d5c4b9f67e28e80d3a20c42e046a413c.ppt
- Количество слайдов: 23

Hadasit Bio-Holdings Ltd. – “HBL” (Tel Aviv Stock Exchange: HDST) July 2008

Forward Looking Statements This document/presentation contains forward looking statements. Words such as “estimates, ” “expects, ” “intends, ” “plans, ” “believes” and terms of similar substance used in connection with any discussion of future operational performance or financial results, identify forward looking statements. Such statements are subject to risks and uncertainties that could cause the actual results to differ materially from these statements. The forward-looking statements contained herein. speak only as of the date of this document The Company expressly disclaims any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in the Company's expectations with regard thereto or to reflect any change in events, conditions or circumstances on which any such forward-looking statement is based, in whole. or in part This document does not constitute or form any part of any offer or invitation to sell, or any solicitation of any offer to purchase or subscribe for any shares in Hadasit Bio-Holdings Ltd. or any of its affiliated entities nor shall it form the basis of, or be relied on in connection with, . any contract therefore 2
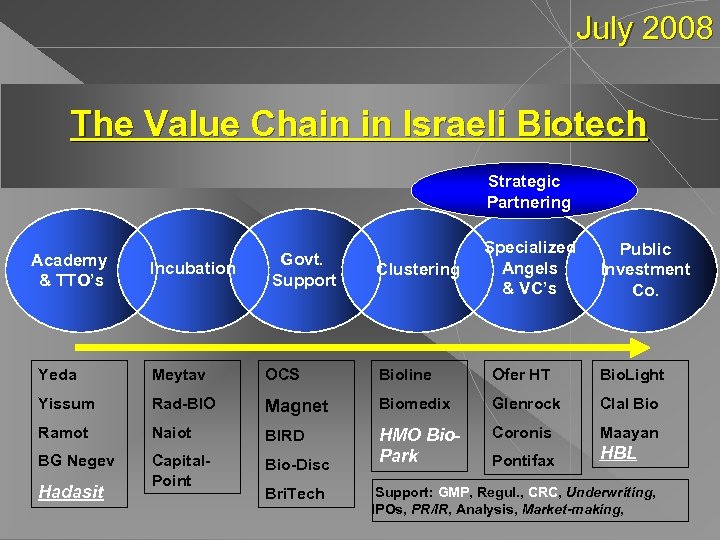
July 2008 The Value Chain in Israeli Biotech Strategic Partnering Academy & TTO’s Incubation Govt. Support Clustering Specialized Angels & VC’s Public Investment Co. Yeda Meytav OCS Bioline Ofer HT Bio. Light Yissum Rad-BIO Magnet Biomedix Glenrock Clal Bio Ramot Naiot BIRD Coronis Maayan BG Negev Capital. Point Bio-Disc HMO Bio. Park Pontifax HBL Hadasit Bri. Tech Support: GMP, Regul. , CRC, Underwriting, IPOs, PR/IR, Analysis, Market-making,

HBL - Company Profile Holdings in early-stage biotech companies (until completion of Phase I) • 10 companies – All IP developed at Hadassah • 3 areas (Oncology, Auto-immune diseases, Tissue engineering / Stem cells) • Criteria – Demonstration of Proof of Concept, Blockbuster Market • Exit strategy - at end of Phase I Leveraging Hadassah Hospital • Source of Intellectual Property and future investment opportunities • Skilled personnel, infrastructure, GMP facilities and clinical trials • • 4 Established: September 2005 Traded: Tel Aviv Stock Exchange (HDST) – since January 2006 Investment rounds: • IPO (January 2006) – $ 8 m • Convertible Bonds (August 2006) – $ 10 m , $ 5 m converted (Feb. 2007) • Strategic Investment (May 2007) - $ 2 m (Consensus Business Group) Stockholders: Hadasit (owned by Hadassah) (52%), public (43%), CBG - (5%)

HBL Structure (7/08) 100% Public 43% Drug Engineering Conju. Gate 22% Thrombo tech 52% 30% 66% Incure C. B. G 5% H. B. L Ltd. 25% TK Signal (UK) Oncology 34% 4. 5% 75% Cell Cure Forticell Bio. (Nasdaq) Verto Tissue Engineering and Stem-cell Medicine 100% Prot. Ab 80% KAHR 92% Tolarex Auto-immune Disease
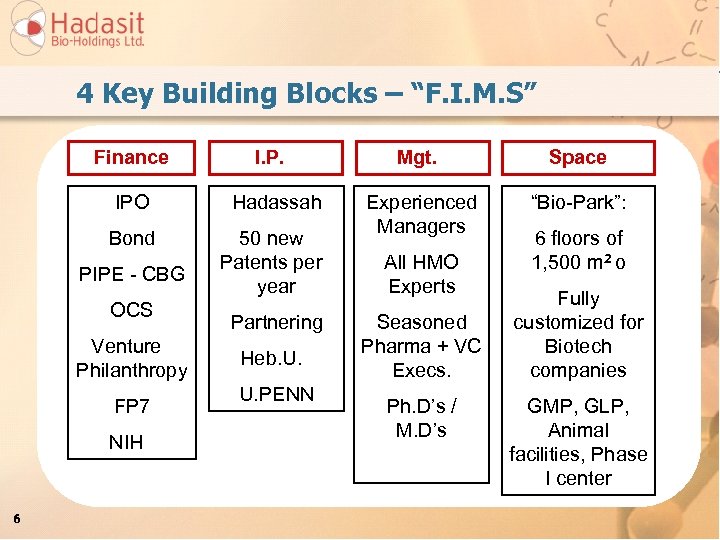
4 Key Building Blocks – “F. I. M. S” Finance I. P. IPO Hadassah Bond 50 new Patents per year PIPE - CBG OCS Venture Philanthropy FP 7 NIH 6 Partnering Heb. U. U. PENN Mgt. Space Experienced Managers “Bio-Park”: All HMO Experts Seasoned Pharma + VC Execs. Ph. D’s / M. D’s 6 floors of 1, 500 m 2 o Fully customized for Biotech companies GMP, GLP, Animal facilities, Phase I center

2008 – Value Adding Milestones § KAHR – HBL Investment, 5 New Compounds from UPENN, OCS approval for $ 400 K, new VP, external investment § Tolarex – HBL Investment, IND approval from FDA, Phase I start, external investment § Prot. Ab – Completion of humanization – external investment § TK Signal – Preparation for Phase I, Phase I start § Verto – Completion of Phase I trial, strategic partnership § Incure – Expansion of trial data, Exit via strategic partnership § Conju. Gate – External Investment, Manuf. , Phase I start § Bioline. Rx – 2 X Phase II completions (BL 1020, BL 1040) § HBL – Investment ($ 5 -7 M) 7

Hadassah Bio-Park - 2008 § § § § 8 First medical center “on-site” bio park – custom built for life science companies (including labs, Phase I center) Building upon infrastructure of Hebrew U. Med. School and Hadassah University Hospital (conducting more than 50% of all translational medicine research in Israel) HU conducts over 40% of all Biotechnology research in Israel. Hadassah Ein-Kerem campus includes Med. and Dentistry schools, Nursing, Pharmacy & HBL Public Health schools First building to include 6 floors: total available space – 10, 000 m 2 Jerusalem municipality and JDA provide significant financial incentives: tax breaks and grants 5 HBL companies (+ HQ) to relocate

Investment Strategy § High return on investment through investments before the first clinical phase (Phase I) – – – § § After proof of concept in animals and before completion of Phase I Value multipliers: X 10 to X 50 Midway Exit planned after Phase I, clear dividend policy Product target markets measured in the billions of dollars (“Blockbuster”) – – 1 2 Oncology, tissue engineering, stem-cell medicine, auto-immune diseases Answer to Unmet Clinical Needs (not improvement of existing drugs) Risk Management 3 – – – Reducing company “burn-rate” by utilizing the infrastructure of Hadassah Hospital for development and clinical research – 9 Dispersing risk through a portfolio of 9 companies in different fields Huge pool of future investment options (IP) – getting in at “ground level” with rapid increase in value Professional identification of investments and guidance of the companies by a team of technology and business experts
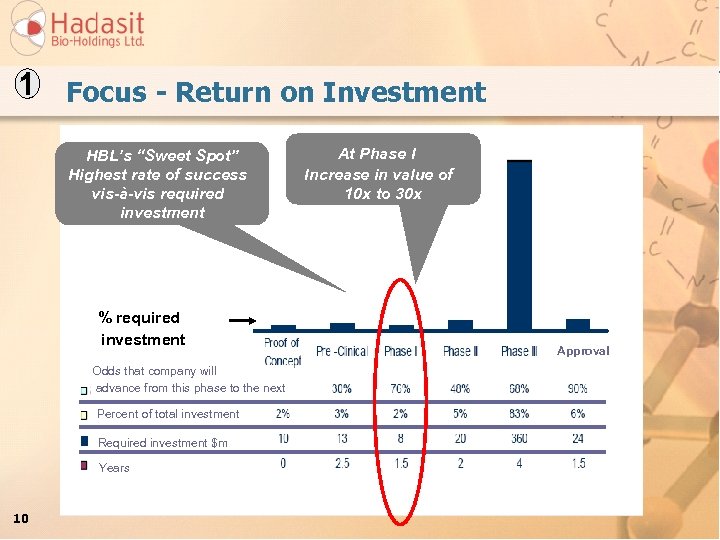
1 Focus - Return on Investment HBL’s “Sweet Spot” Highest rate of success vis-à-vis required investment % required investment Odds that company will advance from this phase to the next Percent of total investment Required investment $m Years 10 At Phase I Increase in value of 10 x to 30 x Approval
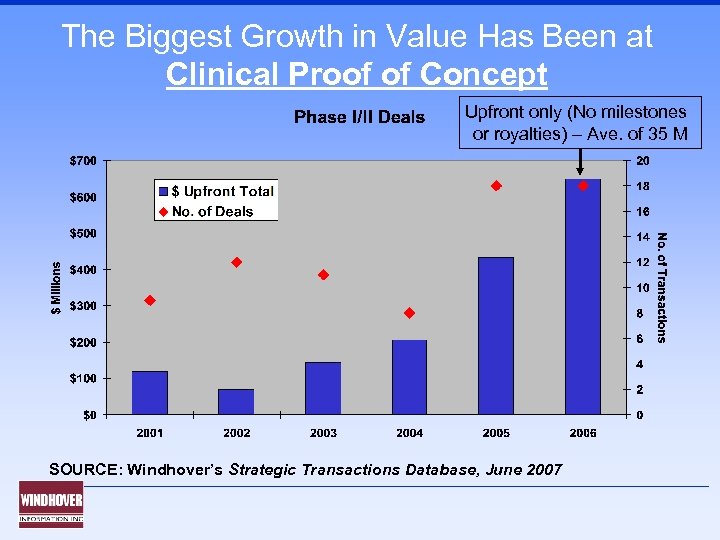
The Biggest Growth in Value Has Been at Clinical Proof of Concept Upfront only (No milestones or royalties) – Ave. of 35 M SOURCE: Windhover’s Strategic Transactions Database, June 2007
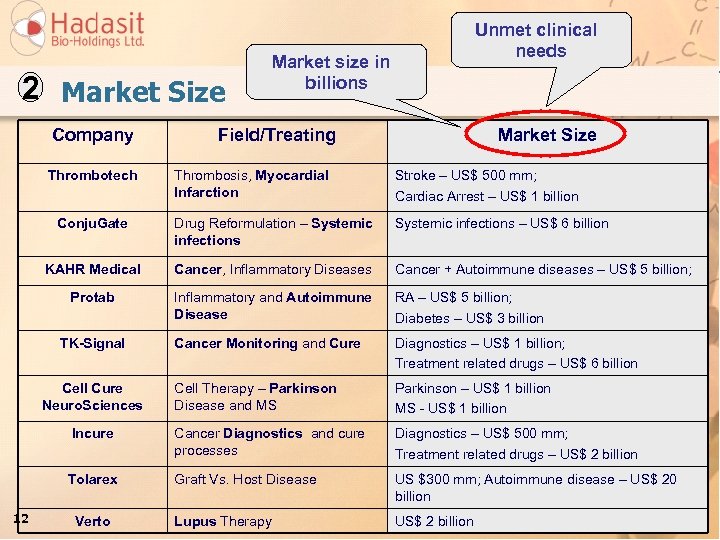
2 Market Size Company Thrombotech Unmet clinical needs Market size in billions Field/Treating Market Size Thrombosis, Myocardial Infarction Stroke – US$ 500 mm; Cardiac Arrest – US$ 1 billion Conju. Gate Drug Reformulation – Systemic infections – US$ 6 billion KAHR Medical Cancer, Inflammatory Diseases Cancer + Autoimmune diseases – US$ 5 billion; Protab Inflammatory and Autoimmune Disease RA – US$ 5 billion; Diabetes – US$ 3 billion Cancer Monitoring and Cure Diagnostics – US$ 1 billion; Treatment related drugs – US$ 6 billion Cell Therapy – Parkinson Disease and MS Parkinson – US$ 1 billion MS - US$ 1 billion Incure Cancer Diagnostics and cure processes Diagnostics – US$ 500 mm; Treatment related drugs – US$ 2 billion Tolarex Graft Vs. Host Disease US $300 mm; Autoimmune disease – US$ 20 billion Lupus Therapy US$ 2 billion TK-Signal Cell Cure Neuro. Sciences 12 Verto

Management Dr. Rafi Hofstein - Chairman of the Board. President and CEO of Hadasit Ltd. , the Technology Holdings Company of Hadassah Medical Organization. Ex-President of Mindsense Biosystems Ltd. Business Unit Director of Ecogen Inc. , Langhorne PA after serving as the Scientific Director of its Israel office. Ph. D. and M. Sc. in lifesciences and chemistry from the Weizmann Institute of Science, B. Sc. in chemistry and physics from the Hebrew University in Jerusalem. Dr. Hofstein completed postdoctoral training at the Harvard Medical School in Boston, MA, in the departments of biological chemistry and neurobiology. Founder of ILSI, advisor to Israeli Govt. , Bo. Ds: Breath ID, Evogene, Bioline, Conju. Gate, KAHR, Prot. Ab, Tolarex. Adv. Ophir Shahaf - CEO. Appointed March 2007 after serving as VP of the Company since Jan. 2006. Ex-VP business development at Protalix Biotherapeutics Ltd. Part of the founding and management team of Clal Biotechnology Industries Ltd. as VP and general counsel. LL. B. from Tel Aviv University, MBA from the Stern School of Business at New York University. Teaches biotechnology entrepreneurship and management, fellow of the Merage Foundation at Irvine, CA. Bo. Ds: Tolarex, Prot. Ab, Biokine, Thrombotech, Conju. Gate. 13

Board of Directors / Advisory Board § § § 14 Prof. Shlomo Mor-Yosef, HMO Director General Yuval Cohen, Fortissimo Private Equity Doron Debbie (Consensus Business Group) Tsipi Gal-Yam (External) Yaron Kulas (External) Prof. Ruth Arnon (Weizmann) Steven G. Burrill – Burrill & Co. Tamar Howson (Bristol Myers Squibb, Lex. Pharma) Mary Tanner (Biogen, NY Hedge Funds) Jonathan Silverstein (Orbimed VC) Jonathan Fleming (Oxford Biopartners VC) Cheryl Reicin (Torys LLP) All department heads and leading physicians of HMO

External Investments in Companies 2 Major Strategic Investors / Partners have Invested in HBL Portfolio Companies, : § TEVA Pharmaceutical Industries - $ 1 M in Cell Cure Neuro. Sciences (Aug. 2007) – Recognition of leadership in HESC field – Attractive terms for company (value, rights) – Significant role in R&D, production and regulation § Clal Biotechnology (CBI) – Option Exercise + additional in Thrombotech (Sept. 2007) – $ 750 K exercise fee + $ 500 K in follow on round – Major shareholder participation (Total $ 1. 75 M) – Sufficient to complete Phase I trials (Stroke / MI) – Experienced CEO and Chairman recruited 15
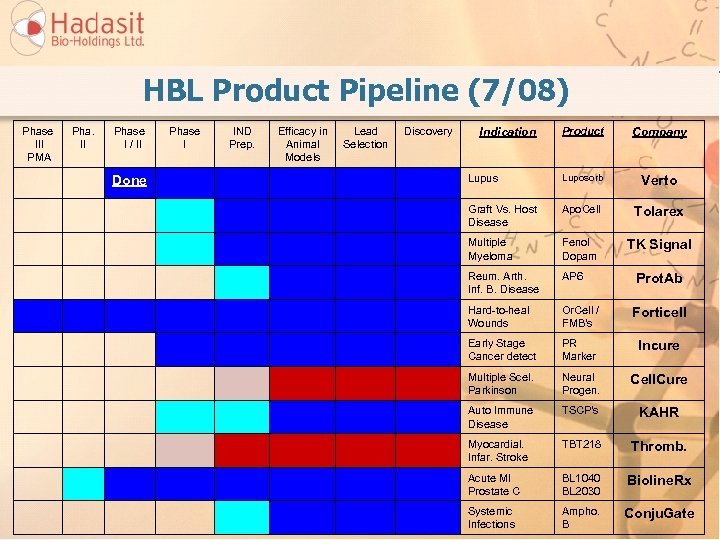
HBL Product Pipeline (7/08) Phase III PMA Pha. II Phase I / II Done Phase I IND Prep. Efficacy in Animal Models Lead Selection Discovery Product Company Lupus Luposorb Verto Graft Vs. Host Disease Apo. Cell Tolarex Multiple Myeloma Fenol Dopam TK Signal Reum. Arth. Inf. B. Disease AP 6 Hard-to-heal Wounds Or. Cell / FMB's Forticell Early Stage Cancer detect PR Marker Incure Multiple Scel. Parkinson Neural Progen. Cell. Cure Auto Immune Disease TSCP's KAHR Myocardial. Infar. Stroke TBT 218 Thromb. Acute MI Prostate C BL 1040 BL 2030 Bioline. Rx Systemic Infections Ampho. B Conju. Gate Indication Prot. Ab

Progress in Portfolio Companies (I) Cell Cure Neuro. Sciences • Investment agreement with TEVA ($ 1 M at $ 13 pre-$) • HBL holdings increased to 34. 5% • All IP related to Neurological Disorders (Parkinson, MS, Alzheimer’s) – permanently released and owned by the company • Receipt of $ 2 M in "soft funding" – Michael J. Fox, ALS, Beresheet, OCS • New VP of R&D, 4 additional researchers (>20 employees overall) Verto • • 17 Completion of Phase I/II trial – 10 patients w. no side effects, measurable therapeutic benefits. Trial complete – analysis and preparation of final report - ongoing. Discussions with potential strategic partners awaiting additional clinical data

Progress in Portfolio Companies (II) Prot. Ab (PTK) • Humanization and production of the lead antibody underway (Antitope – Cambridge, UK). First samples arrived and tested. • OCS approved 60% of R&D budget ($ 500 K). • Discussions with private and VC investors. • Significant interest from strategic partners. Thrombotech • Closed investment following option agreement with CBI ($ 1. 75 M), including 300 K from HBL. • Recruited experienced CEO and Chairman. • Full set of pre-clinical and toxicology trials – repeated in "external" lab with excellent results 18

Progress in Portfolio Companies (III) Tolarex (PTK) • Advanced preparations for clinical trial – Q 1/08, Passed IRB, awaiting MOH approval and pre-IND at the FDA • Additions of personnel (7) and equipment. OCS approved R&D budget of $ 1. 1 M (60% support) • CEO – experience in drug development (Alon Moran – Bio. Mass) KAHR Medical (PTK) • Negotiations with U. Penn for significant expansion of IP basis (5 new Compounds, based on TSCP’s - immune-related proteins) • Additional investment by HBL, Application for OCS support • Manufacturing contracts signed with Cobra and Invitrogen • CEO – Ex-VP of Compugen (Dr. Noam Shani) 19

Progress in Portfolio Companies (IV) Incure • Expansion of ELISA study – 40 patients. Groups added: elders, smokers, pregnant. • Data showing detection of cancer metastases via blood sample. • Negotiation with US strategic partner – licensing & financing deal. • Next step – commercialization via design of diagnostic kit Conju. Gate Ltd. • Platform targeted at unmet need – highly toxic drugs with adverse side effects • Products to improve efficacy while eliminating toxicity, close to phase I with lead product • Leading scientists, strong IP, 3 batches already produced. • Significant equity (33%), BOD representation (2 of 5) and leading position in BD effort. 20

Financial Data § § § $ 3. 0 in cash, as of 1/6/08 3 Leading companies (PTK) raising between $ 3 -5 M each All companies show major progress since HBL Investment Timeline: 3 companies in Phase I within 12 -18 months Pipeline Potential: exclusive mandate (“first look”) for IP from 1, 200 MD’s/Ph. D’s at Hadassah, 250 patents yearly § Top-notch science, IP protection, management § HMO infrastructure allows for efficient financing, low BR Current PIPE § $ 5 -7 M at par + warrants. Commitment from Hadassah, CBG and other key shareholders to participate. 21

Summary § § § § 22 All companies are funded by HBL, partners Leverage of “soft funds”: OCS, research foundations Science, IP and Infrastructure – HMO Key milestone events expected (Phase I, analyst coverage and external investment) Validation of strategic model via external strategic investors (CBG, Teva, CBI) Clear Exit strategy at end of Phase I NAV = 65% discount. Target of 7. 5 NIS. Share price allows major appreciation linked to milestones

Thank You Ophir Shahaf, CEO Hadasit Bio-Holdings Ltd. Tel. : +972 -54 -520 -1177 23
d5c4b9f67e28e80d3a20c42e046a413c.ppt