002) suggest impaired mitochondrial energy metabolism in schizophrenia.
A direct link to phosphocreatine and ATP energy systems came from studies using 31 P-MRS with or without chemical shift imaging, which enabled the measurement of ATP, phosphocreatine and inorganic phosphate. These studies showed reduced ATP in the frontal lobe and in left temporal lobe of schizophrenic patients as compared to controls (Volz et al. 2000). Altered brain energy metabolism could be due to impairment of mitochondria and a variety of studies reviewed recently by Ben Shachar (2002) suggest impaired mitochondrial energy metabolism in schizophrenia.
 J Neurochem. 2002 Dec; 83(6): 1241 -51. Mitochondrial dysfunction in schizophrenia: a possible linkage to dopamine. Ben-Shachar et al
J Neurochem. 2002 Dec; 83(6): 1241 -51. Mitochondrial dysfunction in schizophrenia: a possible linkage to dopamine. Ben-Shachar et al
 Creatine as a New Treatment Strategy in Schizophrenia : A Double-Blind Trial Kaptsan A, Odessky A, Osher Y, Belmaker RH and Levine J Ben Gurion University of the Negev, Beersheva, Israel
Creatine as a New Treatment Strategy in Schizophrenia : A Double-Blind Trial Kaptsan A, Odessky A, Osher Y, Belmaker RH and Levine J Ben Gurion University of the Negev, Beersheva, Israel
 Methods Twelve patients were treated with creatine monohydrate or placebo, each for 3 months in a double-blind crossover design. Rating scales included scales for assessing negative and positive symptoms of schizophrenia, clinical global impressions scale, scales for side–effects and a cognitive battery
Methods Twelve patients were treated with creatine monohydrate or placebo, each for 3 months in a double-blind crossover design. Rating scales included scales for assessing negative and positive symptoms of schizophrenia, clinical global impressions scale, scales for side–effects and a cognitive battery
 Results Creatine treatment was not superior over placebo in reducing the score of PANSS, CGI and the neurocognitive tests applied. Side effects of creatine treatment were few and included nausea and vomiting
Results Creatine treatment was not superior over placebo in reducing the score of PANSS, CGI and the neurocognitive tests applied. Side effects of creatine treatment were few and included nausea and vomiting
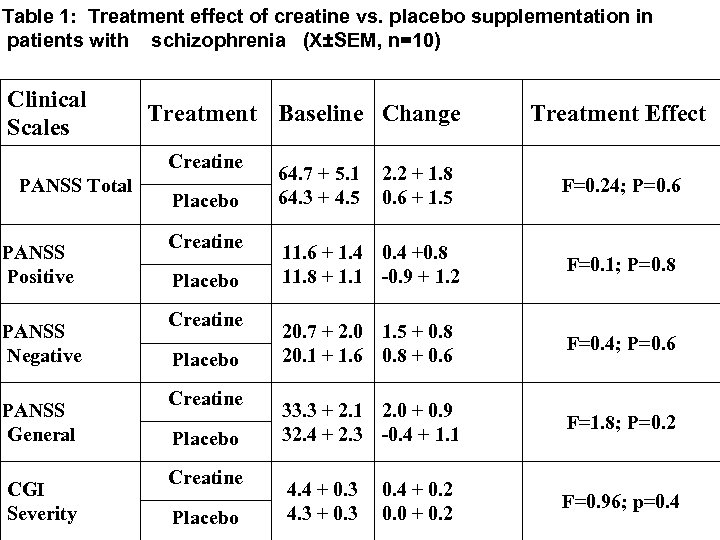 Table 1: Treatment effect of creatine vs. placebo supplementation in patients with schizophrenia (X±SEM, n=10) Clinical Scales Treatment Baseline Change Creatine PANSS Total PANSS Positive PANSS Negative PANSS General CGI Severity Placebo Creatine Placebo 64. 7 + 5. 1 64. 3 + 4. 5 2. 2 + 1. 8 0. 6 + 1. 5 Treatment Effect F=0. 24; P=0. 6 11. 6 + 1. 4 0. 4 +0. 8 11. 8 + 1. 1 -0. 9 + 1. 2 F=0. 1; P=0. 8 20. 7 + 2. 0 1. 5 + 0. 8 20. 1 + 1. 6 0. 8 + 0. 6 F=0. 4; P=0. 6 33. 3 + 2. 1 2. 0 + 0. 9 32. 4 + 2. 3 -0. 4 + 1. 1 F=1. 8; P=0. 2 4. 4 + 0. 3 4. 3 + 0. 3 F=0. 96; p=0. 4 + 0. 2 0. 0 + 0. 2
Table 1: Treatment effect of creatine vs. placebo supplementation in patients with schizophrenia (X±SEM, n=10) Clinical Scales Treatment Baseline Change Creatine PANSS Total PANSS Positive PANSS Negative PANSS General CGI Severity Placebo Creatine Placebo 64. 7 + 5. 1 64. 3 + 4. 5 2. 2 + 1. 8 0. 6 + 1. 5 Treatment Effect F=0. 24; P=0. 6 11. 6 + 1. 4 0. 4 +0. 8 11. 8 + 1. 1 -0. 9 + 1. 2 F=0. 1; P=0. 8 20. 7 + 2. 0 1. 5 + 0. 8 20. 1 + 1. 6 0. 8 + 0. 6 F=0. 4; P=0. 6 33. 3 + 2. 1 2. 0 + 0. 9 32. 4 + 2. 3 -0. 4 + 1. 1 F=1. 8; P=0. 2 4. 4 + 0. 3 4. 3 + 0. 3 F=0. 96; p=0. 4 + 0. 2 0. 0 + 0. 2
 Conclusions • This study (creatine - 5 grams daily for 3 months) failed to report an effect of creatine monohydrate treatment on the symptomatology and cognitive functions of patients with schizophrenia. • Higher doses of creatine administered for longer periods of time may be still effective in schizophrenia. • Alternatively, creatine is suggested to globally enhance brain energy metabolism. This does not necessarily refute the future use of agents with more specific effects on brain energy metabolism, affecting hypometabolic frontal brain regions, whereas sparing other normal or hypermetabolic brain areas.
Conclusions • This study (creatine - 5 grams daily for 3 months) failed to report an effect of creatine monohydrate treatment on the symptomatology and cognitive functions of patients with schizophrenia. • Higher doses of creatine administered for longer periods of time may be still effective in schizophrenia. • Alternatively, creatine is suggested to globally enhance brain energy metabolism. This does not necessarily refute the future use of agents with more specific effects on brain energy metabolism, affecting hypometabolic frontal brain regions, whereas sparing other normal or hypermetabolic brain areas.
 Creatine Monohydrate in Resistant Depression: a preliminary study Roitman S, Green T, Osher Y, Karni N and Joseph Levine Faculty of Health Sciences, Ben Gurion University of the Negev, Beersheva, Israel
Creatine Monohydrate in Resistant Depression: a preliminary study Roitman S, Green T, Osher Y, Karni N and Joseph Levine Faculty of Health Sciences, Ben Gurion University of the Negev, Beersheva, Israel
 Accumulated evidence suggests the possible involvement of hypoactive prefrontal cerebral energy metabolism in the pathophysiology of unipolar and bipolar depression (Ketter et al, 2001) as well as decreased brain creatine containing compounds in depressed patients (Kato et al, 1992; Dager et al, 2004). In this regard, Kato et al (1992)reported decreased brain phosphocreatine in severely (as opposed to mildly) depressed patients and Dager et al (2004) studying depressed or mixed-state bipolar patients reported an inverse correlation between severity of depression and white matter creatine levels.
Accumulated evidence suggests the possible involvement of hypoactive prefrontal cerebral energy metabolism in the pathophysiology of unipolar and bipolar depression (Ketter et al, 2001) as well as decreased brain creatine containing compounds in depressed patients (Kato et al, 1992; Dager et al, 2004). In this regard, Kato et al (1992)reported decreased brain phosphocreatine in severely (as opposed to mildly) depressed patients and Dager et al (2004) studying depressed or mixed-state bipolar patients reported an inverse correlation between severity of depression and white matter creatine levels.
 Several studies also suggest that agents with reported antidepressant activity may increase brain levels of creatine containing compounds Silveri et al S-adenosyl-L-methionine: effects on brain bioenergetic status and transverse relaxation time in healthy subjects. Biol Psychiatry 2003; 54(8): 833 -9.
Several studies also suggest that agents with reported antidepressant activity may increase brain levels of creatine containing compounds Silveri et al S-adenosyl-L-methionine: effects on brain bioenergetic status and transverse relaxation time in healthy subjects. Biol Psychiatry 2003; 54(8): 833 -9.
 31 P-MRS study of acetyl-L-carnitine treatment in geriatric depression: preliminary results. Pettegrew JW, Levine J, Gershon S et al Neurophysics Laboratory, Department of Psychiatry, School of Medicine, University of Pittsburgh, PA, USA. A 12 -week study of two elderly, depressed subjects investigated the effect of acetyl-L-carnitine (ALCAR) treatment on the Hamilton Depression Rating Scale (HDRS) and on measures of high-energy phosphate and membrane phospholipid metabolism. High-energy and membrane phospholipid metabolites were measured by phosphorus magnetic resonance spectroscopic imaging (31 P MRSI) analysis. HDRS and 31 P MRSI measurements were taken at entry, 6 and 12 weeks for the depressed subjects. 31 P MRSI analysis revealed that ALCAR treatment resulted in increasing levels of the prefrontal phosphocreatine (PCr), which correlated with HDRS. . Bipolar Disord. 2002 Feb; 4(1): 61 -6.
31 P-MRS study of acetyl-L-carnitine treatment in geriatric depression: preliminary results. Pettegrew JW, Levine J, Gershon S et al Neurophysics Laboratory, Department of Psychiatry, School of Medicine, University of Pittsburgh, PA, USA. A 12 -week study of two elderly, depressed subjects investigated the effect of acetyl-L-carnitine (ALCAR) treatment on the Hamilton Depression Rating Scale (HDRS) and on measures of high-energy phosphate and membrane phospholipid metabolism. High-energy and membrane phospholipid metabolites were measured by phosphorus magnetic resonance spectroscopic imaging (31 P MRSI) analysis. HDRS and 31 P MRSI measurements were taken at entry, 6 and 12 weeks for the depressed subjects. 31 P MRSI analysis revealed that ALCAR treatment resulted in increasing levels of the prefrontal phosphocreatine (PCr), which correlated with HDRS. . Bipolar Disord. 2002 Feb; 4(1): 61 -6.
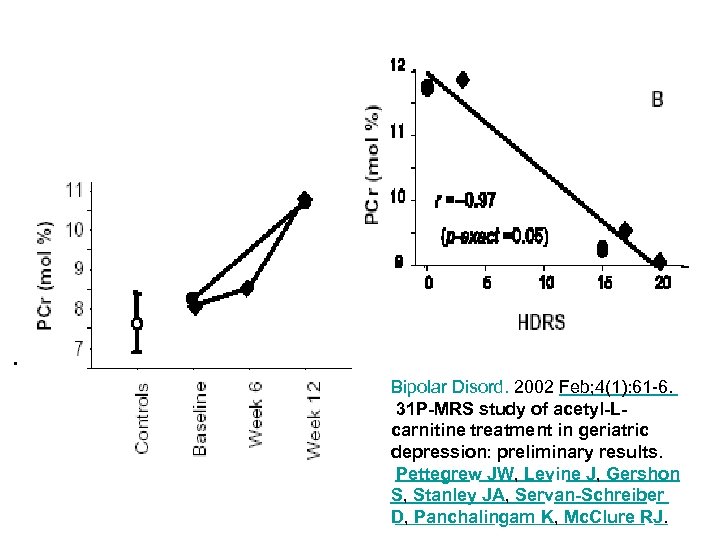 Bipolar Disord. 2002 Feb; 4(1): 61 -6. 31 P-MRS study of acetyl-Lcarnitine treatment in geriatric depression: preliminary results. Pettegrew JW, Levine J, Gershon S, Stanley JA, Servan-Schreiber D, Panchalingam K, Mc. Clure RJ.
Bipolar Disord. 2002 Feb; 4(1): 61 -6. 31 P-MRS study of acetyl-Lcarnitine treatment in geriatric depression: preliminary results. Pettegrew JW, Levine J, Gershon S, Stanley JA, Servan-Schreiber D, Panchalingam K, Mc. Clure RJ.
 • Taken together, these findings suggest the possibility of using oral creatine supplementation to increase brain creatine containing compounds and modify brain high-energy phosphate metabolism in key hypoactive brain areas in subjects with unipolar and bipolar depression.
• Taken together, these findings suggest the possibility of using oral creatine supplementation to increase brain creatine containing compounds and modify brain high-energy phosphate metabolism in key hypoactive brain areas in subjects with unipolar and bipolar depression.
 Methods The study was an open, 4 week clinical add-on trial examining the effect of creatine monohydrate in the treatment of resistant depression. All 10 patients except one bipolar patient had been treated with antidepressants in adequate doses for at least 6 weeks prior to participation in the study, without any clinically significant improvement. Three patients had comorbid medically stable hypertension and/or diabetes mellitus type II. Patients had no history of alcohol or drug abuse.
Methods The study was an open, 4 week clinical add-on trial examining the effect of creatine monohydrate in the treatment of resistant depression. All 10 patients except one bipolar patient had been treated with antidepressants in adequate doses for at least 6 weeks prior to participation in the study, without any clinically significant improvement. Three patients had comorbid medically stable hypertension and/or diabetes mellitus type II. Patients had no history of alcohol or drug abuse.
 Methods Creatine monohydrate was administered for 4 weeks (3 g. daily in the first week followed by 5 g. daily for another 3 weeks). Ongoing psychotropic treatment was not changed during the study.
Methods Creatine monohydrate was administered for 4 weeks (3 g. daily in the first week followed by 5 g. daily for another 3 weeks). Ongoing psychotropic treatment was not changed during the study.
 The Hamilton Depression Scale (HDS), Hamilton Anxiety Scale (HAS), and Clinical Global Impression (CGI) scores were recorded at baseline and at weeks one, two, three, and four.
The Hamilton Depression Scale (HDS), Hamilton Anxiety Scale (HAS), and Clinical Global Impression (CGI) scores were recorded at baseline and at weeks one, two, three, and four.
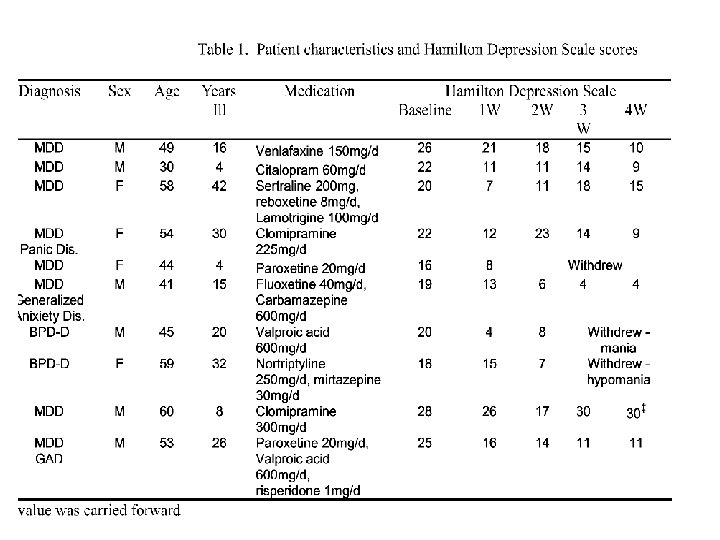
 Results Seven patients completed at least three weeks of the study. One way repeated measures ANOVA showed significant improvement on all scales: Mean(+ SD) CGI , score decreased from 4. 43(0. 5) at baseline to 3. 00(1. 4), at week 4 [p=0. 02]. Mean(+ SD) HDS score decreased from 23. 14(3. 3) at baseline to 12. 57(8. 3) at week 4 [p=0. 002]. Mean(+ SD) HAS scores decreased from 18. 71(3. 1) at baseline to 12. 00(6. 2) at week 4 [p=0. 016]. LSD post-hoc testing revealed that each of the outcome measures improved significantly (p<0. 012) over baseline by week one.
Results Seven patients completed at least three weeks of the study. One way repeated measures ANOVA showed significant improvement on all scales: Mean(+ SD) CGI , score decreased from 4. 43(0. 5) at baseline to 3. 00(1. 4), at week 4 [p=0. 02]. Mean(+ SD) HDS score decreased from 23. 14(3. 3) at baseline to 12. 57(8. 3) at week 4 [p=0. 002]. Mean(+ SD) HAS scores decreased from 18. 71(3. 1) at baseline to 12. 00(6. 2) at week 4 [p=0. 016]. LSD post-hoc testing revealed that each of the outcome measures improved significantly (p<0. 012) over baseline by week one.
 Results Two female patients (ED & ZC) showed transient increases in HDS scores following dose increase to 5 g/d. In both cases, when creatine dose was returned to 3 g/d, improvement was noted by the following week. One patient (MH) did not improve while on creatine treatment and withdrew from the study after week three. Three patients did not complete at least three weeks of the study: two bipolar patients showed improvement of depression first but then dropped out due to development of mania or hypomania; the third patient improved considerably during the first week and discontinued treatment.
Results Two female patients (ED & ZC) showed transient increases in HDS scores following dose increase to 5 g/d. In both cases, when creatine dose was returned to 3 g/d, improvement was noted by the following week. One patient (MH) did not improve while on creatine treatment and withdrew from the study after week three. Three patients did not complete at least three weeks of the study: two bipolar patients showed improvement of depression first but then dropped out due to development of mania or hypomania; the third patient improved considerably during the first week and discontinued treatment.
 Adverse reactions were few and mild. Two patients complained of transient nuasea, in one case including transient flatus and constipation. These complaints disappeared by week four. . Follow up: after termination of the study three of the seven completers reported a worsening of their depressive and anxiety symptoms. They then restarted creatine with considerable improvement in their condition within one to two weeks.
Adverse reactions were few and mild. Two patients complained of transient nuasea, in one case including transient flatus and constipation. These complaints disappeared by week four. . Follow up: after termination of the study three of the seven completers reported a worsening of their depressive and anxiety symptoms. They then restarted creatine with considerable improvement in their condition within one to two weeks.
 Discussion This preliminary open label augmentation study of creatine monohydrate – an agent which enhances brain energy metabolism- demonstrated a beneficial effect in the treatment of resistant depression. Five out of the seven completers achieved a reduction of greater than 50% in baseline HDS scores.
Discussion This preliminary open label augmentation study of creatine monohydrate – an agent which enhances brain energy metabolism- demonstrated a beneficial effect in the treatment of resistant depression. Five out of the seven completers achieved a reduction of greater than 50% in baseline HDS scores.
 Discussion Two of our subjects have shown a transient increase in HDS. A reduction in Cr treatment from 5 to 3 grams was associated in these subjects with a renewed decrease in HDS. While only indicative, this may suggest an inverted U shape response for creatine. This study included two bipolar I patients. Each developed mania or hypomania while treated with Cr. Such a phenomenon may be of interest regarding the pathogenesis of mania. Is the induction of mania associated with the enhancement of brain energy by creatine in certain key brain structures? In this context, SAMe, an antidepressant and a precursor of creatine, was reported to be associated with high rate of manic/hypomanic switch in bipolar patients (Lipinski et al, 2003) and to increase brain phosphocreatine (Silveri et al, 1984). Silveri MM, Parow AM, Villafuerte RA, Damico KE, Goren J, Stoll AL, Cohen BM, Renshaw PF: Sadenosyl-L-methionine: effects on brain bioenergetic status and transverse relaxation time in healthy subjects. Biol Psychiatry 2003; 54(8): 833 -9. Lipinski JF, Cohen BM, Frankenburg F, Tohen M, Waternaux C, Altesman R, Jones B, Harris P: Open trial of S-adenosylmethionine for treatment of depression. Am J Psychiatry 1984; 141(3): 448 -
Discussion Two of our subjects have shown a transient increase in HDS. A reduction in Cr treatment from 5 to 3 grams was associated in these subjects with a renewed decrease in HDS. While only indicative, this may suggest an inverted U shape response for creatine. This study included two bipolar I patients. Each developed mania or hypomania while treated with Cr. Such a phenomenon may be of interest regarding the pathogenesis of mania. Is the induction of mania associated with the enhancement of brain energy by creatine in certain key brain structures? In this context, SAMe, an antidepressant and a precursor of creatine, was reported to be associated with high rate of manic/hypomanic switch in bipolar patients (Lipinski et al, 2003) and to increase brain phosphocreatine (Silveri et al, 1984). Silveri MM, Parow AM, Villafuerte RA, Damico KE, Goren J, Stoll AL, Cohen BM, Renshaw PF: Sadenosyl-L-methionine: effects on brain bioenergetic status and transverse relaxation time in healthy subjects. Biol Psychiatry 2003; 54(8): 833 -9. Lipinski JF, Cohen BM, Frankenburg F, Tohen M, Waternaux C, Altesman R, Jones B, Harris P: Open trial of S-adenosylmethionine for treatment of depression. Am J Psychiatry 1984; 141(3): 448 -