16d6858d2e46ccc304d58032f4c2da72.ppt
- Количество слайдов: 90
 HIV In Mothers and Children
HIV In Mothers and Children
 What Is HIV/AIDS? • Acquired immunodeficiency syndrome (AIDS) is caused by the human immunodeficiency virus (HIV). • HIV attacks and destroys white blood cells, causing a defect in the body’s immune system. 2
What Is HIV/AIDS? • Acquired immunodeficiency syndrome (AIDS) is caused by the human immunodeficiency virus (HIV). • HIV attacks and destroys white blood cells, causing a defect in the body’s immune system. 2
 What Is HIV/AIDS? • The immune system of an HIV-infected person becomes so weakened that it cannot protect itself from serious infections. When this happens, the person clinically has AIDS. • AIDS may manifest as early as 2 years or as late as 10 years after infection with HIV. 3
What Is HIV/AIDS? • The immune system of an HIV-infected person becomes so weakened that it cannot protect itself from serious infections. When this happens, the person clinically has AIDS. • AIDS may manifest as early as 2 years or as late as 10 years after infection with HIV. 3
 Number of People with HIV/AIDS by Region North America 890, 000 Caribbean 330, 000 Latin America 1. 4 million Source: UNAIDS/WHO 1998. Western Europe 500, 000 North Africa & Middle East 210, 000 Sub-Saharan Africa 22. 5 million Eastern Europe & Central Asia 270, 000 East Asia & Pacific 560, 000 South and South East Asia 6. 7 million Australia and New Zealand 12, 000 4
Number of People with HIV/AIDS by Region North America 890, 000 Caribbean 330, 000 Latin America 1. 4 million Source: UNAIDS/WHO 1998. Western Europe 500, 000 North Africa & Middle East 210, 000 Sub-Saharan Africa 22. 5 million Eastern Europe & Central Asia 270, 000 East Asia & Pacific 560, 000 South and South East Asia 6. 7 million Australia and New Zealand 12, 000 4
 HIV Transmission Through Sexual Contact • Of every 100 HIV infected adults, 75 -85 have been infected through unprotected intercourse – 70% of these infections are from heterosexual intercourse • STDs, especially ulcerative lesions in STDs genitalia, increase risk of transmission Source: UNAIDS/WHO 1996. 5
HIV Transmission Through Sexual Contact • Of every 100 HIV infected adults, 75 -85 have been infected through unprotected intercourse – 70% of these infections are from heterosexual intercourse • STDs, especially ulcerative lesions in STDs genitalia, increase risk of transmission Source: UNAIDS/WHO 1996. 5
 Modes of HIV Transmission • Sexual intercourse • Accidental exposure to blood/blood products (e. g. , blood transfusions, shared needles, contaminated instruments) • Mother to child during: – pregnancy – birth – breastfeeding 6
Modes of HIV Transmission • Sexual intercourse • Accidental exposure to blood/blood products (e. g. , blood transfusions, shared needles, contaminated instruments) • Mother to child during: – pregnancy – birth – breastfeeding 6
 Women and HIV Social Risk Factors – Illiteracy – Lack of awareness of preventive measures Biological risk factors – Twice as easy for women to contract HIV from men – Physiology of women (e. g. , menstruation, intercourse) – Pregnancy-associated conditions (e. g. , anemia, menorrhagia and hemorrhage) increase the need 7 for blood transfusion
Women and HIV Social Risk Factors – Illiteracy – Lack of awareness of preventive measures Biological risk factors – Twice as easy for women to contract HIV from men – Physiology of women (e. g. , menstruation, intercourse) – Pregnancy-associated conditions (e. g. , anemia, menorrhagia and hemorrhage) increase the need 7 for blood transfusion
 HIV and Contraception • Contraception with protection – Male condom (latex and vinyl) – Female condom – Nonoxynol-9 (antiviral spermicidal cream)1 – Diaphragm 1 • Methods appropriate for use by women with HIV. They should use a condom for their partner’s protection. – Hormonals (COCs, Implants, PICs) –Partial protection if used without condom Voluntary sterilization 1 8
HIV and Contraception • Contraception with protection – Male condom (latex and vinyl) – Female condom – Nonoxynol-9 (antiviral spermicidal cream)1 – Diaphragm 1 • Methods appropriate for use by women with HIV. They should use a condom for their partner’s protection. – Hormonals (COCs, Implants, PICs) –Partial protection if used without condom Voluntary sterilization 1 8
 Effect of AIDS on Pregnancy • • Infertility Repeated abortions Prematurity Intrauterine growth retardation Stillbirths Congenital abnormalities Embryopathies 9
Effect of AIDS on Pregnancy • • Infertility Repeated abortions Prematurity Intrauterine growth retardation Stillbirths Congenital abnormalities Embryopathies 9
 HIV Transmission from Mother to Infant • Antenatal – In utero by transplacental passage • Intranatal – Exposure to maternal blood and vaginal secretions during labor and delivery • Postnatal – Postpartum through breastfeeding Source: UNAIDS/WHO 1996; UNAIDS/WHO 1998. 10
HIV Transmission from Mother to Infant • Antenatal – In utero by transplacental passage • Intranatal – Exposure to maternal blood and vaginal secretions during labor and delivery • Postnatal – Postpartum through breastfeeding Source: UNAIDS/WHO 1996; UNAIDS/WHO 1998. 10
 HIV Transmission from Mother to Infant • 25 -35% of all infants born to HIV-infected women in developing countries become infected • 90% of HIV-infected infants and children were infected by mother Source: UNAIDS/WHO 1996; UNAIDS/WHO 1998. 11
HIV Transmission from Mother to Infant • 25 -35% of all infants born to HIV-infected women in developing countries become infected • 90% of HIV-infected infants and children were infected by mother Source: UNAIDS/WHO 1996; UNAIDS/WHO 1998. 11
 • approximately 600, 000 HIV-infected infants are born every year–at least 1, 600 every day– in resource-constrained countries. • Transmission occurs during pregnancy, labor and delivery, and breastfeeding. • The rate of mother to child transmission has been reduced to less than 5 percent among the limited number of HIV-infected women in developed countries.
• approximately 600, 000 HIV-infected infants are born every year–at least 1, 600 every day– in resource-constrained countries. • Transmission occurs during pregnancy, labor and delivery, and breastfeeding. • The rate of mother to child transmission has been reduced to less than 5 percent among the limited number of HIV-infected women in developed countries.
 • high rates are largely due to the lack of access to: – HIV voluntary counseling and testing – replacement feeding – selective caesarean section – antiretroviral drug therapy
• high rates are largely due to the lack of access to: – HIV voluntary counseling and testing – replacement feeding – selective caesarean section – antiretroviral drug therapy
 HIV Transmission HIV cannot be transmitted by: – Casual person to person contact at home or work or in social or public places – Food, air, water – Insect/mosquito bites – Coughing, sneezing, spitting – Shaking hands, touching, dry kissing or hugging – Swimming pools, toilets, etc. 14
HIV Transmission HIV cannot be transmitted by: – Casual person to person contact at home or work or in social or public places – Food, air, water – Insect/mosquito bites – Coughing, sneezing, spitting – Shaking hands, touching, dry kissing or hugging – Swimming pools, toilets, etc. 14
 AIDS and Infants • Symptoms generally develop by 6 months of age – Diarrhea – Failure to thrive • Most of these children die before their second birthday • Children born to HIV-infected parents are likely to become orphans 15
AIDS and Infants • Symptoms generally develop by 6 months of age – Diarrhea – Failure to thrive • Most of these children die before their second birthday • Children born to HIV-infected parents are likely to become orphans 15
 Reducing pediatric HIV infection and disease involves three stages: • preventing HIV infection among women of childbearing age • preventing unwanted pregnancy among HIV-positive women • preventing mother to child transmission during pregnancy, labor and delivery, and breastfeeding
Reducing pediatric HIV infection and disease involves three stages: • preventing HIV infection among women of childbearing age • preventing unwanted pregnancy among HIV-positive women • preventing mother to child transmission during pregnancy, labor and delivery, and breastfeeding
 BENEFITS TO HIV TESTING • EARLY COUNSELING AND TREATMENT OF HIV INFECTION • ABILITY TO MAKE DECISIONS REGARDING PREGNANCY • IMPLEMENTATION OF STRATEGIES TO ATTEMPT TO PREVENT TRANSMISSION TO FETUS
BENEFITS TO HIV TESTING • EARLY COUNSELING AND TREATMENT OF HIV INFECTION • ABILITY TO MAKE DECISIONS REGARDING PREGNANCY • IMPLEMENTATION OF STRATEGIES TO ATTEMPT TO PREVENT TRANSMISSION TO FETUS
 WHO SHOULD WE SCREEN? • ALL PREGNANT WOMEN • TARGETED TESTING FAILS TO IDENTIFY A SUBSTANTIAL PROPORTION OF HIV POSITIVE WOMEN
WHO SHOULD WE SCREEN? • ALL PREGNANT WOMEN • TARGETED TESTING FAILS TO IDENTIFY A SUBSTANTIAL PROPORTION OF HIV POSITIVE WOMEN
 Anti-Retroviral Based Prevention Strategies • zidovudine (AZT) administered to the mother from 14 weeks of gestation and to the child during the first seven days after birth, reduced the risk of mother to child transmission among non-breastfeeding mothers by two-thirds • Two similar studies conducted in Côte d’Ivoire and Burkina Faso among breastfeeding mothers demonstrated a 37 percent reduction in mother to child transmission.
Anti-Retroviral Based Prevention Strategies • zidovudine (AZT) administered to the mother from 14 weeks of gestation and to the child during the first seven days after birth, reduced the risk of mother to child transmission among non-breastfeeding mothers by two-thirds • Two similar studies conducted in Côte d’Ivoire and Burkina Faso among breastfeeding mothers demonstrated a 37 percent reduction in mother to child transmission.
 Anti-Retroviral Based Prevention Strategies • A study in Uganda demonstrated a 47 percent reduction in mother to child transmission following the administration of a single dose of nevirapine to the mother at onset of labor and to the baby within 72 hours after birth. • The combination of AZT and lamivudine in a short-course regimen also has been shown to reduce mother to child transmission.
Anti-Retroviral Based Prevention Strategies • A study in Uganda demonstrated a 47 percent reduction in mother to child transmission following the administration of a single dose of nevirapine to the mother at onset of labor and to the baby within 72 hours after birth. • The combination of AZT and lamivudine in a short-course regimen also has been shown to reduce mother to child transmission.
 Protecting Health Care Workers During Labor and Delivery • Precautions during labor: – Protection from blood and amniotic fluids – Protection from sharp instruments • Resuscitation of baby: – No mouth to mouth suction – No mouth to mouth breathing • Precautions following labor: – Proper disinfection of instruments – Proper disposal of placenta and other items 21
Protecting Health Care Workers During Labor and Delivery • Precautions during labor: – Protection from blood and amniotic fluids – Protection from sharp instruments • Resuscitation of baby: – No mouth to mouth suction – No mouth to mouth breathing • Precautions following labor: – Proper disinfection of instruments – Proper disposal of placenta and other items 21
 PRETEST COUNSELING • TAKE RISK HISTORY AND COUNCIL REGARDING RISK REDUCTION • DISCUSS REASONS FOR TEST • PROVIDE INFORMATION TO WOMEN REGARDING TESTING & ILLNESS • RISKS & BENEFITS OF TESTING • CONFIDENTIALITY OF RESULTS • ASSESS WINDOW PERIOD • PERSON HAS RIGHT TO REFUSE TESTING
PRETEST COUNSELING • TAKE RISK HISTORY AND COUNCIL REGARDING RISK REDUCTION • DISCUSS REASONS FOR TEST • PROVIDE INFORMATION TO WOMEN REGARDING TESTING & ILLNESS • RISKS & BENEFITS OF TESTING • CONFIDENTIALITY OF RESULTS • ASSESS WINDOW PERIOD • PERSON HAS RIGHT TO REFUSE TESTING
 POST-TEST COUNSELING • HIV RESULTS SHOULD BE GIVEN IN PERSON • ASSESS PATIENT’S UNDERSTANDING • ENCOURAGE PATIENT TO EXPRESS FEELINGS AND ASK QUESTIONS • NEGATIVE AND INDETERMINATE RESULTS: DISCUSS NEED FOR REPEAT TESTING
POST-TEST COUNSELING • HIV RESULTS SHOULD BE GIVEN IN PERSON • ASSESS PATIENT’S UNDERSTANDING • ENCOURAGE PATIENT TO EXPRESS FEELINGS AND ASK QUESTIONS • NEGATIVE AND INDETERMINATE RESULTS: DISCUSS NEED FOR REPEAT TESTING
 POSITIVE RESULT • • IDENTIFY IMMEDIATE CONCERNS IDENTIFY SUPPORTS EFFECT OF HIV ON PREGNANCY RISK OF TRANSMISSION TO FETUS DURING PREGNANCY, L&D, BF • MEASURES TO DECREASE HIV TRANSMISSION
POSITIVE RESULT • • IDENTIFY IMMEDIATE CONCERNS IDENTIFY SUPPORTS EFFECT OF HIV ON PREGNANCY RISK OF TRANSMISSION TO FETUS DURING PREGNANCY, L&D, BF • MEASURES TO DECREASE HIV TRANSMISSION

 ANTENATAL CARE
ANTENATAL CARE
 INTRODUCTION • MULTIDISCIPLINARY TEAM APPROACH • MEDICAL NEEDS • SOCIAL AND PSYCHOLOGICAL NEEDS
INTRODUCTION • MULTIDISCIPLINARY TEAM APPROACH • MEDICAL NEEDS • SOCIAL AND PSYCHOLOGICAL NEEDS
 ANTENATAL CARE • SIMILAR TO CARE FOR HIV NEGATIVE WOMEN • PREGNANCY NOT HIGH RISK • SAME NUMBER OF ANTENATAL VISITS • AVOID INVASIVE ANTENATAL TESTS OR PROCEDURES
ANTENATAL CARE • SIMILAR TO CARE FOR HIV NEGATIVE WOMEN • PREGNANCY NOT HIGH RISK • SAME NUMBER OF ANTENATAL VISITS • AVOID INVASIVE ANTENATAL TESTS OR PROCEDURES
 FIRST VISIT • PATIENT HISTORY • • DATES OF 1 ST POSITIVE HIV TEST HIV RISK FACTORS HIV CARE AT TIME OF CONCEPTION SEROLOGIC STATUS OF PARTNER OTHER STD’S OPPORTUNISTIC INFECTIONS DRUG HISTORY
FIRST VISIT • PATIENT HISTORY • • DATES OF 1 ST POSITIVE HIV TEST HIV RISK FACTORS HIV CARE AT TIME OF CONCEPTION SEROLOGIC STATUS OF PARTNER OTHER STD’S OPPORTUNISTIC INFECTIONS DRUG HISTORY
 FIRST VISIT • INVESTIGATIONS • CBC & DIFFERENTIAL • LYTES, GLUCOSE, RFT’S, LIVER ENZYMES • CD 4+ COUNT, CD 8 COUNT, CD 4/CD 8 • VIRAL LOAD • SEROLOGY FOR HEP A, B, C, SYPHILIS, RUBELLA, TOXO, CMV • TB SKIN TEST
FIRST VISIT • INVESTIGATIONS • CBC & DIFFERENTIAL • LYTES, GLUCOSE, RFT’S, LIVER ENZYMES • CD 4+ COUNT, CD 8 COUNT, CD 4/CD 8 • VIRAL LOAD • SEROLOGY FOR HEP A, B, C, SYPHILIS, RUBELLA, TOXO, CMV • TB SKIN TEST
 FOLLOW UP VISITS • STANDARD OBSTETRICAL ROUTINE • INCREASE SURVEILLANCE ONLY IF WARRANTED • LABS EVERY 3 MONTHS • CD 4+ COUNT • VIRAL LOAD • SEROLOGY FOR TOXOPLASMOSIS AND SYPHILIS
FOLLOW UP VISITS • STANDARD OBSTETRICAL ROUTINE • INCREASE SURVEILLANCE ONLY IF WARRANTED • LABS EVERY 3 MONTHS • CD 4+ COUNT • VIRAL LOAD • SEROLOGY FOR TOXOPLASMOSIS AND SYPHILIS
 OPPORTUNISTIC INFECTIONS • PROPHYLAXIS SHOULD BE OFFERED IN PREGNANCY FOR THE FOLLOWING • • • PNEUMOCYSTIS CARINII PNEUMONIA TOXOPLASMOSIS TUBERCULOSIS MYCOBACTERIUM AVIUM COMPLEX VARICELLA ZOSTER HEPATITIS A, B
OPPORTUNISTIC INFECTIONS • PROPHYLAXIS SHOULD BE OFFERED IN PREGNANCY FOR THE FOLLOWING • • • PNEUMOCYSTIS CARINII PNEUMONIA TOXOPLASMOSIS TUBERCULOSIS MYCOBACTERIUM AVIUM COMPLEX VARICELLA ZOSTER HEPATITIS A, B
 CONCLUSION • HIV IN PREGNANCY SHOULD BE MANAGED BY MULTIDISCIPLINARY TEAM • ANTENATAL CARE IS SIMILAR TO THAT OF HIV POSITIVE WOMEN • PREGNANCY NOT CONSIDERED HIGH RISK SIMPLY BY VIRTUE OF HIV INFECTION
CONCLUSION • HIV IN PREGNANCY SHOULD BE MANAGED BY MULTIDISCIPLINARY TEAM • ANTENATAL CARE IS SIMILAR TO THAT OF HIV POSITIVE WOMEN • PREGNANCY NOT CONSIDERED HIGH RISK SIMPLY BY VIRTUE OF HIV INFECTION
 ANTIRETROVIRAL USE
ANTIRETROVIRAL USE
 ANTEPARTUM ANTIRETROVIRAL USE • GOALS: – CONTROL DISEASE IN MOTHER – REDUCE PERINATAL TRANSMISSION • VERY LITTLE DATA AVAILABLE ON EFFECTS IN PREGNANCY • MOST DATA ASSESSES ZIDOVUDINE • LITTLE DATA ON OTHER DRUGS
ANTEPARTUM ANTIRETROVIRAL USE • GOALS: – CONTROL DISEASE IN MOTHER – REDUCE PERINATAL TRANSMISSION • VERY LITTLE DATA AVAILABLE ON EFFECTS IN PREGNANCY • MOST DATA ASSESSES ZIDOVUDINE • LITTLE DATA ON OTHER DRUGS
 CONCLUSIONS • ZIDOVUDINE REDUCES PERINATAL TRANSMISSION IN WOMEN AT DIFFERENT STAGES OF DISEASE • LONG AS WELL AS SHORTER REGIMENS EFFECTIVE • STILL EFFECTIVE IN BREASTFEEDING POPULATIONS • USE OF OTHER ANTIRETROVIRALS IN COMBINATION WITH ZDV PROMISING, STILL INVESTIGATIONAL
CONCLUSIONS • ZIDOVUDINE REDUCES PERINATAL TRANSMISSION IN WOMEN AT DIFFERENT STAGES OF DISEASE • LONG AS WELL AS SHORTER REGIMENS EFFECTIVE • STILL EFFECTIVE IN BREASTFEEDING POPULATIONS • USE OF OTHER ANTIRETROVIRALS IN COMBINATION WITH ZDV PROMISING, STILL INVESTIGATIONAL
 IN UTERO EXPOSURE
IN UTERO EXPOSURE
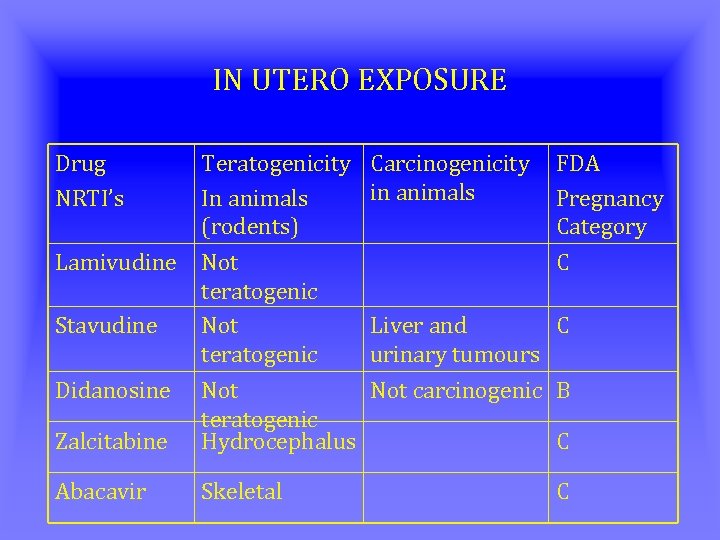 IN UTERO EXPOSURE Drug NRTI’s Zalcitabine Teratogenicity In animals (rodents) Not teratogenic Hydrocephalus Abacavir Skeletal Lamivudine Stavudine Didanosine Carcinogenicity in animals FDA Pregnancy Category C Liver and C urinary tumours Not carcinogenic B C C
IN UTERO EXPOSURE Drug NRTI’s Zalcitabine Teratogenicity In animals (rodents) Not teratogenic Hydrocephalus Abacavir Skeletal Lamivudine Stavudine Didanosine Carcinogenicity in animals FDA Pregnancy Category C Liver and C urinary tumours Not carcinogenic B C C
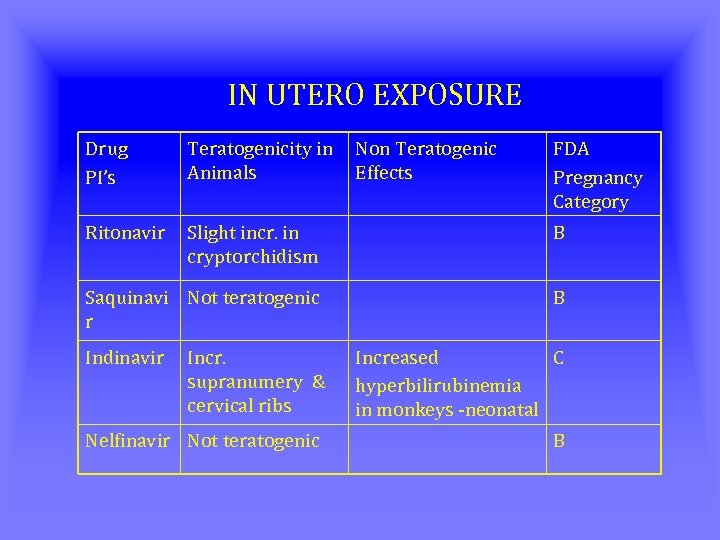 IN UTERO EXPOSURE Drug PI’s Teratogenicity in Animals Ritonavir Slight incr. in cryptorchidism B Saquinavi Not teratogenic r B Indinavir Non Teratogenic Effects FDA Pregnancy Category Incr. supranumery & cervical ribs Increased C hyperbilirubinemia in monkeys -neonatal Nelfinavir Not teratogenic B
IN UTERO EXPOSURE Drug PI’s Teratogenicity in Animals Ritonavir Slight incr. in cryptorchidism B Saquinavi Not teratogenic r B Indinavir Non Teratogenic Effects FDA Pregnancy Category Incr. supranumery & cervical ribs Increased C hyperbilirubinemia in monkeys -neonatal Nelfinavir Not teratogenic B
 ANTIRETROVIRAL THERAPY DURING LABOR & DELIVERY
ANTIRETROVIRAL THERAPY DURING LABOR & DELIVERY
 IV ZIDOVUDINE • ZDV LOADING DOSE AT ONSET OF LABOR 2 MG/KG OVER 1 HR • CONTINUOUS INFUSION WHILE IN LABOR 1 MG/KG/HR
IV ZIDOVUDINE • ZDV LOADING DOSE AT ONSET OF LABOR 2 MG/KG OVER 1 HR • CONTINUOUS INFUSION WHILE IN LABOR 1 MG/KG/HR
 • INCREASING EVIDENCE THAT MOST PERINATAL TRANSMISSION OCCURS NEAR TIME OF OR DURING DELIVERY • REDUCTION OF PERINATAL TRANSMISSION DUE TO SYSTEMIC ANTIRETROVIRAL DRUG LEVELS IN NEONATE AT TIME OF DELIVERY
• INCREASING EVIDENCE THAT MOST PERINATAL TRANSMISSION OCCURS NEAR TIME OF OR DURING DELIVERY • REDUCTION OF PERINATAL TRANSMISSION DUE TO SYSTEMIC ANTIRETROVIRAL DRUG LEVELS IN NEONATE AT TIME OF DELIVERY
 IV ZIDOVUDINE • ZDV READILY CROSSES PLACENTA • INITIAL IV DOSE RESULTS IN VIRUCIDAL LEVELS IN MOM & INFANT • CONTINUOUS INFUSION ENSURES STABLE DRUG LEVELS IN INFANT DURING BIRTH
IV ZIDOVUDINE • ZDV READILY CROSSES PLACENTA • INITIAL IV DOSE RESULTS IN VIRUCIDAL LEVELS IN MOM & INFANT • CONTINUOUS INFUSION ENSURES STABLE DRUG LEVELS IN INFANT DURING BIRTH
 ORAL ZIDOVUDINE • IF IV ZDV NOT AVAILABLE, ORAL ZDV MAY BE USED INTRAPARTUM • ZDV 600 MG PO @ ONSET OF LABOR • 300 MG PO Q 3 H IN LABOR
ORAL ZIDOVUDINE • IF IV ZDV NOT AVAILABLE, ORAL ZDV MAY BE USED INTRAPARTUM • ZDV 600 MG PO @ ONSET OF LABOR • 300 MG PO Q 3 H IN LABOR
 BANGKOK, LANCET 1999 • RANDOMIZED PLACEBO CONTROLLED • ZDV 300 MG PO BID FROM 36 WKS GA UNTIL ONSET OF LABOR • 300 MG PO Q 3 H WHILE IN LABOR • ALL WOMEN ADVISED NOT TO BREASTFEED • TRANSMISSION RATES: 9. 4% IN RX GROUP; 18. 9% IN CONTROL GROUP
BANGKOK, LANCET 1999 • RANDOMIZED PLACEBO CONTROLLED • ZDV 300 MG PO BID FROM 36 WKS GA UNTIL ONSET OF LABOR • 300 MG PO Q 3 H WHILE IN LABOR • ALL WOMEN ADVISED NOT TO BREASTFEED • TRANSMISSION RATES: 9. 4% IN RX GROUP; 18. 9% IN CONTROL GROUP
 ABIDJAN, LANCET 1999 • SIMILAR TRIAL TO BANGKOK, BUT IN BREASTFEEDING WOMEN
ABIDJAN, LANCET 1999 • SIMILAR TRIAL TO BANGKOK, BUT IN BREASTFEEDING WOMEN
 COTE D’IVOIRE & BURKINA FASO, LANCET 1999 • PLACEBO VS ZDV STARTED @ 36 -38 WKS GA • 300 MG PO DAILY • 600 MG PO AT ONSET OF LABOR • 300 MG PO BID UNTIL 7 DAYS PP • >85% OF INFANTS BREASTFED >3 MOS • 18% VS 27. 5 % TRANSMISSION @ 6 MOS (38% EFFICACY)
COTE D’IVOIRE & BURKINA FASO, LANCET 1999 • PLACEBO VS ZDV STARTED @ 36 -38 WKS GA • 300 MG PO DAILY • 600 MG PO AT ONSET OF LABOR • 300 MG PO BID UNTIL 7 DAYS PP • >85% OF INFANTS BREASTFED >3 MOS • 18% VS 27. 5 % TRANSMISSION @ 6 MOS (38% EFFICACY)
 • RESULTS SHOW SHORT-COURSE PO ZDV SAFE & EFFECTIVE IN ING RISK OF MOTHER-TO-CHILD TRANSMISSION • PREVENTION RATES NOT AS HIGH AS WITH IV ZDV
• RESULTS SHOW SHORT-COURSE PO ZDV SAFE & EFFECTIVE IN ING RISK OF MOTHER-TO-CHILD TRANSMISSION • PREVENTION RATES NOT AS HIGH AS WITH IV ZDV
 ORAL NEVIRAPINE • NON-NUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITOR • VERY LONG HALF-LIFE • RAPID DEV’T OF DRUG RESISTANCE
ORAL NEVIRAPINE • NON-NUCLEOSIDE REVERSE TRANSCRIPTASE INHIBITOR • VERY LONG HALF-LIFE • RAPID DEV’T OF DRUG RESISTANCE
 HIVNET 012 STUDY GUAY ET AL - 1999 • 13626 RANDOMIZED - NVP VS ZDV • NVP REGIMEN • 200 MG PO AT ONSET OF LABOR • 2 MG/KG PO DOSE TO BABY 72 HR DEL’Y • ZDV REGIMEN • 600 MG PO AT ONSET OF LABOR • 300 MG PO Q 3 H DURING LABOR • 4 MG/KG BID x 7 DAYS TO INFANTS
HIVNET 012 STUDY GUAY ET AL - 1999 • 13626 RANDOMIZED - NVP VS ZDV • NVP REGIMEN • 200 MG PO AT ONSET OF LABOR • 2 MG/KG PO DOSE TO BABY 72 HR DEL’Y • ZDV REGIMEN • 600 MG PO AT ONSET OF LABOR • 300 MG PO Q 3 H DURING LABOR • 4 MG/KG BID x 7 DAYS TO INFANTS
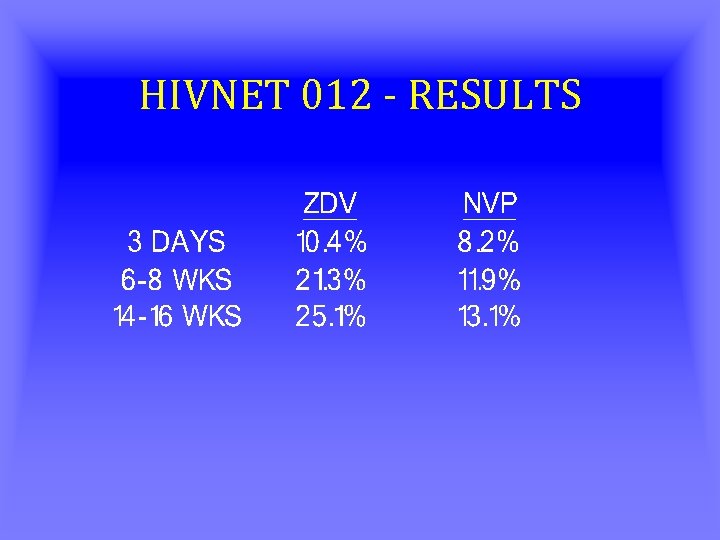 HIVNET 012 - RESULTS
HIVNET 012 - RESULTS
 SO WHAT? • EFFICACY OF SHORT-COURSE NVP 47% GREATER THAN SHORT COURSE ZDV • CURRENTLY SHORT-COURSE PO NVP NOT COMPARED TO IV ZDV FOR TRANSMISSION PREVENTION
SO WHAT? • EFFICACY OF SHORT-COURSE NVP 47% GREATER THAN SHORT COURSE ZDV • CURRENTLY SHORT-COURSE PO NVP NOT COMPARED TO IV ZDV FOR TRANSMISSION PREVENTION
 CONCLUSIONS • DURING LABOR - ZDV 2 MG/KG IV LOADING DOSE, THEN 1 MG/KG/HR • IF IV ZDV NOT AVAILABLE CONSIDER PO REGIMEN • MAY CONSIDER ADDITION OF NVP 200 MG PO TO IV ZDV @ ONSET OF LABOR
CONCLUSIONS • DURING LABOR - ZDV 2 MG/KG IV LOADING DOSE, THEN 1 MG/KG/HR • IF IV ZDV NOT AVAILABLE CONSIDER PO REGIMEN • MAY CONSIDER ADDITION OF NVP 200 MG PO TO IV ZDV @ ONSET OF LABOR
 OBSTETRICAL PRACTICE
OBSTETRICAL PRACTICE
 OBSTETRICAL PRACTICE • 70 % OF HIV TRANSMISSION OCCURS INTRAPARTUM. • THE GOAL OF OBSTETRICAL MANAGEMENT OF THE HIV PATIENT IS TO AVOID THOSE PRACTICES THAT INCREASE RISK OF TRANSMISSION.
OBSTETRICAL PRACTICE • 70 % OF HIV TRANSMISSION OCCURS INTRAPARTUM. • THE GOAL OF OBSTETRICAL MANAGEMENT OF THE HIV PATIENT IS TO AVOID THOSE PRACTICES THAT INCREASE RISK OF TRANSMISSION.
 OBSTETRICAL PRACTICE RUPTURE OF MEMBRANES LANDESMAN ET AL. , 1996 • RUPTURED MEMBRANES ONE OF MANY VARIABLES EXAMINED • 281 MOTHER-CHILD PAIRS WITH MEMBRANES RUPTURED LESS THAN 4 HOURS • 206 MOTHER-CHILD PAIRS WITH MEMBRANES RUPTURED MORE THAN 4 HOURS
OBSTETRICAL PRACTICE RUPTURE OF MEMBRANES LANDESMAN ET AL. , 1996 • RUPTURED MEMBRANES ONE OF MANY VARIABLES EXAMINED • 281 MOTHER-CHILD PAIRS WITH MEMBRANES RUPTURED LESS THAN 4 HOURS • 206 MOTHER-CHILD PAIRS WITH MEMBRANES RUPTURED MORE THAN 4 HOURS
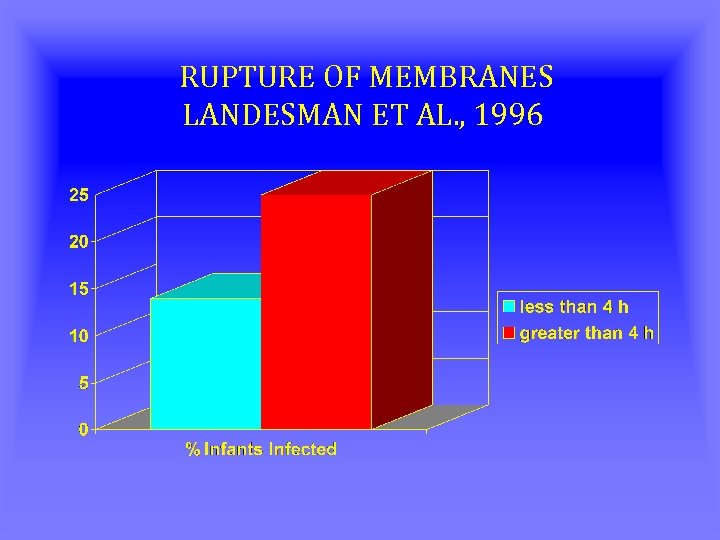 RUPTURE OF MEMBRANES LANDESMAN ET AL. , 1996
RUPTURE OF MEMBRANES LANDESMAN ET AL. , 1996
 OBSTETRICAL PRACTICE MODE OF DELIVERY - VAGINAL • ARTIFICIAL RUPTURE OF MEMBRANES SHOULD BE AVOIDED • RUPTURE OF MEMBRANES PAST 4 HOURS SHOULD BE AVOIDED • FETAL SCALP SAMPLING AND THE USE OF SCALP ELECTRODES SHOULD BE AVOIDED
OBSTETRICAL PRACTICE MODE OF DELIVERY - VAGINAL • ARTIFICIAL RUPTURE OF MEMBRANES SHOULD BE AVOIDED • RUPTURE OF MEMBRANES PAST 4 HOURS SHOULD BE AVOIDED • FETAL SCALP SAMPLING AND THE USE OF SCALP ELECTRODES SHOULD BE AVOIDED
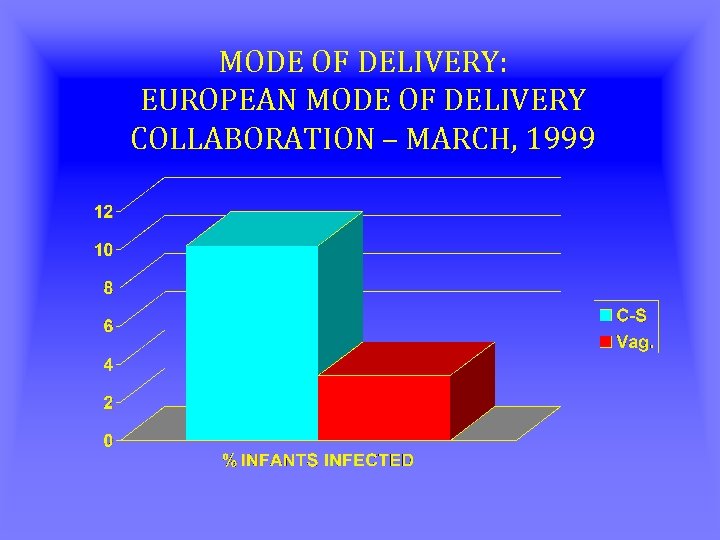
 MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999 • • RANDOMIZED CLINICAL TRIAL 370 MOTHER-CHILD PAIRS ANALYZED 203 DELIVERED BY C-S 167 DELIVERED VAGINALLY
MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999 • • RANDOMIZED CLINICAL TRIAL 370 MOTHER-CHILD PAIRS ANALYZED 203 DELIVERED BY C-S 167 DELIVERED VAGINALLY
 MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999
MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999
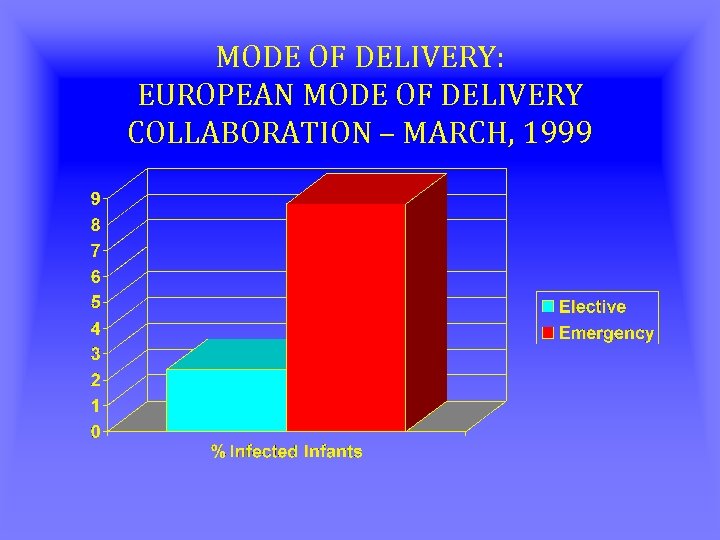
 MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999 • 203 C-S PERFORMED • 165 WERE PERFORMED ELECTIVELY • 31 WERE PERFORMED EMERGENTLY
MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999 • 203 C-S PERFORMED • 165 WERE PERFORMED ELECTIVELY • 31 WERE PERFORMED EMERGENTLY
 MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999
MODE OF DELIVERY: EUROPEAN MODE OF DELIVERY COLLABORATION – MARCH, 1999
 MODE OF DELIVERY: META-ANALYSIS THE INTERNATIONAL PERINATAL HIV GROUP, APRIL 1999 • 15 PROSPECTIVE COHORT STUDIES • 8533 MOTHER-CHILD PAIRS • REDUCTION OF TRANSMISSION 50% (OR 0. 43, 95% CI, 0. 33 – 0. 56) WITH ELECTIVE C-S VS. OTHER MODES OF DELIVERY • REDUCTION OF TRANSMISSION 87% (OR 0. 13, 95% CI, 0. 09 – 0. 19) WITH ELECTIVE C-S & PACTG 076
MODE OF DELIVERY: META-ANALYSIS THE INTERNATIONAL PERINATAL HIV GROUP, APRIL 1999 • 15 PROSPECTIVE COHORT STUDIES • 8533 MOTHER-CHILD PAIRS • REDUCTION OF TRANSMISSION 50% (OR 0. 43, 95% CI, 0. 33 – 0. 56) WITH ELECTIVE C-S VS. OTHER MODES OF DELIVERY • REDUCTION OF TRANSMISSION 87% (OR 0. 13, 95% CI, 0. 09 – 0. 19) WITH ELECTIVE C-S & PACTG 076
 MODE OF DELIVERY – CAESAREAN SECTION • HIV INFECTED WOMEN SHOULD BE COUNSELLED ABOUT ELECTIVE C-S • VERTICAL TRANSMISSION IS REDUCED TO 2% WITH PACTG 076 THERAPY AND ELECTIVE C-S • WOMEN WITH HIGH VIRAL LOADS MAY BENEFIT MOST FROM C-S • TO AVOID SROM & ONSET OF LABOUR, ELECTIVE C-S IS PERFORMED AT 38 WEEKS • AFTER SROM OR ONSET OF LABOUR C-S IS LESS PROTECTIVE • TO AVOID C-S MORBIDITY, ANTIBIOTIC PROPHYLAXIS SHOULD BE CONSIDERED
MODE OF DELIVERY – CAESAREAN SECTION • HIV INFECTED WOMEN SHOULD BE COUNSELLED ABOUT ELECTIVE C-S • VERTICAL TRANSMISSION IS REDUCED TO 2% WITH PACTG 076 THERAPY AND ELECTIVE C-S • WOMEN WITH HIGH VIRAL LOADS MAY BENEFIT MOST FROM C-S • TO AVOID SROM & ONSET OF LABOUR, ELECTIVE C-S IS PERFORMED AT 38 WEEKS • AFTER SROM OR ONSET OF LABOUR C-S IS LESS PROTECTIVE • TO AVOID C-S MORBIDITY, ANTIBIOTIC PROPHYLAXIS SHOULD BE CONSIDERED
 VIRAL LOAD
VIRAL LOAD
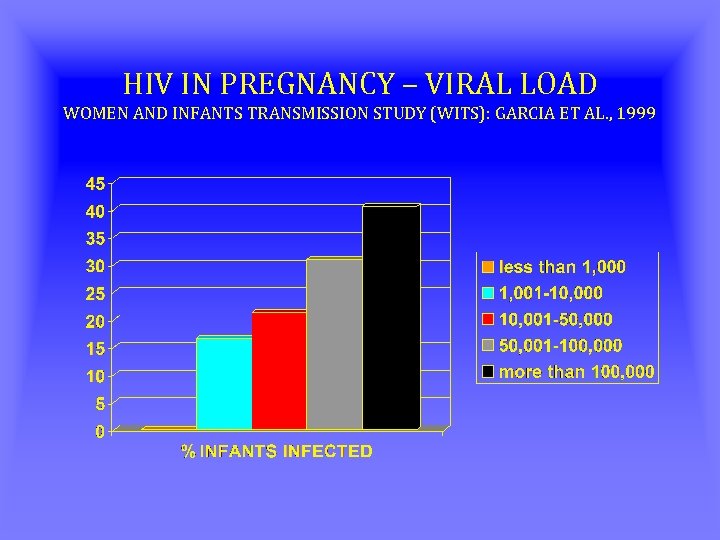
 HIV IN PREGNANCY – VIRAL LOAD WOMEN AND INFANTS TRANSMISSION STUDY (WITS): GARCIA ET AL. , 1999 HIV Viral Load (Copies per m. L) Less than 1, 000 Number of HIV Transmissions 0 of 57 1, 000 – 10, 000 32 of 193 10, 001 – 50, 000 39 of 183 50, 001 – 100, 000 17 of 54 Greater than 100, 000 26 of 64
HIV IN PREGNANCY – VIRAL LOAD WOMEN AND INFANTS TRANSMISSION STUDY (WITS): GARCIA ET AL. , 1999 HIV Viral Load (Copies per m. L) Less than 1, 000 Number of HIV Transmissions 0 of 57 1, 000 – 10, 000 32 of 193 10, 001 – 50, 000 39 of 183 50, 001 – 100, 000 17 of 54 Greater than 100, 000 26 of 64
 HIV IN PREGNANCY – VIRAL LOAD WOMEN AND INFANTS TRANSMISSION STUDY (WITS): GARCIA ET AL. , 1999
HIV IN PREGNANCY – VIRAL LOAD WOMEN AND INFANTS TRANSMISSION STUDY (WITS): GARCIA ET AL. , 1999
 BREASTFEEDING IN HIV POSITIVE WOMEN
BREASTFEEDING IN HIV POSITIVE WOMEN
 INTRODUCTION • HIV DNA PRESENT IN BREAST MILK • HIV TRANSMISSION CAN OCCUR THROUGH BREASTFEEDING • BREASTFEEDING IS AN INDEPENDENT RISK FACTOR FOR HIV TRANSMISSION
INTRODUCTION • HIV DNA PRESENT IN BREAST MILK • HIV TRANSMISSION CAN OCCUR THROUGH BREASTFEEDING • BREASTFEEDING IS AN INDEPENDENT RISK FACTOR FOR HIV TRANSMISSION
 EVIDENCE TO SUPPORT TRANSMISSION • ISOLATION OF HIV-1 FROM CELLULAR & NON-CELLULAR FRACTIONS OF BREAST MILK • CASE REPORTS OF INFECTED CHILDREN BREASTFED BY MOTHERS WHO ACQUIRED HIV POSTPARTUM
EVIDENCE TO SUPPORT TRANSMISSION • ISOLATION OF HIV-1 FROM CELLULAR & NON-CELLULAR FRACTIONS OF BREAST MILK • CASE REPORTS OF INFECTED CHILDREN BREASTFED BY MOTHERS WHO ACQUIRED HIV POSTPARTUM
 EVIDENCE TO SUPPORT TRANSMISSION • DOCUMENTATION OF OTHER RETROVIRUSES TRANSMITTED THROUGH BREAST MILK • CASE REPORTS OF BREAST FED CHILDREN WHO WERE INITIALLY HIV NEGATIVE BUT SEROCONVERTED DURING BREASTFEEDING
EVIDENCE TO SUPPORT TRANSMISSION • DOCUMENTATION OF OTHER RETROVIRUSES TRANSMITTED THROUGH BREAST MILK • CASE REPORTS OF BREAST FED CHILDREN WHO WERE INITIALLY HIV NEGATIVE BUT SEROCONVERTED DURING BREASTFEEDING
 POLICIES • AVOIDANCE OF BREASTFEEDING IS CONTROVERSIAL AND DEPENDS ON INTERNAL MILIEU • DEVELOPING COUNTRIES VS INDUSTRIALIZED COUNTRIES
POLICIES • AVOIDANCE OF BREASTFEEDING IS CONTROVERSIAL AND DEPENDS ON INTERNAL MILIEU • DEVELOPING COUNTRIES VS INDUSTRIALIZED COUNTRIES
 POLICIES • UNAIDS REVISED STATEMENT 1998: WOMEN SHOULD BE OFFERED HIV COUNSELING AND TESTING, BE INFORMED OF RISKS AND BENEFITS OF BREASTFEEDING IF THE MOTHER IS HIV POSITIVE, AND SHOULD MAKE A DECISION THAT TAKES INTO ACCOUNT THE INDIVIDUAL &FAMILY SITUATIONS
POLICIES • UNAIDS REVISED STATEMENT 1998: WOMEN SHOULD BE OFFERED HIV COUNSELING AND TESTING, BE INFORMED OF RISKS AND BENEFITS OF BREASTFEEDING IF THE MOTHER IS HIV POSITIVE, AND SHOULD MAKE A DECISION THAT TAKES INTO ACCOUNT THE INDIVIDUAL &FAMILY SITUATIONS
 MECHANISM OF TRANSMISSION • EXACT MECHANISM OF TRANSMISSION THROUGH BREAST MILK STILL NOT WELL UNDERSTOOD • INFECTION VIA CELL-FREE HIV IN BREAST MILK OR VIA HIV-INFECTED CELLS • SUSCEPTIBILITY OF IMMATURE NEONATAL GI TRACT TO VIRUS • GI TRACT MUCOSAL DAMAGE
MECHANISM OF TRANSMISSION • EXACT MECHANISM OF TRANSMISSION THROUGH BREAST MILK STILL NOT WELL UNDERSTOOD • INFECTION VIA CELL-FREE HIV IN BREAST MILK OR VIA HIV-INFECTED CELLS • SUSCEPTIBILITY OF IMMATURE NEONATAL GI TRACT TO VIRUS • GI TRACT MUCOSAL DAMAGE
 DURATION OF BREASTFEEDING • STUDIES - IN TRANSMISSION WITH INCREASING DURATION OF BREASTFEEDING
DURATION OF BREASTFEEDING • STUDIES - IN TRANSMISSION WITH INCREASING DURATION OF BREASTFEEDING
 MALAWI, JAMA 1999 • CUMULATIVE INFECTION RISK WHILE BREASTFEEDING • • • 3. 5% AT END OF 5 MONTHS 7. 0% AT END OF 11 MONTHS 8. 9% AT END OF 17 MONTHS 10. 3% AT END OF 23 MONTHS NO FURTHER TRANSMISSION AFTER BREASTFEEDING STOPPED
MALAWI, JAMA 1999 • CUMULATIVE INFECTION RISK WHILE BREASTFEEDING • • • 3. 5% AT END OF 5 MONTHS 7. 0% AT END OF 11 MONTHS 8. 9% AT END OF 17 MONTHS 10. 3% AT END OF 23 MONTHS NO FURTHER TRANSMISSION AFTER BREASTFEEDING STOPPED
 MULTICENTER STUDY, LANCET 1998 • CUMULATIVE INFECTION RISK WHILE BREASTFEEDING • • • 0. 7% AT END OF 6 MONTHS 0. 95% AT END OF 9 MONTHS 2. 5% AT END OF 12 MONTHS 6. 3% AT END OF 18 MONTHS 7. 4% AT END OF 24 MONTHS 9. 2% AT END OF 36 MONTHS
MULTICENTER STUDY, LANCET 1998 • CUMULATIVE INFECTION RISK WHILE BREASTFEEDING • • • 0. 7% AT END OF 6 MONTHS 0. 95% AT END OF 9 MONTHS 2. 5% AT END OF 12 MONTHS 6. 3% AT END OF 18 MONTHS 7. 4% AT END OF 24 MONTHS 9. 2% AT END OF 36 MONTHS
 DURATION OF BREASTFEEDING • ? EARLY WEANING POLICY • PROBLEMS WITH EARLY WEANING • ADVERSE NEONATAL EFFECTS • COLOSTRUM HIGHLY INFECTIOUS
DURATION OF BREASTFEEDING • ? EARLY WEANING POLICY • PROBLEMS WITH EARLY WEANING • ADVERSE NEONATAL EFFECTS • COLOSTRUM HIGHLY INFECTIOUS
 EXCLUSIVITY OF BRESTFEEDING • STUDIES - INFANTS EXCLUSIVELY BREAST FED AT LOWER RISK OF ACQUIRING HIV THAN THOSE FED WITH OTHER TYPES OF MILK, TEA, OR JUICE WHILE BEING BREAST FED
EXCLUSIVITY OF BRESTFEEDING • STUDIES - INFANTS EXCLUSIVELY BREAST FED AT LOWER RISK OF ACQUIRING HIV THAN THOSE FED WITH OTHER TYPES OF MILK, TEA, OR JUICE WHILE BEING BREAST FED
 BRAZIL STUDY, 1998 • CHILDREN FED WITH OTHER TYPES OF MILK WHILE BEING BREASTFED WERE AT 2. 2 -FOLD GREATER RISK OF HIV INFECTION THAN THOSE EXCLUSIVELY BREASTFED • CHILDREN FED WITH TEA OR FRUIT JUICE WHLE BEING BREASTFED WERE AT 2. 6 -FOLD GREATER RISK OF INFECTION
BRAZIL STUDY, 1998 • CHILDREN FED WITH OTHER TYPES OF MILK WHILE BEING BREASTFED WERE AT 2. 2 -FOLD GREATER RISK OF HIV INFECTION THAN THOSE EXCLUSIVELY BREASTFED • CHILDREN FED WITH TEA OR FRUIT JUICE WHLE BEING BREASTFED WERE AT 2. 6 -FOLD GREATER RISK OF INFECTION
 DURBAN (SOUTH AFRICA), LANCET 1999 • 3 GROUPS OF CHILDREN - NEVER BREASTFED, EXCLUSIVELY BREASTFED, MIXED FEEDING • NO SIGNIFICANT DIFFERENCE IN TRANSMISSION BETWEEN NEVER AND EXCLUSIVELY BREASTFED GROUPS • SIGNIFICANTLY INCREASED RISK OF TRANSMISSION FOR MIXED FEEDING
DURBAN (SOUTH AFRICA), LANCET 1999 • 3 GROUPS OF CHILDREN - NEVER BREASTFED, EXCLUSIVELY BREASTFED, MIXED FEEDING • NO SIGNIFICANT DIFFERENCE IN TRANSMISSION BETWEEN NEVER AND EXCLUSIVELY BREASTFED GROUPS • SIGNIFICANTLY INCREASED RISK OF TRANSMISSION FOR MIXED FEEDING
 INTERPRETATION • IMMUNE FACTORS IN BREAST MILK • GROWTH FACTORS IN BREAST MILK • MUCOSAL DAMAGE WITH MIXED FEEDING
INTERPRETATION • IMMUNE FACTORS IN BREAST MILK • GROWTH FACTORS IN BREAST MILK • MUCOSAL DAMAGE WITH MIXED FEEDING
 MATERNAL FACTORS • CRACKED NIPPLES • BLEEDING NIPPLES • PARITY
MATERNAL FACTORS • CRACKED NIPPLES • BLEEDING NIPPLES • PARITY
 CONCLUSION • PRECISE RISK FACTORS AND MECHANISM OF TRANSMISSION STILL NOT WELL UNDERSTOOD • WOMEN WHO ARE HIV POSITIVE SHOULD BE ADVISED TO AVOID BREASTFEEDING • WOMEN WHO BREASTFEED SHOULD BE INFORMED THAT TRANSMISSION CAN OCCUR
CONCLUSION • PRECISE RISK FACTORS AND MECHANISM OF TRANSMISSION STILL NOT WELL UNDERSTOOD • WOMEN WHO ARE HIV POSITIVE SHOULD BE ADVISED TO AVOID BREASTFEEDING • WOMEN WHO BREASTFEED SHOULD BE INFORMED THAT TRANSMISSION CAN OCCUR
 SUMMARY
SUMMARY
 HIV SCREENING • ALL PREGNANT WOMEN SHOULD BE OFFERRED HIV TESTING • PRE- & POST- TEST COUNSELING FOR ALL PREGNANT WOMEN • TARGETED TESTING OF PREGNANT WOMEN WHO REPORT HIGH RISK BEHAVIOR NOT RECOMMENDED
HIV SCREENING • ALL PREGNANT WOMEN SHOULD BE OFFERRED HIV TESTING • PRE- & POST- TEST COUNSELING FOR ALL PREGNANT WOMEN • TARGETED TESTING OF PREGNANT WOMEN WHO REPORT HIGH RISK BEHAVIOR NOT RECOMMENDED
 ANTENATAL CARE • HIV IN PREGNANCY REQUIRES MULTIDISCIPLINARY APPROACH • ANTENATAL CARE IS SIMILAR TO THAT OF HIV -VE WOMEN • PREGNANCY NOT HIGH RISK • AVOID INVASIVE PROCEDURES • MONITOR CD 4+ AND VIRAL LOAD AT LEAST EVERY 3 MONTHS IF ABLE TO PROVIDE ANTIRETROVIRAL THERAPY
ANTENATAL CARE • HIV IN PREGNANCY REQUIRES MULTIDISCIPLINARY APPROACH • ANTENATAL CARE IS SIMILAR TO THAT OF HIV -VE WOMEN • PREGNANCY NOT HIGH RISK • AVOID INVASIVE PROCEDURES • MONITOR CD 4+ AND VIRAL LOAD AT LEAST EVERY 3 MONTHS IF ABLE TO PROVIDE ANTIRETROVIRAL THERAPY
 ANTIRETROVIRAL USE • Zidovudine reduces perinatal transmission in women at different stages of disease • long (ante, peri, and postnatal) as well as shorter regimens effective • still effective in breastfeeding populations • Use of other antiretrovirals in combination with ZDV promising, still investigational
ANTIRETROVIRAL USE • Zidovudine reduces perinatal transmission in women at different stages of disease • long (ante, peri, and postnatal) as well as shorter regimens effective • still effective in breastfeeding populations • Use of other antiretrovirals in combination with ZDV promising, still investigational
 INTRAPARTUM ANTIRETROVIRAL THERAPY • DURING LABOR - ZDV 2 MG/KG IV LOADING DOSE, THEN 1 MG/KG/HR • IF IV ZDV NOT AVAILABLE CONSIDER PO REGIMEN • MAY CONSIDER ADDITION OF NVP 200 MG PO TO IV ZDV @ ONSET OF LABOR
INTRAPARTUM ANTIRETROVIRAL THERAPY • DURING LABOR - ZDV 2 MG/KG IV LOADING DOSE, THEN 1 MG/KG/HR • IF IV ZDV NOT AVAILABLE CONSIDER PO REGIMEN • MAY CONSIDER ADDITION OF NVP 200 MG PO TO IV ZDV @ ONSET OF LABOR
 BREASTFEEDING • PRECISE RISK FACTORS AND MECHANISM OF TRANSMISSION STILL NOT WELL UNDERSTOOD • WOMEN WHO ARE HIV POSITIVE SHOULD BE ADVISED TO AVOID BREASTFEEDING • WOMEN WHO BREASTFEED SHOULD BE INFORMED THAT TRANSMISSION CAN OCCUR
BREASTFEEDING • PRECISE RISK FACTORS AND MECHANISM OF TRANSMISSION STILL NOT WELL UNDERSTOOD • WOMEN WHO ARE HIV POSITIVE SHOULD BE ADVISED TO AVOID BREASTFEEDING • WOMEN WHO BREASTFEED SHOULD BE INFORMED THAT TRANSMISSION CAN OCCUR


