f141cc31100c27aafef8f7d56fc1ba7b.ppt
- Количество слайдов: 45
HIV infection in Pregnancy รองศาสตราจารย นายแพทย อตวทธ กมทมาศ สาขาสตศาสตรและนรเวชวทยา คณะแพทยศาสตร มหาวทยาลยธรรมศาสตร

Natural history l l l The principal target=T lymphocytes Specific at CD 4 surface antigen (receptor for the virus) Monocyte-macrophages may be infected Incubation period ; days to weeks Acute retroviral syndrome ; fever, night sweats, fatigue, rash, headache, lymphadenophathy, pharyngitis, myalgias, arthralgias, nausea, vomiting, diarrhea ; lasts < 10 days

l After symptoms abate ; chronic viremia l Median time = 10 years --- AIDS l AIDS; generalized lymphadenopathy, oral hairy leukoplakia, aphthous ulcer, thrombocytopenia, opportunistic infections (candida, HSV, TB, CMV, HPV, PCP, toxo), Kaposi sarcoma, non-Hodgkin lymphoma l Death
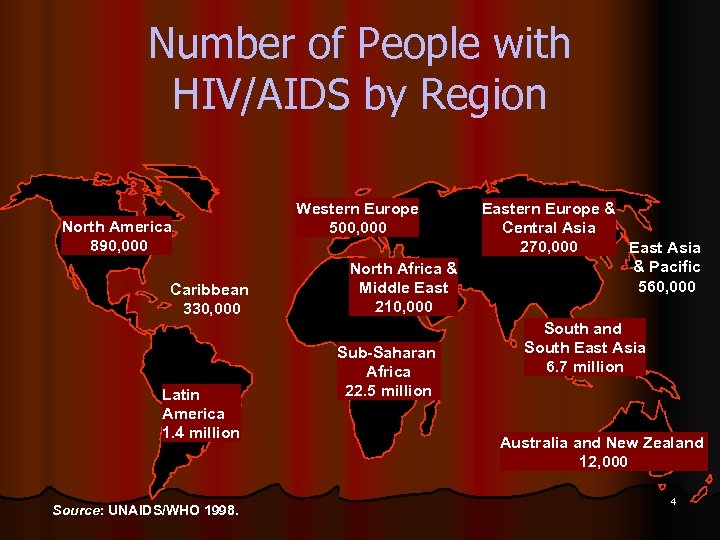
Number of People with HIV/AIDS by Region North America 890, 000 Caribbean 330, 000 Latin America 1. 4 million Source: UNAIDS/WHO 1998. Western Europe 500, 000 North Africa & Middle East 210, 000 Sub-Saharan Africa 22. 5 million Eastern Europe & Central Asia 270, 000 East Asia & Pacific 560, 000 South and South East Asia 6. 7 million Australia and New Zealand 12, 000 4
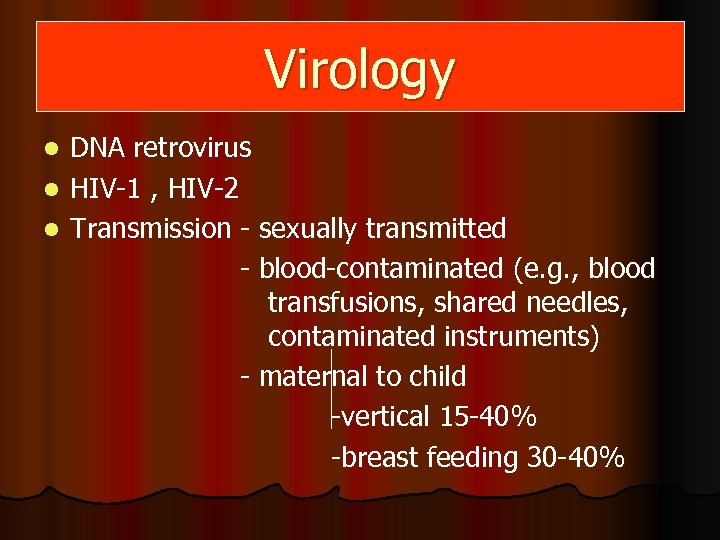
Virology DNA retrovirus l HIV-1 , HIV-2 l Transmission - sexually transmitted - blood-contaminated (e. g. , blood transfusions, shared needles, contaminated instruments) - maternal to child -vertical 15 -40% -breast feeding 30 -40% l
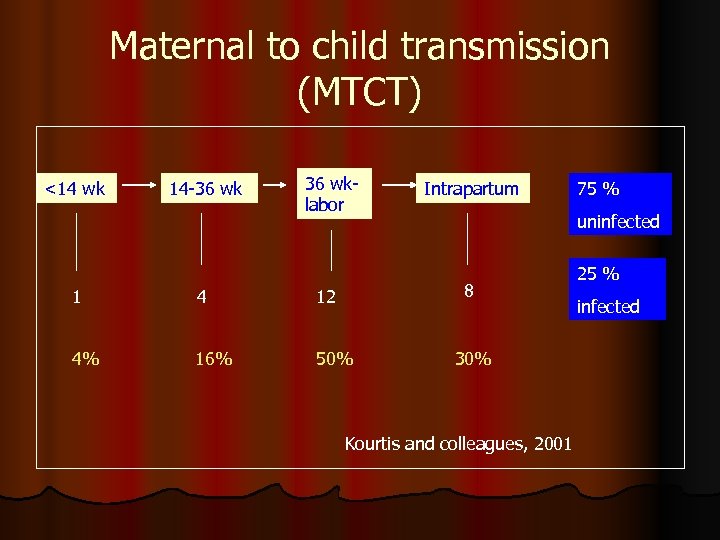
Maternal to child transmission (MTCT) <14 wk 14 -36 wklabor Intrapartum uninfected 8 1 4 12 4% 16% 50% 75 % 30% Kourtis and colleagues, 2001 25 % infected

Risk factors for vertical transmission 1. Preterm birth (3. 7 relative risk for intrapartum transmission ; Kuhn and assoc 1999) 2. Prolonged membrane rupture (increase rate from 15 to 25% in ROM > 4 hr ; Landesman and co-workers 1996) 3. Placental inflammation, chorioamnionitis, concurrent syphylis (Mwanyumba 2002)
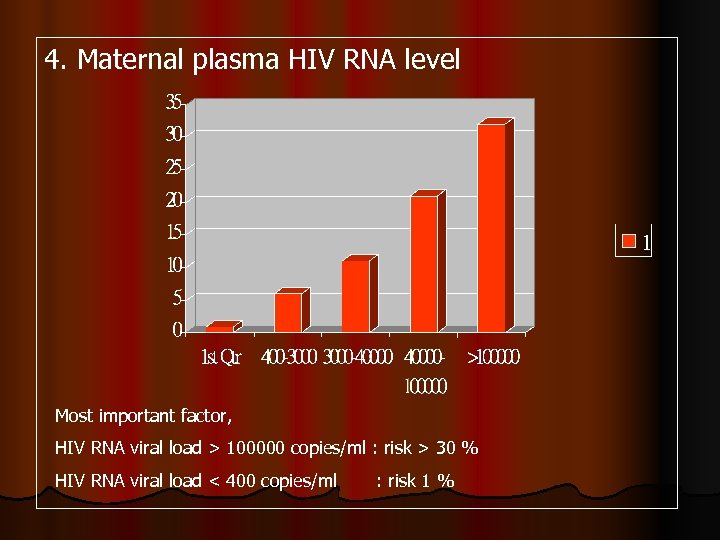
4. Maternal plasma HIV RNA level Most important factor, HIV RNA viral load > 100000 copies/ml : risk > 30 % HIV RNA viral load < 400 copies/ml : risk 1 %
5. Stage of disease 6. CD 4+ T-cell count 7. Mode of delivery cesarean section vs vaginal delivery 8. Breast feeding (risk 30 -40%)

Pregnancy on HIV infection Pregnancy : slightly immunosuppressive : minimal effect on CD 4 count : minimal effect on HIV RNA level : does not have significant effect on the clinical or immunological course of HIV infection (Minkoff 2003) l Maternal morbidity and mortality : not increased l HIV infection on pregnancy • Slightly increase rate of preterm birth • Slightly increase rate of IUGR • Slightly increase rate of PROM • Fetal and neonatal infection varies from 25 -40 percent

Adverse Pregnancy Outcomes and Relationship to HIV Infection Pregnancy Outcome Relationship to HIV Infection Spontaneous abortion Limited data, but evidence of possible increased risk Stillbirth No association noted in developed countries; evidence of increased risk in developing countries Perinatal mortality No association noted in developed countries, but data limited; evidence of increased risk in developing countries Newborn mortality Limited data in developed countries; evidence of increased risk in developing countries Intra-uterine growth Evidence of possible increased risk retardation Anderson 2001.

Adverse Pregnancy Outcomes and Relationship to HIV Infection (continued) Pregnancy Outcome Relationship to HIV Infection Low birth weight Evidence of possible increased risk Preterm delivery Evidence of possible increased risk, especially w/ more advanced disease Pre-eclampsia No data Gestational diabetes No data Amnionitis Limited data; more recent studies do not suggest an increased risk; some earlier studies found increased histologic placental inflammation, particularly in those with preterm deliveries Oligohydramnios Minimal data Fetal malformation No evidence of increased risk Anderson 2001.

Management during pregnancy l l Therapeutic goals ; maximal suppression of viral load and restoration of immunological function ; prevention of maternal to child transmission ARV therapy should be offered to all HIV infected pregnant women regardless of CD 4 cell count or HIV RNA level To treat the mother as well as to reduce the risk of perinatal transmission Holistic care : antepartum / intrapartum / postpartum : mother / fetus-baby : psycho / bio / social

Antepartum care 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. Posttest counseling / psychological support History taking Physical examination Per vaginal examination Oral health examination Ophthalmic examination Lab tests Tuberculin test Chest X-ray Prenatal care in high risk clinic Nutrition support / vitamin supplementation Ultrasound Prevention of opportunistic infection Immunization Anteretroviral administration

Intrapartum care 1. 2. 3. 4. 5. ARV during labor period ; minimum viral load Mode of delivery Labor augmentation is used when needed to shorten the interval to delivery / but avoid ARM Minimize operative obstetrics : scalp electrode, fetal scalp blood sampling, forceps extraction, vacuum extraction Universal precaution ; percutaneous exposure of needle=0. 3%, mucous membrane exposure=0. 09%, atraumatic needle, absorbable suture, non-touch technique, 0. 5% sodium hypochloride, room for isolation

l Cesarean section ; decrease vertical transmission one-half compared with vaginal delivery (metaanalysis of 15 prospective cohort studies by the international perinatal HIV group 1999) l Combined cesarean section with ARV reduced the risk 87 % l ACOG 2000 ; recommended C/S when HIV RNA viral loads > 1000 copies/ml l Scheduled C/S is recommended at 38 wk l If viral load < 1000 copies/ml ; data insufficient to estimate benefit of C/S (ACOG 2000)

Postpartum care 1. ARV Mother: l AIDS, HIV infection with CD 4<200 ; continue ARV treatment l CD 4 200 -350 ; controversial for ARV l CD 4 > 350 ; stop ARV , monitoring CD 4 Baby: ARV 1 / 6 weeks If delivery occurs before treatment is given, the newborn can receive prophylaxis for 6 weeks with zidovudine, or in some cases combination antiretroviral treatment 2. Contraceptives ; condom + OCP l points of interest ; TR, injectable, norplant, IUD

3. Breast feeding l Not recommended l Africa ; breast feeding with continuation of ARV prophylaxis 4. Postpartum clinic and pap smear ; 6 mo / 1 year

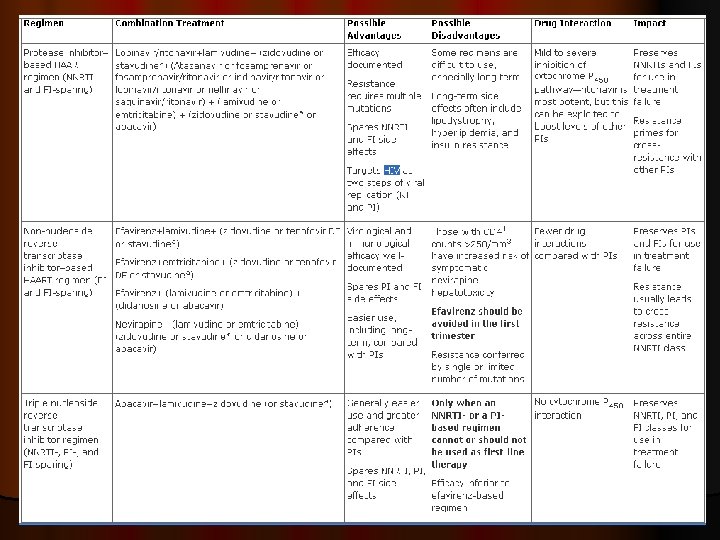
Guidelines for ARV in pregnancy 1. Classes of ARV drugs By FDA pregnancy category classification e-text Mc. Graw. Hill Cunningham FG, Leveno KJ, Bloom SL, Hauth JC, Gilstrap III L, Wenstrom KD. Williams Obstetrics. 22 nd ed. New York: Mc. GRAW-HILL; 2005.
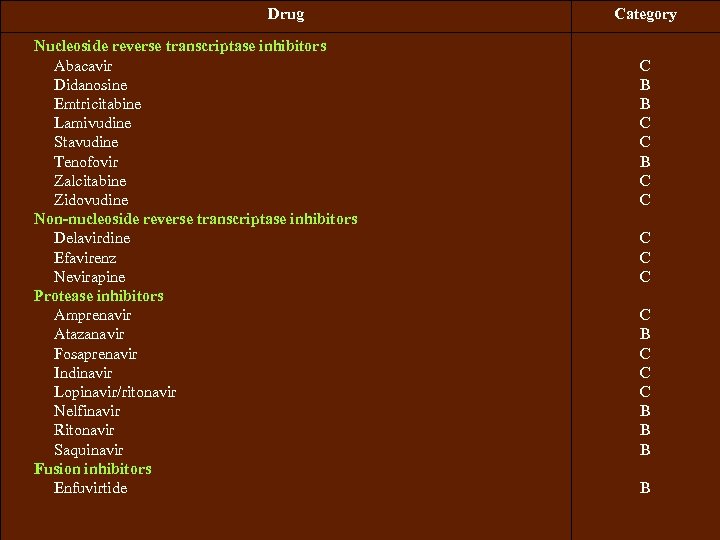
Drug Nucleoside reverse transcriptase inhibitors Abacavir Didanosine Emtricitabine Lamivudine Stavudine Tenofovir Zalcitabine Zidovudine Non-nucleoside reverse transcriptase inhibitors Delavirdine Efavirenz Nevirapine Protease inhibitors Amprenavir Atazanavir Fosaprenavir Indinavir Lopinavir/ritonavir Nelfinavir Ritonavir Saquinavir Fusion inhibitors Enfuvirtide Category C B B C C C C B B
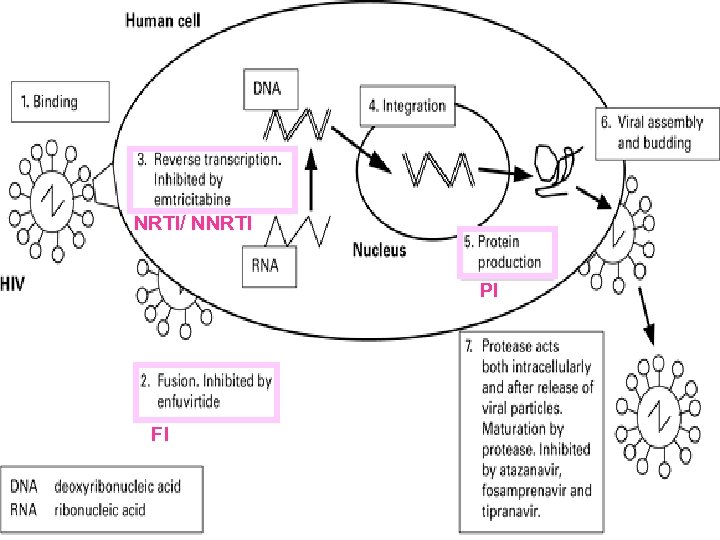
NRTI/ NNRTI PI FI

2. Regimens l NNRTI-based l PI-based l Triple NRTI-based l FI-based l http: //AIDSinfo. nih. gov

3. Monitoring l CD 4 count at initiation then CD 4 count every 3 months l HIV RNA levels at 4 weeks after initiation of treatment then HIV RNA levels monthly until undetectable, then every 3 months l HIV RNA level at GA 36 weeks
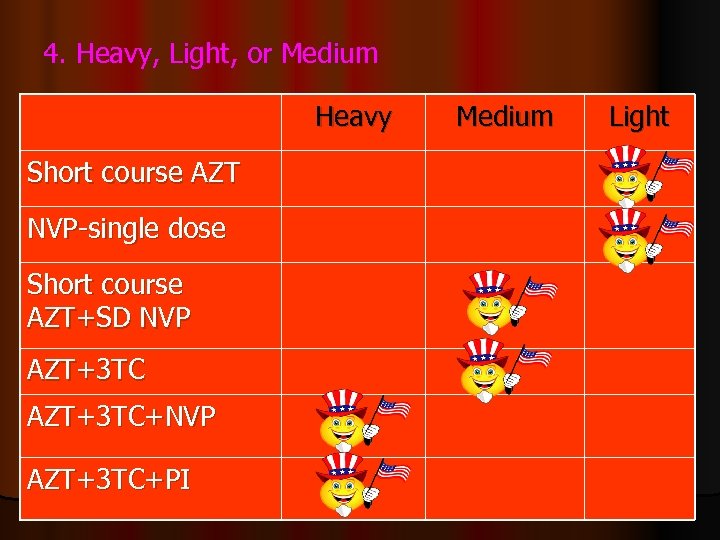
4. Heavy, Light, or Medium Heavy Short course AZT NVP-single dose Short course AZT+SD NVP AZT+3 TC+NVP AZT+3 TC+PI Medium Light
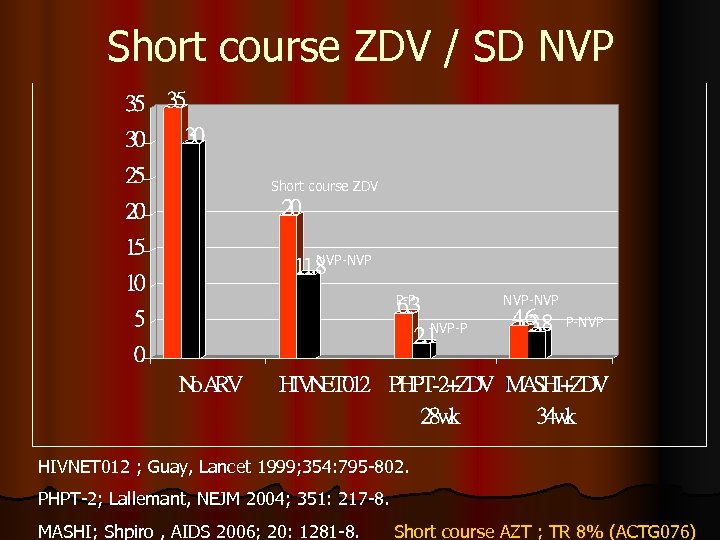
Short course ZDV / SD NVP Short course ZDV NVP-NVP P-P NVP-NVP NVP-P P-NVP HIVNET 012 ; Guay, Lancet 1999; 354: 795 -802. PHPT-2; Lallemant, NEJM 2004; 351: 217 -8. MASHI; Shpiro , AIDS 2006; 20: 1281 -8. Short course AZT ; TR 8% (ACTG 076)


NVP concentration after SD-NVP • Median T 1/2 = 61. 3 hours • Drug can be detected up to 19 days • Lower limit assay quantification 50 ng/ml ; 3 -4 weeks postpartum Cressey TR. JAIDS 2005; 38: 283 -8. • SD NVP covering the tail ; ZDV/3 TC 7 days : reduce resistance from 60 % to 10 -12 % TOPH Trial, SA

Two drugs regimen l Short course AZT (28 week)+SD NVP : TR 6. 3% (PHPT-2) l AZT+dd. I (36 wk to 1 wk PP) : TR 6. 9% (SIMBA trial) l AZT+3 TC 32 wk add SD NVP : TR 4. 7% (Ditrame Plus) l AZT 36 wk + SD NVP : TR 6. 5%

HAART l l Depend on immune status of mother -low CD 4 <200 ; start for maternal health -high CD 4 ; consider -pro ; low TR (PACTG 316, TR 1. 5%) -con ; high risk of NVP toxicity, increase risk of GDM with PI, risk of preterm delivery (controversial) Which HAART? -NNRTI based HAART -PI based HAART

Toxicities concerned NVP -rash ; women>men (3. 7 x) -more common with high CD 4 > 250 (10 X increase in women) l Hepatotoxicity -symptomatic hepatotoxicity ; CD 4 <250 : 0. 51. 7%, CD 4 > 250: 10. 0 -11. 3% -fetal hepatic events ; CD 4 250 -400 : 0. 42%, CD 4 >400 : 1. 1%) -TRC cohort ; low : high CD 4 2. 9% versus 7. 7% l
5. Factors for selection Mother Child Medical services

Action reports from Thammasat Hospital

ทมงาน อายรแพทย (อ. อนชา ( n สตแพทย (อ. อตวทธ ( n กมารแพทย (อ. อจฉรา ( n ทมพยาบาลหนวยเอดสธรรมศาสต ร (คณกรองทพย ( n ทมอาสาสมครผตดเชอ (คณวนใหม ( n เภสชกร n นกสงคมสงเคราะห n อน ๆ n

n < 2540 ; no ARV important role of a termination of pregnancy n 2541 -2546 ; AZT + SD NVP n > 2547 ; HAART

ขอมลจาก หนวยเอดสธรรมศาสตร โรงพยาบาลธรรมศาสตรเฉลมพระเกยรต

AZT regimen Prevalence of HIV infection pregnancy in TUH = 1 -2 percent n AZT alone = infection rate 3. 9 percent n AZT regimen from other studies ; infection rate 5 -8 percent, ACTG 076 protocol = 8% n Cesarean section = beneficial n MPH ; still using AZT regimen n

Regimens ; Pediatrics AIDS clinical trials group, USA n Antepartum: 100 mg 5 times/day, initiating at 14 -34 wk, continue throughout pregnancy (or 200 mg 3 times/day, 300 mg twice a day) n Intrapartum: IV Zidovudine in a 1 -hr initial dose of 2 mg/kg, followed by a continuous infusion of 1 mg/kg/hr until delivery n Neonates: begin at 8 -12 hr after birth, and give syrup at 2 mg/kg every 6 hr for 6 weeks
Regimen MPH n Antepartum; 300 mg twice a day, initiating at 14 -34 (28) wk, continue throughout pregnancy (regardless of CD 4 count) n Intrapartum; AZT 300 mg every 3 hr and single dose NVP 200 mg orally n Postpartum; AZT 300 mg+3 TC 150 mg twice a day for 2 weeks n Neonates; NVP 2 mg/kg single dose and AZT 2 mg/kg every 6 hr for 6 weeks n Disadvantages (compare to HARRT) : n n Higher transmission rate High incidence of NVP resistance
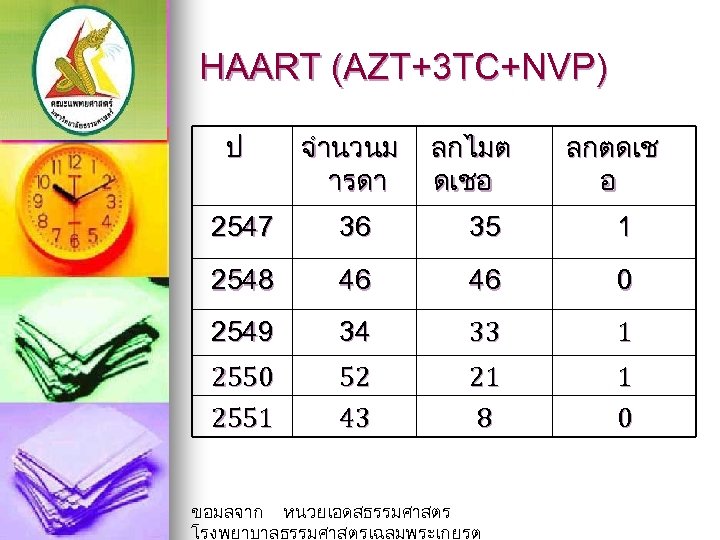
HAART (AZT+3 TC+NVP) ป จำนวนม ารดา ลกไมต ดเชอ 2547 36 35 1 2548 46 46 0 2549 34 33 1 2550 2551 52 43 21 8 1 0 ขอมลจาก หนวยเอดสธรรมศาสตร โรงพยาบาลธรรมศาสตรเฉลมพระเกยรต ลกตดเช อ

Triple agents (HARRT) Thammasat Hospital ; transmission rate 1. 2 % n Other studies ; transmission rate 1 -1. 5% n

Regimen : TUH 1. CD 4 ≤ 200 / GA 14 weeks n Antepartum; AZT(300)/3 TC(150) q 12 hr + NVP(200) OD for 2 wk then AZT(300)/3 TC(150) +NVP(200) q 12 hr n Intrapartum; AZT 300 mg q 3 hr and AZT(300)/3 TC(150) +NVP(200) q 12 hr n Postpartum; AZT(300)/3 TC(150) +NVP(200) q 12 hr n Neonates; AZT 2 mg/kg q 6 hrx 6 wk

2. CD 4 > 200 / GA 28 weeks Antepartum; AZT(300)/3 TC(150) q 12 hr + NVP(200) OD for 2 wk then AZT(300)/3 TC(150) +NVP(200) q 12 hr Intrapartum; AZT 300 mg q 3 hr and AZT(300)/3 TC(150) +NVP(200) q 12 hr Postpartum; AZT(300)/3 TC(150) q 12 hr x 14 days, stop NVP Neonates; AZT 2 mg/kg q 6 hrx 6 wk

Alternative regimens; n AZT/3 TC/Nelfinavir(NLF) (250 mg 5 tabs q 12 hr, no need for test dose, no covering tail) n AZT/3 TC/Efavirenz (GA>24 wk) n GPOvir(3 TC/d 4 T/NVP)(follow the protocol AZT/3 TC/NVP and test doses NVP for 2 wk) In case C/S n Start AZT with 30 cc of water since NPO then NPO except medicine with water until delivery and postop care period 12 -24 hr For no ANC patients n Intrapartum; Anti. HIV stat, NVP 200 mg single dose (immediately) and AZT 300 mg q 3 hr regardless of CD 4 count n Postpartum; AZT 300/3 TC 150 q 12 hrx 14 wk n Neonates; NVP 2 mg/kg single dose + AZT 2 mg/kg q 6 hr x 6 wk (start immediately)

thank you for your attention until the end of the session
f141cc31100c27aafef8f7d56fc1ba7b.ppt