30542eca11a463f19012ab90029f2576.ppt
- Количество слайдов: 31
 Green Chemistry at Pfizer Peter Dunn Pfizer Green Chemistry Lead
Green Chemistry at Pfizer Peter Dunn Pfizer Green Chemistry Lead
 Agenda § Introduction to Green Chemistry at Pfizer ü What it is, what it encompasses § Making a Difference through Green Chemistry ü Engagement and alignment across the company ü Internal tools – helping chemists “go green” -- solvent guides, reagent guide, acid/base guide, metrics tool. ü Education ü Supporting and influencing academic research ü Results ü Solvent reduction program across our Research Division ü Pregabalin (Lyrica®) Process Development Program ü Atorvastatin (Lipitor®) Process Development Program § Future Directions
Agenda § Introduction to Green Chemistry at Pfizer ü What it is, what it encompasses § Making a Difference through Green Chemistry ü Engagement and alignment across the company ü Internal tools – helping chemists “go green” -- solvent guides, reagent guide, acid/base guide, metrics tool. ü Education ü Supporting and influencing academic research ü Results ü Solvent reduction program across our Research Division ü Pregabalin (Lyrica®) Process Development Program ü Atorvastatin (Lipitor®) Process Development Program § Future Directions
 Pfizer Green Chemistry Mission u u To introduce, educate and promote application of Green Chemistry across Pfizer. Key Philosophy: Voluntary restraint is better than enforced constraint. Green Chemistry includes protection of the environment and worker safety. Informing and influencing the Green Chemistry research agenda.
Pfizer Green Chemistry Mission u u To introduce, educate and promote application of Green Chemistry across Pfizer. Key Philosophy: Voluntary restraint is better than enforced constraint. Green Chemistry includes protection of the environment and worker safety. Informing and influencing the Green Chemistry research agenda.
 Pfizer Green Chemistry – Engagement & Alignment § Success required attention to Green Chemistry across all our locations: research, scale-up, and manufacturing facilities. § We have: ü ü A full-time GC leader with a company-wide responsibility A company GC Policy and Steering Committee (responsible for the strategic plan, communications plans, key policy decisions, and monitoring of performance). Research site GC teams – Medicinal Chemists, Process Chemists and EHS colleagues, set annual objectives, manage site-based awards programs, raise awareness, and drive behavior change. Integrated GC into our co-development process with manufacturing and initiated Manufacturing GC Awards.
Pfizer Green Chemistry – Engagement & Alignment § Success required attention to Green Chemistry across all our locations: research, scale-up, and manufacturing facilities. § We have: ü ü A full-time GC leader with a company-wide responsibility A company GC Policy and Steering Committee (responsible for the strategic plan, communications plans, key policy decisions, and monitoring of performance). Research site GC teams – Medicinal Chemists, Process Chemists and EHS colleagues, set annual objectives, manage site-based awards programs, raise awareness, and drive behavior change. Integrated GC into our co-development process with manufacturing and initiated Manufacturing GC Awards.
 Use of Internal Tools – Med. Chem. Solvent Selection Guide Preferred Usable Undesirable Water Acetone Ethanol 2 -Propanol 1 -Propanol Ethyl Acetate Isopropyl acetate Methanol MEK 1 -Butanol t-Butanol Cyclohexane Heptane Toluene Methylcyclohexane TBME Isooctane Acetonitrile 2 -Me. THF Xylenes DMSO Acetic Acid Ethylene Glycol Pentane Hexane(s) Di-isopropyl ether Diethyl ether Dichloromethane Dichloroethane Chloroform NMP DMF Pyridine DMAc Dioxane Dimethoxyethane Benzene Carbon tetrachloride
Use of Internal Tools – Med. Chem. Solvent Selection Guide Preferred Usable Undesirable Water Acetone Ethanol 2 -Propanol 1 -Propanol Ethyl Acetate Isopropyl acetate Methanol MEK 1 -Butanol t-Butanol Cyclohexane Heptane Toluene Methylcyclohexane TBME Isooctane Acetonitrile 2 -Me. THF Xylenes DMSO Acetic Acid Ethylene Glycol Pentane Hexane(s) Di-isopropyl ether Diethyl ether Dichloromethane Dichloroethane Chloroform NMP DMF Pyridine DMAc Dioxane Dimethoxyethane Benzene Carbon tetrachloride
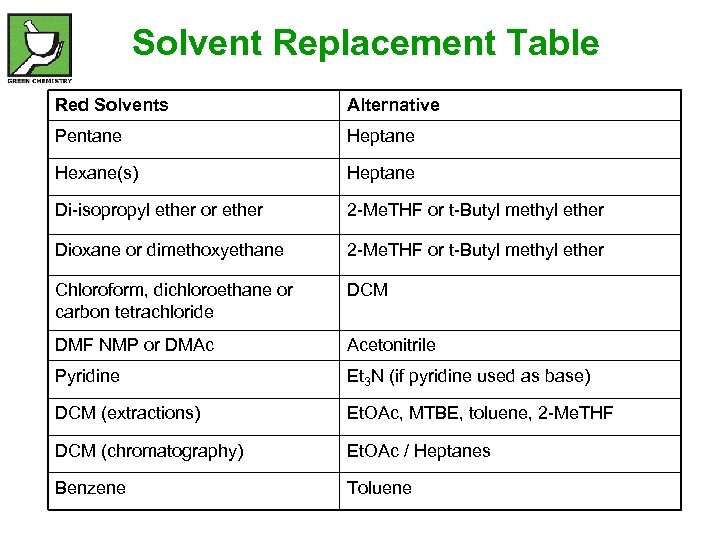 Solvent Replacement Table Red Solvents Alternative Pentane Heptane Hexane(s) Heptane Di-isopropyl ether or ether 2 -Me. THF or t-Butyl methyl ether Dioxane or dimethoxyethane 2 -Me. THF or t-Butyl methyl ether Chloroform, dichloroethane or carbon tetrachloride DCM DMF NMP or DMAc Acetonitrile Pyridine Et 3 N (if pyridine used as base) DCM (extractions) Et. OAc, MTBE, toluene, 2 -Me. THF DCM (chromatography) Et. OAc / Heptanes Benzene Toluene
Solvent Replacement Table Red Solvents Alternative Pentane Heptane Hexane(s) Heptane Di-isopropyl ether or ether 2 -Me. THF or t-Butyl methyl ether Dioxane or dimethoxyethane 2 -Me. THF or t-Butyl methyl ether Chloroform, dichloroethane or carbon tetrachloride DCM DMF NMP or DMAc Acetonitrile Pyridine Et 3 N (if pyridine used as base) DCM (extractions) Et. OAc, MTBE, toluene, 2 -Me. THF DCM (chromatography) Et. OAc / Heptanes Benzene Toluene
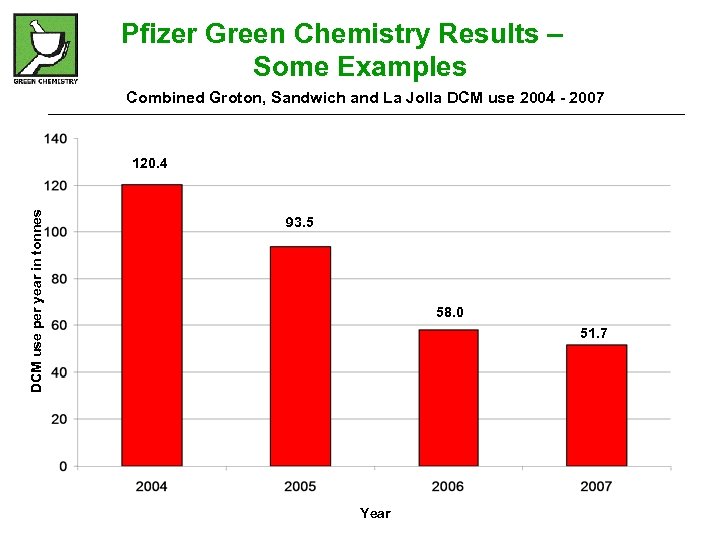 Pfizer Green Chemistry Results – Some Examples Combined Groton, Sandwich and La Jolla DCM use 2004 - 2007 DCM use per year in tonnes 120. 4 93. 5 58. 0 51. 7 Year
Pfizer Green Chemistry Results – Some Examples Combined Groton, Sandwich and La Jolla DCM use 2004 - 2007 DCM use per year in tonnes 120. 4 93. 5 58. 0 51. 7 Year
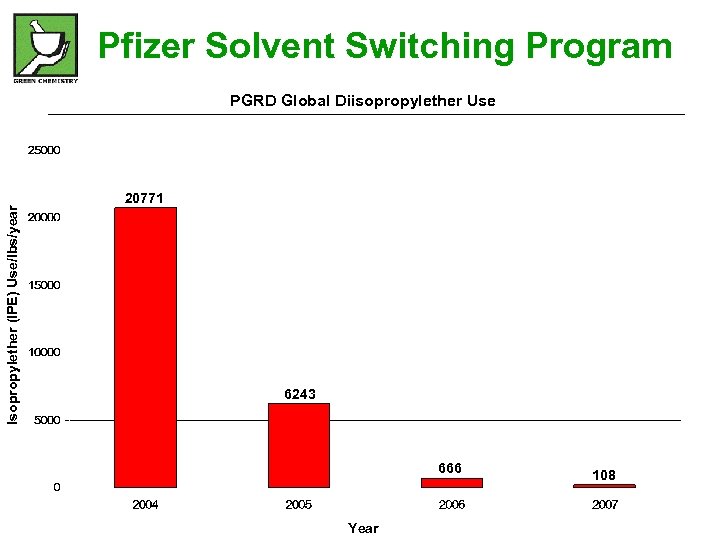 Pfizer Solvent Switching Program Isopropylether (IPE) Use/lbs/year PGRD Global Diisopropylether Use 20771 6243 666 Year 108
Pfizer Solvent Switching Program Isopropylether (IPE) Use/lbs/year PGRD Global Diisopropylether Use 20771 6243 666 Year 108
 Reagent Selection Guide
Reagent Selection Guide
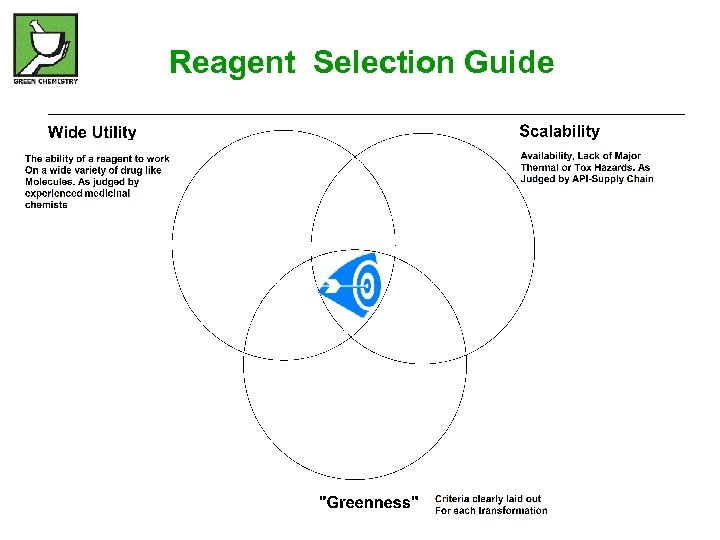 Reagent Selection Guide
Reagent Selection Guide
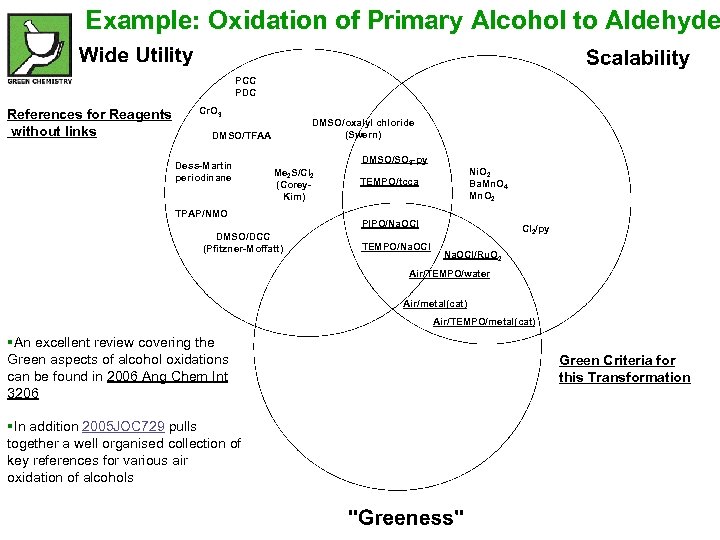 Example: Oxidation of Primary Alcohol to Aldehyde PCC PDC References for Reagents without links Cr. O 3 DMSO/oxalyl chloride (Swern) DMSO/TFAA Dess-Martin periodinane DMSO/SO 3 -py Me 2 S/Cl 2 (Corey. Kim) TPAP/NMO DMSO/DCC (Pfitzner-Moffatt) Ni. O 2 Ba. Mn. O 4 Mn. O 2 TEMPO/tcca PIPO/Na. OCl TEMPO/Na. OCl Cl 2/py Na. OCl/Ru. O 2 Air/TEMPO/water Air/metal(cat) Air/TEMPO/metal(cat) §An excellent review covering the Green aspects of alcohol oxidations can be found in 2006 Ang Chem Int 3206 §In addition 2005 JOC 729 pulls together a well organised collection of key references for various air oxidation of alcohols Green Criteria for this Transformation
Example: Oxidation of Primary Alcohol to Aldehyde PCC PDC References for Reagents without links Cr. O 3 DMSO/oxalyl chloride (Swern) DMSO/TFAA Dess-Martin periodinane DMSO/SO 3 -py Me 2 S/Cl 2 (Corey. Kim) TPAP/NMO DMSO/DCC (Pfitzner-Moffatt) Ni. O 2 Ba. Mn. O 4 Mn. O 2 TEMPO/tcca PIPO/Na. OCl TEMPO/Na. OCl Cl 2/py Na. OCl/Ru. O 2 Air/TEMPO/water Air/metal(cat) Air/TEMPO/metal(cat) §An excellent review covering the Green aspects of alcohol oxidations can be found in 2006 Ang Chem Int 3206 §In addition 2005 JOC 729 pulls together a well organised collection of key references for various air oxidation of alcohols Green Criteria for this Transformation
 § Green Chemistry - Pfizer’s Support and Influence on Academic Research Membership in the ACS GCI Pharmaceutical Roundtable § Let Academics and Govt agencies know of some of the key challenges in Pharmaceutical Manufacturing so they can be addressed (see P. J. Dunn et al. , Green Chemistry, 2007, 9, 411420) § Inform research community, encourage funding agencies. § Selectively fund key research areas (examples include: ) § Amide formation with high economy § Amide reduction (through the Roundtable) § Oxidations without chlorinated solvents § Suzuki reactions without halogenation (through the Roundtable) § Solvent recovery using membrane technology
§ Green Chemistry - Pfizer’s Support and Influence on Academic Research Membership in the ACS GCI Pharmaceutical Roundtable § Let Academics and Govt agencies know of some of the key challenges in Pharmaceutical Manufacturing so they can be addressed (see P. J. Dunn et al. , Green Chemistry, 2007, 9, 411420) § Inform research community, encourage funding agencies. § Selectively fund key research areas (examples include: ) § Amide formation with high economy § Amide reduction (through the Roundtable) § Oxidations without chlorinated solvents § Suzuki reactions without halogenation (through the Roundtable) § Solvent recovery using membrane technology
 Pfizer Green Chemistry - Education § Pfizer believes education is a key to changing behaviors – of present colleagues and future scientists § We … ü Hold GC seminars at all our research sites - by chemists for chemists with prominent chemistry speakers ü Hold GC workshops for university students (St Louis, Connecticut, Puerto-Rico, Ireland, UK) ü Have worked with educational partners to develop a middle school green chemistry (sustainability) curriculum: http: //grogrdapp 66. pfizer. com: 8080/ram/temp_files/2007/Green. Chemistry_6 -12 -07. asx
Pfizer Green Chemistry - Education § Pfizer believes education is a key to changing behaviors – of present colleagues and future scientists § We … ü Hold GC seminars at all our research sites - by chemists for chemists with prominent chemistry speakers ü Hold GC workshops for university students (St Louis, Connecticut, Puerto-Rico, Ireland, UK) ü Have worked with educational partners to develop a middle school green chemistry (sustainability) curriculum: http: //grogrdapp 66. pfizer. com: 8080/ram/temp_files/2007/Green. Chemistry_6 -12 -07. asx
 Pfizer Green Chemistry Results – External Recognition § Institute of Chemical Engineers (IChem. E)- Astra. Zeneca Award “ Excellence in Green Chemistry and Engineering Award” (2006) For Lyrica® revised synthesis – significant reductions in waste by using a enzymatic process, and performing all reaction steps in water § UK Institute of Chemical Engineers (IChem. E) “Crystal Faraday Award for Green Chemical Technology" (2003) For process redesign of Viagra® (sildenafil citrate) – “Sets a new benchmark standard for minimising solvent use in Pharmaceutical Manufacturing” § U. S. Environmental Protection Agency (EPA) “Presidential Green Chemistry Award” (2002) Revised manufacturing process for Zoloft® (sertraline hydrochloride) doubled product yield, and significantly reduced environmental impacts (use of resources, waste minimization)
Pfizer Green Chemistry Results – External Recognition § Institute of Chemical Engineers (IChem. E)- Astra. Zeneca Award “ Excellence in Green Chemistry and Engineering Award” (2006) For Lyrica® revised synthesis – significant reductions in waste by using a enzymatic process, and performing all reaction steps in water § UK Institute of Chemical Engineers (IChem. E) “Crystal Faraday Award for Green Chemical Technology" (2003) For process redesign of Viagra® (sildenafil citrate) – “Sets a new benchmark standard for minimising solvent use in Pharmaceutical Manufacturing” § U. S. Environmental Protection Agency (EPA) “Presidential Green Chemistry Award” (2002) Revised manufacturing process for Zoloft® (sertraline hydrochloride) doubled product yield, and significantly reduced environmental impacts (use of resources, waste minimization)
 Green Chemistry in Process Dev. § Pregabalin (Lyrica®) is a Drug for the treatment of Neuropathic Pain § Launched in the US in September 2005 § Sales $1. 16 billion (2006), $1. 8 billion (2007)
Green Chemistry in Process Dev. § Pregabalin (Lyrica®) is a Drug for the treatment of Neuropathic Pain § Launched in the US in September 2005 § Sales $1. 16 billion (2006), $1. 8 billion (2007)
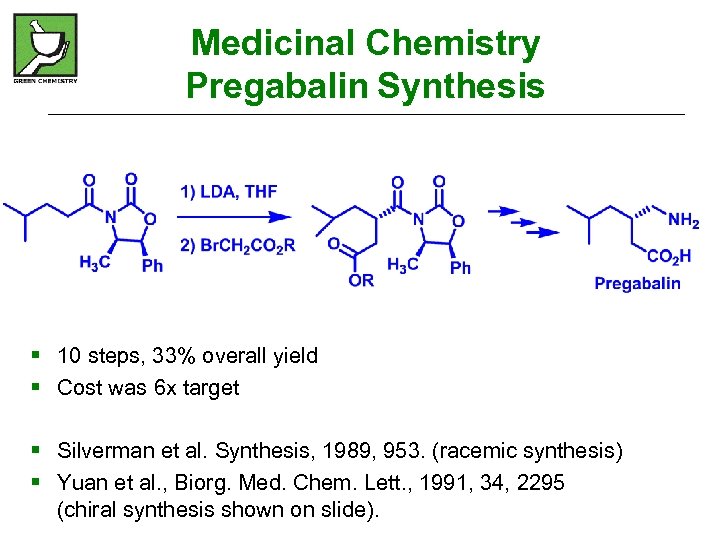 Medicinal Chemistry Pregabalin Synthesis § 10 steps, 33% overall yield § Cost was 6 x target § Silverman et al. Synthesis, 1989, 953. (racemic synthesis) § Yuan et al. , Biorg. Med. Chem. Lett. , 1991, 34, 2295 (chiral synthesis shown on slide).
Medicinal Chemistry Pregabalin Synthesis § 10 steps, 33% overall yield § Cost was 6 x target § Silverman et al. Synthesis, 1989, 953. (racemic synthesis) § Yuan et al. , Biorg. Med. Chem. Lett. , 1991, 34, 2295 (chiral synthesis shown on slide).
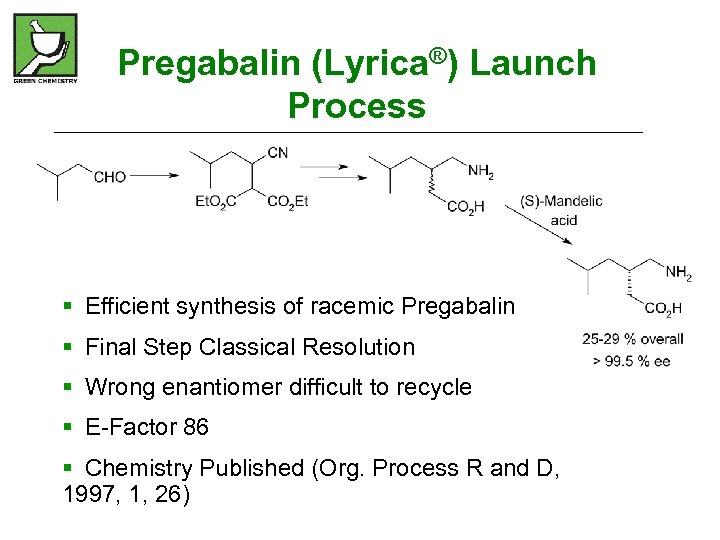 Pregabalin (Lyrica®) Launch Process § Efficient synthesis of racemic Pregabalin § Final Step Classical Resolution § Wrong enantiomer difficult to recycle § E-Factor 86 § Chemistry Published (Org. Process R and D, 1997, 1, 26)
Pregabalin (Lyrica®) Launch Process § Efficient synthesis of racemic Pregabalin § Final Step Classical Resolution § Wrong enantiomer difficult to recycle § E-Factor 86 § Chemistry Published (Org. Process R and D, 1997, 1, 26)
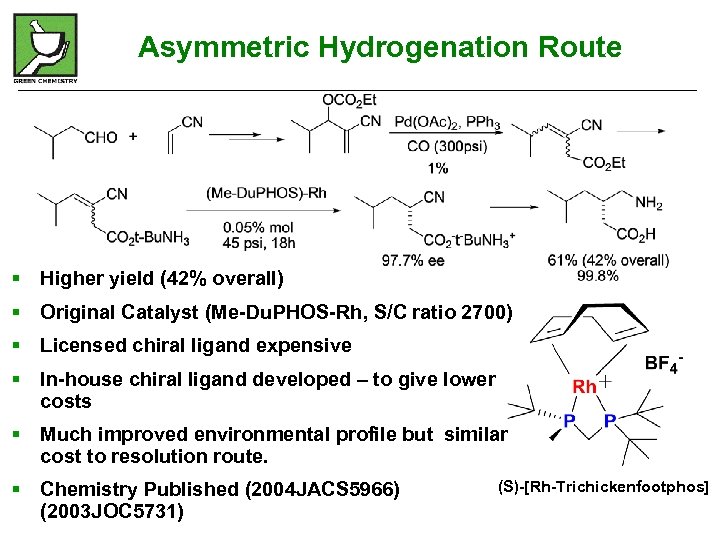 Asymmetric Hydrogenation Route § Higher yield (42% overall) § Original Catalyst (Me-Du. PHOS-Rh, S/C ratio 2700) § Licensed chiral ligand expensive § In-house chiral ligand developed – to give lower costs § Much improved environmental profile but similar cost to resolution route. § Chemistry Published (2004 JACS 5966) (2003 JOC 5731) (S)-[Rh-Trichickenfootphos]
Asymmetric Hydrogenation Route § Higher yield (42% overall) § Original Catalyst (Me-Du. PHOS-Rh, S/C ratio 2700) § Licensed chiral ligand expensive § In-house chiral ligand developed – to give lower costs § Much improved environmental profile but similar cost to resolution route. § Chemistry Published (2004 JACS 5966) (2003 JOC 5731) (S)-[Rh-Trichickenfootphos]
 Enzymatic Resolution of CNDE § Enzymatic hydrolysis of Cyano diester enabled early resolution of chiral center § Enzyme screen revealed 2 (S)-selective hits with E>200: § Thermomyces lanuginosus lipase (Novozymes) § Rhizopus delemar lipase (Amano)
Enzymatic Resolution of CNDE § Enzymatic hydrolysis of Cyano diester enabled early resolution of chiral center § Enzyme screen revealed 2 (S)-selective hits with E>200: § Thermomyces lanuginosus lipase (Novozymes) § Rhizopus delemar lipase (Amano)
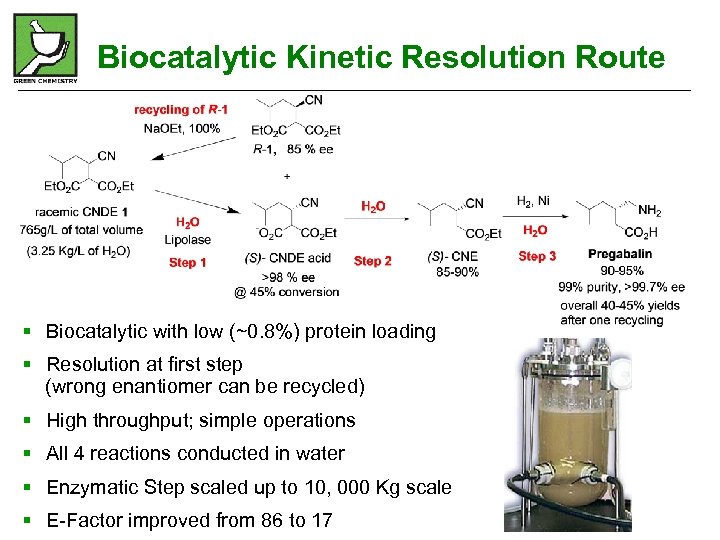 Biocatalytic Kinetic Resolution Route § Biocatalytic with low (~0. 8%) protein loading § Resolution at first step (wrong enantiomer can be recycled) § High throughput; simple operations § All 4 reactions conducted in water § Enzymatic Step scaled up to 10, 000 Kg scale § E-Factor improved from 86 to 17
Biocatalytic Kinetic Resolution Route § Biocatalytic with low (~0. 8%) protein loading § Resolution at first step (wrong enantiomer can be recycled) § High throughput; simple operations § All 4 reactions conducted in water § Enzymatic Step scaled up to 10, 000 Kg scale § E-Factor improved from 86 to 17
 Comparison of Pregabalin Processes § Chemoenzymatic route uses >5 x less inputs than 1 st generation route
Comparison of Pregabalin Processes § Chemoenzymatic route uses >5 x less inputs than 1 st generation route
 Pregabalin Synthetic Improvements § By replacing all reaction solvents with water, bringing the Resolution to the beginning, and the Raney nickel reduction to the end, the proposed improvements will yield annual improvements of: § Starting material usage reduction of 800 tons § Solvent reductions: § Methanol 1 million gallons § Ethanol 0. 4 million gallons § Tetrahydrofuran 2. 2 million gallons § Isopropanol 2 million gallons § Mandelic Acid usage eliminated – 500 tons
Pregabalin Synthetic Improvements § By replacing all reaction solvents with water, bringing the Resolution to the beginning, and the Raney nickel reduction to the end, the proposed improvements will yield annual improvements of: § Starting material usage reduction of 800 tons § Solvent reductions: § Methanol 1 million gallons § Ethanol 0. 4 million gallons § Tetrahydrofuran 2. 2 million gallons § Isopropanol 2 million gallons § Mandelic Acid usage eliminated – 500 tons
 Pregabalin Summary § § § § § Launched in the US in September 2005 Treatment of Neuropathic pain Sales in 2006 $ 1. 16 billion Sales in 2007 $ 1. 8 billion New enzymatic chemistry successfully scaled up to 10 tonnes scale. Process was switched to the enzymatic route in 3 Q 2006 By making the switch to optimal route very early in the product lifetime, Pfizer ensures close to maximum benefits to the environment. Chemistry has been published Martinez et al. (OPRD, 2008, 11, 392). In 2006 Pfizer received the Astra. Zeneca Award for Excellence in Green Chemistry and Engineering for its work on Pregabalin.
Pregabalin Summary § § § § § Launched in the US in September 2005 Treatment of Neuropathic pain Sales in 2006 $ 1. 16 billion Sales in 2007 $ 1. 8 billion New enzymatic chemistry successfully scaled up to 10 tonnes scale. Process was switched to the enzymatic route in 3 Q 2006 By making the switch to optimal route very early in the product lifetime, Pfizer ensures close to maximum benefits to the environment. Chemistry has been published Martinez et al. (OPRD, 2008, 11, 392). In 2006 Pfizer received the Astra. Zeneca Award for Excellence in Green Chemistry and Engineering for its work on Pregabalin.
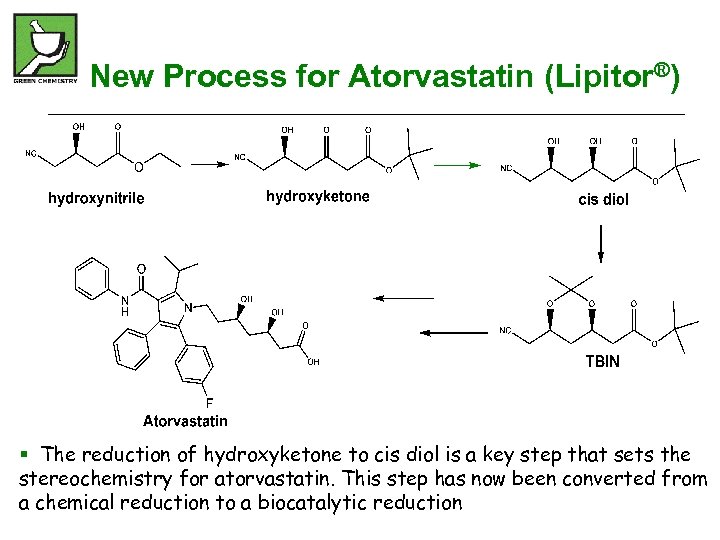 New Process for Atorvastatin (Lipitor®) § The reduction of hydroxyketone to cis diol is a key step that sets the stereochemistry for atorvastatin. This step has now been converted from a chemical reduction to a biocatalytic reduction
New Process for Atorvastatin (Lipitor®) § The reduction of hydroxyketone to cis diol is a key step that sets the stereochemistry for atorvastatin. This step has now been converted from a chemical reduction to a biocatalytic reduction
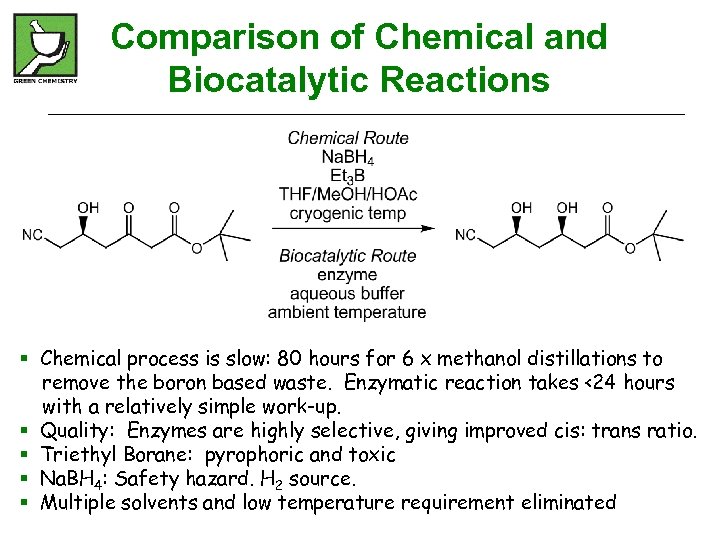 Comparison of Chemical and Biocatalytic Reactions § Chemical process is slow: 80 hours for 6 x methanol distillations to remove the boron based waste. Enzymatic reaction takes <24 hours with a relatively simple work-up. § Quality: Enzymes are highly selective, giving improved cis: trans ratio. § Triethyl Borane: pyrophoric and toxic § Na. BH 4: Safety hazard. H 2 source. § Multiple solvents and low temperature requirement eliminated
Comparison of Chemical and Biocatalytic Reactions § Chemical process is slow: 80 hours for 6 x methanol distillations to remove the boron based waste. Enzymatic reaction takes <24 hours with a relatively simple work-up. § Quality: Enzymes are highly selective, giving improved cis: trans ratio. § Triethyl Borane: pyrophoric and toxic § Na. BH 4: Safety hazard. H 2 source. § Multiple solvents and low temperature requirement eliminated
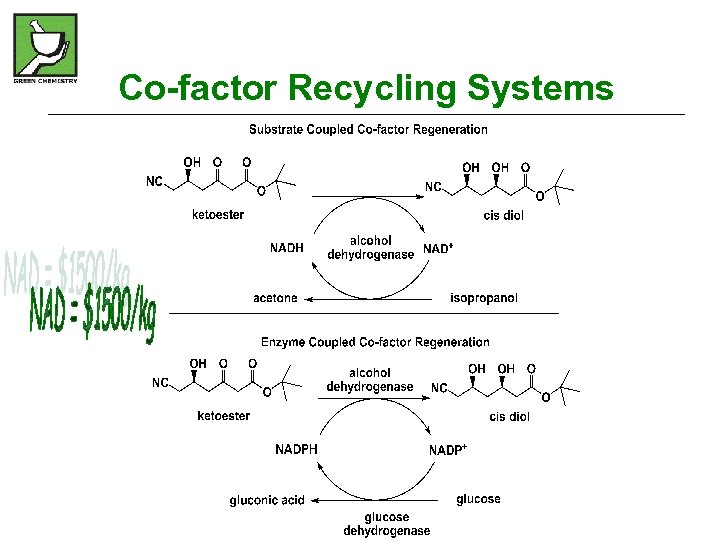 Co-factor Recycling Systems
Co-factor Recycling Systems
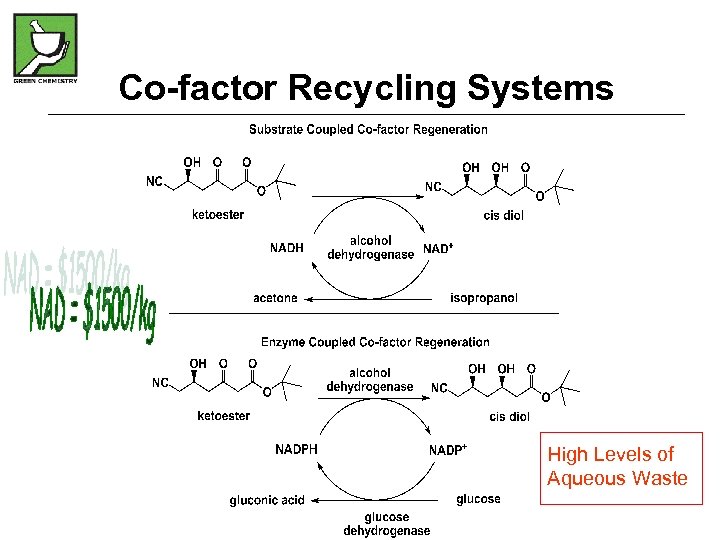 Co-factor Recycling Systems High Levels of Aqueous Waste
Co-factor Recycling Systems High Levels of Aqueous Waste
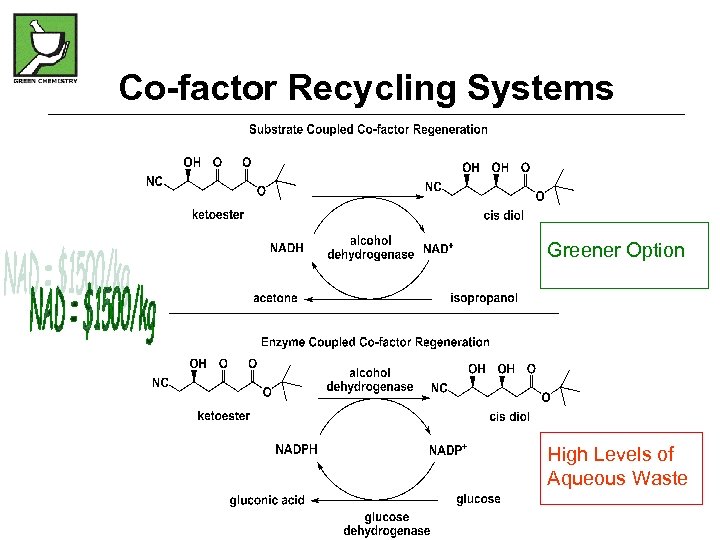 Co-factor Recycling Systems Greener Option High Levels of Aqueous Waste
Co-factor Recycling Systems Greener Option High Levels of Aqueous Waste
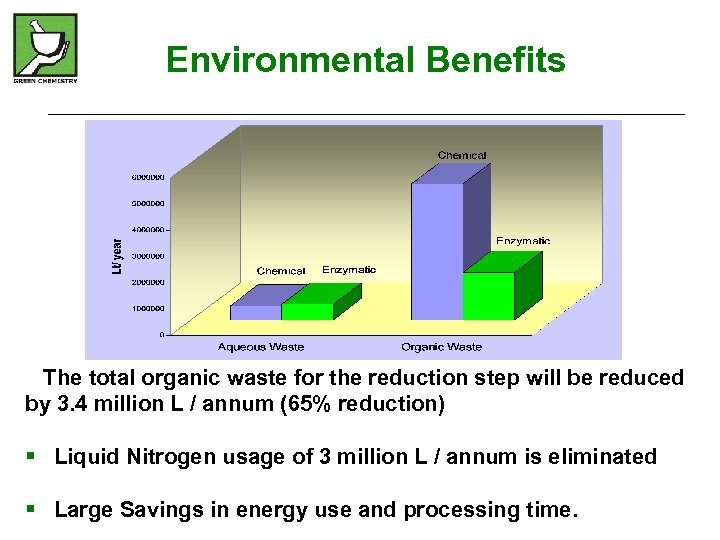 Environmental Benefits The total organic waste for the reduction step will be reduced by 3. 4 million L / annum (65% reduction) § Liquid Nitrogen usage of 3 million L / annum is eliminated § Large Savings in energy use and processing time.
Environmental Benefits The total organic waste for the reduction step will be reduced by 3. 4 million L / annum (65% reduction) § Liquid Nitrogen usage of 3 million L / annum is eliminated § Large Savings in energy use and processing time.
 Where do we go from here ? § Aggressively pursue ultra low E-Factors for our high volume products (especially Celebrex®, Lyrica®, Atorvastatin®). § Use a Metrics based system so that all new commercial products meet a good “dignity level” of environmental performance. § Continue our successful work in minimising the environmental footprint to discover drugs. § Continue with our external education work promoting Green Chemistry.
Where do we go from here ? § Aggressively pursue ultra low E-Factors for our high volume products (especially Celebrex®, Lyrica®, Atorvastatin®). § Use a Metrics based system so that all new commercial products meet a good “dignity level” of environmental performance. § Continue our successful work in minimising the environmental footprint to discover drugs. § Continue with our external education work promoting Green Chemistry.
 Thanks and Acknowledgment § Pregabalin § Enzyme Chemistry –C. Martinez, S. Hu, J, Tao, P. Kellerher § Asymmetric Hydrogenation – G. Hoge, W. Kissel § Energy Calculations – Kevin Hettenbach §Lipitor § D. Bauer, M. Burns, A. Denhole, A. Fahy, C. Healy, O’Shaughnessy, E, Maitiu, F. Stomeo, G. Wittaker, J. Wong § IEP, (Wiesbarden, Germany) § 60 members of the Pfizer Green Chemistry teams § To our partners in education and research § To YOU – today’s audience!
Thanks and Acknowledgment § Pregabalin § Enzyme Chemistry –C. Martinez, S. Hu, J, Tao, P. Kellerher § Asymmetric Hydrogenation – G. Hoge, W. Kissel § Energy Calculations – Kevin Hettenbach §Lipitor § D. Bauer, M. Burns, A. Denhole, A. Fahy, C. Healy, O’Shaughnessy, E, Maitiu, F. Stomeo, G. Wittaker, J. Wong § IEP, (Wiesbarden, Germany) § 60 members of the Pfizer Green Chemistry teams § To our partners in education and research § To YOU – today’s audience!


