cedf0d3de3c5085460d6eb2183d5ec9f.ppt
- Количество слайдов: 37
 Health Canada experiences with early identification of potential carcinogens - An Existing Substances Perspective Sunil Kulkarni Hazard Methodology Division, Existing Substances Risk Assessment Bureau Health Canada, Ottawa, ON
Health Canada experiences with early identification of potential carcinogens - An Existing Substances Perspective Sunil Kulkarni Hazard Methodology Division, Existing Substances Risk Assessment Bureau Health Canada, Ottawa, ON
 Outline • • Brief introduction DSL - Categorization – Tools/Approaches Chemicals Management Plan – Phase I & II Remaining priorities (Q)SAR tools we use Challenges of (Q)SAR models & modelable endpoints (Q)SAR results/analyses
Outline • • Brief introduction DSL - Categorization – Tools/Approaches Chemicals Management Plan – Phase I & II Remaining priorities (Q)SAR tools we use Challenges of (Q)SAR models & modelable endpoints (Q)SAR results/analyses
 Existing Substances under CEPA 1999 • Approximately 23, 000 substances (e. g. , industrial chemicals) on the Domestic Substances List (DSL) • Includes substances used for commercial manufacturing or manufactured or imported in Canada at >100 kg/year between Jan 1, 1984 and Dec 31, 1986
Existing Substances under CEPA 1999 • Approximately 23, 000 substances (e. g. , industrial chemicals) on the Domestic Substances List (DSL) • Includes substances used for commercial manufacturing or manufactured or imported in Canada at >100 kg/year between Jan 1, 1984 and Dec 31, 1986
 Categorization • Identify substances on the basis of exposure or hazard to consider further for screening assessment and to determine if they pose “harm to human health” or not • A variety of tools including those based on (Q)SAR approaches were applied
Categorization • Identify substances on the basis of exposure or hazard to consider further for screening assessment and to determine if they pose “harm to human health” or not • A variety of tools including those based on (Q)SAR approaches were applied
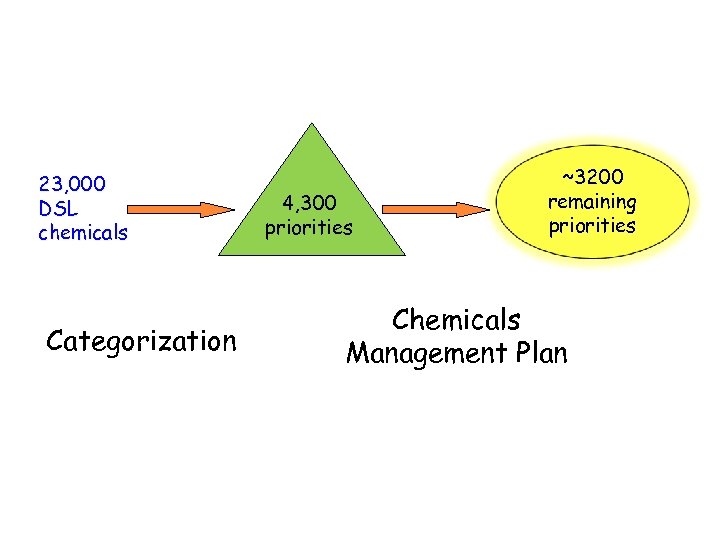 23, 000 DSL chemicals Categorization 4, 300 priorities ~3200 remaining priorities Chemicals Management Plan
23, 000 DSL chemicals Categorization 4, 300 priorities ~3200 remaining priorities Chemicals Management Plan
 Chemicals Management Plan (CMP) • To assess and manage the risks associated with 4300 legacy substances identified through categorization by 2020 • 4300 substances were prioritized into high (~500), medium (~3200) and low concern substances (~550) • CMP brings all existing federal programs together into a single strategy to ensure that chemicals are managed appropriately to prevent harm to Canadians and their environment • It is science-based and specifically designed to protect human health and the environment through four major areas of action: • • Taking action on chemical substances of high concern Taking action on specific industry sectors Investing in research and biomonitoring Improving the information base for decision-making through mandatory submission of use and volume information
Chemicals Management Plan (CMP) • To assess and manage the risks associated with 4300 legacy substances identified through categorization by 2020 • 4300 substances were prioritized into high (~500), medium (~3200) and low concern substances (~550) • CMP brings all existing federal programs together into a single strategy to ensure that chemicals are managed appropriately to prevent harm to Canadians and their environment • It is science-based and specifically designed to protect human health and the environment through four major areas of action: • • Taking action on chemical substances of high concern Taking action on specific industry sectors Investing in research and biomonitoring Improving the information base for decision-making through mandatory submission of use and volume information
 Historical use of (Q)SAR applications 2000 -06 DSL Categorization Commercial (Q)SAR models; basis for decision making (prioritization) 2006 -11 Ministerial Challenge Phase CMP (high priorities) Commercial and some public domain (Q)SAR models, Metabolism, Analogue identification, Read-across; basis for decision making but mainly supportive evidence 2011 - CMP II (includes data poor substances) Commercial and public domain (Q)SAR models, Analogue identification, Chemical categories, Read-across, Metabolism, in-house models/tools
Historical use of (Q)SAR applications 2000 -06 DSL Categorization Commercial (Q)SAR models; basis for decision making (prioritization) 2006 -11 Ministerial Challenge Phase CMP (high priorities) Commercial and some public domain (Q)SAR models, Metabolism, Analogue identification, Read-across; basis for decision making but mainly supportive evidence 2011 - CMP II (includes data poor substances) Commercial and public domain (Q)SAR models, Analogue identification, Chemical categories, Read-across, Metabolism, in-house models/tools
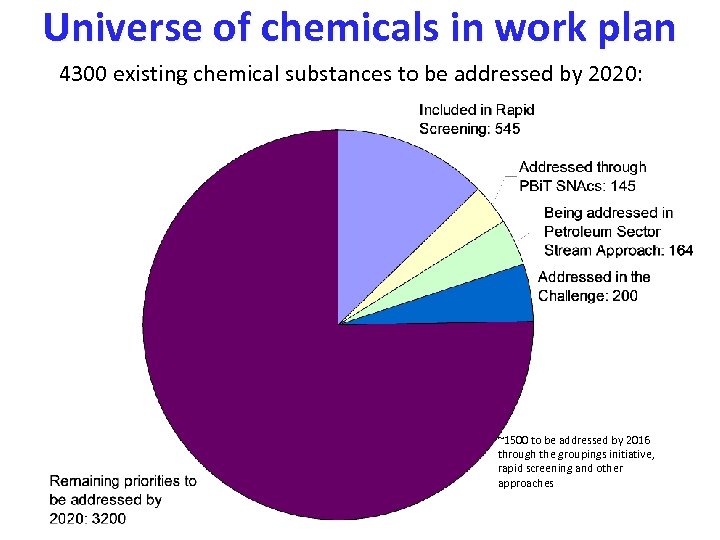 Universe of chemicals in work plan 4300 existing chemical substances to be addressed by 2020: ~1500 to be addressed by 2016 through the groupings initiative, rapid screening and other approaches
Universe of chemicals in work plan 4300 existing chemical substances to be addressed by 2020: ~1500 to be addressed by 2016 through the groupings initiative, rapid screening and other approaches
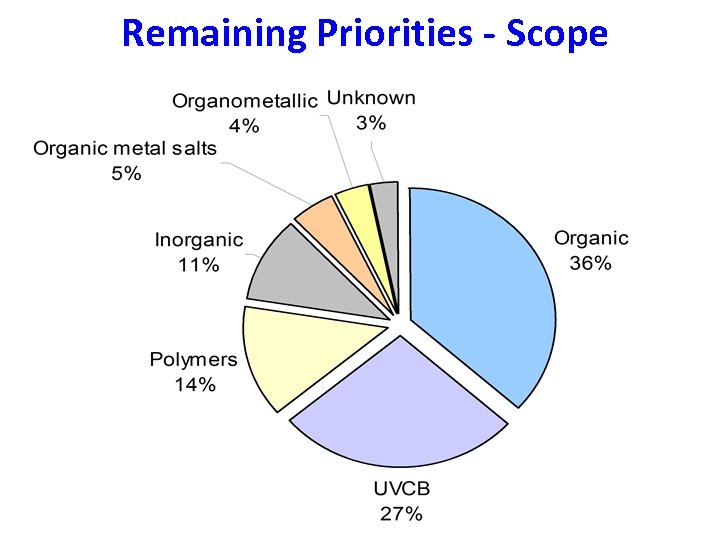 Remaining Priorities - Scope
Remaining Priorities - Scope
 (Q)SAR tools are generally only applicable to discrete organics!
(Q)SAR tools are generally only applicable to discrete organics!
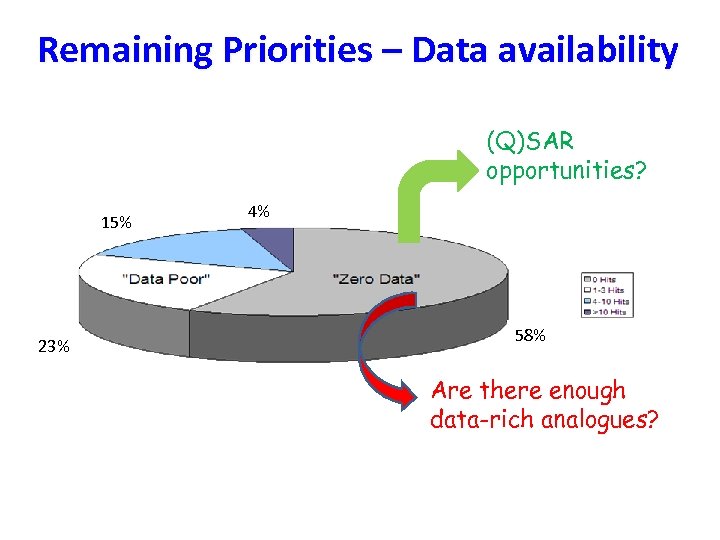 Remaining Priorities – Data availability (Q)SAR opportunities? 15% 23% 4% 58% Are there enough data-rich analogues?
Remaining Priorities – Data availability (Q)SAR opportunities? 15% 23% 4% 58% Are there enough data-rich analogues?
 Approach
Approach
 Human health risk assessment • Chemical’s inherent toxicity & potential human exposure • Assess a range of endpoints including genotoxicity, carcinogenicity, developmental toxicity, reproductive toxicity & skin sensitization • (Q)SAR approaches, including analogue/chemical category read across are used to support our assessments (line of evidence) • Apply weight of evidence and precaution in our decision-making
Human health risk assessment • Chemical’s inherent toxicity & potential human exposure • Assess a range of endpoints including genotoxicity, carcinogenicity, developmental toxicity, reproductive toxicity & skin sensitization • (Q)SAR approaches, including analogue/chemical category read across are used to support our assessments (line of evidence) • Apply weight of evidence and precaution in our decision-making
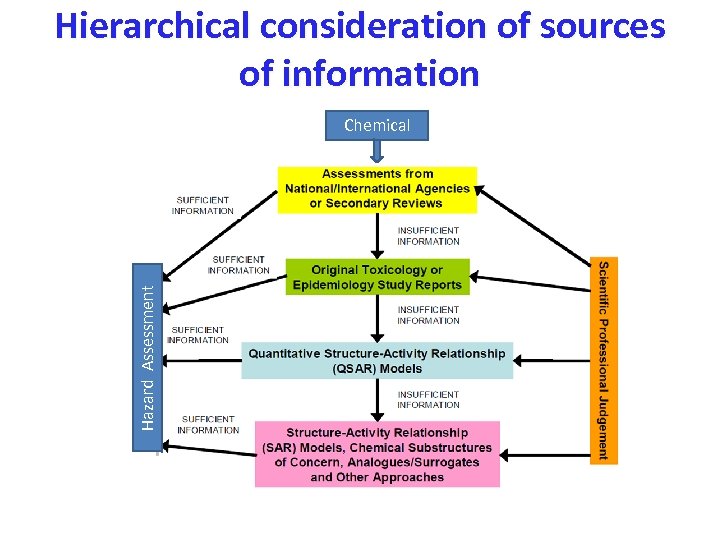 Hierarchical consideration of sources of information Hazard Assessment Chemical
Hierarchical consideration of sources of information Hazard Assessment Chemical
 Predictive tools for hazard assessment Non-commercial Commercial • • • Casetox Topkat Derek Model Applier Oasis Times • • • OECD QSAR Toolbox Toxtree Onco. Logic Caesar (Vega) lazar Supporting tools • Leadscope Hosted - chemical data miner • Pipeline Pilot – cheminformatics and workflow builder
Predictive tools for hazard assessment Non-commercial Commercial • • • Casetox Topkat Derek Model Applier Oasis Times • • • OECD QSAR Toolbox Toxtree Onco. Logic Caesar (Vega) lazar Supporting tools • Leadscope Hosted - chemical data miner • Pipeline Pilot – cheminformatics and workflow builder
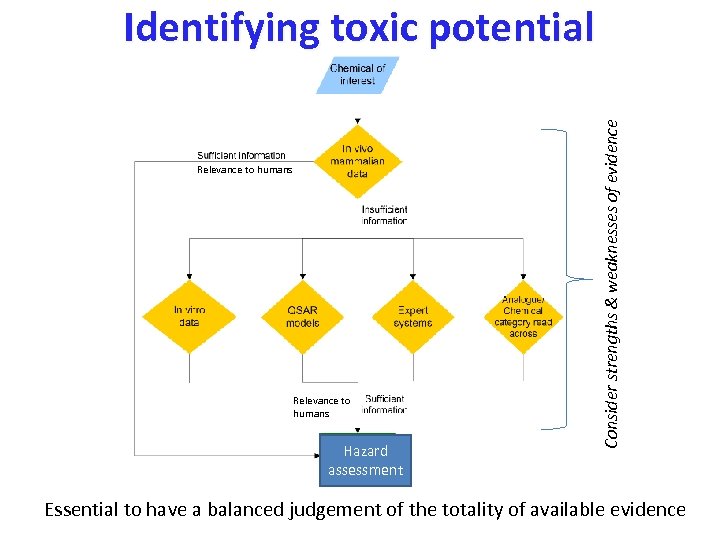 Relevance to humans Hazard assessment Consider strengths & weaknesses of evidence Identifying toxic potential Essential to have a balanced judgement of the totality of available evidence
Relevance to humans Hazard assessment Consider strengths & weaknesses of evidence Identifying toxic potential Essential to have a balanced judgement of the totality of available evidence
 Reliability of estimations • Minimizing uncertainties and maximizing confidence in predictions considering multiple factors: - OECD QSAR Validation principles - accuracy of input - quality of underlying biological data - multiple models based on different predictive paradigms or methodologies - mechanistic understanding - inputs from in vitro/in vivo tests (if available) • Professional judgement of expert(s)
Reliability of estimations • Minimizing uncertainties and maximizing confidence in predictions considering multiple factors: - OECD QSAR Validation principles - accuracy of input - quality of underlying biological data - multiple models based on different predictive paradigms or methodologies - mechanistic understanding - inputs from in vitro/in vivo tests (if available) • Professional judgement of expert(s)
 (Q)SAR tools/approaches to identify potential genotoxic carcinogens • QSAR Toolbox profiler flags- DNA/Protein binding, Benigni-Bossa, Onco. Logic • Metabolic simulators (Toolbox/TIMES) + DNA/Protein binding/Benigni-Bossa flags • Combination of (Q)SAR models for genotoxicity & carcinogenicity (Casetox, Model Applier, Derek, Times, Toxtree, Caesar, Topkat) • Genotox - Salmonella (Ames) models for different strains, Chrom ab, Micronuclei Ind, Mouse Lymphoma mut with metabolic activation • Carcinogenicity – Male & female rats, mice, rodent
(Q)SAR tools/approaches to identify potential genotoxic carcinogens • QSAR Toolbox profiler flags- DNA/Protein binding, Benigni-Bossa, Onco. Logic • Metabolic simulators (Toolbox/TIMES) + DNA/Protein binding/Benigni-Bossa flags • Combination of (Q)SAR models for genotoxicity & carcinogenicity (Casetox, Model Applier, Derek, Times, Toxtree, Caesar, Topkat) • Genotox - Salmonella (Ames) models for different strains, Chrom ab, Micronuclei Ind, Mouse Lymphoma mut with metabolic activation • Carcinogenicity – Male & female rats, mice, rodent
 (Q)SAR tools/approaches to identify potential non-genotoxic carcinogens • Flags from QSAR Toolbox profilers – Benigni-Bossa flags • QSAR models based on in vitro Cell Transformation assays such as Syrian Hamster Embryo, BALB/c-3 T 3, C 3 H 10 T 1/2 • Expert rule based systems Derek and Toxtree
(Q)SAR tools/approaches to identify potential non-genotoxic carcinogens • Flags from QSAR Toolbox profilers – Benigni-Bossa flags • QSAR models based on in vitro Cell Transformation assays such as Syrian Hamster Embryo, BALB/c-3 T 3, C 3 H 10 T 1/2 • Expert rule based systems Derek and Toxtree
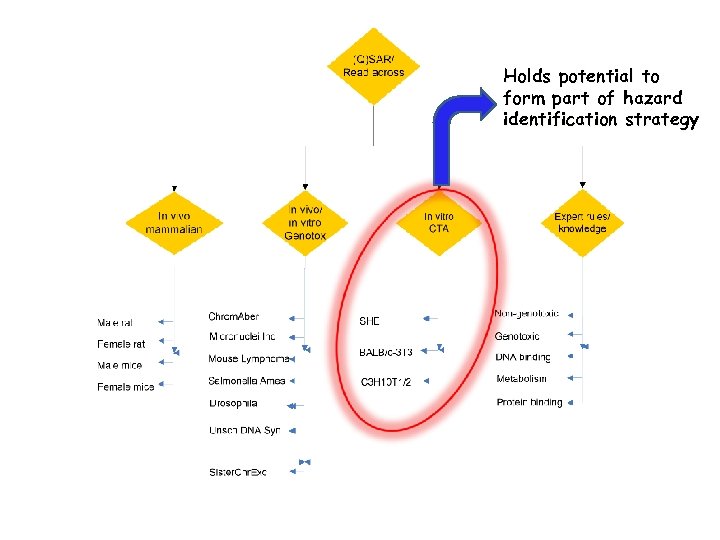 Holds potential to form part of hazard identification strategy
Holds potential to form part of hazard identification strategy
 Helpful to have a better understanding of Cell Transformation in mechanistic interpretation of (non -genotoxic) carcinogenicity
Helpful to have a better understanding of Cell Transformation in mechanistic interpretation of (non -genotoxic) carcinogenicity
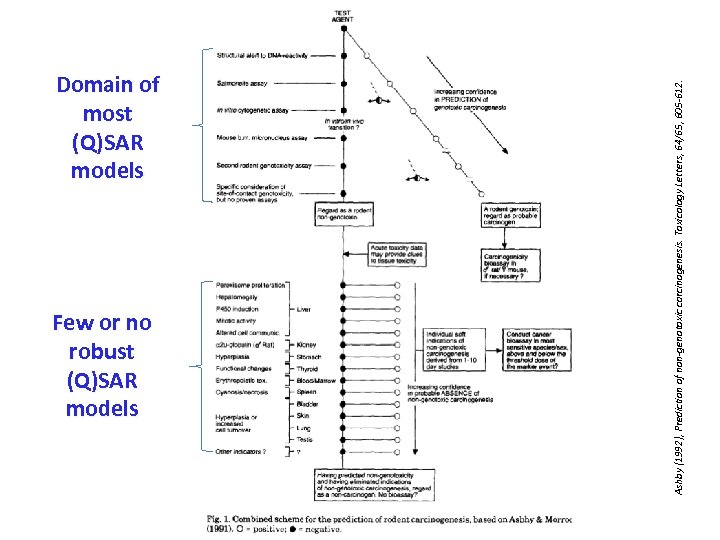 Few or no robust (Q)SAR models Ashby (1992), Prediction of non-genotoxic carcinogenesis. Toxicology Letters, 64/65, 605 -612. Domain of most (Q)SAR models
Few or no robust (Q)SAR models Ashby (1992), Prediction of non-genotoxic carcinogenesis. Toxicology Letters, 64/65, 605 -612. Domain of most (Q)SAR models
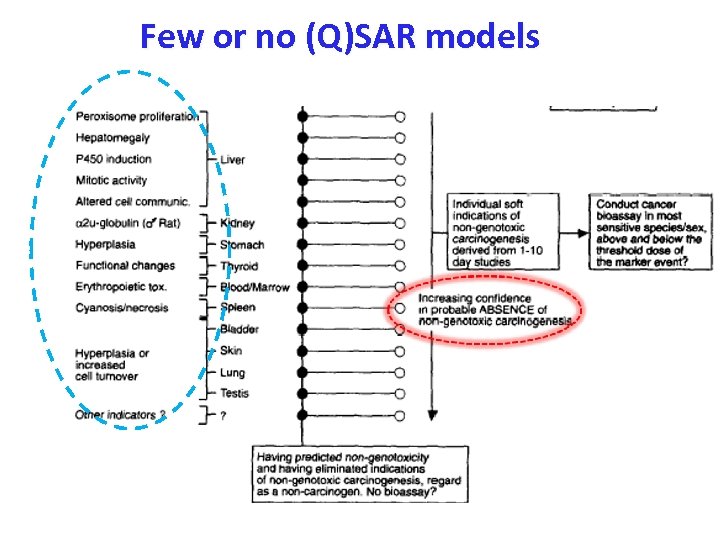 Few or no (Q)SAR models
Few or no (Q)SAR models
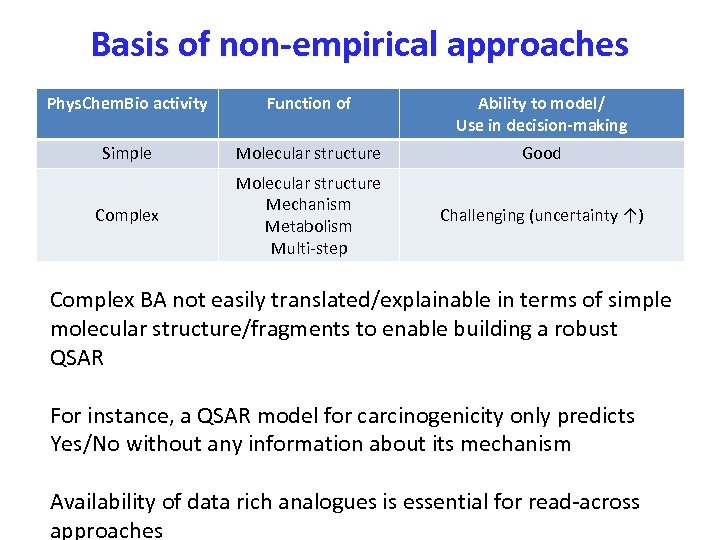 Basis of non-empirical approaches Phys. Chem. Bio activity Function of Ability to model/ Use in decision-making Simple Molecular structure Good Complex Molecular structure Mechanism Metabolism Multi-step Challenging (uncertainty ↑) Complex BA not easily translated/explainable in terms of simple molecular structure/fragments to enable building a robust QSAR For instance, a QSAR model for carcinogenicity only predicts Yes/No without any information about its mechanism Availability of data rich analogues is essential for read-across approaches
Basis of non-empirical approaches Phys. Chem. Bio activity Function of Ability to model/ Use in decision-making Simple Molecular structure Good Complex Molecular structure Mechanism Metabolism Multi-step Challenging (uncertainty ↑) Complex BA not easily translated/explainable in terms of simple molecular structure/fragments to enable building a robust QSAR For instance, a QSAR model for carcinogenicity only predicts Yes/No without any information about its mechanism Availability of data rich analogues is essential for read-across approaches
 (Q)SAR analysis
(Q)SAR analysis
 Performance of some (Q)SAR models • A set of chemicals with in vitro and in vivo data on genotoxicity and carcinogenicity was chosen • Predictions were obtained for different human health relevant endpoints by running these through a variety of (Q)SAR models • Performance of models to discriminate carcinogenic and non -carcinogenic chemicals was evaluated by analysing the results • Structural analysis of chemicals incorrectly classified by all models revealed a diverse group of chemicals with few trends (we are working on that) • Failure of models/expert systems to flag them as “Out of domain”
Performance of some (Q)SAR models • A set of chemicals with in vitro and in vivo data on genotoxicity and carcinogenicity was chosen • Predictions were obtained for different human health relevant endpoints by running these through a variety of (Q)SAR models • Performance of models to discriminate carcinogenic and non -carcinogenic chemicals was evaluated by analysing the results • Structural analysis of chemicals incorrectly classified by all models revealed a diverse group of chemicals with few trends (we are working on that) • Failure of models/expert systems to flag them as “Out of domain”
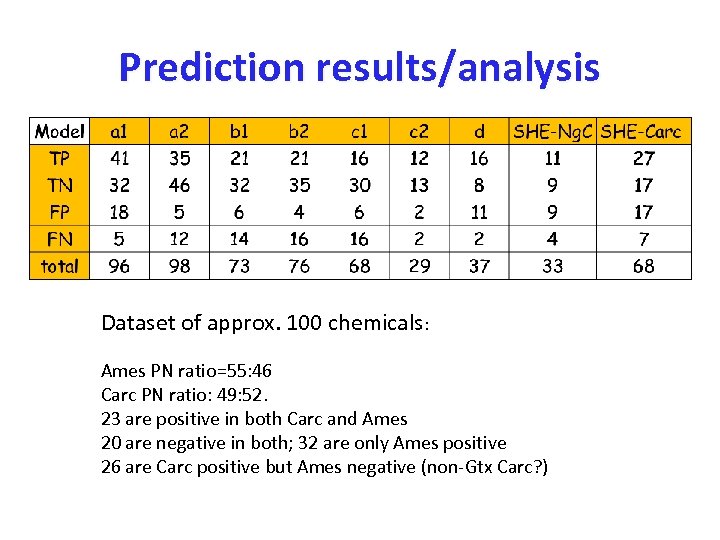 Prediction results/analysis Dataset of approx. 100 chemicals: Ames PN ratio=55: 46 Carc PN ratio: 49: 52. 23 are positive in both Carc and Ames 20 are negative in both; 32 are only Ames positive 26 are Carc positive but Ames negative (non-Gtx Carc? )
Prediction results/analysis Dataset of approx. 100 chemicals: Ames PN ratio=55: 46 Carc PN ratio: 49: 52. 23 are positive in both Carc and Ames 20 are negative in both; 32 are only Ames positive 26 are Carc positive but Ames negative (non-Gtx Carc? )
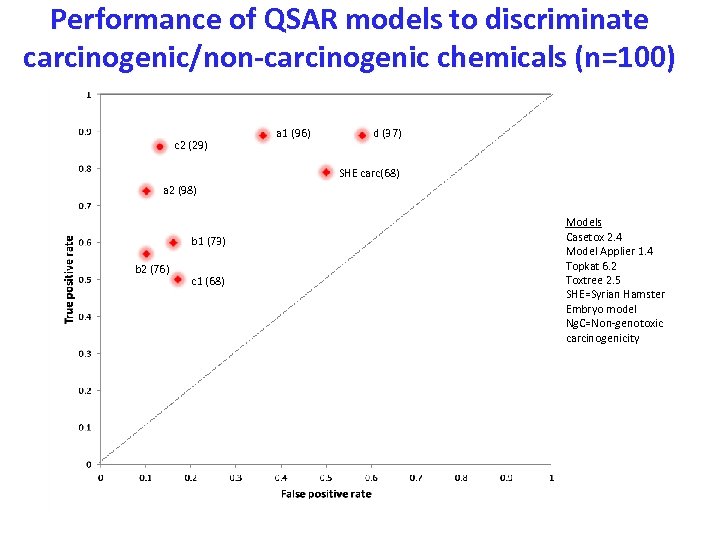 Performance of QSAR models to discriminate carcinogenic/non-carcinogenic chemicals (n=100) c 2 (29) a 1 (96) d (37) SHE carc(68) a 2 (98) b 1 (73) b 2 (76) c 1 (68) Models Casetox 2. 4 Model Applier 1. 4 Topkat 6. 2 Toxtree 2. 5 SHE=Syrian Hamster Embryo model Ng. C=Non-genotoxic carcinogenicity
Performance of QSAR models to discriminate carcinogenic/non-carcinogenic chemicals (n=100) c 2 (29) a 1 (96) d (37) SHE carc(68) a 2 (98) b 1 (73) b 2 (76) c 1 (68) Models Casetox 2. 4 Model Applier 1. 4 Topkat 6. 2 Toxtree 2. 5 SHE=Syrian Hamster Embryo model Ng. C=Non-genotoxic carcinogenicity
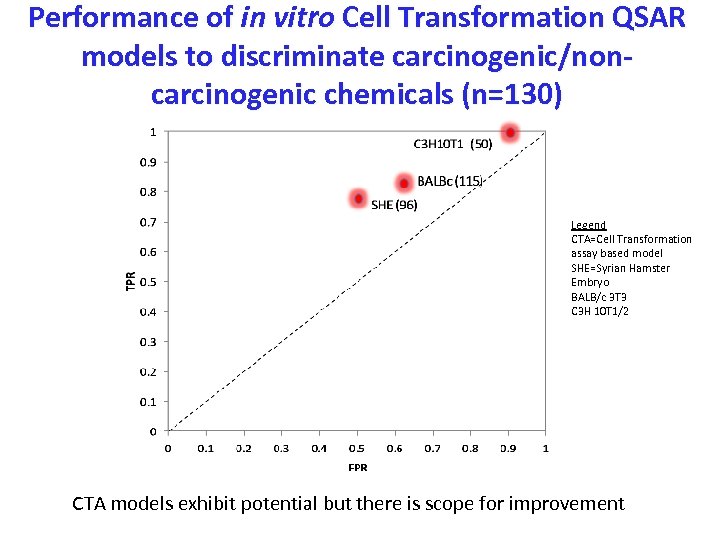 Performance of in vitro Cell Transformation QSAR models to discriminate carcinogenic/noncarcinogenic chemicals (n=130) Legend CTA=Cell Transformation assay based model SHE=Syrian Hamster Embryo BALB/c 3 T 3 C 3 H 10 T 1/2 CTA models exhibit potential but there is scope for improvement
Performance of in vitro Cell Transformation QSAR models to discriminate carcinogenic/noncarcinogenic chemicals (n=130) Legend CTA=Cell Transformation assay based model SHE=Syrian Hamster Embryo BALB/c 3 T 3 C 3 H 10 T 1/2 CTA models exhibit potential but there is scope for improvement
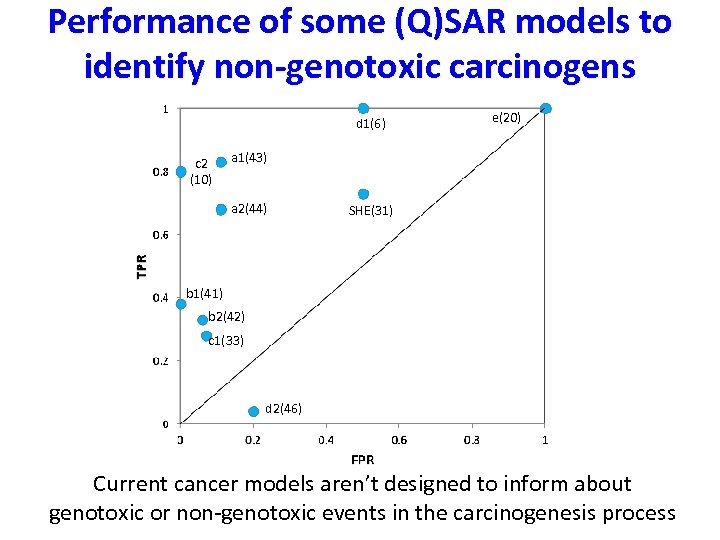 Performance of some (Q)SAR models to identify non-genotoxic carcinogens d 1(6) c 2 (10) e(20) a 1(43) a 2(44) SHE(31) b 1(41) b 2(42) c 1(33) d 2(46) Current cancer models aren’t designed to inform about genotoxic or non-genotoxic events in the carcinogenesis process
Performance of some (Q)SAR models to identify non-genotoxic carcinogens d 1(6) c 2 (10) e(20) a 1(43) a 2(44) SHE(31) b 1(41) b 2(42) c 1(33) d 2(46) Current cancer models aren’t designed to inform about genotoxic or non-genotoxic events in the carcinogenesis process
 Data analysis
Data analysis
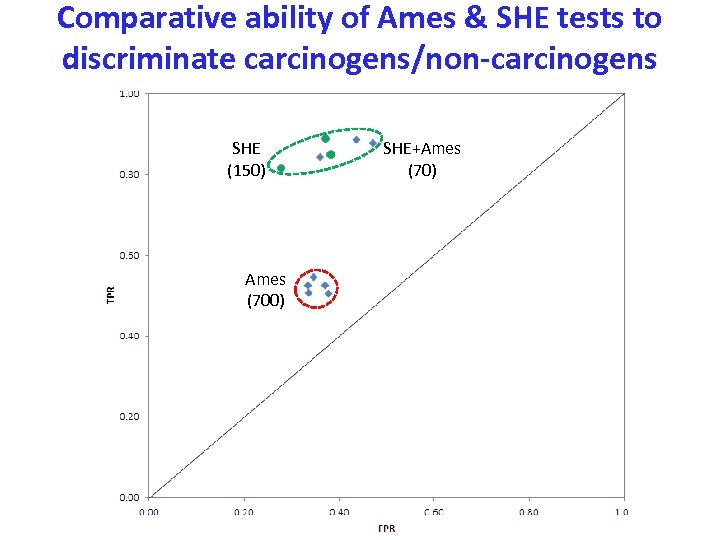 Comparative ability of Ames & SHE tests to discriminate carcinogens/non-carcinogens SHE (150) Ames (700) SHE+Ames (70)
Comparative ability of Ames & SHE tests to discriminate carcinogens/non-carcinogens SHE (150) Ames (700) SHE+Ames (70)
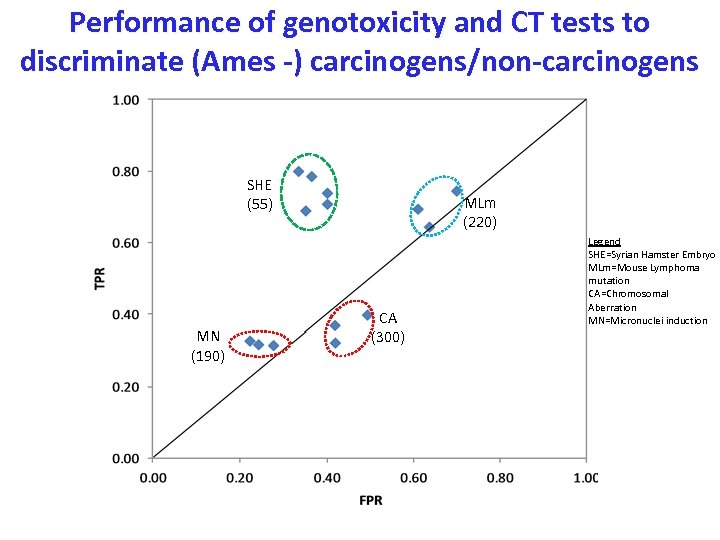 Performance of genotoxicity and CT tests to discriminate (Ames -) carcinogens/non-carcinogens SHE (55) MN (190) MLm (220) CA (300) Legend SHE=Syrian Hamster Embryo MLm=Mouse Lymphoma mutation CA=Chromosomal Aberration MN=Micronuclei induction
Performance of genotoxicity and CT tests to discriminate (Ames -) carcinogens/non-carcinogens SHE (55) MN (190) MLm (220) CA (300) Legend SHE=Syrian Hamster Embryo MLm=Mouse Lymphoma mutation CA=Chromosomal Aberration MN=Micronuclei induction
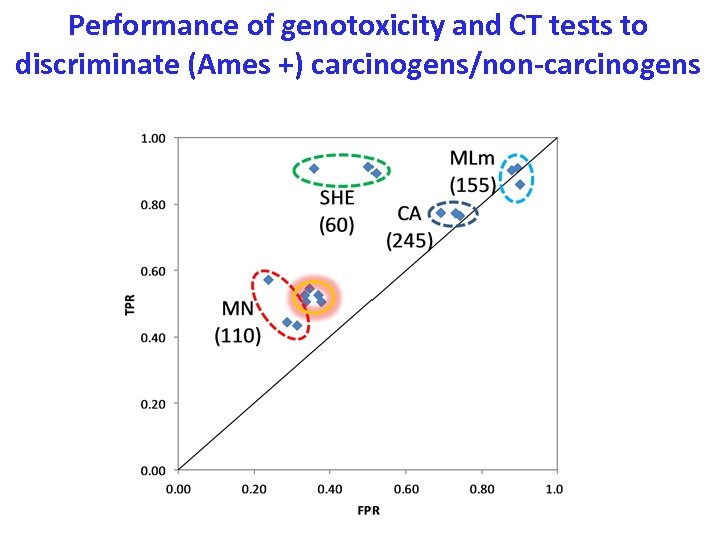 Performance of genotoxicity and CT tests to discriminate (Ames +) carcinogens/non-carcinogens
Performance of genotoxicity and CT tests to discriminate (Ames +) carcinogens/non-carcinogens
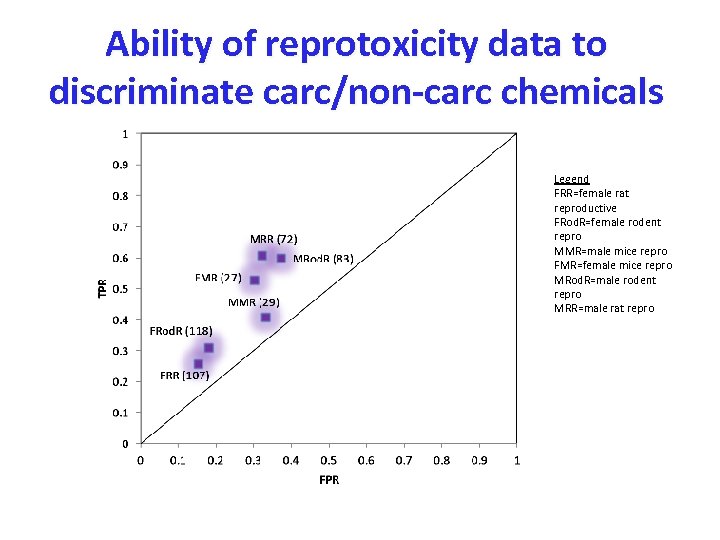 Ability of reprotoxicity data to discriminate carc/non-carc chemicals Legend FRR=female rat reproductive FRod. R=female rodent repro MMR=male mice repro FMR=female mice repro MRod. R=male rodent repro MRR=male rat repro
Ability of reprotoxicity data to discriminate carc/non-carc chemicals Legend FRR=female rat reproductive FRod. R=female rodent repro MMR=male mice repro FMR=female mice repro MRod. R=male rodent repro MRR=male rat repro
 Finally………. . tpr Scope for improvement nt rre Cu ce an m or f er p fpr
Finally………. . tpr Scope for improvement nt rre Cu ce an m or f er p fpr
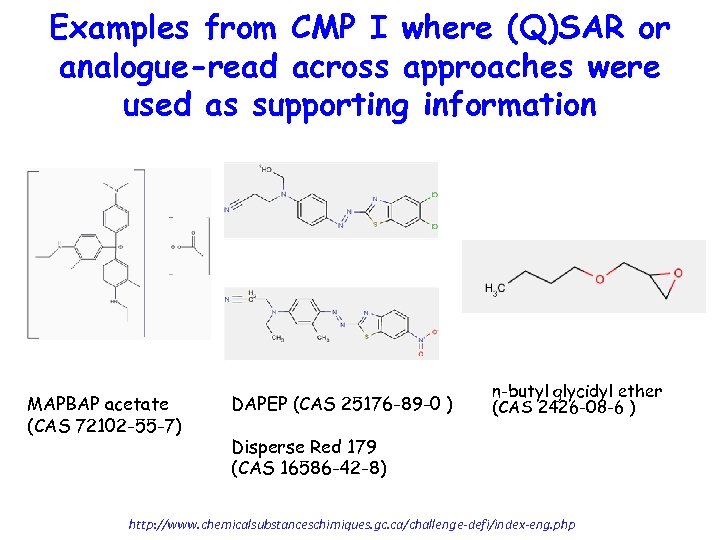 Examples from CMP I where (Q)SAR or analogue-read across approaches were used as supporting information MAPBAP acetate (CAS 72102 -55 -7) DAPEP (CAS 25176 -89 -0 ) n-butyl glycidyl ether (CAS 2426 -08 -6 ) Disperse Red 179 (CAS 16586 -42 -8) http: //www. chemicalsubstanceschimiques. gc. ca/challenge-defi/index-eng. php
Examples from CMP I where (Q)SAR or analogue-read across approaches were used as supporting information MAPBAP acetate (CAS 72102 -55 -7) DAPEP (CAS 25176 -89 -0 ) n-butyl glycidyl ether (CAS 2426 -08 -6 ) Disperse Red 179 (CAS 16586 -42 -8) http: //www. chemicalsubstanceschimiques. gc. ca/challenge-defi/index-eng. php


