fae4dc84180b6c946468399137b91b92.ppt
- Количество слайдов: 24
 HIV variants and US licensed assays Indira Hewlett, Ph. D Chief, Lab. of Molecular Virology CBER/FDA XIX SOGAT, 2006
HIV variants and US licensed assays Indira Hewlett, Ph. D Chief, Lab. of Molecular Virology CBER/FDA XIX SOGAT, 2006
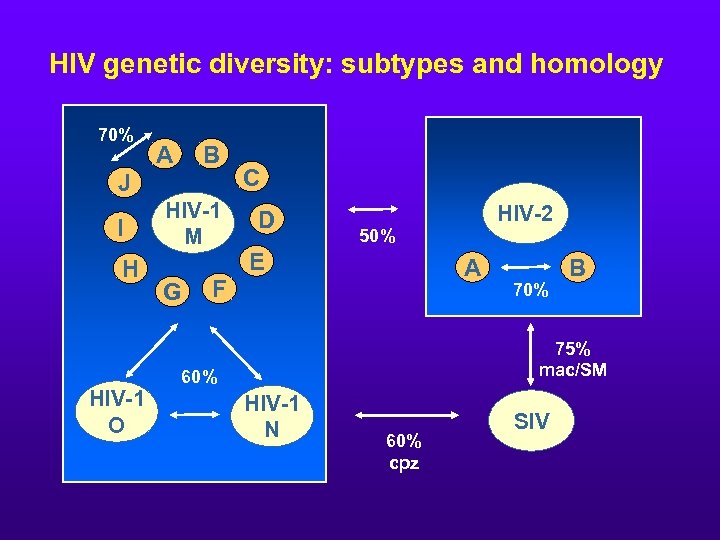 HIV genetic diversity: subtypes and homology 70% A B J I H HIV-1 O HIV-1 M G C D HIV-2 50% E A F 70% B 75% mac/SM 60% HIV-1 N 60% cpz SIV
HIV genetic diversity: subtypes and homology 70% A B J I H HIV-1 O HIV-1 M G C D HIV-2 50% E A F 70% B 75% mac/SM 60% HIV-1 N 60% cpz SIV
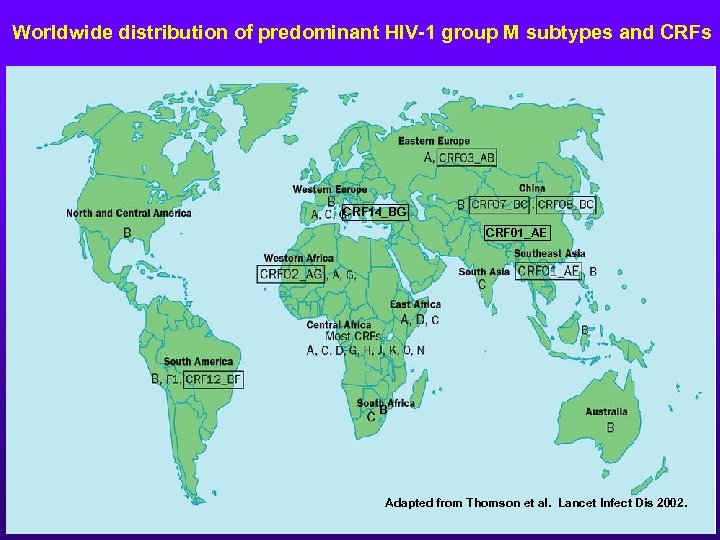 Worldwide distribution of predominant HIV-1 group M subtypes and CRFs CRF 14_BG CRF 01_AE B Adapted from Thomson et al. Lancet Infect Dis 2002.
Worldwide distribution of predominant HIV-1 group M subtypes and CRFs CRF 14_BG CRF 01_AE B Adapted from Thomson et al. Lancet Infect Dis 2002.
 Diagnostic implications • New serologic and NAT assays have limited representation of viral epitopes and sequences • Potential impact on sensitivity for new variants HIV variants • CBER initiated collaboration with Cameroonian Ministry of Health to study HIV diversity and test performance • Cameroon has all known subtypes and new variants
Diagnostic implications • New serologic and NAT assays have limited representation of viral epitopes and sequences • Potential impact on sensitivity for new variants HIV variants • CBER initiated collaboration with Cameroonian Ministry of Health to study HIV diversity and test performance • Cameroon has all known subtypes and new variants
 Study goals § Evaluate sensitivity of existing and new blood screening, rapid and other diagnostic tests for diverse subtypes § Characterize and genotype HIV variants in a region of high genetic diversity § Identify samples to serve as candidate reference reagents
Study goals § Evaluate sensitivity of existing and new blood screening, rapid and other diagnostic tests for diverse subtypes § Characterize and genotype HIV variants in a region of high genetic diversity § Identify samples to serve as candidate reference reagents
 Study Plan and Methods • Blood samples (240 samples) collected from sites around Yaounde tested by a rapid HIV assay used to screen blood donors in Cameroon. • Samples tested by 9 FDA licensed assays • 4 HIV antibody EIAs, 1 p 24 antigen, one IFA, one Wblot, 2 qualitative and one quanitative nucleic acid tests (NAT) • Discordant samples analyzed by in-house test for group O • Genotype analysis performed on HIV positive samples
Study Plan and Methods • Blood samples (240 samples) collected from sites around Yaounde tested by a rapid HIV assay used to screen blood donors in Cameroon. • Samples tested by 9 FDA licensed assays • 4 HIV antibody EIAs, 1 p 24 antigen, one IFA, one Wblot, 2 qualitative and one quanitative nucleic acid tests (NAT) • Discordant samples analyzed by in-house test for group O • Genotype analysis performed on HIV positive samples
 Results • 149/240 were found to be reactive by the test used in Cameroon • 133/149 of samples were confirmed as positive on the basis of reactivity on all tests • 5/149 were negative on all tests • 9/149 were discordant among assays • 2/149 were HIV-2 reactive • 3/149 samples positive by p 24 assays
Results • 149/240 were found to be reactive by the test used in Cameroon • 133/149 of samples were confirmed as positive on the basis of reactivity on all tests • 5/149 were negative on all tests • 9/149 were discordant among assays • 2/149 were HIV-2 reactive • 3/149 samples positive by p 24 assays
 Results – con’t • 91/240 were negative according to tests in Cameroon • 60 negative on all tests; 2/91 were positive • 17/91 were discordant amongst all assays • 12/91 were reactive on HIV-2 assay • 25 samples from both previously screened antibody positive and negative samples were discordant between assays • No HIV group O was detected in discordant samples using an in-house ELISA.
Results – con’t • 91/240 were negative according to tests in Cameroon • 60 negative on all tests; 2/91 were positive • 17/91 were discordant amongst all assays • 12/91 were reactive on HIV-2 assay • 25 samples from both previously screened antibody positive and negative samples were discordant between assays • No HIV group O was detected in discordant samples using an in-house ELISA.
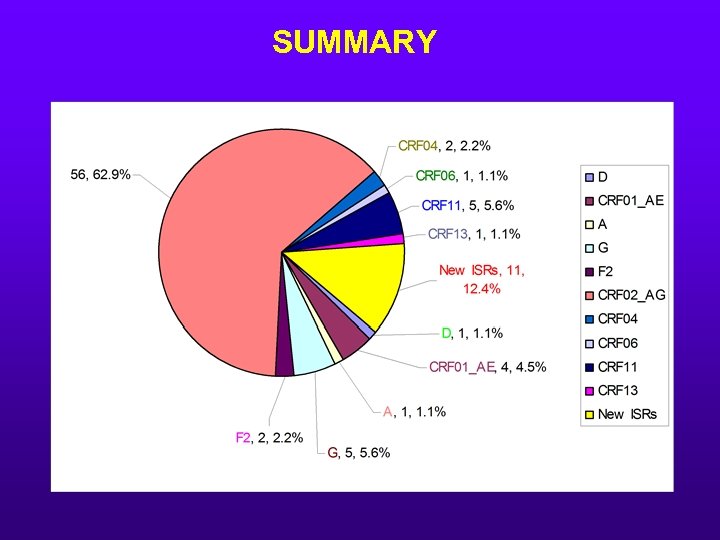 SUMMARY
SUMMARY
 Summary • Current licensed HIV NAT and Ab were able to detect most subtypes and recombinant HIV variants § However a small number of CRF 02 AG were not detected by at least one manufacturer’s assay • CRF 02_AG most prevalent viral strain in Cameroon (62. 9%) • New ISRs identified in this study; reactive in NAT assays. • High reactive rates seen with HIV negative Cameroonian samples
Summary • Current licensed HIV NAT and Ab were able to detect most subtypes and recombinant HIV variants § However a small number of CRF 02 AG were not detected by at least one manufacturer’s assay • CRF 02_AG most prevalent viral strain in Cameroon (62. 9%) • New ISRs identified in this study; reactive in NAT assays. • High reactive rates seen with HIV negative Cameroonian samples
 Regulatory implications • HIV genetic diversity appears to be evolving globally at a fairly rapid rate • Different rates of disease progression for different subtypes recently reported • Continued surveillance for existing and new emerging variants and development of reference reagents may be warranted • HIV variant Samples should be included in the evaluation of HIV and other human retroviral tests
Regulatory implications • HIV genetic diversity appears to be evolving globally at a fairly rapid rate • Different rates of disease progression for different subtypes recently reported • Continued surveillance for existing and new emerging variants and development of reference reagents may be warranted • HIV variant Samples should be included in the evaluation of HIV and other human retroviral tests
 Current PHS efforts • Continued FDA surveillance for viral variants and screening and diagnostic assays • PHS working group formed to monitor emerging natural and drug resistant HIV variants in global setting • Evaluate implications for diagnosis (conventional and rapid), blood screening, therapy and vaccine development • Develop repositories to aid in the evaluation of new diagnostic and blood screening tests, vaccines and new therapies
Current PHS efforts • Continued FDA surveillance for viral variants and screening and diagnostic assays • PHS working group formed to monitor emerging natural and drug resistant HIV variants in global setting • Evaluate implications for diagnosis (conventional and rapid), blood screening, therapy and vaccine development • Develop repositories to aid in the evaluation of new diagnostic and blood screening tests, vaccines and new therapies
 Application of Nanotechnology to diagnostics
Application of Nanotechnology to diagnostics
 Nano-Scale Diagnostics • Nanotechnology offers some potentially unique features based on the size (1 -100 nm scale) and properties that could permit rapid, sensitive detection of multiple pathogens and analytes simultaneously • Nanotechnology-based approaches could potentially provide a new generation of diagnostic assays • Nano-scale detection could permit miniaturization allowing small volumes of sample to be tested with a high degree of sensitivity
Nano-Scale Diagnostics • Nanotechnology offers some potentially unique features based on the size (1 -100 nm scale) and properties that could permit rapid, sensitive detection of multiple pathogens and analytes simultaneously • Nanotechnology-based approaches could potentially provide a new generation of diagnostic assays • Nano-scale detection could permit miniaturization allowing small volumes of sample to be tested with a high degree of sensitivity
 Nanoparticle-Based Bio-Barcode Amplification (BCA) Assay • Based on chemical probes) labeled on the nanoparticles (NPs) and magnetic microparticles (MMPs) • Use barcode DNA-modified NPs for signal amplification and MMPs for easy separation • Particle-initiated Ag developing technique for signal enhancement • High sensitivity but without enzymatic amplification • Microarray format (or could be adapted to ELISA format) • Multiplex assay system for rapid and sensitive detection
Nanoparticle-Based Bio-Barcode Amplification (BCA) Assay • Based on chemical probes) labeled on the nanoparticles (NPs) and magnetic microparticles (MMPs) • Use barcode DNA-modified NPs for signal amplification and MMPs for easy separation • Particle-initiated Ag developing technique for signal enhancement • High sensitivity but without enzymatic amplification • Microarray format (or could be adapted to ELISA format) • Multiplex assay system for rapid and sensitive detection
 Application of BCA to HIV detection • BCA had been applied to detection of PSA • Applicability of BCA to infectious disease testing was explored using HIV p 24 antigen as proof of concept • Potential use in settings where NAT is less available
Application of BCA to HIV detection • BCA had been applied to detection of PSA • Applicability of BCA to infectious disease testing was explored using HIV p 24 antigen as proof of concept • Potential use in settings where NAT is less available
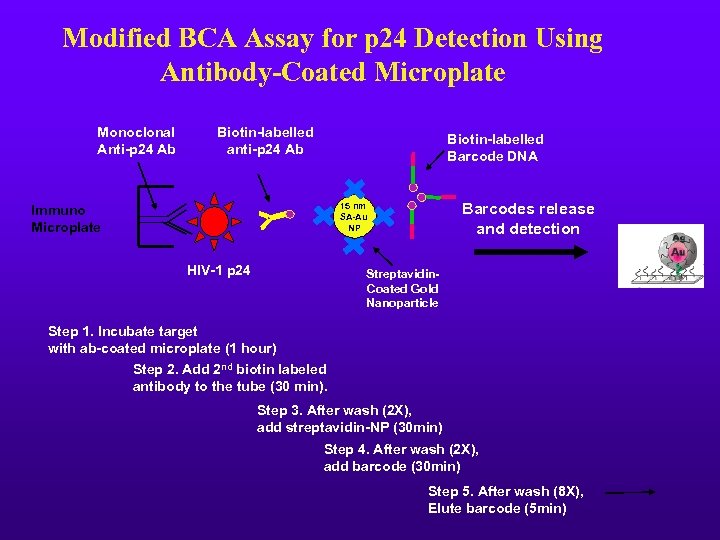 Modified BCA Assay for p 24 Detection Using Antibody-Coated Microplate Monoclonal Anti-p 24 Ab Biotin-labelled anti-p 24 Ab Biotin-labelled Barcode DNA Barcodes release and detection 15 nm SA-Au NP Immuno Microplate HIV-1 p 24 Streptavidin. Coated Gold Nanoparticle Step 1. Incubate target with ab-coated microplate (1 hour) Step 2. Add 2 nd biotin labeled antibody to the tube (30 min). Step 3. After wash (2 X), add streptavidin-NP (30 min) Step 4. After wash (2 X), add barcode (30 min) Step 5. After wash (8 X), Elute barcode (5 min)
Modified BCA Assay for p 24 Detection Using Antibody-Coated Microplate Monoclonal Anti-p 24 Ab Biotin-labelled anti-p 24 Ab Biotin-labelled Barcode DNA Barcodes release and detection 15 nm SA-Au NP Immuno Microplate HIV-1 p 24 Streptavidin. Coated Gold Nanoparticle Step 1. Incubate target with ab-coated microplate (1 hour) Step 2. Add 2 nd biotin labeled antibody to the tube (30 min). Step 3. After wash (2 X), add streptavidin-NP (30 min) Step 4. After wash (2 X), add barcode (30 min) Step 5. After wash (8 X), Elute barcode (5 min)
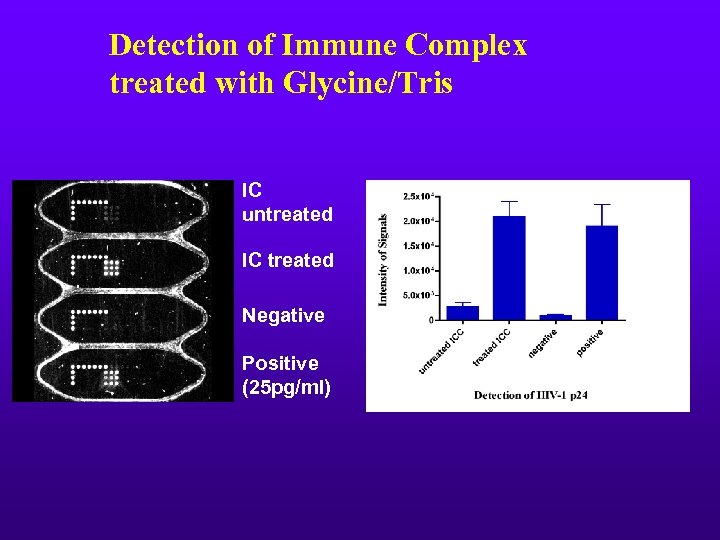 Detection of Immune Complex treated with Glycine/Tris IC untreated IC treated Negative Positive (25 pg/ml)
Detection of Immune Complex treated with Glycine/Tris IC untreated IC treated Negative Positive (25 pg/ml)
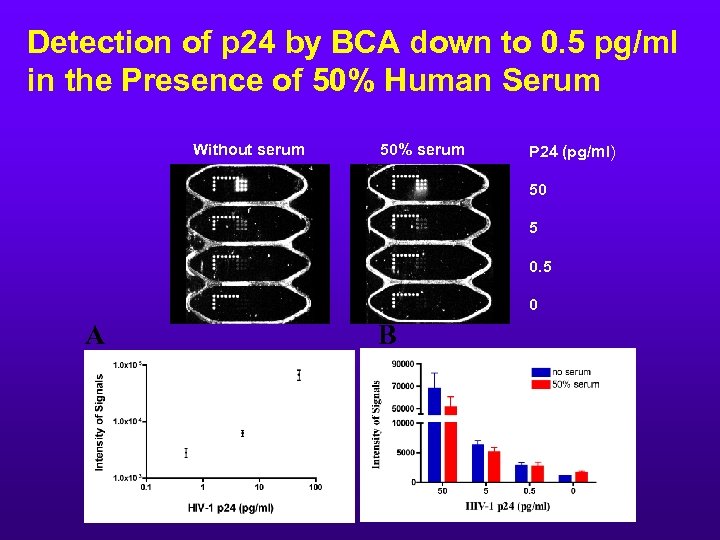 Detection of p 24 by BCA down to 0. 5 pg/ml in the Presence of 50% Human Serum Without serum 50% serum P 24 (pg/ml) 50 5 0 A B
Detection of p 24 by BCA down to 0. 5 pg/ml in the Presence of 50% Human Serum Without serum 50% serum P 24 (pg/ml) 50 5 0 A B
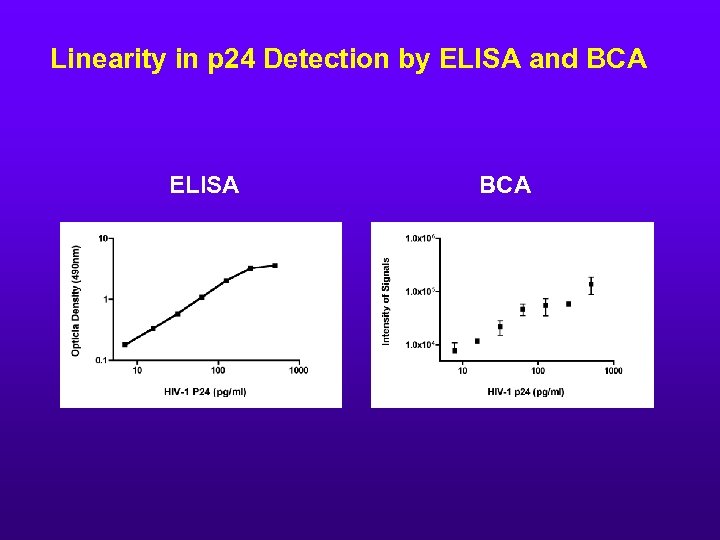 Linearity in p 24 Detection by ELISA and BCA ELISA BCA
Linearity in p 24 Detection by ELISA and BCA ELISA BCA
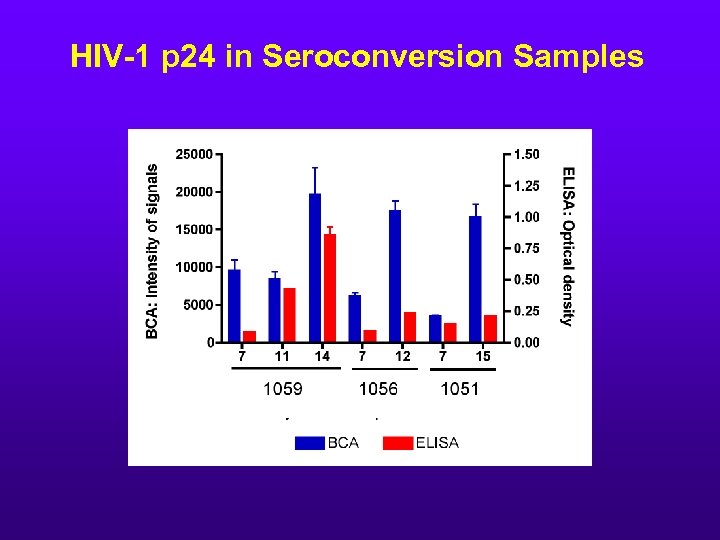 HIV-1 p 24 in Seroconversion Samples
HIV-1 p 24 in Seroconversion Samples
 Preliminary data on performance of BCA with clinical samples Samples tested + - Total + 14 1 15 - 1 30 31 Total 15 31 46 BCA Sensitivity = 14/15 = 93. 3% Specificity = 30/31 = 96. 7% Concordance = 44/46 = 95. 7%
Preliminary data on performance of BCA with clinical samples Samples tested + - Total + 14 1 15 - 1 30 31 Total 15 31 46 BCA Sensitivity = 14/15 = 93. 3% Specificity = 30/31 = 96. 7% Concordance = 44/46 = 95. 7%
 Conclusion • BCA assay could detect 0. 5 pg/ml of HIV-1 p 24 antigen compared with 10 ~ 50 pg of conventional p 24 antigen capture assays (ELISA). • There is a linear relationship between concentration of p 24 and the signal intensities at the range of 0. 5 ~ 500 pg / ml. • BCA may be approx. 20 ~ 100 fold more sensitive than ELISA. • 22 HIV negative samples tested were non reactive when tested by BCA • In seroconversion panels, BCA detected HIV p 24 earlier than current p 24 assay and at the same time as PCR
Conclusion • BCA assay could detect 0. 5 pg/ml of HIV-1 p 24 antigen compared with 10 ~ 50 pg of conventional p 24 antigen capture assays (ELISA). • There is a linear relationship between concentration of p 24 and the signal intensities at the range of 0. 5 ~ 500 pg / ml. • BCA may be approx. 20 ~ 100 fold more sensitive than ELISA. • 22 HIV negative samples tested were non reactive when tested by BCA • In seroconversion panels, BCA detected HIV p 24 earlier than current p 24 assay and at the same time as PCR
 Cameroon Ministry of Health Leopold Zekeng Bih Awazi CBER/FDA Ana Machuca Jinjie Hu Shixing Tang Arindam Dhar Owen Wood Sherwin Lee Steve Kerby Maria Rios NIH/NHLBI G. Nemo L. Harvath Northwestern University Chad Mirkin Stephen Wolinsky Nanosphere James Storhoff Blood Systems Research Institute Philip Norris
Cameroon Ministry of Health Leopold Zekeng Bih Awazi CBER/FDA Ana Machuca Jinjie Hu Shixing Tang Arindam Dhar Owen Wood Sherwin Lee Steve Kerby Maria Rios NIH/NHLBI G. Nemo L. Harvath Northwestern University Chad Mirkin Stephen Wolinsky Nanosphere James Storhoff Blood Systems Research Institute Philip Norris


