3e07fd3cb2514a802615b2b4da6423a6.ppt
- Количество слайдов: 73
 HEARTLINE 2014 IRCCS S. Martino Genoa Cardiology Meeting SCA-NSTEMI Trattamento antipiastrinico ed eccesso di sanguinamento : dove portano i risultati dell’ ACCOAST ? Luigi Oltrona Visconti Divisione di Cardiologia IRCCS Fondazione Policlinico S. Matteo Pavia
HEARTLINE 2014 IRCCS S. Martino Genoa Cardiology Meeting SCA-NSTEMI Trattamento antipiastrinico ed eccesso di sanguinamento : dove portano i risultati dell’ ACCOAST ? Luigi Oltrona Visconti Divisione di Cardiologia IRCCS Fondazione Policlinico S. Matteo Pavia
 2011
2011
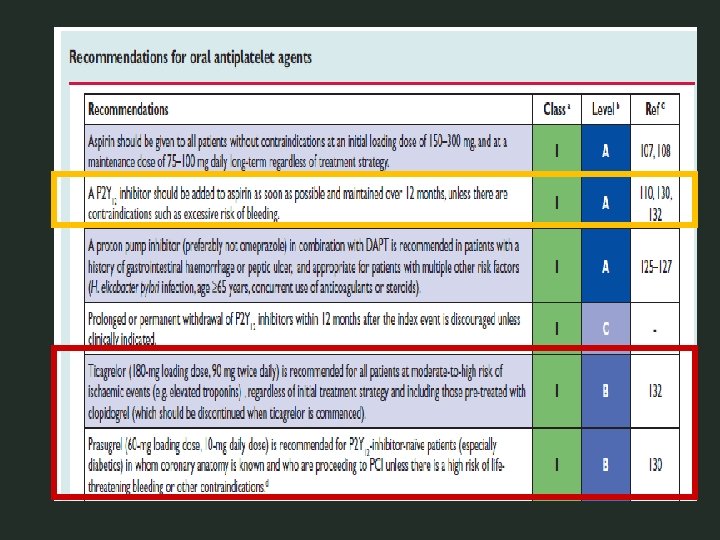
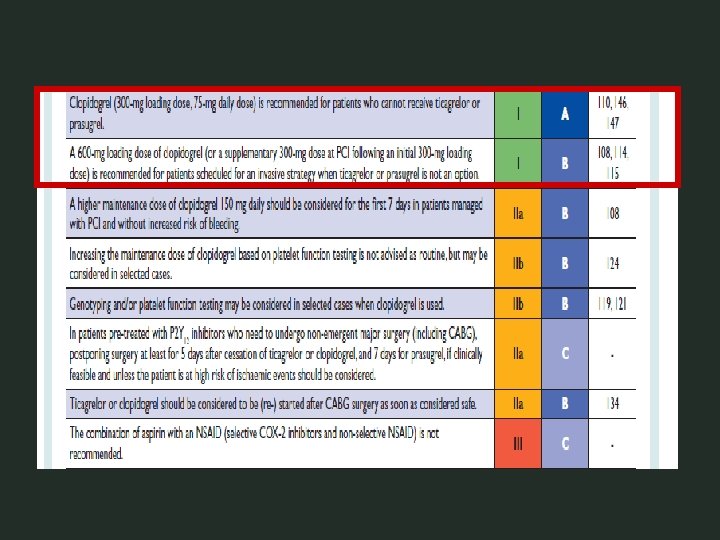
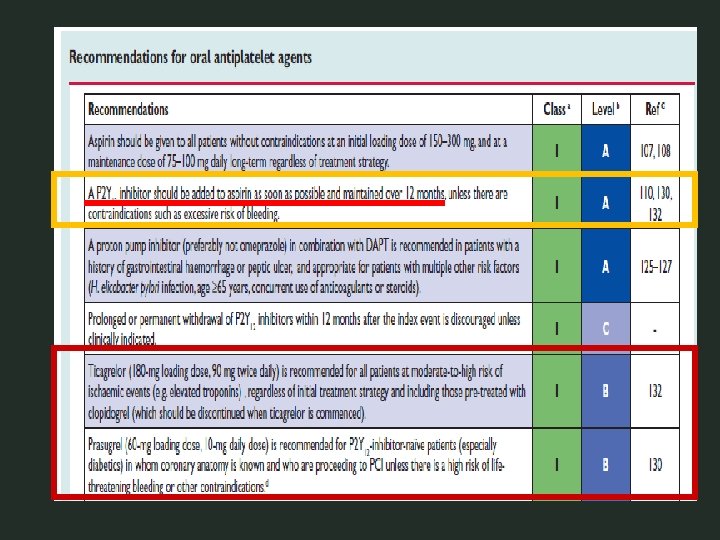
 PCI-CURE Principali risultati
PCI-CURE Principali risultati
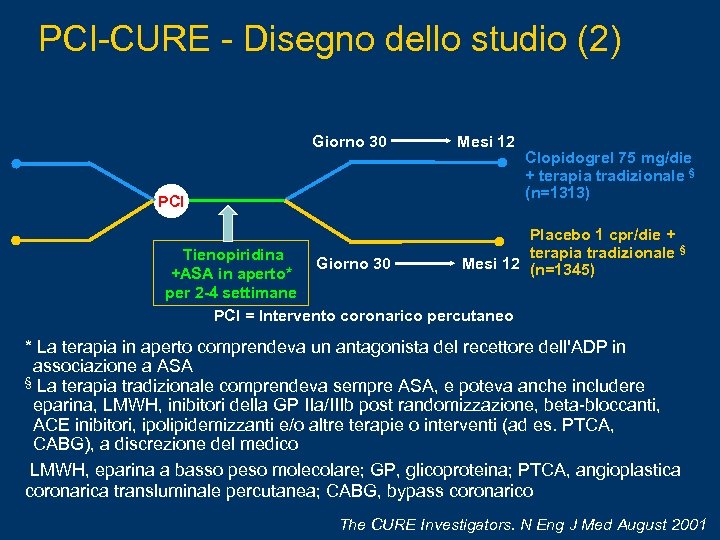 PCI-CURE - Disegno dello studio (2) Giorno 30 Mesi 12 PCI Clopidogrel 75 mg/die + terapia tradizionale § (n=1313) Placebo 1 cpr/die + terapia tradizionale § Mesi 12 (n=1345) Tienopiridina Giorno 30 +ASA in aperto* per 2 -4 settimane PCI = Intervento coronarico percutaneo * La terapia in aperto comprendeva un antagonista del recettore dell'ADP in associazione a ASA § La terapia tradizionale comprendeva sempre ASA, e poteva anche includere eparina, LMWH, inibitori della GP IIa/IIIb post randomizzazione, beta-bloccanti, ACE inibitori, ipolipidemizzanti e/o altre terapie o interventi (ad es. PTCA, CABG), a discrezione del medico LMWH, eparina a basso peso molecolare; GP, glicoproteina; PTCA, angioplastica coronarica transluminale percutanea; CABG, bypass coronarico The CURE Investigators. N Eng J Med August 2001
PCI-CURE - Disegno dello studio (2) Giorno 30 Mesi 12 PCI Clopidogrel 75 mg/die + terapia tradizionale § (n=1313) Placebo 1 cpr/die + terapia tradizionale § Mesi 12 (n=1345) Tienopiridina Giorno 30 +ASA in aperto* per 2 -4 settimane PCI = Intervento coronarico percutaneo * La terapia in aperto comprendeva un antagonista del recettore dell'ADP in associazione a ASA § La terapia tradizionale comprendeva sempre ASA, e poteva anche includere eparina, LMWH, inibitori della GP IIa/IIIb post randomizzazione, beta-bloccanti, ACE inibitori, ipolipidemizzanti e/o altre terapie o interventi (ad es. PTCA, CABG), a discrezione del medico LMWH, eparina a basso peso molecolare; GP, glicoproteina; PTCA, angioplastica coronarica transluminale percutanea; CABG, bypass coronarico The CURE Investigators. N Eng J Med August 2001
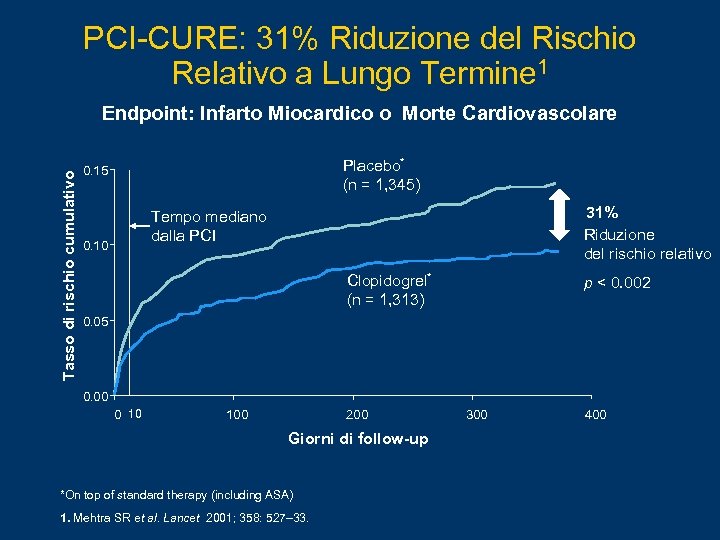 PCI-CURE: 31% Riduzione del Rischio Relativo a Lungo Termine 1 Tasso di rischio cumulativo Endpoint: Infarto Miocardico o Morte Cardiovascolare Placebo* (n = 1, 345) 0. 15 31% Riduzione del rischio relativo Tempo mediano dalla PCI 0. 10 Clopidogrel* (n = 1, 313) p < 0. 002 0. 05 0. 00 0 10 100 200 Giorni di follow-up *On top of standard therapy (including ASA) 1. Mehtra SR et al. Lancet 2001; 358: 527– 33. 300 400
PCI-CURE: 31% Riduzione del Rischio Relativo a Lungo Termine 1 Tasso di rischio cumulativo Endpoint: Infarto Miocardico o Morte Cardiovascolare Placebo* (n = 1, 345) 0. 15 31% Riduzione del rischio relativo Tempo mediano dalla PCI 0. 10 Clopidogrel* (n = 1, 313) p < 0. 002 0. 05 0. 00 0 10 100 200 Giorni di follow-up *On top of standard therapy (including ASA) 1. Mehtra SR et al. Lancet 2001; 358: 527– 33. 300 400
 The CREDO Trial Clopidogrel for the Reduction of Events During Observation
The CREDO Trial Clopidogrel for the Reduction of Events During Observation
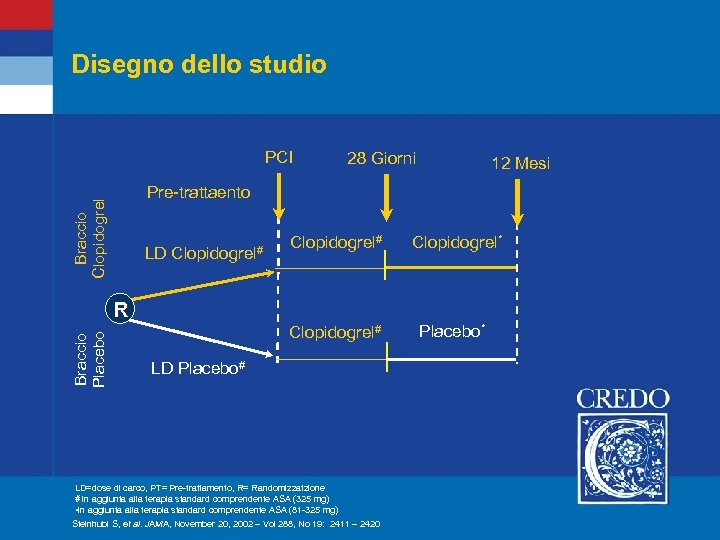 Disegno dello studio PCI 28 Giorni 12 Mesi Braccio Clopidogrel Pre-trattaento LD Clopidogrel# Clopidogrel* Braccio Placebo R Clopidogrel# LD Placebo# LD=dose di carco, PT= Pre-trattamento, R= Randomizzatzione # in aggiunta alla terapia standard comprendente ASA (325 mg) • in aggiunta alla terapia standard comprendente ASA (81 -325 mg) Steinhubl S, et al. JAMA, November 20, 2002 – Vol 288, No 19: 2411 – 2420 Placebo*
Disegno dello studio PCI 28 Giorni 12 Mesi Braccio Clopidogrel Pre-trattaento LD Clopidogrel# Clopidogrel* Braccio Placebo R Clopidogrel# LD Placebo# LD=dose di carco, PT= Pre-trattamento, R= Randomizzatzione # in aggiunta alla terapia standard comprendente ASA (325 mg) • in aggiunta alla terapia standard comprendente ASA (81 -325 mg) Steinhubl S, et al. JAMA, November 20, 2002 – Vol 288, No 19: 2411 – 2420 Placebo*
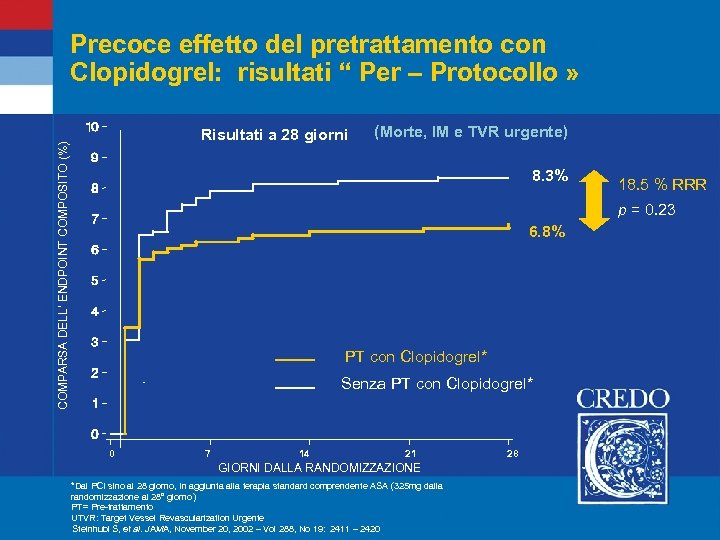 Precoce effetto del pretrattamento con Clopidogrel: risultati “ Per – Protocollo » COMPARSA DELL’ ENDPOINT COMPOSITO (%) Risultati a 28 giorni (Morte, IM e TVR urgente) 8. 3% 18. 5 % RRR p = 0. 23 6. 8% PT con Clopidogrel* Senza PT con Clopidogrel* 0 7 14 21 GIORNI DALLA RANDOMIZZAZIONE *Dal PCI sino al 28 giorno, In aggiunta alla terapia standard comprendente ASA (325 mg dalla randomizzazione al 28° giorno) PT= Pre-trattamento UTVR: Target Vessel Revascularization Urgente Steinhubl S, et al. JAMA, November 20, 2002 – Vol 288, No 19: 2411 – 2420 28
Precoce effetto del pretrattamento con Clopidogrel: risultati “ Per – Protocollo » COMPARSA DELL’ ENDPOINT COMPOSITO (%) Risultati a 28 giorni (Morte, IM e TVR urgente) 8. 3% 18. 5 % RRR p = 0. 23 6. 8% PT con Clopidogrel* Senza PT con Clopidogrel* 0 7 14 21 GIORNI DALLA RANDOMIZZAZIONE *Dal PCI sino al 28 giorno, In aggiunta alla terapia standard comprendente ASA (325 mg dalla randomizzazione al 28° giorno) PT= Pre-trattamento UTVR: Target Vessel Revascularization Urgente Steinhubl S, et al. JAMA, November 20, 2002 – Vol 288, No 19: 2411 – 2420 28
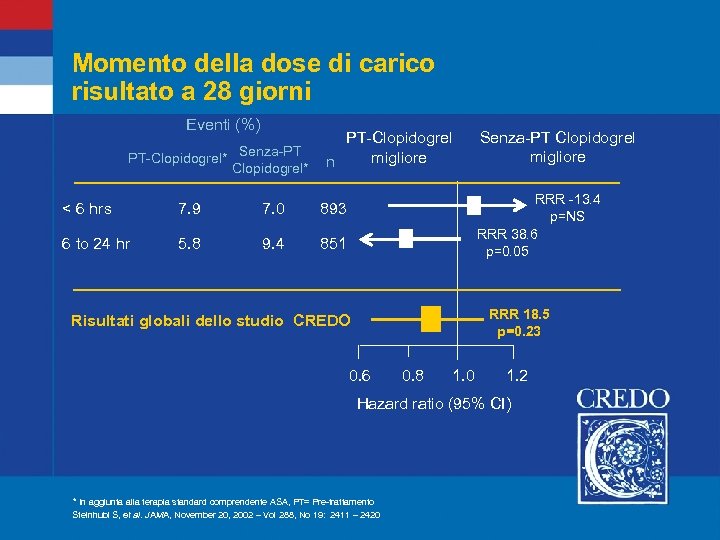 Momento della dose di carico risultato a 28 giorni Eventi (%) PT-Clopidogrel* Senza-PT Clopidogrel* PT-Clopidogrel migliore n < 6 hrs 7. 9 7. 0 5. 8 9. 4 RRR -13. 4 p=NS RRR 38. 6 p=0. 05 893 6 to 24 hr Senza-PT Clopidogrel migliore 851 RRR 18. 5 p=0. 23 Risultati globali dello studio CREDO 0. 6 0. 8 1. 0 1. 2 Hazard ratio (95% CI) * In aggiunta alla terapia standard comprendente ASA, PT= Pre-trattamento Steinhubl S, et al. JAMA, November 20, 2002 – Vol 288, No 19: 2411 – 2420
Momento della dose di carico risultato a 28 giorni Eventi (%) PT-Clopidogrel* Senza-PT Clopidogrel* PT-Clopidogrel migliore n < 6 hrs 7. 9 7. 0 5. 8 9. 4 RRR -13. 4 p=NS RRR 38. 6 p=0. 05 893 6 to 24 hr Senza-PT Clopidogrel migliore 851 RRR 18. 5 p=0. 23 Risultati globali dello studio CREDO 0. 6 0. 8 1. 0 1. 2 Hazard ratio (95% CI) * In aggiunta alla terapia standard comprendente ASA, PT= Pre-trattamento Steinhubl S, et al. JAMA, November 20, 2002 – Vol 288, No 19: 2411 – 2420
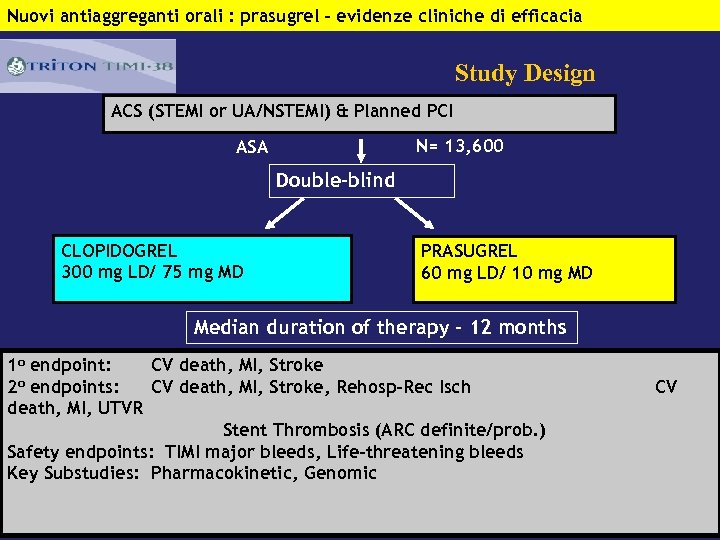 Nuovi antiaggreganti orali : prasugrel – evidenze cliniche di efficacia Study Design ACS (STEMI or UA/NSTEMI) & Planned PCI N= 13, 600 ASA Double-blind CLOPIDOGREL 300 mg LD/ 75 mg MD PRASUGREL 60 mg LD/ 10 mg MD Median duration of therapy - 12 months 1 o endpoint: CV death, MI, Stroke 2 o endpoints: CV death, MI, Stroke, Rehosp-Rec Isch death, MI, UTVR Stent Thrombosis (ARC definite/prob. ) Safety endpoints: TIMI major bleeds, Life-threatening bleeds Key Substudies: Pharmacokinetic, Genomic CV
Nuovi antiaggreganti orali : prasugrel – evidenze cliniche di efficacia Study Design ACS (STEMI or UA/NSTEMI) & Planned PCI N= 13, 600 ASA Double-blind CLOPIDOGREL 300 mg LD/ 75 mg MD PRASUGREL 60 mg LD/ 10 mg MD Median duration of therapy - 12 months 1 o endpoint: CV death, MI, Stroke 2 o endpoints: CV death, MI, Stroke, Rehosp-Rec Isch death, MI, UTVR Stent Thrombosis (ARC definite/prob. ) Safety endpoints: TIMI major bleeds, Life-threatening bleeds Key Substudies: Pharmacokinetic, Genomic CV
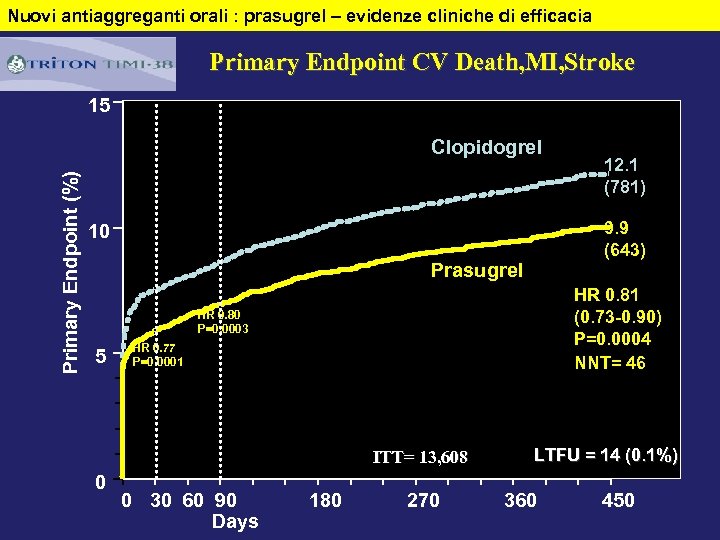 Nuovi antiaggreganti orali : prasugrel – evidenze cliniche di efficacia Primary Endpoint CV Death, MI, Stroke 15 Primary Endpoint (%) Clopidogrel 9. 9 (643) 10 Prasugrel HR 0. 81 (0. 73 -0. 90) P=0. 0004 NNT= 46 HR 0. 80 P=0. 0003 5 HR 0. 77 P=0. 0001 ITT= 13, 608 0 0 30 60 90 Days 12. 1 (781) 180 270 LTFU = 14 (0. 1%) 360 450
Nuovi antiaggreganti orali : prasugrel – evidenze cliniche di efficacia Primary Endpoint CV Death, MI, Stroke 15 Primary Endpoint (%) Clopidogrel 9. 9 (643) 10 Prasugrel HR 0. 81 (0. 73 -0. 90) P=0. 0004 NNT= 46 HR 0. 80 P=0. 0003 5 HR 0. 77 P=0. 0001 ITT= 13, 608 0 0 30 60 90 Days 12. 1 (781) 180 270 LTFU = 14 (0. 1%) 360 450
 A Comparison of Prasugrel at the Time of Percutaneous Coronary Intervention Or as Pre-treatment At the Time of Diagnosis in Patients with Non-ST-Elevation Myocardial Infarction (NSTEMI)
A Comparison of Prasugrel at the Time of Percutaneous Coronary Intervention Or as Pre-treatment At the Time of Diagnosis in Patients with Non-ST-Elevation Myocardial Infarction (NSTEMI)
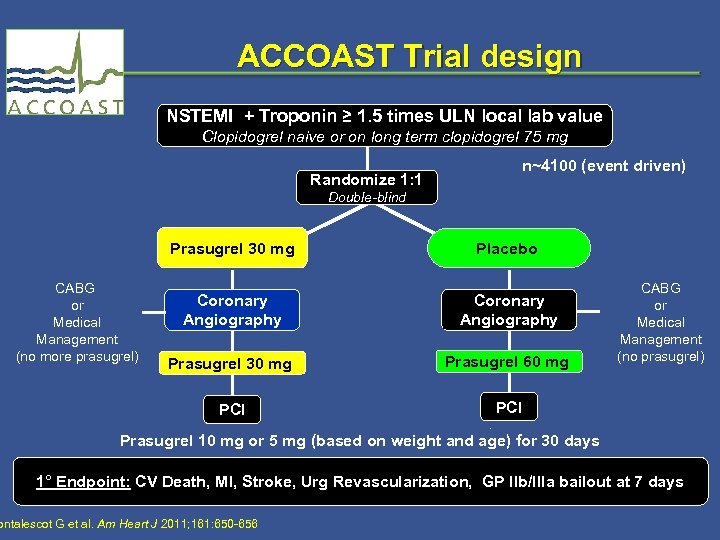 ACCOAST Trial design NSTEMI + Troponin ≥ 1. 5 times ULN local lab value Clopidogrel naive or on long term clopidogrel 75 mg Randomize 1: 1 n~4100 (event driven) Double-blind Prasugrel 30 mg Coronary Angiography Prasugrel 30 mg Prasugrel 60 mg PCI CABG or Medical Management (no more prasugrel) Placebo PCI CABG or Medical Management (no prasugrel) Prasugrel 10 mg or 5 mg (based on weight and age) for 30 days 1° Endpoint: CV Death, MI, Stroke, Urg Revascularization, GP IIb/IIIa bailout at 7 days ontalescot G et al. Am Heart J 2011; 161: 650 -656
ACCOAST Trial design NSTEMI + Troponin ≥ 1. 5 times ULN local lab value Clopidogrel naive or on long term clopidogrel 75 mg Randomize 1: 1 n~4100 (event driven) Double-blind Prasugrel 30 mg Coronary Angiography Prasugrel 30 mg Prasugrel 60 mg PCI CABG or Medical Management (no more prasugrel) Placebo PCI CABG or Medical Management (no prasugrel) Prasugrel 10 mg or 5 mg (based on weight and age) for 30 days 1° Endpoint: CV Death, MI, Stroke, Urg Revascularization, GP IIb/IIIa bailout at 7 days ontalescot G et al. Am Heart J 2011; 161: 650 -656
 NEJM 2013; 369: 999 -1010
NEJM 2013; 369: 999 -1010
 Clinical questions and rationale of the study To answer the question: IN NSTEMI patients, does having good inhibition of P 2 Y 12 mediated platelet activation and aggregation prior to the start of PCI reduce the incidence of ischemic events compared to administration of a fast acting inhibitor (prasugrel) on the table? ” Potential benefits of pretreatment: Prevention of ischemic events in NSTEMI patients while waiting for the PCI, during and after PCI. Potential risks of pretreatment: Bleeding risks need to be considered when starting platelet inhibition in NSTEMI patients before the coronary anatomy is known (eg, the patient will not be a candidate for PCI- medical management or CABG).
Clinical questions and rationale of the study To answer the question: IN NSTEMI patients, does having good inhibition of P 2 Y 12 mediated platelet activation and aggregation prior to the start of PCI reduce the incidence of ischemic events compared to administration of a fast acting inhibitor (prasugrel) on the table? ” Potential benefits of pretreatment: Prevention of ischemic events in NSTEMI patients while waiting for the PCI, during and after PCI. Potential risks of pretreatment: Bleeding risks need to be considered when starting platelet inhibition in NSTEMI patients before the coronary anatomy is known (eg, the patient will not be a candidate for PCI- medical management or CABG).
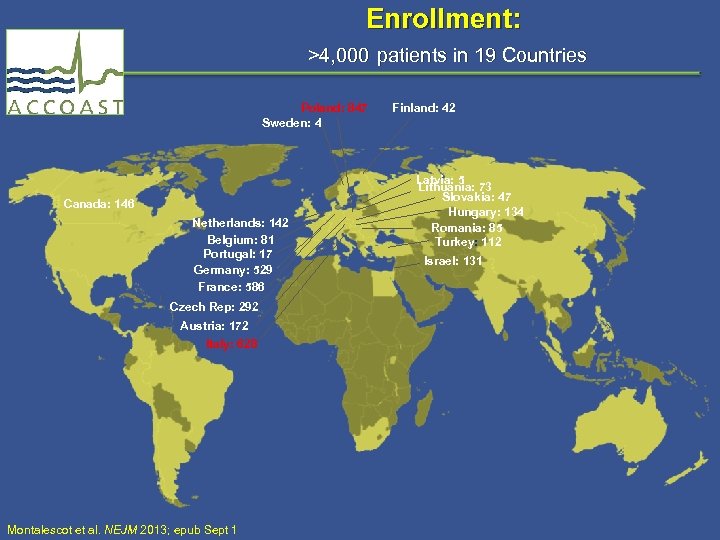 Enrollment: >4, 000 patients in 19 Countries Poland: 847 Sweden: 4 Canada: 146 Netherlands: 142 Belgium: 81 Portugal: 17 Germany: 529 France: 586 Czech Rep: 292 Austria: 172 Italy: 628 Montalescot et al. NEJM 2013; epub Sept 1 Finland: 42 Latvia: 5 Lithuania: 73 Slovakia: 47 Hungary: 134 Romania: 85 Turkey: 112 Israel: 131
Enrollment: >4, 000 patients in 19 Countries Poland: 847 Sweden: 4 Canada: 146 Netherlands: 142 Belgium: 81 Portugal: 17 Germany: 529 France: 586 Czech Rep: 292 Austria: 172 Italy: 628 Montalescot et al. NEJM 2013; epub Sept 1 Finland: 42 Latvia: 5 Lithuania: 73 Slovakia: 47 Hungary: 134 Romania: 85 Turkey: 112 Israel: 131
 Toni Badia, Ospedale Misericordia e Dolce, Prato; Sergio Berti, Fondazione Toscana G. Monasterio -Ospedale del Cuore G. Pasquinucci, Massa; Leonardo Bolognese, Cardiovascular and Neurological Department Azienda Ospedaliera Arezzo; Francesco Maria Bovenzi, Ospedale Campo di Marte, Lucca; Paola Camisasca, Nuovo Ospedale San Gerardo, Monza, Milano; Claudio Cavallini, Ospedale Santa Maria della Misericordia, Perugia; Raffaele De Caterina, Ospedale SS. Annunziata, Chieti; Stefano De Servi, Ospedale di Legnano, Milano; Giuseppe Fantini, Policlinico Universitario Modena, Modena; Claudio Fresco, Azienda Ospedaliero Universitaria Santa Maria della Misericordia, Udine; Antonio Manari, Azienda Ospedaliera-IRCCS S. Maria Nuova, Reggio Emilia; Sebastiano Marra, Azienda Ospedaliera S. Giovanni Battista, Torino; Ciro Mauro, Azienda. Ospedaliera Antonio Cardarelli, Napoli; Luca Olivotti, Ospedale Santa Corona, Pietra Ligure; Anna Sonia Petronio, Stabilimento Ospedaliero di Cisanello, Pisa; Francesco Prati, S. Giovanni Hospital, Rome; Bernhard Reimers, Ospedale Civile di Mirano, Venezia; Massimo Santini, Ospedale S. Filippo Neri, Roma; Silva Severi, Ospedale Misericordia, Grosseto; Luigi Oltrona Visconti, Division of Cardiology, Fondazione IRCCS Policlinico San Matteo, Pavia; Corrado Tamburino, Ospedale Ferrarotto, Catania; Roberto Zanini, Ospedale Civile Carlo Poma, Mantova.
Toni Badia, Ospedale Misericordia e Dolce, Prato; Sergio Berti, Fondazione Toscana G. Monasterio -Ospedale del Cuore G. Pasquinucci, Massa; Leonardo Bolognese, Cardiovascular and Neurological Department Azienda Ospedaliera Arezzo; Francesco Maria Bovenzi, Ospedale Campo di Marte, Lucca; Paola Camisasca, Nuovo Ospedale San Gerardo, Monza, Milano; Claudio Cavallini, Ospedale Santa Maria della Misericordia, Perugia; Raffaele De Caterina, Ospedale SS. Annunziata, Chieti; Stefano De Servi, Ospedale di Legnano, Milano; Giuseppe Fantini, Policlinico Universitario Modena, Modena; Claudio Fresco, Azienda Ospedaliero Universitaria Santa Maria della Misericordia, Udine; Antonio Manari, Azienda Ospedaliera-IRCCS S. Maria Nuova, Reggio Emilia; Sebastiano Marra, Azienda Ospedaliera S. Giovanni Battista, Torino; Ciro Mauro, Azienda. Ospedaliera Antonio Cardarelli, Napoli; Luca Olivotti, Ospedale Santa Corona, Pietra Ligure; Anna Sonia Petronio, Stabilimento Ospedaliero di Cisanello, Pisa; Francesco Prati, S. Giovanni Hospital, Rome; Bernhard Reimers, Ospedale Civile di Mirano, Venezia; Massimo Santini, Ospedale S. Filippo Neri, Roma; Silva Severi, Ospedale Misericordia, Grosseto; Luigi Oltrona Visconti, Division of Cardiology, Fondazione IRCCS Policlinico San Matteo, Pavia; Corrado Tamburino, Ospedale Ferrarotto, Catania; Roberto Zanini, Ospedale Civile Carlo Poma, Mantova.
 Main Inclusion/Exclusion Criteria Inclusion ¨ NSTEMI symptoms within 48 hours prior to study entry ¨ Elevated troponin (≥ 1. 5 times ULN) per local lab(s) ¨ Patient to be scheduled for coronary angiography and PCI within 2 hours to 24 hours of randomization and no later than 48 hours after randomization Exclusion ¨ STEMI patients ¨ Medical history contraindicating therapy with prasugrel ¨ History of stroke or transient ischemic attack (TIA) ¨ LD of any P 2 Y 12 antagonist ≤ 7 days of study entry Montalescot G et al. Am Heart J 2011; 161: 650656. e 1
Main Inclusion/Exclusion Criteria Inclusion ¨ NSTEMI symptoms within 48 hours prior to study entry ¨ Elevated troponin (≥ 1. 5 times ULN) per local lab(s) ¨ Patient to be scheduled for coronary angiography and PCI within 2 hours to 24 hours of randomization and no later than 48 hours after randomization Exclusion ¨ STEMI patients ¨ Medical history contraindicating therapy with prasugrel ¨ History of stroke or transient ischemic attack (TIA) ¨ LD of any P 2 Y 12 antagonist ≤ 7 days of study entry Montalescot G et al. Am Heart J 2011; 161: 650656. e 1
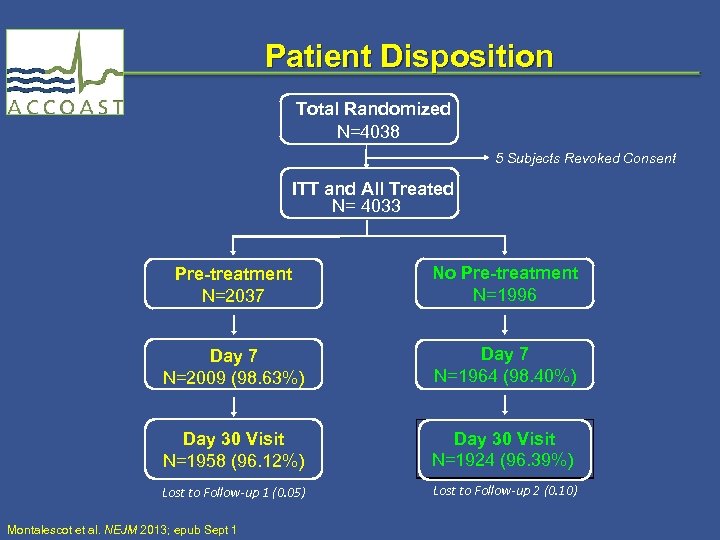 Patient Disposition Total Randomized N=4038 5 Subjects Revoked Consent ITT and All Treated N= 4033 Pre-treatment N=2037 No Pre-treatment N=1996 Day 7 N=2009 (98. 63%) Day 7 N=1964 (98. 40%) Day 30 Visit N=1958 (96. 12%) Day 30 Visit N=1924 (96. 39%) Lost to Follow-up 1 (0. 05) Lost to Follow-up 2 (0. 10) Montalescot et al. NEJM 2013; epub Sept 1
Patient Disposition Total Randomized N=4038 5 Subjects Revoked Consent ITT and All Treated N= 4033 Pre-treatment N=2037 No Pre-treatment N=1996 Day 7 N=2009 (98. 63%) Day 7 N=1964 (98. 40%) Day 30 Visit N=1958 (96. 12%) Day 30 Visit N=1924 (96. 39%) Lost to Follow-up 1 (0. 05) Lost to Follow-up 2 (0. 10) Montalescot et al. NEJM 2013; epub Sept 1
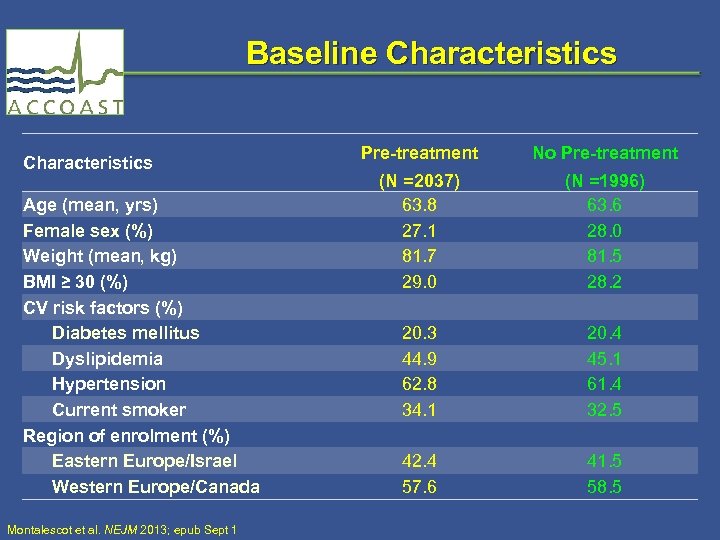 Baseline Characteristics Age (mean, yrs) Female sex (%) Weight (mean, kg) BMI ≥ 30 (%) CV risk factors (%) Diabetes mellitus Dyslipidemia Hypertension Current smoker Region of enrolment (%) Eastern Europe/Israel Western Europe/Canada Montalescot et al. NEJM 2013; epub Sept 1 Pre-treatment No Pre-treatment (N =2037) 63. 8 27. 1 81. 7 29. 0 20. 3 44. 9 62. 8 34. 1 42. 4 57. 6 (N =1996) 63. 6 28. 0 81. 5 28. 2 20. 4 45. 1 61. 4 32. 5 41. 5 58. 5
Baseline Characteristics Age (mean, yrs) Female sex (%) Weight (mean, kg) BMI ≥ 30 (%) CV risk factors (%) Diabetes mellitus Dyslipidemia Hypertension Current smoker Region of enrolment (%) Eastern Europe/Israel Western Europe/Canada Montalescot et al. NEJM 2013; epub Sept 1 Pre-treatment No Pre-treatment (N =2037) 63. 8 27. 1 81. 7 29. 0 20. 3 44. 9 62. 8 34. 1 42. 4 57. 6 (N =1996) 63. 6 28. 0 81. 5 28. 2 20. 4 45. 1 61. 4 32. 5 41. 5 58. 5
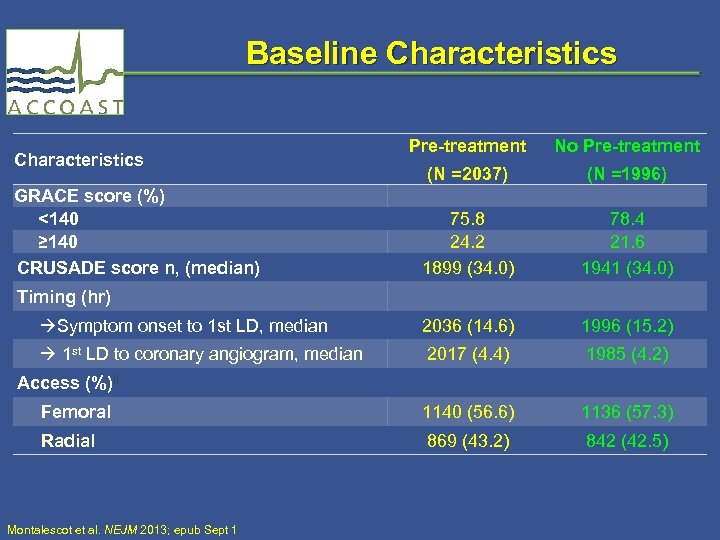 Baseline Characteristics Pre-treatment No Pre-treatment (N =2037) 75. 8 24. 2 1899 (34. 0) (N =1996) 78. 4 21. 6 1941 (34. 0) Symptom onset to 1 st LD, median 2036 (14. 6) 1996 (15. 2) 1 st LD to coronary angiogram, median 2017 (4. 4) 1985 (4. 2) Access (%)‖ Femoral 1140 (56. 6) 1136 (57. 3) Radial 869 (43. 2) 842 (42. 5) Characteristics GRACE score (%) <140 ≥ 140 CRUSADE score n, (median) Timing (hr) Montalescot et al. NEJM 2013; epub Sept 1
Baseline Characteristics Pre-treatment No Pre-treatment (N =2037) 75. 8 24. 2 1899 (34. 0) (N =1996) 78. 4 21. 6 1941 (34. 0) Symptom onset to 1 st LD, median 2036 (14. 6) 1996 (15. 2) 1 st LD to coronary angiogram, median 2017 (4. 4) 1985 (4. 2) Access (%)‖ Femoral 1140 (56. 6) 1136 (57. 3) Radial 869 (43. 2) 842 (42. 5) Characteristics GRACE score (%) <140 ≥ 140 CRUSADE score n, (median) Timing (hr) Montalescot et al. NEJM 2013; epub Sept 1
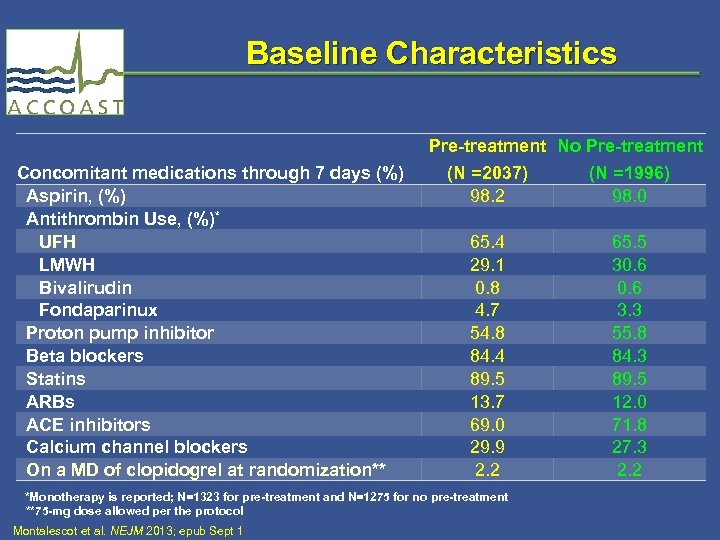 Baseline Characteristics Pre-treatment No Pre-treatment Concomitant medications through 7 days (%) Aspirin, (%) Antithrombin Use, (%)* UFH LMWH Bivalirudin Fondaparinux Proton pump inhibitor Beta blockers Statins ARBs ACE inhibitors Calcium channel blockers On a MD of clopidogrel at randomization** (N =2037) 98. 2 65. 4 29. 1 0. 8 4. 7 54. 8 84. 4 89. 5 13. 7 69. 0 29. 9 2. 2 *Monotherapy is reported; N=1323 for pre-treatment and N=1275 for no pre-treatment **75 -mg dose allowed per the protocol Montalescot et al. NEJM 2013; epub Sept 1 (N =1996) 98. 0 65. 5 30. 6 3. 3 55. 8 84. 3 89. 5 12. 0 71. 8 27. 3 2. 2
Baseline Characteristics Pre-treatment No Pre-treatment Concomitant medications through 7 days (%) Aspirin, (%) Antithrombin Use, (%)* UFH LMWH Bivalirudin Fondaparinux Proton pump inhibitor Beta blockers Statins ARBs ACE inhibitors Calcium channel blockers On a MD of clopidogrel at randomization** (N =2037) 98. 2 65. 4 29. 1 0. 8 4. 7 54. 8 84. 4 89. 5 13. 7 69. 0 29. 9 2. 2 *Monotherapy is reported; N=1323 for pre-treatment and N=1275 for no pre-treatment **75 -mg dose allowed per the protocol Montalescot et al. NEJM 2013; epub Sept 1 (N =1996) 98. 0 65. 5 30. 6 3. 3 55. 8 84. 3 89. 5 12. 0 71. 8 27. 3 2. 2
 Efficacy Results
Efficacy Results
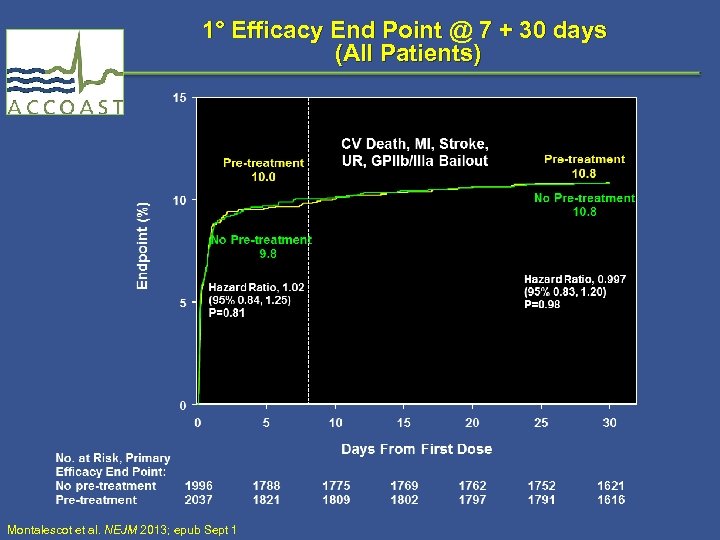 1° Efficacy End Point @ 7 + 30 days (All Patients) Montalescot et al. NEJM 2013; epub Sept 1
1° Efficacy End Point @ 7 + 30 days (All Patients) Montalescot et al. NEJM 2013; epub Sept 1
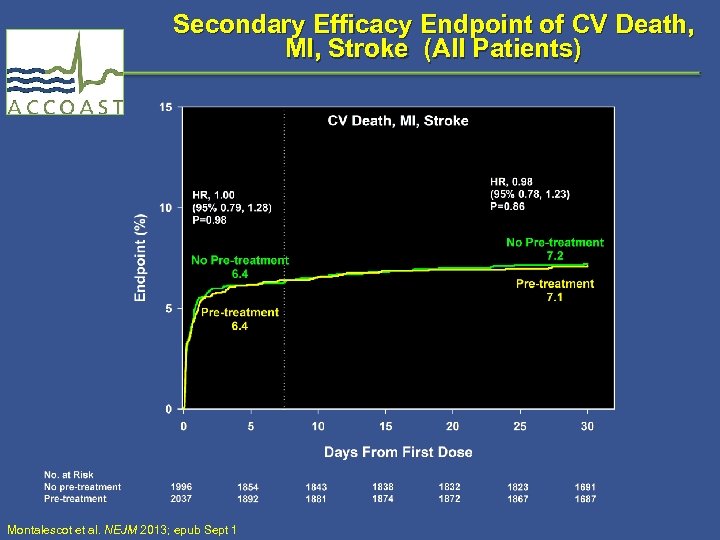 Secondary Efficacy Endpoint of CV Death, MI, Stroke (All Patients) Montalescot et al. NEJM 2013; epub Sept 1
Secondary Efficacy Endpoint of CV Death, MI, Stroke (All Patients) Montalescot et al. NEJM 2013; epub Sept 1
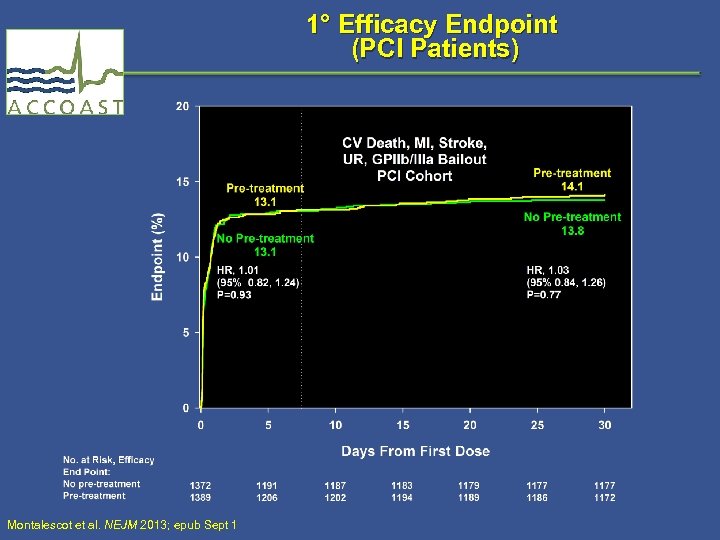 1° Efficacy Endpoint (PCI Patients) Montalescot et al. NEJM 2013; epub Sept 1
1° Efficacy Endpoint (PCI Patients) Montalescot et al. NEJM 2013; epub Sept 1
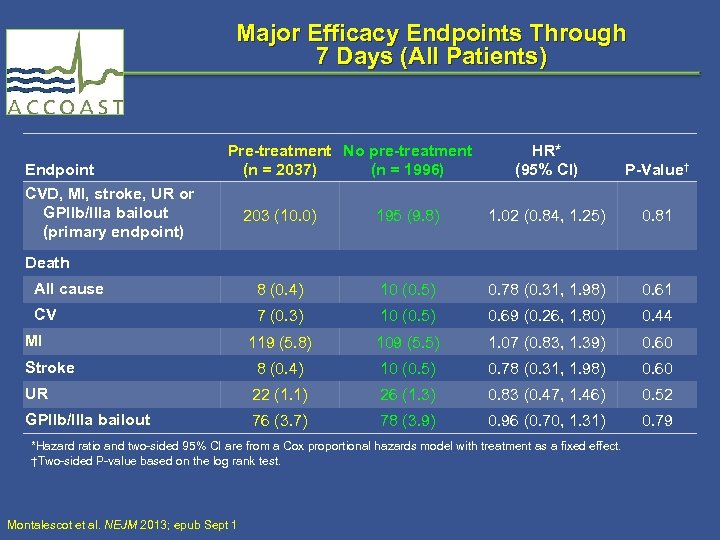 Major Efficacy Endpoints Through 7 Days (All Patients) Endpoint CVD, MI, stroke, UR or GPIIb/IIIa bailout (primary endpoint) Pre-treatment No pre-treatment (n = 2037) (n = 1996) HR* (95% CI) P-Value† 203 (10. 0) 195 (9. 8) 1. 02 (0. 84, 1. 25) 0. 81 All cause 8 (0. 4) 10 (0. 5) 0. 78 (0. 31, 1. 98) 0. 61 CV 7 (0. 3) 10 (0. 5) 0. 69 (0. 26, 1. 80) 0. 44 119 (5. 8) 109 (5. 5) 1. 07 (0. 83, 1. 39) 0. 60 Stroke 8 (0. 4) 10 (0. 5) 0. 78 (0. 31, 1. 98) 0. 60 UR 22 (1. 1) 26 (1. 3) 0. 83 (0. 47, 1. 46) 0. 52 GPIIb/IIIa bailout 76 (3. 7) 78 (3. 9) 0. 96 (0. 70, 1. 31) 0. 79 Death MI *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Montalescot et al. NEJM 2013; epub Sept 1
Major Efficacy Endpoints Through 7 Days (All Patients) Endpoint CVD, MI, stroke, UR or GPIIb/IIIa bailout (primary endpoint) Pre-treatment No pre-treatment (n = 2037) (n = 1996) HR* (95% CI) P-Value† 203 (10. 0) 195 (9. 8) 1. 02 (0. 84, 1. 25) 0. 81 All cause 8 (0. 4) 10 (0. 5) 0. 78 (0. 31, 1. 98) 0. 61 CV 7 (0. 3) 10 (0. 5) 0. 69 (0. 26, 1. 80) 0. 44 119 (5. 8) 109 (5. 5) 1. 07 (0. 83, 1. 39) 0. 60 Stroke 8 (0. 4) 10 (0. 5) 0. 78 (0. 31, 1. 98) 0. 60 UR 22 (1. 1) 26 (1. 3) 0. 83 (0. 47, 1. 46) 0. 52 GPIIb/IIIa bailout 76 (3. 7) 78 (3. 9) 0. 96 (0. 70, 1. 31) 0. 79 Death MI *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Montalescot et al. NEJM 2013; epub Sept 1
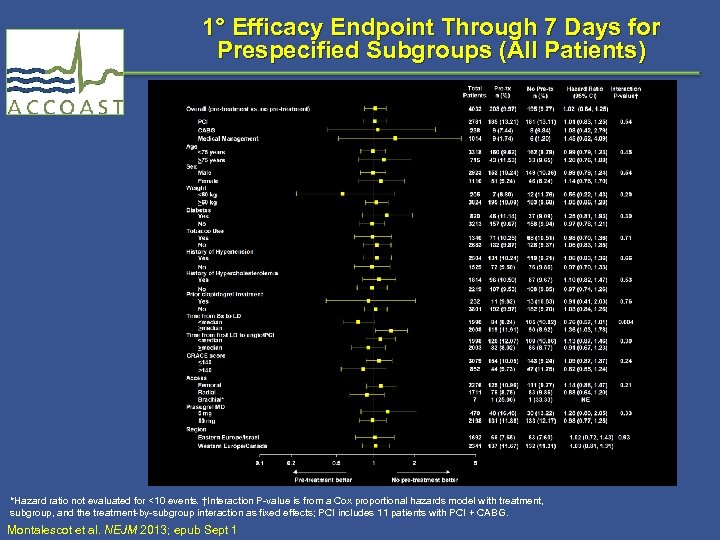 1° Efficacy Endpoint Through 7 Days for Prespecified Subgroups (All Patients) *Hazard ratio not evaluated for <10 events. †Interaction P-value is from a Cox proportional hazards model with treatment, subgroup, and the treatment-by-subgroup interaction as fixed effects; PCI includes 11 patients with PCI + CABG. Montalescot et al. NEJM 2013; epub Sept 1
1° Efficacy Endpoint Through 7 Days for Prespecified Subgroups (All Patients) *Hazard ratio not evaluated for <10 events. †Interaction P-value is from a Cox proportional hazards model with treatment, subgroup, and the treatment-by-subgroup interaction as fixed effects; PCI includes 11 patients with PCI + CABG. Montalescot et al. NEJM 2013; epub Sept 1
 Safety Results
Safety Results
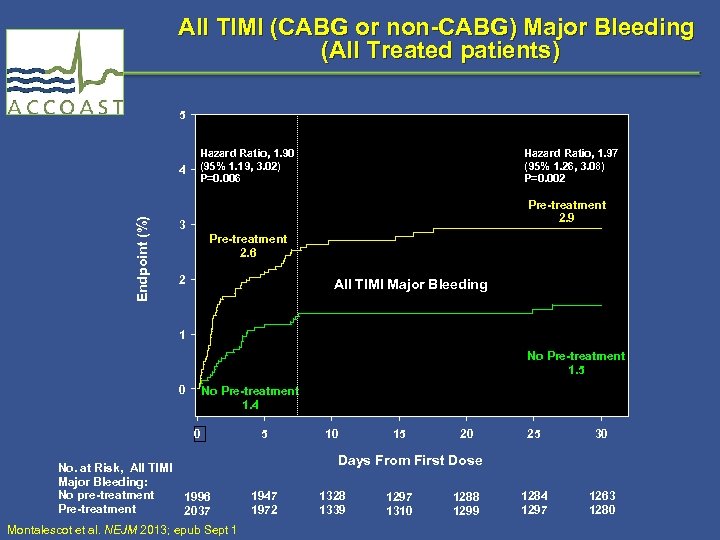 All TIMI (CABG or non-CABG) Major Bleeding (All Treated patients) 5 Endpoint (%) 4 Hazard Ratio, 1. 90 (95% 1. 19, 3. 02) P=0. 006 Hazard Ratio, 1. 97 (95% 1. 26, 3. 08) P=0. 002 Pre-treatment 2. 9 3 Pre-treatment 2. 6 2 All TIMI Major Bleeding 1 No Pre-treatment 1. 5 0 No Pre-treatment 1. 4 0 No. at Risk, All TIMI Major Bleeding: No pre-treatment 1996 Pre-treatment 2037 Montalescot et al. NEJM 2013; epub Sept 1 5 10 15 20 25 30 1284 1297 1263 1280 Days From First Dose 1947 1972 1328 1339 1297 1310 1288 1299
All TIMI (CABG or non-CABG) Major Bleeding (All Treated patients) 5 Endpoint (%) 4 Hazard Ratio, 1. 90 (95% 1. 19, 3. 02) P=0. 006 Hazard Ratio, 1. 97 (95% 1. 26, 3. 08) P=0. 002 Pre-treatment 2. 9 3 Pre-treatment 2. 6 2 All TIMI Major Bleeding 1 No Pre-treatment 1. 5 0 No Pre-treatment 1. 4 0 No. at Risk, All TIMI Major Bleeding: No pre-treatment 1996 Pre-treatment 2037 Montalescot et al. NEJM 2013; epub Sept 1 5 10 15 20 25 30 1284 1297 1263 1280 Days From First Dose 1947 1972 1328 1339 1297 1310 1288 1299
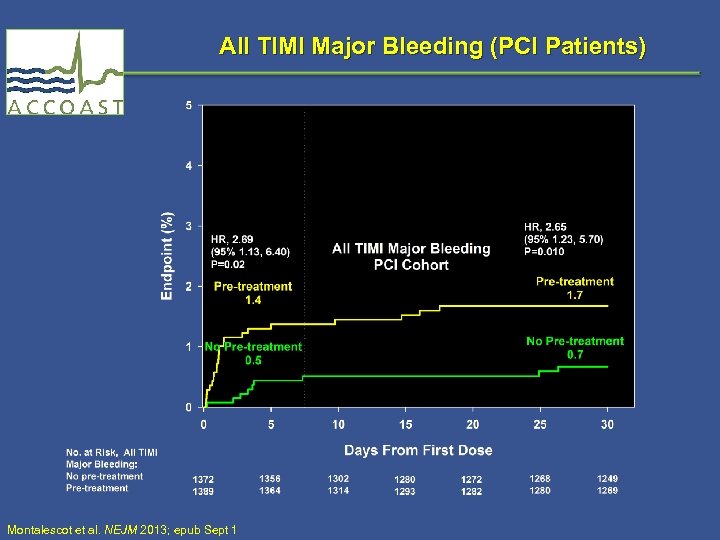 All TIMI Major Bleeding (PCI Patients) Montalescot et al. NEJM 2013; epub Sept 1
All TIMI Major Bleeding (PCI Patients) Montalescot et al. NEJM 2013; epub Sept 1
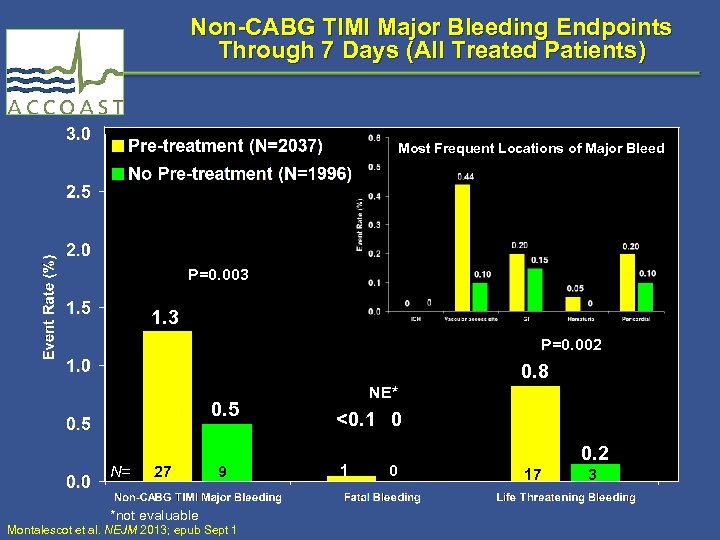 Non-CABG TIMI Major Bleeding Endpoints Through 7 Days (All Treated Patients) Most Frequent Locations of Major Bleed P=0. 003 1. 3 P=0. 002 0. 8 0. 5 N= 27 *not evaluable 9 Montalescot et al. NEJM 2013; epub Sept 1 NE* <0. 1 0 0. 2 17 3
Non-CABG TIMI Major Bleeding Endpoints Through 7 Days (All Treated Patients) Most Frequent Locations of Major Bleed P=0. 003 1. 3 P=0. 002 0. 8 0. 5 N= 27 *not evaluable 9 Montalescot et al. NEJM 2013; epub Sept 1 NE* <0. 1 0 0. 2 17 3
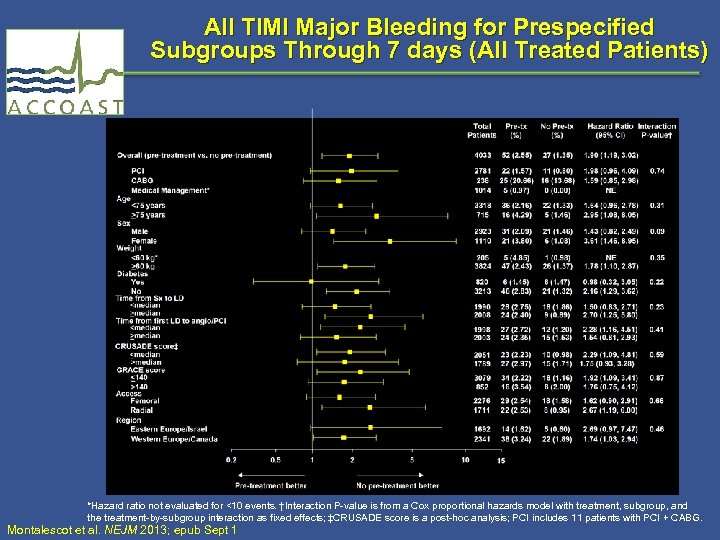 All TIMI Major Bleeding for Prespecified Subgroups Through 7 days (All Treated Patients) *Hazard ratio not evaluated for <10 events. †Interaction P-value is from a Cox proportional hazards model with treatment, subgroup, and the treatment-by-subgroup interaction as fixed effects; ‡CRUSADE score is a post-hoc analysis; PCI includes 11 patients with PCI + CABG. Montalescot et al. NEJM 2013; epub Sept 1
All TIMI Major Bleeding for Prespecified Subgroups Through 7 days (All Treated Patients) *Hazard ratio not evaluated for <10 events. †Interaction P-value is from a Cox proportional hazards model with treatment, subgroup, and the treatment-by-subgroup interaction as fixed effects; ‡CRUSADE score is a post-hoc analysis; PCI includes 11 patients with PCI + CABG. Montalescot et al. NEJM 2013; epub Sept 1
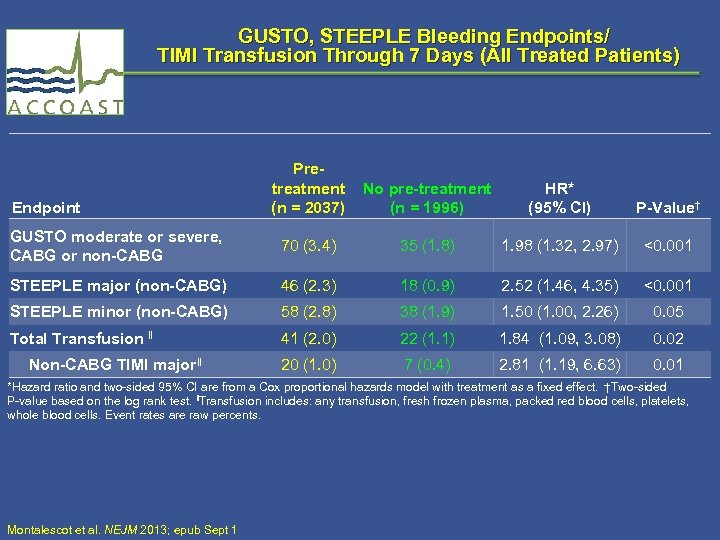 GUSTO, STEEPLE Bleeding Endpoints/ TIMI Transfusion Through 7 Days (All Treated Patients) Pretreatment (n = 2037) No pre-treatment (n = 1996) HR* (95% CI) P-Value† GUSTO moderate or severe, CABG or non-CABG 70 (3. 4) 35 (1. 8) 1. 98 (1. 32, 2. 97) <0. 001 STEEPLE major (non-CABG) 46 (2. 3) 18 (0. 9) 2. 52 (1. 46, 4. 35) <0. 001 STEEPLE minor (non-CABG) 58 (2. 8) 38 (1. 9) 1. 50 (1. 00, 2. 26) 0. 05 Total Transfusion ‖ 41 (2. 0) 22 (1. 1) 1. 84 (1. 09, 3. 08) 0. 02 20 (1. 0) 7 (0. 4) 2. 81 (1. 19, 6. 63) 0. 01 Endpoint Non-CABG TIMI major‖ *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. ‖Transfusion includes: any transfusion, fresh frozen plasma, packed red blood cells, platelets, whole blood cells. Event rates are raw percents. Montalescot et al. NEJM 2013; epub Sept 1
GUSTO, STEEPLE Bleeding Endpoints/ TIMI Transfusion Through 7 Days (All Treated Patients) Pretreatment (n = 2037) No pre-treatment (n = 1996) HR* (95% CI) P-Value† GUSTO moderate or severe, CABG or non-CABG 70 (3. 4) 35 (1. 8) 1. 98 (1. 32, 2. 97) <0. 001 STEEPLE major (non-CABG) 46 (2. 3) 18 (0. 9) 2. 52 (1. 46, 4. 35) <0. 001 STEEPLE minor (non-CABG) 58 (2. 8) 38 (1. 9) 1. 50 (1. 00, 2. 26) 0. 05 Total Transfusion ‖ 41 (2. 0) 22 (1. 1) 1. 84 (1. 09, 3. 08) 0. 02 20 (1. 0) 7 (0. 4) 2. 81 (1. 19, 6. 63) 0. 01 Endpoint Non-CABG TIMI major‖ *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. ‖Transfusion includes: any transfusion, fresh frozen plasma, packed red blood cells, platelets, whole blood cells. Event rates are raw percents. Montalescot et al. NEJM 2013; epub Sept 1
 Overall Conclusions ¨ In NSTEMI patients managed invasively within 48 hours of randomization, pre-treatment with prasugrel does not reduce major ischemic events up to 30 days and increases major bleeding complications. ¨ The efficacy and safety results are consistent among patients undergoing PCI ¨ No subgroup appears to have a favorable risk/benefit ratio with pre-treatment.
Overall Conclusions ¨ In NSTEMI patients managed invasively within 48 hours of randomization, pre-treatment with prasugrel does not reduce major ischemic events up to 30 days and increases major bleeding complications. ¨ The efficacy and safety results are consistent among patients undergoing PCI ¨ No subgroup appears to have a favorable risk/benefit ratio with pre-treatment.
 Considerations
Considerations
 Overall Conclusions (L. Oltrona) ¨ in intermediate risk NSTEMI patients ¨ early invasively managed (median 4. 3 hrs post randomisation) ¨ pre-treatment with 30 mg of prasugrel (+ 30 mg at the time of PCI) does not reduce major ischemic events up to 30 days but increases major bleeding complications. ¨ ( The efficacy and safety results are consistent among patients undergoing PCI. No subgroup appears to have a favorable risk/benefit ratio with pre-treatment) ¨ Drug failure or strategy failure ?
Overall Conclusions (L. Oltrona) ¨ in intermediate risk NSTEMI patients ¨ early invasively managed (median 4. 3 hrs post randomisation) ¨ pre-treatment with 30 mg of prasugrel (+ 30 mg at the time of PCI) does not reduce major ischemic events up to 30 days but increases major bleeding complications. ¨ ( The efficacy and safety results are consistent among patients undergoing PCI. No subgroup appears to have a favorable risk/benefit ratio with pre-treatment) ¨ Drug failure or strategy failure ?
 The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
 The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
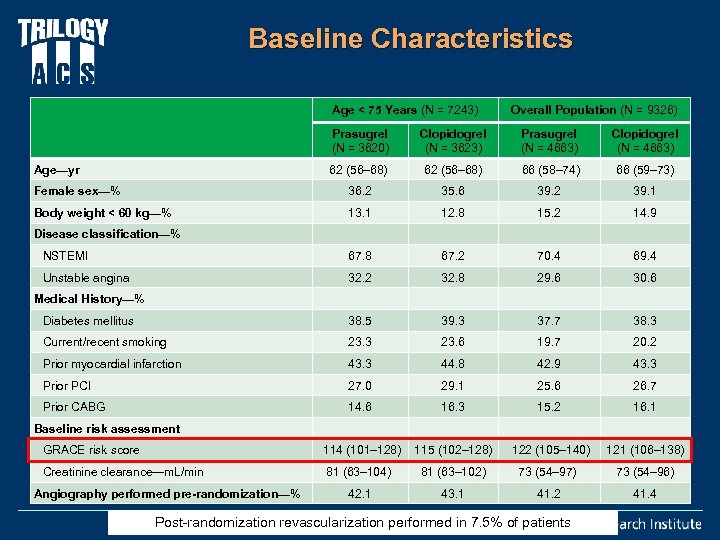 Baseline Characteristics Age < 75 Years (N = 7243) Overall Population (N = 9326) Prasugrel (N = 3620) Clopidogrel (N = 3623) Prasugrel (N = 4663) Clopidogrel (N = 4663) Age—yr 62 (56– 68) 66 (58– 74) 66 (59– 73) Female sex—% 36. 2 35. 6 39. 2 39. 1 Body weight < 60 kg—% 13. 1 12. 8 15. 2 14. 9 NSTEMI 67. 8 67. 2 70. 4 69. 4 Unstable angina 32. 2 32. 8 29. 6 30. 6 Medical History—% Diabetes mellitus 38. 5 39. 3 37. 7 38. 3 Current/recent smoking 23. 3 23. 6 19. 7 20. 2 Prior myocardial infarction 43. 3 44. 8 42. 9 43. 3 Prior PCI 27. 0 29. 1 25. 6 26. 7 Prior CABG 14. 6 16. 3 15. 2 16. 1 114 (101– 128) 115 (102– 128) 122 (105– 140) 121 (106– 138) 81 (63– 104) 81 (63– 102) 73 (54– 97) 73 (54– 96) 42. 1 43. 1 41. 2 41. 4 Disease classification—% Baseline risk assessment GRACE risk score Creatinine clearance—m. L/min Angiography performed pre-randomization—% Post-randomization revascularization performed in 7. 5% of patients
Baseline Characteristics Age < 75 Years (N = 7243) Overall Population (N = 9326) Prasugrel (N = 3620) Clopidogrel (N = 3623) Prasugrel (N = 4663) Clopidogrel (N = 4663) Age—yr 62 (56– 68) 66 (58– 74) 66 (59– 73) Female sex—% 36. 2 35. 6 39. 2 39. 1 Body weight < 60 kg—% 13. 1 12. 8 15. 2 14. 9 NSTEMI 67. 8 67. 2 70. 4 69. 4 Unstable angina 32. 2 32. 8 29. 6 30. 6 Medical History—% Diabetes mellitus 38. 5 39. 3 37. 7 38. 3 Current/recent smoking 23. 3 23. 6 19. 7 20. 2 Prior myocardial infarction 43. 3 44. 8 42. 9 43. 3 Prior PCI 27. 0 29. 1 25. 6 26. 7 Prior CABG 14. 6 16. 3 15. 2 16. 1 114 (101– 128) 115 (102– 128) 122 (105– 140) 121 (106– 138) 81 (63– 104) 81 (63– 102) 73 (54– 97) 73 (54– 96) 42. 1 43. 1 41. 2 41. 4 Disease classification—% Baseline risk assessment GRACE risk score Creatinine clearance—m. L/min Angiography performed pre-randomization—% Post-randomization revascularization performed in 7. 5% of patients
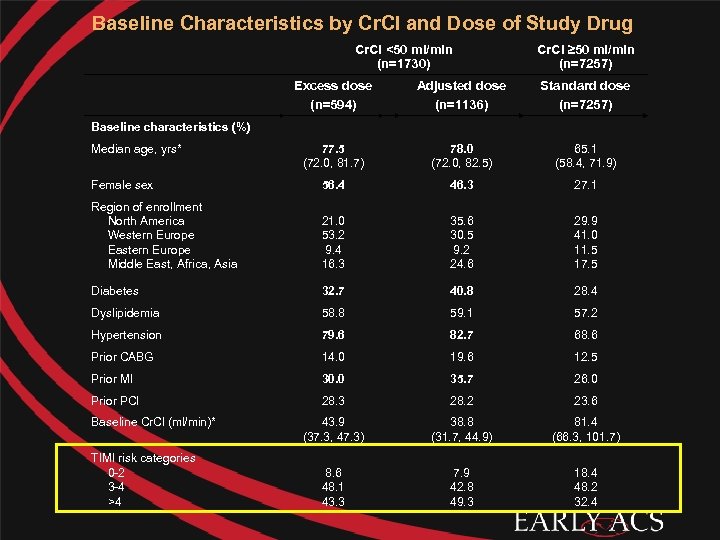 Baseline Characteristics by Cr. Cl and Dose of Study Drug Cr. Cl <50 ml/min (n=1730) Cr. Cl ≥ 50 ml/min (n=7257) Excess dose (n=594) Adjusted dose (n=1136) Standard dose (n=7257) 77. 5 (72. 0, 81. 7) 78. 0 (72. 0, 82. 5) 65. 1 (58. 4, 71. 9) Female sex 56. 4 46. 3 27. 1 Region of enrollment North America Western Europe Eastern Europe Middle East, Africa, Asia 21. 0 53. 2 9. 4 16. 3 35. 6 30. 5 9. 2 24. 6 29. 9 41. 0 11. 5 17. 5 Diabetes 32. 7 40. 8 28. 4 Dyslipidemia 58. 8 59. 1 57. 2 Hypertension 79. 6 82. 7 68. 6 Prior CABG 14. 0 19. 6 12. 5 Prior MI 30. 0 35. 7 26. 0 Prior PCI 28. 3 28. 2 23. 6 43. 9 (37. 3, 47. 3) 38. 8 (31. 7, 44. 9) 81. 4 (66. 3, 101. 7) 8. 6 48. 1 43. 3 7. 9 42. 8 49. 3 18. 4 48. 2 32. 4 Baseline characteristics (%) Median age, yrs* Baseline Cr. Cl (ml/min)* TIMI risk categories 0 -2 3 -4 >4
Baseline Characteristics by Cr. Cl and Dose of Study Drug Cr. Cl <50 ml/min (n=1730) Cr. Cl ≥ 50 ml/min (n=7257) Excess dose (n=594) Adjusted dose (n=1136) Standard dose (n=7257) 77. 5 (72. 0, 81. 7) 78. 0 (72. 0, 82. 5) 65. 1 (58. 4, 71. 9) Female sex 56. 4 46. 3 27. 1 Region of enrollment North America Western Europe Eastern Europe Middle East, Africa, Asia 21. 0 53. 2 9. 4 16. 3 35. 6 30. 5 9. 2 24. 6 29. 9 41. 0 11. 5 17. 5 Diabetes 32. 7 40. 8 28. 4 Dyslipidemia 58. 8 59. 1 57. 2 Hypertension 79. 6 82. 7 68. 6 Prior CABG 14. 0 19. 6 12. 5 Prior MI 30. 0 35. 7 26. 0 Prior PCI 28. 3 28. 2 23. 6 43. 9 (37. 3, 47. 3) 38. 8 (31. 7, 44. 9) 81. 4 (66. 3, 101. 7) 8. 6 48. 1 43. 3 7. 9 42. 8 49. 3 18. 4 48. 2 32. 4 Baseline characteristics (%) Median age, yrs* Baseline Cr. Cl (ml/min)* TIMI risk categories 0 -2 3 -4 >4
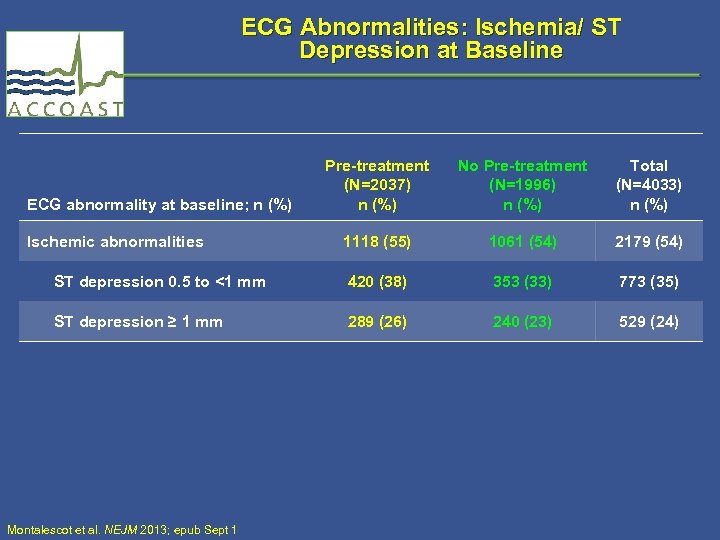 ECG Abnormalities: Ischemia/ ST Depression at Baseline Pre-treatment (N=2037) n (%) No Pre-treatment (N=1996) n (%) Total (N=4033) n (%) 1118 (55) 1061 (54) 2179 (54) ST depression 0. 5 to <1 mm 420 (38) 353 (33) 773 (35) ST depression ≥ 1 mm 289 (26) 240 (23) 529 (24) ECG abnormality at baseline; n (%) Ischemic abnormalities Montalescot et al. NEJM 2013; epub Sept 1
ECG Abnormalities: Ischemia/ ST Depression at Baseline Pre-treatment (N=2037) n (%) No Pre-treatment (N=1996) n (%) Total (N=4033) n (%) 1118 (55) 1061 (54) 2179 (54) ST depression 0. 5 to <1 mm 420 (38) 353 (33) 773 (35) ST depression ≥ 1 mm 289 (26) 240 (23) 529 (24) ECG abnormality at baseline; n (%) Ischemic abnormalities Montalescot et al. NEJM 2013; epub Sept 1
 Montalescot et al. Am Heart J 2011; 161 : 650 -656
Montalescot et al. Am Heart J 2011; 161 : 650 -656
 The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
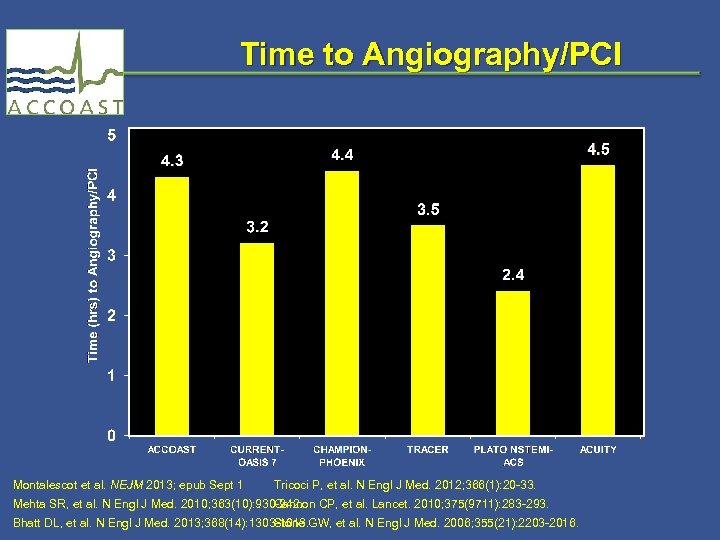 Time to Angiography/PCI Montalescot et al. NEJM 2013; epub Sept 1 Tricoci P, et al. N Engl J Med. 2012; 366(1): 20 -33. Mehta SR, et al. N Engl J Med. 2010; 363(10): 930 -942. Cannon CP, et al. Lancet. 2010; 375(9711): 283 -293. Bhatt DL, et al. N Engl J Med. 2013; 368(14): 1303 -1013. Stone GW, et al. N Engl J Med. 2006; 355(21): 2203 -2016.
Time to Angiography/PCI Montalescot et al. NEJM 2013; epub Sept 1 Tricoci P, et al. N Engl J Med. 2012; 366(1): 20 -33. Mehta SR, et al. N Engl J Med. 2010; 363(10): 930 -942. Cannon CP, et al. Lancet. 2010; 375(9711): 283 -293. Bhatt DL, et al. N Engl J Med. 2013; 368(14): 1303 -1013. Stone GW, et al. N Engl J Med. 2006; 355(21): 2203 -2016.
 No differences between quartiles of time to PCI (highest quartile > 15 hours)
No differences between quartiles of time to PCI (highest quartile > 15 hours)
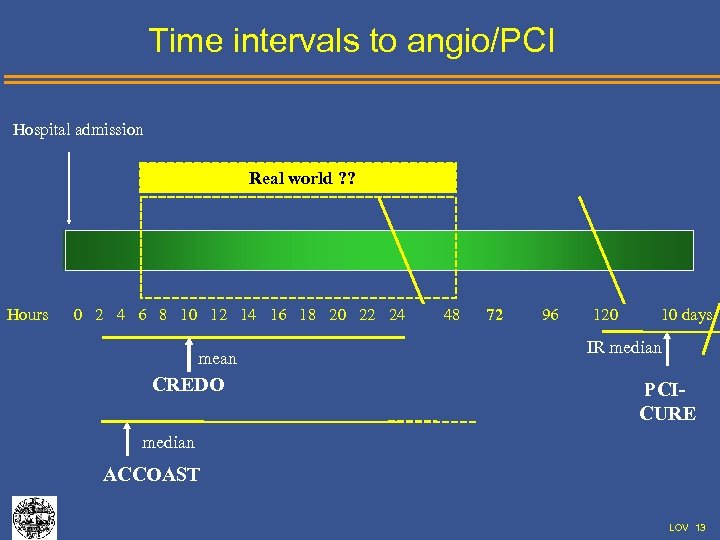 Time intervals to angio/PCI Hospital admission Real world ? ? Hours 0 2 4 6 8 10 12 14 16 18 20 22 24 mean CREDO 48 72 96 120 10 days IR median PCICURE median ACCOAST LOV 13
Time intervals to angio/PCI Hospital admission Real world ? ? Hours 0 2 4 6 8 10 12 14 16 18 20 22 24 mean CREDO 48 72 96 120 10 days IR median PCICURE median ACCOAST LOV 13
 The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
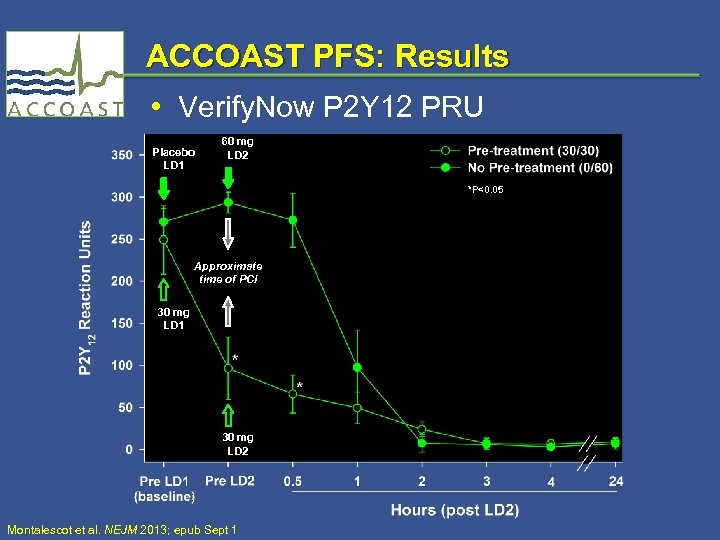 ACCOAST PFS: Results • Verify. Now P 2 Y 12 PRU Placebo LD 1 60 mg LD 2 *P<0. 05 Approximate time of PCI 30 mg LD 1 30 mg LD 2 Montalescot et al. NEJM 2013; epub Sept 1
ACCOAST PFS: Results • Verify. Now P 2 Y 12 PRU Placebo LD 1 60 mg LD 2 *P<0. 05 Approximate time of PCI 30 mg LD 1 30 mg LD 2 Montalescot et al. NEJM 2013; epub Sept 1
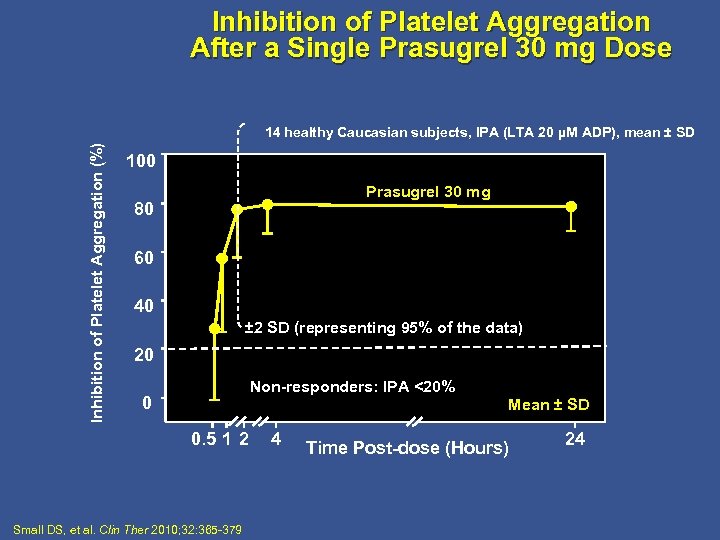 Inhibition of Platelet Aggregation After a Single Prasugrel 30 mg Dose Inhibition of Platelet Aggregation (%) 14 healthy Caucasian subjects, IPA (LTA 20 µM ADP), mean ± SD 100 Prasugrel 30 mg 80 60 40 ± 2 SD (representing 95% of the data) 20 Non-responders: IPA <20% 0 Mean ± SD 0. 5 1 2 Small DS, et al. Clin Ther 2010; 32: 365 -379 4 Time Post-dose (Hours) 24
Inhibition of Platelet Aggregation After a Single Prasugrel 30 mg Dose Inhibition of Platelet Aggregation (%) 14 healthy Caucasian subjects, IPA (LTA 20 µM ADP), mean ± SD 100 Prasugrel 30 mg 80 60 40 ± 2 SD (representing 95% of the data) 20 Non-responders: IPA <20% 0 Mean ± SD 0. 5 1 2 Small DS, et al. Clin Ther 2010; 32: 365 -379 4 Time Post-dose (Hours) 24
 The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
The most frequent questions 1. Risk-level of population 2. Timing of PCI 3. Drug dosage (pharmacodynamic considerations) 4. Clopidogrel pretreated 5. Medically managed pts 6. Femoral vs radial approach
 Montalescot et al. Am Heart J 2011; 161 : 650 -656
Montalescot et al. Am Heart J 2011; 161 : 650 -656
 Backups
Backups
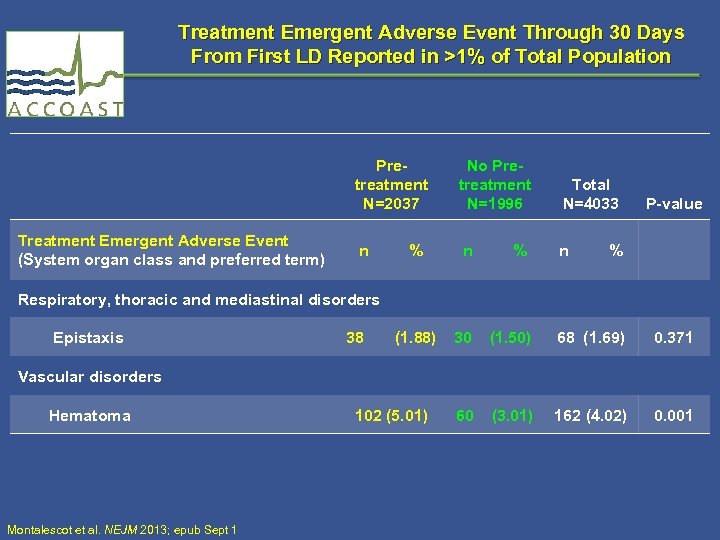 Treatment Emergent Adverse Event Through 30 Days From First LD Reported in >1% of Total Population Pretreatment N=2037 Treatment Emergent Adverse Event (System organ class and preferred term) No Pretreatment N=1996 Total N=4033 n % 38 (1. 88) 30 (1. 50) 68 (1. 69) 0. 371 102 (5. 01) 60 (3. 01) 162 (4. 02) 0. 001 P-value Respiratory, thoracic and mediastinal disorders Epistaxis Vascular disorders Hematoma Montalescot et al. NEJM 2013; epub Sept 1
Treatment Emergent Adverse Event Through 30 Days From First LD Reported in >1% of Total Population Pretreatment N=2037 Treatment Emergent Adverse Event (System organ class and preferred term) No Pretreatment N=1996 Total N=4033 n % 38 (1. 88) 30 (1. 50) 68 (1. 69) 0. 371 102 (5. 01) 60 (3. 01) 162 (4. 02) 0. 001 P-value Respiratory, thoracic and mediastinal disorders Epistaxis Vascular disorders Hematoma Montalescot et al. NEJM 2013; epub Sept 1
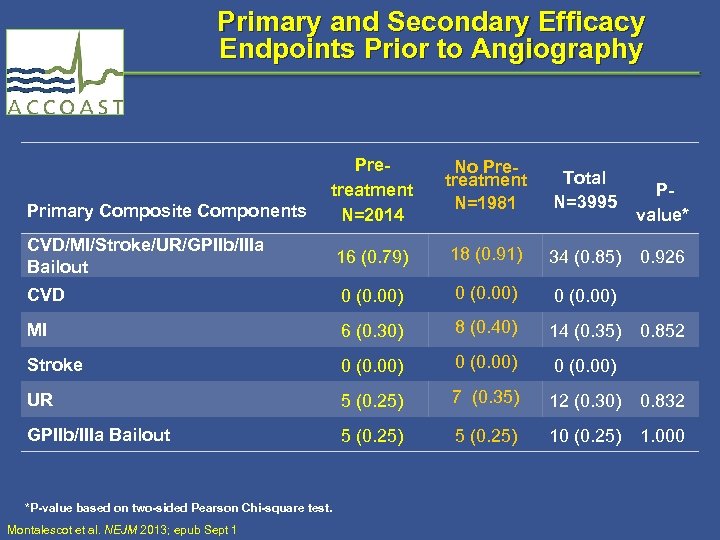 Primary and Secondary Efficacy Endpoints Prior to Angiography Primary Composite Components Pretreatment N=2014 No Pretreatment N=1981 Total N=3995 Pvalue* CVD/MI/Stroke/UR/GPIIb/IIIa Bailout 16 (0. 79) 18 (0. 91) 34 (0. 85) 0. 926 CVD 0 (0. 00) MI 6 (0. 30) 8 (0. 40) 14 (0. 35) 0. 852 Stroke 0 (0. 00) UR 5 (0. 25) 7 (0. 35) 12 (0. 30) 0. 832 GPIIb/IIIa Bailout 5 (0. 25) 10 (0. 25) 1. 000 *P-value based on two-sided Pearson Chi-square test. Montalescot et al. NEJM 2013; epub Sept 1
Primary and Secondary Efficacy Endpoints Prior to Angiography Primary Composite Components Pretreatment N=2014 No Pretreatment N=1981 Total N=3995 Pvalue* CVD/MI/Stroke/UR/GPIIb/IIIa Bailout 16 (0. 79) 18 (0. 91) 34 (0. 85) 0. 926 CVD 0 (0. 00) MI 6 (0. 30) 8 (0. 40) 14 (0. 35) 0. 852 Stroke 0 (0. 00) UR 5 (0. 25) 7 (0. 35) 12 (0. 30) 0. 832 GPIIb/IIIa Bailout 5 (0. 25) 10 (0. 25) 1. 000 *P-value based on two-sided Pearson Chi-square test. Montalescot et al. NEJM 2013; epub Sept 1
 TIMI Bleeding Criteria Non-CABG Related Bleeding: 1. Major • Any intracranial bleeding (excluding microhemorrhages <10 mm evident only on gradient-echo MRI) • Clinically overt signs of hemorrhage associated with a drop in hemoglobin of ≥ 5 g/d. L or a ≥ 15% absolute decrease in haematocrit • Fatal bleeding (bleeding that directly results in death within 7 d) 2. Minor • Clinically overt (including imaging), resulting in hemoglobin drop of 3 to <5 g/d. L or ≥ 10% decrease in haematocrit • No observed blood loss: ≥ 4 g/d. L decrease in the haemoglobin concentration or ≥ 12% decrease in haematocrit • Any overt sign of hemorrhage that meets one of the following criteria and does not meet criteria for a major or minor bleeding event, as defined above • Requiring intervention (medical practitioner-guided medical or surgical treatment to stop or treat bleeding, including temporarily or permanently discontinuing or changing the dose of a medication or study drug) • Leading to or prolonging hospitalization • Prompting evaluation (leading to an unscheduled visit to a healthcare professional and diagnostic testing, either laboratory or imaging) 3. Minimal • Any overt bleeding event that does not meet the criteria above • Any clinically overt sign of haemorrhage (including imaging) associated with a <3 g/d. L decrease in haemoglobin concentration or <9% decrease in haematocrit
TIMI Bleeding Criteria Non-CABG Related Bleeding: 1. Major • Any intracranial bleeding (excluding microhemorrhages <10 mm evident only on gradient-echo MRI) • Clinically overt signs of hemorrhage associated with a drop in hemoglobin of ≥ 5 g/d. L or a ≥ 15% absolute decrease in haematocrit • Fatal bleeding (bleeding that directly results in death within 7 d) 2. Minor • Clinically overt (including imaging), resulting in hemoglobin drop of 3 to <5 g/d. L or ≥ 10% decrease in haematocrit • No observed blood loss: ≥ 4 g/d. L decrease in the haemoglobin concentration or ≥ 12% decrease in haematocrit • Any overt sign of hemorrhage that meets one of the following criteria and does not meet criteria for a major or minor bleeding event, as defined above • Requiring intervention (medical practitioner-guided medical or surgical treatment to stop or treat bleeding, including temporarily or permanently discontinuing or changing the dose of a medication or study drug) • Leading to or prolonging hospitalization • Prompting evaluation (leading to an unscheduled visit to a healthcare professional and diagnostic testing, either laboratory or imaging) 3. Minimal • Any overt bleeding event that does not meet the criteria above • Any clinically overt sign of haemorrhage (including imaging) associated with a <3 g/d. L decrease in haemoglobin concentration or <9% decrease in haematocrit
 • TIMI Bleeding Criteria • Bleeding in the Setting of CABG: • Fatal bleeding (bleeding that directly results in death) • Perioperative intracranial bleeding • Reoperation after closure of the sternotomy incision for the purpose of controlling bleeding • Transfusion of ≥ 5 U PRBCs or whole blood within a 48 -h period; cell saver transfusion will not be counted in calculations of blood products. • Chest tube output >2 L within a 24 -h period
• TIMI Bleeding Criteria • Bleeding in the Setting of CABG: • Fatal bleeding (bleeding that directly results in death) • Perioperative intracranial bleeding • Reoperation after closure of the sternotomy incision for the purpose of controlling bleeding • Transfusion of ≥ 5 U PRBCs or whole blood within a 48 -h period; cell saver transfusion will not be counted in calculations of blood products. • Chest tube output >2 L within a 24 -h period
 Steeple Bleeding Criteria Major Bleeding • Fatal bleeding • Retroperitoneal, intracranial, or intraocular bleeding • Bleeding that causes hemodynamic compromise requiring specific treatment • Bleeding that requires intervention (surgical or endoscopic) or decompression of a closed space to stop or control the event • Clinically overt bleeding, requiring any transfusion of ≥ 1 U PRBC or whole blood • Clinically overt bleeding, causing a decrease in hemoglobin of ≥ 3 g/d. L (or, if hemoglobin level is not available, a decrease in hematocrit of ≥ 10%) Minor • Gross hematuria not associated with trauma (eg, from instrumentation) • Epistaxis that is prolonged, is repeated, or requires plugging or intervention • Gastrointestinal hemorrhage • Hemoptysis • Subconjunctival hemorrhage • Hematoma >5 cm or leading to prolonged or new hospitalization • Clinically overt bleeding, causing a decrease in hemoglobin of 2 to 3 g/d. L Uncontrolled bleeding requiring protamine sulfate administration
Steeple Bleeding Criteria Major Bleeding • Fatal bleeding • Retroperitoneal, intracranial, or intraocular bleeding • Bleeding that causes hemodynamic compromise requiring specific treatment • Bleeding that requires intervention (surgical or endoscopic) or decompression of a closed space to stop or control the event • Clinically overt bleeding, requiring any transfusion of ≥ 1 U PRBC or whole blood • Clinically overt bleeding, causing a decrease in hemoglobin of ≥ 3 g/d. L (or, if hemoglobin level is not available, a decrease in hematocrit of ≥ 10%) Minor • Gross hematuria not associated with trauma (eg, from instrumentation) • Epistaxis that is prolonged, is repeated, or requires plugging or intervention • Gastrointestinal hemorrhage • Hemoptysis • Subconjunctival hemorrhage • Hematoma >5 cm or leading to prolonged or new hospitalization • Clinically overt bleeding, causing a decrease in hemoglobin of 2 to 3 g/d. L Uncontrolled bleeding requiring protamine sulfate administration
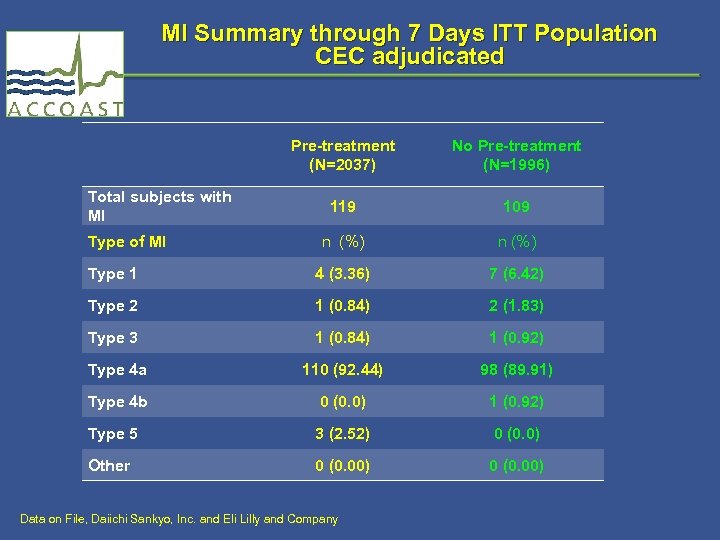 MI Summary through 7 Days ITT Population CEC adjudicated Pre-treatment (N=2037) No Pre-treatment (N=1996) 119 109 n (%) Type 1 4 (3. 36) 7 (6. 42) Type 2 1 (0. 84) 2 (1. 83) Type 3 1 (0. 84) 1 (0. 92) Type 4 a 110 (92. 44) 98 (89. 91) Type 4 b 0 (0. 0) 1 (0. 92) Type 5 3 (2. 52) 0 (0. 0) Other 0 (0. 00) Total subjects with MI Type of MI Data on File, Daiichi Sankyo, Inc. and Eli Lilly and Company
MI Summary through 7 Days ITT Population CEC adjudicated Pre-treatment (N=2037) No Pre-treatment (N=1996) 119 109 n (%) Type 1 4 (3. 36) 7 (6. 42) Type 2 1 (0. 84) 2 (1. 83) Type 3 1 (0. 84) 1 (0. 92) Type 4 a 110 (92. 44) 98 (89. 91) Type 4 b 0 (0. 0) 1 (0. 92) Type 5 3 (2. 52) 0 (0. 0) Other 0 (0. 00) Total subjects with MI Type of MI Data on File, Daiichi Sankyo, Inc. and Eli Lilly and Company
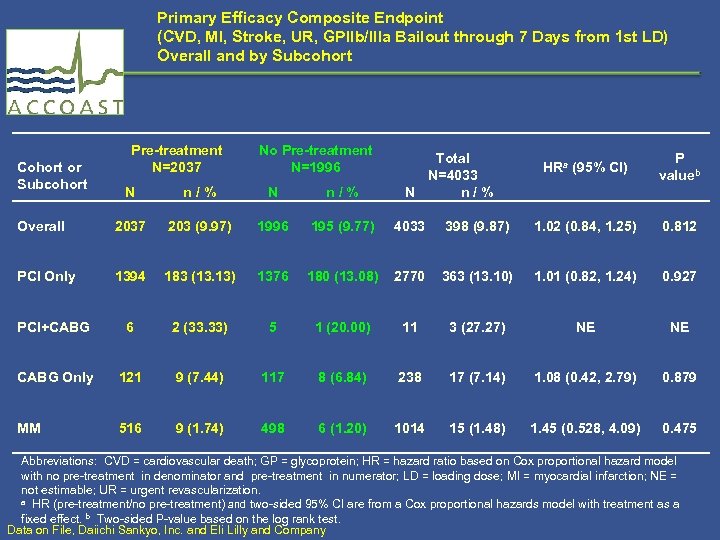 Primary Efficacy Composite Endpoint (CVD, MI, Stroke, UR, GPIIb/IIIa Bailout through 7 Days from 1 st LD) Overall and by Subcohort Cohort or Subcohort Pre-treatment N=2037 No Pre-treatment N=1996 Total N=4033 N n / % HRa (95% CI) P valueb N n / % Overall 2037 203 (9. 97) 1996 195 (9. 77) 4033 398 (9. 87) 1. 02 (0. 84, 1. 25) 0. 812 PCI Only 1394 183 (13. 13) 1376 180 (13. 08) 2770 363 (13. 10) 1. 01 (0. 82, 1. 24) 0. 927 PCI+CABG 6 2 (33. 33) 5 1 (20. 00) 11 3 (27. 27) NE NE CABG Only 121 9 (7. 44) 117 8 (6. 84) 238 17 (7. 14) 1. 08 (0. 42, 2. 79) 0. 879 MM 516 9 (1. 74) 498 6 (1. 20) 1014 15 (1. 48) 1. 45 (0. 528, 4. 09) 0. 475 Abbreviations: CVD = cardiovascular death; GP = glycoprotein; HR = hazard ratio based on Cox proportional hazard model with no pre-treatment in denominator and pre-treatment in numerator; LD = loading dose; MI = myocardial infarction; NE = not estimable; UR = urgent revascularization. a HR (pre-treatment/no pre-treatment) and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. b Two-sided P-value based on the log rank test. Data on File, Daiichi Sankyo, Inc. and Eli Lilly and Company
Primary Efficacy Composite Endpoint (CVD, MI, Stroke, UR, GPIIb/IIIa Bailout through 7 Days from 1 st LD) Overall and by Subcohort Cohort or Subcohort Pre-treatment N=2037 No Pre-treatment N=1996 Total N=4033 N n / % HRa (95% CI) P valueb N n / % Overall 2037 203 (9. 97) 1996 195 (9. 77) 4033 398 (9. 87) 1. 02 (0. 84, 1. 25) 0. 812 PCI Only 1394 183 (13. 13) 1376 180 (13. 08) 2770 363 (13. 10) 1. 01 (0. 82, 1. 24) 0. 927 PCI+CABG 6 2 (33. 33) 5 1 (20. 00) 11 3 (27. 27) NE NE CABG Only 121 9 (7. 44) 117 8 (6. 84) 238 17 (7. 14) 1. 08 (0. 42, 2. 79) 0. 879 MM 516 9 (1. 74) 498 6 (1. 20) 1014 15 (1. 48) 1. 45 (0. 528, 4. 09) 0. 475 Abbreviations: CVD = cardiovascular death; GP = glycoprotein; HR = hazard ratio based on Cox proportional hazard model with no pre-treatment in denominator and pre-treatment in numerator; LD = loading dose; MI = myocardial infarction; NE = not estimable; UR = urgent revascularization. a HR (pre-treatment/no pre-treatment) and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. b Two-sided P-value based on the log rank test. Data on File, Daiichi Sankyo, Inc. and Eli Lilly and Company
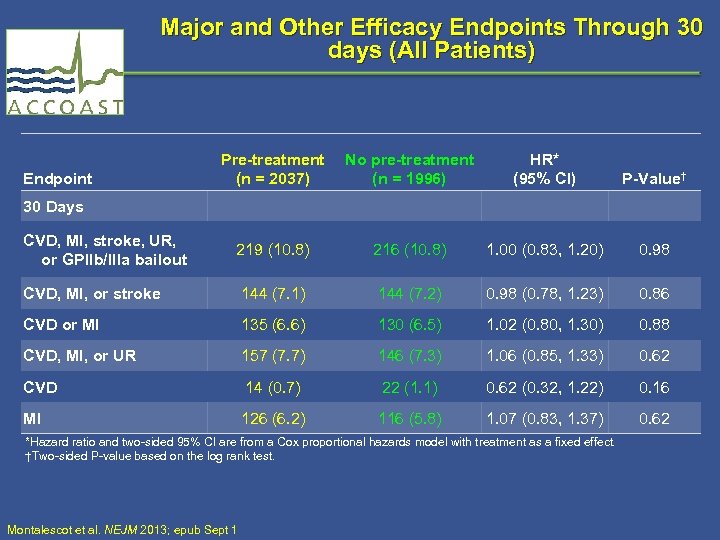 Major and Other Efficacy Endpoints Through 30 days (All Patients) Endpoint Pre-treatment (n = 2037) No pre-treatment (n = 1996) HR* (95% CI) P-Value† 30 Days CVD, MI, stroke, UR, or GPIIb/IIIa bailout 219 (10. 8) 216 (10. 8) 1. 00 (0. 83, 1. 20) 0. 98 CVD, MI, or stroke 144 (7. 1) 144 (7. 2) 0. 98 (0. 78, 1. 23) 0. 86 CVD or MI 135 (6. 6) 130 (6. 5) 1. 02 (0. 80, 1. 30) 0. 88 CVD, MI, or UR 157 (7. 7) 146 (7. 3) 1. 06 (0. 85, 1. 33) 0. 62 CVD 14 (0. 7) 22 (1. 1) 0. 62 (0. 32, 1. 22) 0. 16 MI 126 (6. 2) 116 (5. 8) 1. 07 (0. 83, 1. 37) 0. 62 *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Montalescot et al. NEJM 2013; epub Sept 1
Major and Other Efficacy Endpoints Through 30 days (All Patients) Endpoint Pre-treatment (n = 2037) No pre-treatment (n = 1996) HR* (95% CI) P-Value† 30 Days CVD, MI, stroke, UR, or GPIIb/IIIa bailout 219 (10. 8) 216 (10. 8) 1. 00 (0. 83, 1. 20) 0. 98 CVD, MI, or stroke 144 (7. 1) 144 (7. 2) 0. 98 (0. 78, 1. 23) 0. 86 CVD or MI 135 (6. 6) 130 (6. 5) 1. 02 (0. 80, 1. 30) 0. 88 CVD, MI, or UR 157 (7. 7) 146 (7. 3) 1. 06 (0. 85, 1. 33) 0. 62 CVD 14 (0. 7) 22 (1. 1) 0. 62 (0. 32, 1. 22) 0. 16 MI 126 (6. 2) 116 (5. 8) 1. 07 (0. 83, 1. 37) 0. 62 *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Montalescot et al. NEJM 2013; epub Sept 1
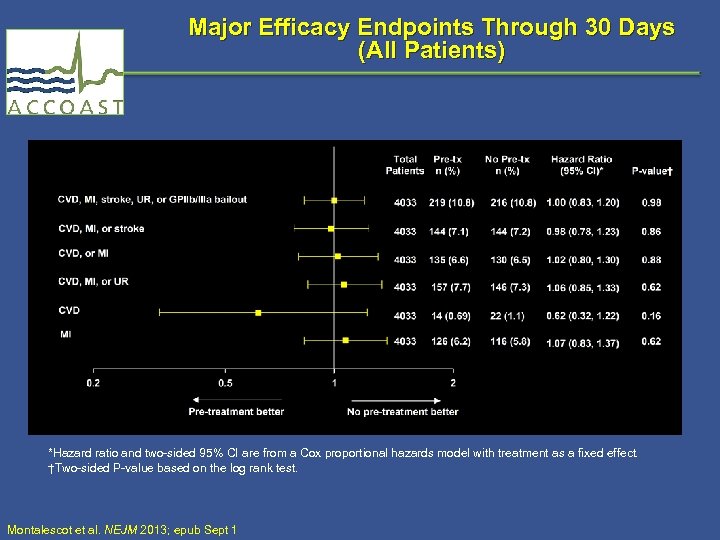 Major Efficacy Endpoints Through 30 Days (All Patients) *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Montalescot et al. NEJM 2013; epub Sept 1
Major Efficacy Endpoints Through 30 Days (All Patients) *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Montalescot et al. NEJM 2013; epub Sept 1
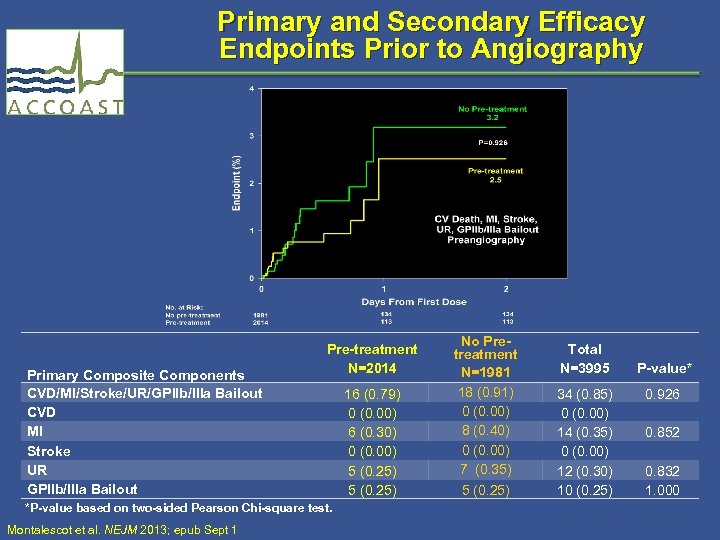 Primary and Secondary Efficacy Endpoints Prior to Angiography Primary Composite Components CVD/MI/Stroke/UR/GPIIb/IIIa Bailout CVD MI Stroke UR GPIIb/IIIa Bailout Pre-treatment N=2014 *P-value based on two-sided Pearson Chi-square test. Montalescot et al. NEJM 2013; epub Sept 1 16 (0. 79) 0 (0. 00) 6 (0. 30) 0 (0. 00) 5 (0. 25) No Pretreatment N=1981 18 (0. 91) 0 (0. 00) 8 (0. 40) 0 (0. 00) 7 (0. 35) 5 (0. 25) Total N=3995 P-value* 34 (0. 85) 0 (0. 00) 14 (0. 35) 0 (0. 00) 12 (0. 30) 10 (0. 25) 0. 926 0. 852 0. 832 1. 000
Primary and Secondary Efficacy Endpoints Prior to Angiography Primary Composite Components CVD/MI/Stroke/UR/GPIIb/IIIa Bailout CVD MI Stroke UR GPIIb/IIIa Bailout Pre-treatment N=2014 *P-value based on two-sided Pearson Chi-square test. Montalescot et al. NEJM 2013; epub Sept 1 16 (0. 79) 0 (0. 00) 6 (0. 30) 0 (0. 00) 5 (0. 25) No Pretreatment N=1981 18 (0. 91) 0 (0. 00) 8 (0. 40) 0 (0. 00) 7 (0. 35) 5 (0. 25) Total N=3995 P-value* 34 (0. 85) 0 (0. 00) 14 (0. 35) 0 (0. 00) 12 (0. 30) 10 (0. 25) 0. 926 0. 852 0. 832 1. 000
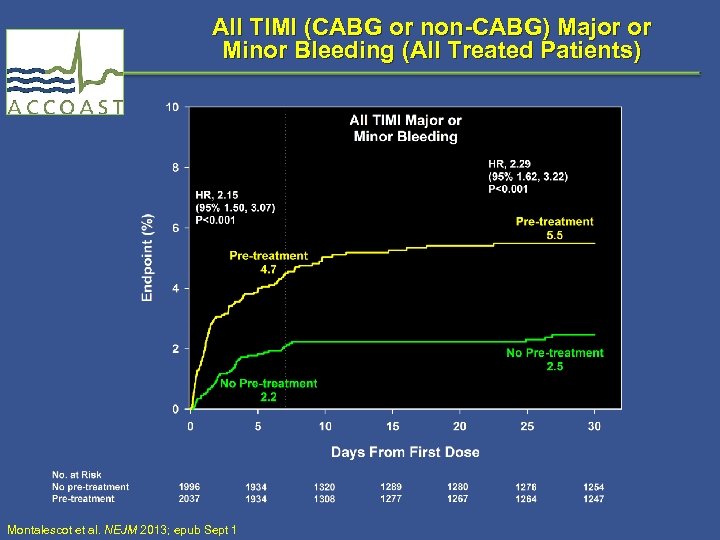 All TIMI (CABG or non-CABG) Major or Minor Bleeding (All Treated Patients) Montalescot et al. NEJM 2013; epub Sept 1
All TIMI (CABG or non-CABG) Major or Minor Bleeding (All Treated Patients) Montalescot et al. NEJM 2013; epub Sept 1
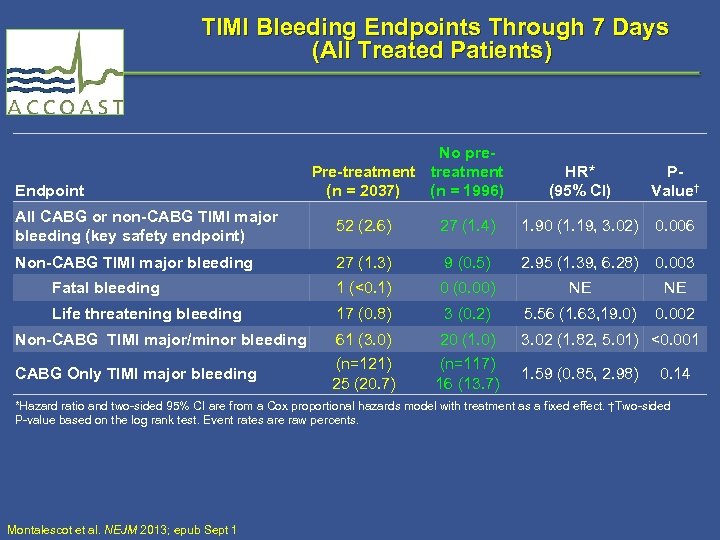 TIMI Bleeding Endpoints Through 7 Days (All Treated Patients) Endpoint No pre. Pre-treatment (n = 2037) (n = 1996) HR* (95% CI) PValue† All CABG or non-CABG TIMI major bleeding (key safety endpoint) 52 (2. 6) 27 (1. 4) 1. 90 (1. 19, 3. 02) 0. 006 Non-CABG TIMI major bleeding 27 (1. 3) 9 (0. 5) 2. 95 (1. 39, 6. 28) 0. 003 Fatal bleeding 1 (<0. 1) 0 (0. 00) NE NE Life threatening bleeding 17 (0. 8) 3 (0. 2) 5. 56 (1. 63, 19. 0) 0. 002 Non-CABG TIMI major/minor bleeding 61 (3. 0) 20 (1. 0) 3. 02 (1. 82, 5. 01) <0. 001 CABG Only TIMI major bleeding (n=121) 25 (20. 7) (n=117) 16 (13. 7) 1. 59 (0. 85, 2. 98) 0. 14 *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Event rates are raw percents. Montalescot et al. NEJM 2013; epub Sept 1
TIMI Bleeding Endpoints Through 7 Days (All Treated Patients) Endpoint No pre. Pre-treatment (n = 2037) (n = 1996) HR* (95% CI) PValue† All CABG or non-CABG TIMI major bleeding (key safety endpoint) 52 (2. 6) 27 (1. 4) 1. 90 (1. 19, 3. 02) 0. 006 Non-CABG TIMI major bleeding 27 (1. 3) 9 (0. 5) 2. 95 (1. 39, 6. 28) 0. 003 Fatal bleeding 1 (<0. 1) 0 (0. 00) NE NE Life threatening bleeding 17 (0. 8) 3 (0. 2) 5. 56 (1. 63, 19. 0) 0. 002 Non-CABG TIMI major/minor bleeding 61 (3. 0) 20 (1. 0) 3. 02 (1. 82, 5. 01) <0. 001 CABG Only TIMI major bleeding (n=121) 25 (20. 7) (n=117) 16 (13. 7) 1. 59 (0. 85, 2. 98) 0. 14 *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided P-value based on the log rank test. Event rates are raw percents. Montalescot et al. NEJM 2013; epub Sept 1
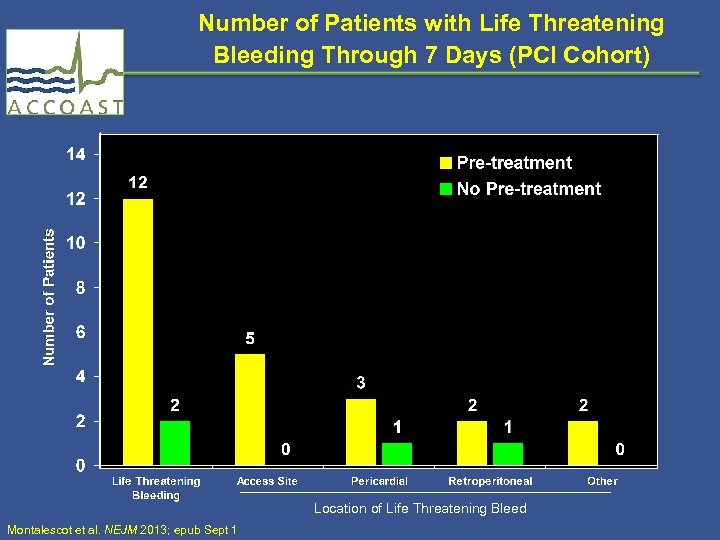 Number of Patients with Life Threatening Bleeding Through 7 Days (PCI Cohort) Location of Life Threatening Bleed Montalescot et al. NEJM 2013; epub Sept 1
Number of Patients with Life Threatening Bleeding Through 7 Days (PCI Cohort) Location of Life Threatening Bleed Montalescot et al. NEJM 2013; epub Sept 1
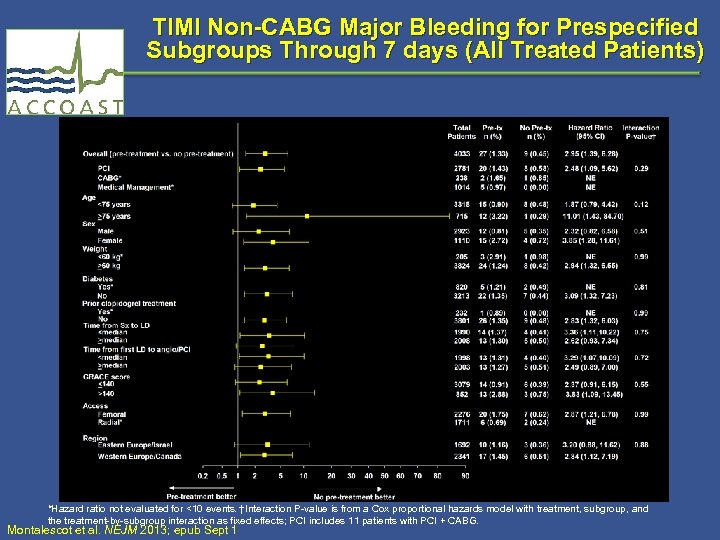 TIMI Non-CABG Major Bleeding for Prespecified Subgroups Through 7 days (All Treated Patients) *Hazard ratio not evaluated for <10 events. †Interaction P-value is from a Cox proportional hazards model with treatment, subgroup, and the treatment-by-subgroup interaction as fixed effects; PCI includes 11 patients with PCI + CABG. Montalescot et al. NEJM 2013; epub Sept 1
TIMI Non-CABG Major Bleeding for Prespecified Subgroups Through 7 days (All Treated Patients) *Hazard ratio not evaluated for <10 events. †Interaction P-value is from a Cox proportional hazards model with treatment, subgroup, and the treatment-by-subgroup interaction as fixed effects; PCI includes 11 patients with PCI + CABG. Montalescot et al. NEJM 2013; epub Sept 1
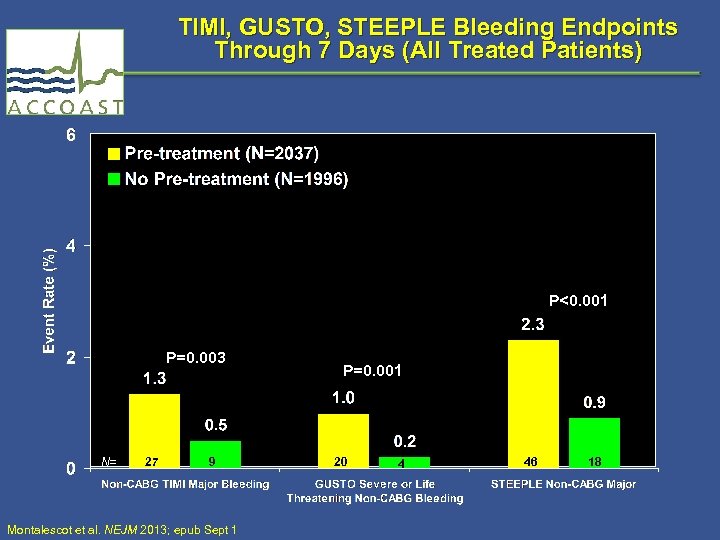 TIMI, GUSTO, STEEPLE Bleeding Endpoints Through 7 Days (All Treated Patients) P<0. 001 P=0. 003 N= 27 9 Montalescot et al. NEJM 2013; epub Sept 1 P=0. 001 20 4 46 18
TIMI, GUSTO, STEEPLE Bleeding Endpoints Through 7 Days (All Treated Patients) P<0. 001 P=0. 003 N= 27 9 Montalescot et al. NEJM 2013; epub Sept 1 P=0. 001 20 4 46 18
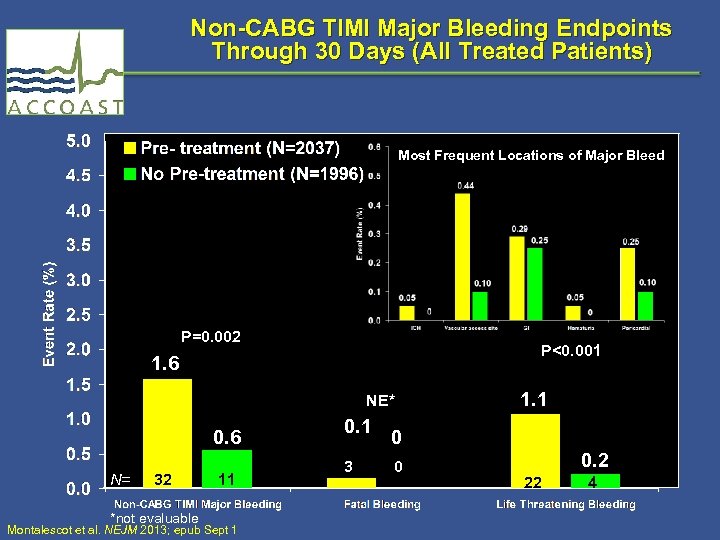 Non-CABG TIMI Major Bleeding Endpoints Through 30 Days (All Treated Patients) Most Frequent Locations of Major Bleed P=0. 002 P<0. 001 1. 6 NE* 0. 6 N= 32 *not evaluable 11 Montalescot et al. NEJM 2013; epub Sept 1 0. 1 3 1. 1 0 0 0. 2 22 4
Non-CABG TIMI Major Bleeding Endpoints Through 30 Days (All Treated Patients) Most Frequent Locations of Major Bleed P=0. 002 P<0. 001 1. 6 NE* 0. 6 N= 32 *not evaluable 11 Montalescot et al. NEJM 2013; epub Sept 1 0. 1 3 1. 1 0 0 0. 2 22 4
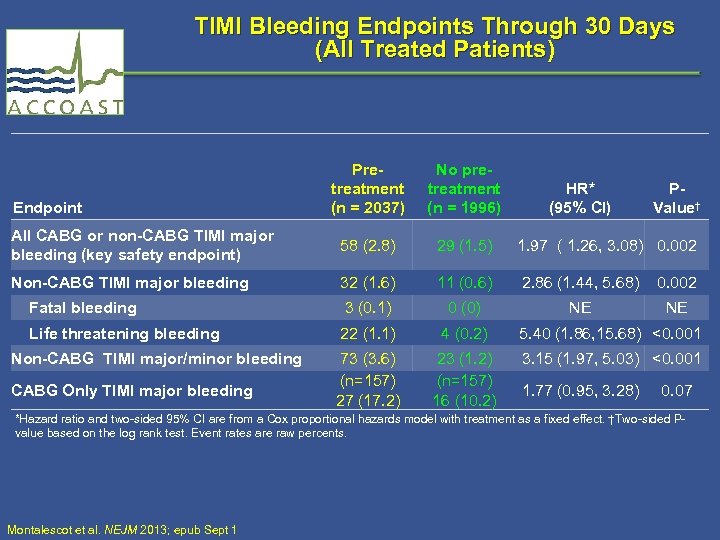 TIMI Bleeding Endpoints Through 30 Days (All Treated Patients) Pretreatment (n = 2037) No pretreatment (n = 1996) All CABG or non-CABG TIMI major bleeding (key safety endpoint) 58 (2. 8) 29 (1. 5) 1. 97 ( 1. 26, 3. 08) 0. 002 Non-CABG TIMI major bleeding 32 (1. 6) 11 (0. 6) 2. 86 (1. 44, 5. 68) 0. 002 Fatal bleeding 3 (0. 1) 0 (0) NE NE Life threatening bleeding 22 (1. 1) 4 (0. 2) 5. 40 (1. 86, 15. 68) <0. 001 73 (3. 6) (n=157) 27 (17. 2) 23 (1. 2) (n=157) 16 (10. 2) 3. 15 (1. 97, 5. 03) <0. 001 Endpoint Non-CABG TIMI major/minor bleeding CABG Only TIMI major bleeding HR* (95% CI) 1. 77 (0. 95, 3. 28) PValue† 0. 07 *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided Pvalue based on the log rank test. Event rates are raw percents. Montalescot et al. NEJM 2013; epub Sept 1
TIMI Bleeding Endpoints Through 30 Days (All Treated Patients) Pretreatment (n = 2037) No pretreatment (n = 1996) All CABG or non-CABG TIMI major bleeding (key safety endpoint) 58 (2. 8) 29 (1. 5) 1. 97 ( 1. 26, 3. 08) 0. 002 Non-CABG TIMI major bleeding 32 (1. 6) 11 (0. 6) 2. 86 (1. 44, 5. 68) 0. 002 Fatal bleeding 3 (0. 1) 0 (0) NE NE Life threatening bleeding 22 (1. 1) 4 (0. 2) 5. 40 (1. 86, 15. 68) <0. 001 73 (3. 6) (n=157) 27 (17. 2) 23 (1. 2) (n=157) 16 (10. 2) 3. 15 (1. 97, 5. 03) <0. 001 Endpoint Non-CABG TIMI major/minor bleeding CABG Only TIMI major bleeding HR* (95% CI) 1. 77 (0. 95, 3. 28) PValue† 0. 07 *Hazard ratio and two-sided 95% CI are from a Cox proportional hazards model with treatment as a fixed effect. †Two-sided Pvalue based on the log rank test. Event rates are raw percents. Montalescot et al. NEJM 2013; epub Sept 1


