f1f9234275c2d94542664de818a202e4.ppt
- Количество слайдов: 42
 Géntherápia: Még mindig csak álom vagy realitás? Molnár Mária Judit Molekuláris Neurológiai Klinikai és Kutatási Központ Semmelweis Egyetem Magyar Személyre Szabott Medicina Társaság 2. Konferenciája 2011. 09. 23. -24.
Géntherápia: Még mindig csak álom vagy realitás? Molnár Mária Judit Molekuláris Neurológiai Klinikai és Kutatási Központ Semmelweis Egyetem Magyar Személyre Szabott Medicina Társaság 2. Konferenciája 2011. 09. 23. -24.
 The history of the gene therapy 1970 1977 1990 1999 2000 2006 2009 - S. Rogers: gene therapy to treat 2 sisters whith argininemia A gene was successfully delivered into mammalian cells First human gene therapy was approved: SCID J. Gelsinger with OTC deficiency died from organ failure after gene therapy A. Fischer cured children with SCID using retroviral vector, 2 of the children developed leukemia. FDA halted the use of retroviruses in the US Patients was successfully treated with metastatic melanoma using killer T cells genetically retargeted to attack the cancer cells Succesfull gene-based th. for the treatment of HIV: lentiviral vector for delivery of an antisense gene againts HIV envelope Researchers succeeded at halting adrenoleukodystrophy, using a vector derived from HIV to deliver the gene for the missing enzyme
The history of the gene therapy 1970 1977 1990 1999 2000 2006 2009 - S. Rogers: gene therapy to treat 2 sisters whith argininemia A gene was successfully delivered into mammalian cells First human gene therapy was approved: SCID J. Gelsinger with OTC deficiency died from organ failure after gene therapy A. Fischer cured children with SCID using retroviral vector, 2 of the children developed leukemia. FDA halted the use of retroviruses in the US Patients was successfully treated with metastatic melanoma using killer T cells genetically retargeted to attack the cancer cells Succesfull gene-based th. for the treatment of HIV: lentiviral vector for delivery of an antisense gene againts HIV envelope Researchers succeeded at halting adrenoleukodystrophy, using a vector derived from HIV to deliver the gene for the missing enzyme
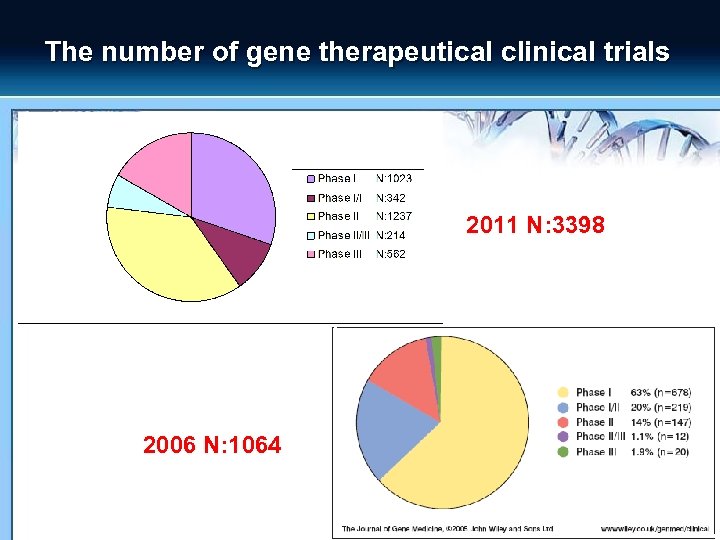 The number of gene therapeutical clinical trials 2011 N: 3398 2006 N: 1064
The number of gene therapeutical clinical trials 2011 N: 3398 2006 N: 1064
 General applications of therapeutic gene transfer 1. Molecular therapy for genetic diseases 2. Establishment of a stable gene reservoir as a source of therapeutic proteins in non - genetic diseases 3. To increase the tumor cells sensitivity to drugs 4. Destruction of malignant cells in neoplasias 5. DNA vaccines
General applications of therapeutic gene transfer 1. Molecular therapy for genetic diseases 2. Establishment of a stable gene reservoir as a source of therapeutic proteins in non - genetic diseases 3. To increase the tumor cells sensitivity to drugs 4. Destruction of malignant cells in neoplasias 5. DNA vaccines
 Therapeutic options in genetic disorders MUTANT GENE 1>> MUTANT RNA 2 > > DEFECTIVE PROTEIN 3 PHENOTYPE 4 Therapeutic targeting of either 1+2+3+4: Classical Pharmacology Therapeutic targeting of 1 : DNA editing, Gene transfer, Gene replacement Therapeutic targeting of 2 : Exon skipping, translational read-through, si. RNA Therapeutic targeting of 3 : Protein replacement, Protein expression Therapeutic targeting of 4 : Pharmacology, physiotherapy, surgery Targeting miscalleneous items : Enhancement of regeneration, tissue replacement by cell therapy
Therapeutic options in genetic disorders MUTANT GENE 1>> MUTANT RNA 2 > > DEFECTIVE PROTEIN 3 PHENOTYPE 4 Therapeutic targeting of either 1+2+3+4: Classical Pharmacology Therapeutic targeting of 1 : DNA editing, Gene transfer, Gene replacement Therapeutic targeting of 2 : Exon skipping, translational read-through, si. RNA Therapeutic targeting of 3 : Protein replacement, Protein expression Therapeutic targeting of 4 : Pharmacology, physiotherapy, surgery Targeting miscalleneous items : Enhancement of regeneration, tissue replacement by cell therapy
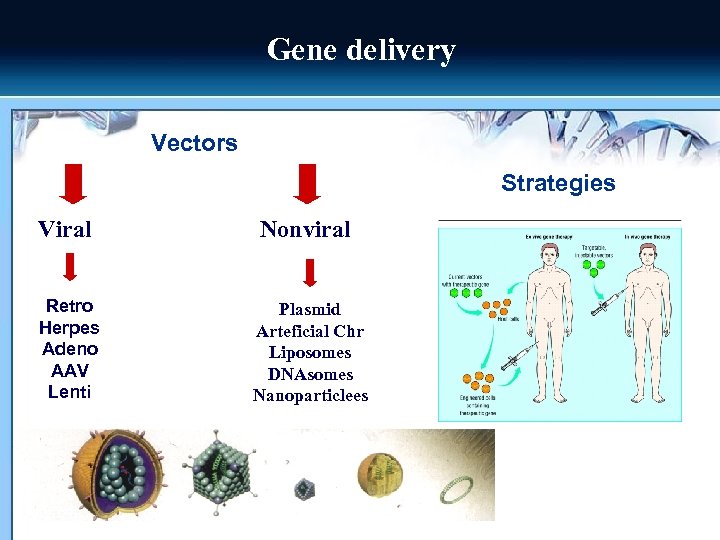 Gene delivery Vectors Strategies Viral Nonviral Retro Herpes Adeno AAV Lenti Plasmid Arteficial Chr Liposomes DNAsomes Nanoparticlees
Gene delivery Vectors Strategies Viral Nonviral Retro Herpes Adeno AAV Lenti Plasmid Arteficial Chr Liposomes DNAsomes Nanoparticlees
 Molecular therapies DNA modulating therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy mt. DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides § Mutant RNA removal § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
Molecular therapies DNA modulating therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy mt. DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides § Mutant RNA removal § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
 Clinical trials with DNA modulation (using viral vectors) § Adrenoleukodystrophy § Alpha Sarcoglycan Deficiency § Gamma Sarcoglycan Deficiency § Late Infantile Neuronal Ceroid Lipofuscinosis: AAVrh 10. - CLN 2 gene Phase I/II § Pompe Disease: r. AAV 1 -CMV-GAA phase I/ II
Clinical trials with DNA modulation (using viral vectors) § Adrenoleukodystrophy § Alpha Sarcoglycan Deficiency § Gamma Sarcoglycan Deficiency § Late Infantile Neuronal Ceroid Lipofuscinosis: AAVrh 10. - CLN 2 gene Phase I/II § Pompe Disease: r. AAV 1 -CMV-GAA phase I/ II
 Adrenoleukodystrophy - 2009 Ex vivo viral correction § § § § Bone marrow stem cells - CD 34+ cells isolated Ex vivo reactivation with cytokine míxture Transfection by lentivirus containing a normal copy of the ABCD 1 gene Patients were treated with CYC and busulfan so that their hemopoetic stem cells containing the ABCD 1 mutation were greatly diminished. Patients were then infused with their own CD 34+ cells engineered to contain normal copies of the ABCD 1 gene The engineered CD 34+ can differentiate into a vast number of different blood cell types. They turned into microglial cells and corrected the deficiencies in the brain responsible for X-ALD Aubourg and Cartier-Lacave 2009
Adrenoleukodystrophy - 2009 Ex vivo viral correction § § § § Bone marrow stem cells - CD 34+ cells isolated Ex vivo reactivation with cytokine míxture Transfection by lentivirus containing a normal copy of the ABCD 1 gene Patients were treated with CYC and busulfan so that their hemopoetic stem cells containing the ABCD 1 mutation were greatly diminished. Patients were then infused with their own CD 34+ cells engineered to contain normal copies of the ABCD 1 gene The engineered CD 34+ can differentiate into a vast number of different blood cell types. They turned into microglial cells and corrected the deficiencies in the brain responsible for X-ALD Aubourg and Cartier-Lacave 2009
 Gamma-Sarco. Glycan Restoration in LGMD 2 C - Phase I Benveniste et al. AAN 2011. 04. 13. § § § Mid-to-high dosages of gamma-sarcoglycan (SGC) genes in AAV 1 9 participants, im injection extensor carpi radialis muscle – The first 2 groups received a single injection – The 2. group received a higher dose than the first group – The 3. group received 3 injections for the highest total dose. The therapy was well tolerated In biopsy specimens 30 days after the inj. showed SGC expression in 5/9 patients, with 4. 7 to 10. 5% of positively stained fibers and detection of SGC m. RNA by RT-PCR in the 3 patients who received the highest dose. The SGC protein became also detectable by Western blot in one patient All patients seropositive for AAV 1, 1 developed a cytotoxic response again AAV 1 capsid
Gamma-Sarco. Glycan Restoration in LGMD 2 C - Phase I Benveniste et al. AAN 2011. 04. 13. § § § Mid-to-high dosages of gamma-sarcoglycan (SGC) genes in AAV 1 9 participants, im injection extensor carpi radialis muscle – The first 2 groups received a single injection – The 2. group received a higher dose than the first group – The 3. group received 3 injections for the highest total dose. The therapy was well tolerated In biopsy specimens 30 days after the inj. showed SGC expression in 5/9 patients, with 4. 7 to 10. 5% of positively stained fibers and detection of SGC m. RNA by RT-PCR in the 3 patients who received the highest dose. The SGC protein became also detectable by Western blot in one patient All patients seropositive for AAV 1, 1 developed a cytotoxic response again AAV 1 capsid
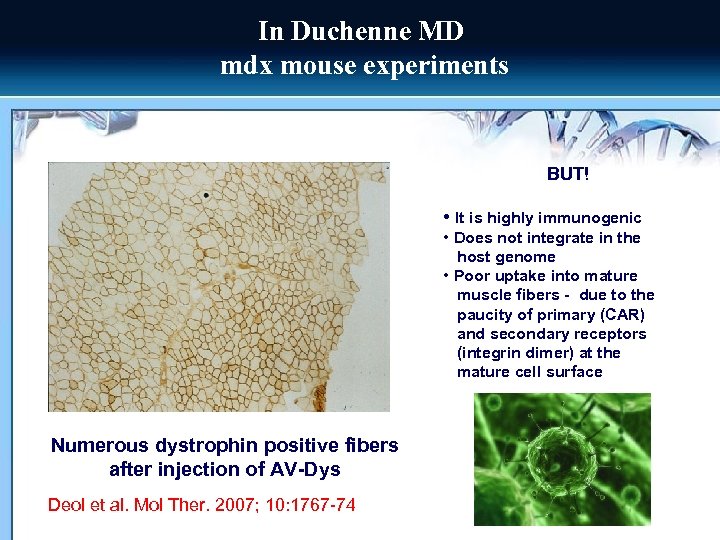 In Duchenne MD mdx mouse experiments BUT! • It is highly immunogenic • Does not integrate in the host genome • Poor uptake into mature muscle fibers - due to the paucity of primary (CAR) and secondary receptors (integrin dimer) at the mature cell surface Numerous dystrophin positive fibers after injection of AV-Dys Deol et al. Mol Ther. 2007; 10: 1767 -74
In Duchenne MD mdx mouse experiments BUT! • It is highly immunogenic • Does not integrate in the host genome • Poor uptake into mature muscle fibers - due to the paucity of primary (CAR) and secondary receptors (integrin dimer) at the mature cell surface Numerous dystrophin positive fibers after injection of AV-Dys Deol et al. Mol Ther. 2007; 10: 1767 -74
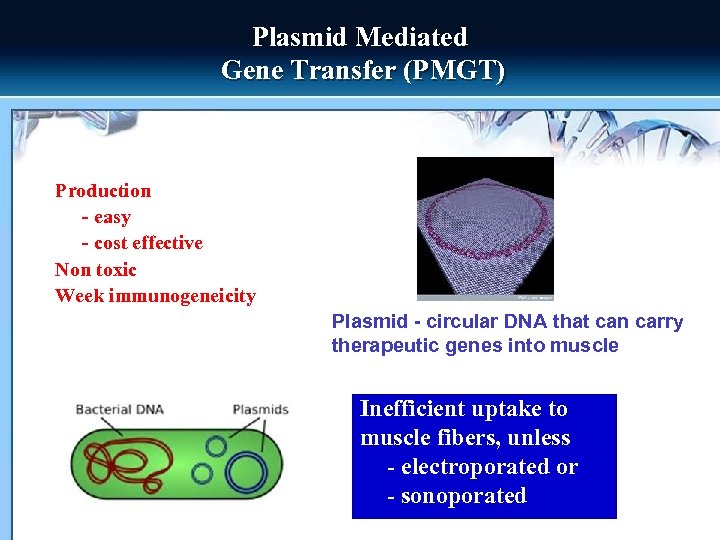 Plasmid Mediated Gene Transfer (PMGT) Production - easy - cost effective Non toxic Week immunogeneicity Plasmid - circular DNA that can carry therapeutic genes into muscle Inefficient uptake to muscle fibers, unless - electroporated or - sonoporated
Plasmid Mediated Gene Transfer (PMGT) Production - easy - cost effective Non toxic Week immunogeneicity Plasmid - circular DNA that can carry therapeutic genes into muscle Inefficient uptake to muscle fibers, unless - electroporated or - sonoporated
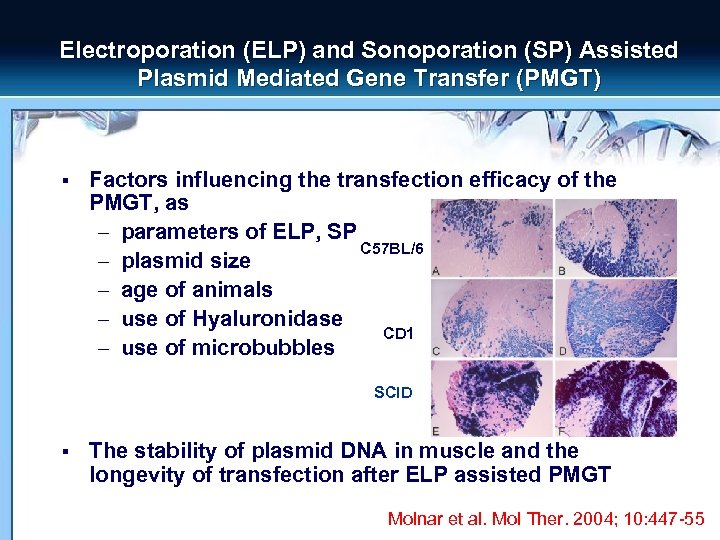 Electroporation (ELP) and Sonoporation (SP) Assisted Plasmid Mediated Gene Transfer (PMGT) § Factors influencing the transfection efficacy of the PMGT, as – parameters of ELP, SP C 57 BL/6 – plasmid size – age of animals – use of Hyaluronidase CD 1 – use of microbubbles SCID § The stability of plasmid DNA in muscle and the longevity of transfection after ELP assisted PMGT Molnar et al. Mol Ther. 2004; 10: 447 -55
Electroporation (ELP) and Sonoporation (SP) Assisted Plasmid Mediated Gene Transfer (PMGT) § Factors influencing the transfection efficacy of the PMGT, as – parameters of ELP, SP C 57 BL/6 – plasmid size – age of animals – use of Hyaluronidase CD 1 – use of microbubbles SCID § The stability of plasmid DNA in muscle and the longevity of transfection after ELP assisted PMGT Molnar et al. Mol Ther. 2004; 10: 447 -55
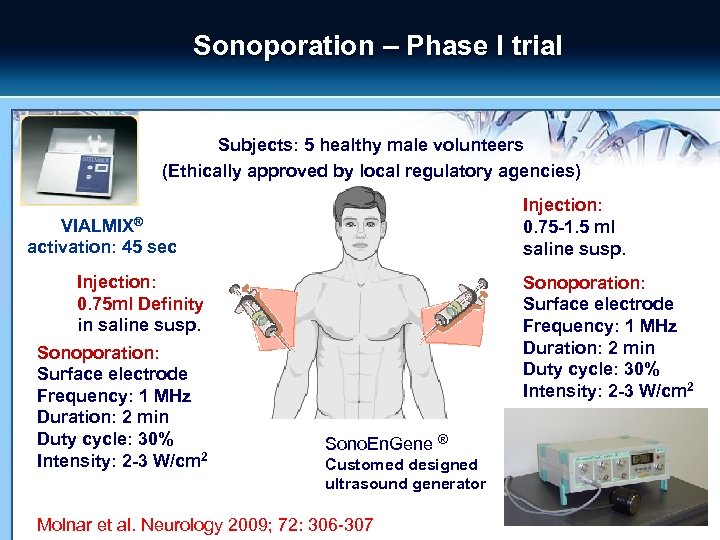 Sonoporation – Phase I trial Subjects: 5 healthy male volunteers (Ethically approved by local regulatory agencies) Injection: 0. 75 -1. 5 ml saline susp. VIALMIX® activation: 45 sec Injection: 0. 75 ml Definity in saline susp. Sonoporation: Surface electrode Frequency: 1 MHz Duration: 2 min Duty cycle: 30% Intensity: 2 -3 W/cm 2 Sono. En. Gene ® Customed designed ultrasound generator Molnar et al. Neurology 2009; 72: 306 -307
Sonoporation – Phase I trial Subjects: 5 healthy male volunteers (Ethically approved by local regulatory agencies) Injection: 0. 75 -1. 5 ml saline susp. VIALMIX® activation: 45 sec Injection: 0. 75 ml Definity in saline susp. Sonoporation: Surface electrode Frequency: 1 MHz Duration: 2 min Duty cycle: 30% Intensity: 2 -3 W/cm 2 Sono. En. Gene ® Customed designed ultrasound generator Molnar et al. Neurology 2009; 72: 306 -307
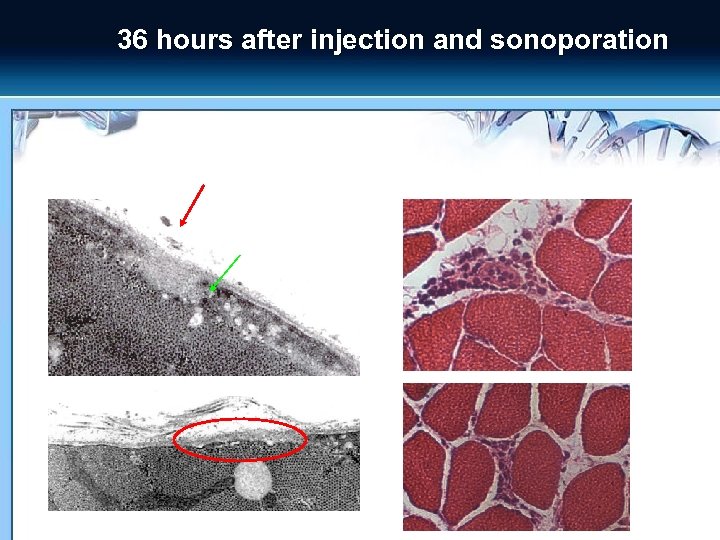 36 hours after injection and sonoporation
36 hours after injection and sonoporation
 Molecular therapies DNA modulating therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides Mutant RNA removal Inactivation of the mutant m. RNA by RNAi Destruction of the mutant m. RNA by ribozymes Translational manipulation § § Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
Molecular therapies DNA modulating therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides Mutant RNA removal Inactivation of the mutant m. RNA by RNAi Destruction of the mutant m. RNA by ribozymes Translational manipulation § § Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
 EXON SKIPPING § § § A mutation-specific therapy Providing personalized medicine Simultaneously may correct all isoforms Maintains the original tissue-specific gene regulation The antisense compounds inducing exon skipping are small synthetic, and highly sequence-specific - 2 -O-methyl phosphorothioate (2 OMe) - phosphorodiamidate morpholino oligomers PMO. - peptide-linked PMO
EXON SKIPPING § § § A mutation-specific therapy Providing personalized medicine Simultaneously may correct all isoforms Maintains the original tissue-specific gene regulation The antisense compounds inducing exon skipping are small synthetic, and highly sequence-specific - 2 -O-methyl phosphorothioate (2 OMe) - phosphorodiamidate morpholino oligomers PMO. - peptide-linked PMO
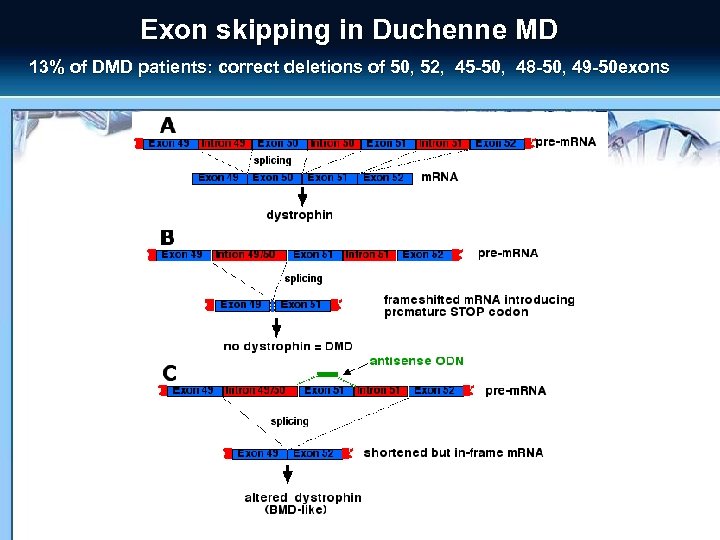 Exon skipping in Duchenne MD 13% of DMD patients: correct deletions of 50, 52, 45 -50, 48 -50, 49 -50 exons
Exon skipping in Duchenne MD 13% of DMD patients: correct deletions of 50, 52, 45 -50, 48 -50, 49 -50 exons
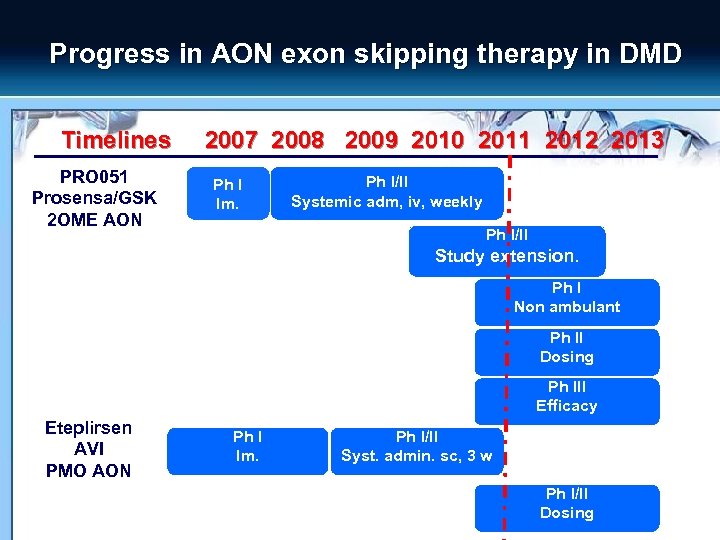 Progress in AON exon skipping therapy in DMD Timelines PRO 051 Prosensa/GSK 2 OME AON 2007 2008 2009 2010 2011 2012 2013 Ph I Im. Ph I/II Systemic adm, iv, weekly Ph I/II Study extension. Ph I Non ambulant Ph II Dosing Ph III Efficacy Eteplirsen AVI PMO AON Ph I Im. Ph I/II Syst. admin. sc, 3 w Ph I/II Dosing
Progress in AON exon skipping therapy in DMD Timelines PRO 051 Prosensa/GSK 2 OME AON 2007 2008 2009 2010 2011 2012 2013 Ph I Im. Ph I/II Systemic adm, iv, weekly Ph I/II Study extension. Ph I Non ambulant Ph II Dosing Ph III Efficacy Eteplirsen AVI PMO AON Ph I Im. Ph I/II Syst. admin. sc, 3 w Ph I/II Dosing
 Systemic administration of PRO 051 in DMD § § § § Weekly sc inj. for 5 weeks in 12 patients, with 4 doses (0. 5, 2, 4, 6 mg/kg) Patients entered a 12 -week open-label extension phase, during which they all received PRO 051 at a dose of 6. 0 mg/kg/ week RNA splicing and protein levels in the tibial anterior muscle were assessed The most common adverse events § irritation at the administration site § mild proteinuria and increased urinary α(1)-microglobulin levels; PRO 051 induced spec. exon-51 skipping at doses of 2 mg/kg or more New dystrophin expression was observed between cc. 60% - 100% of muscle fibers in 10 of the 12 patients, which increased in a dosedependent manner After the 12 -week extension phase, there was improvement of 35. 2± 28. 7 m (from the baseline of 384± 121 m) on the 6 -minute walk test 48 week extension study: no improvement in 6 MWD Goemans et al N. Engl J Med 2011; 364(16): 1513 -22. Goemens et al. AAN, 2011. 04. 13
Systemic administration of PRO 051 in DMD § § § § Weekly sc inj. for 5 weeks in 12 patients, with 4 doses (0. 5, 2, 4, 6 mg/kg) Patients entered a 12 -week open-label extension phase, during which they all received PRO 051 at a dose of 6. 0 mg/kg/ week RNA splicing and protein levels in the tibial anterior muscle were assessed The most common adverse events § irritation at the administration site § mild proteinuria and increased urinary α(1)-microglobulin levels; PRO 051 induced spec. exon-51 skipping at doses of 2 mg/kg or more New dystrophin expression was observed between cc. 60% - 100% of muscle fibers in 10 of the 12 patients, which increased in a dosedependent manner After the 12 -week extension phase, there was improvement of 35. 2± 28. 7 m (from the baseline of 384± 121 m) on the 6 -minute walk test 48 week extension study: no improvement in 6 MWD Goemans et al N. Engl J Med 2011; 364(16): 1513 -22. Goemens et al. AAN, 2011. 04. 13
 Exon skipping trial in DMD with AVI-4658 (eteplirsen) • Eteplirsen was delivered iv. to 19 DMD boys with mutations in the dystrophin gene that can potentially benefit from skipping of exon 51 • 12 weekly infusion (0· 5, 1, 2, 4, 10, 20 mg/kg) • Participants tolerated all doses well • Muscle biopsies were performed at baseline and at 14 weeks • Patients were followed for 26 weeks Cirak et al Lancet 2011 Aug; 378(9791): 595 -605
Exon skipping trial in DMD with AVI-4658 (eteplirsen) • Eteplirsen was delivered iv. to 19 DMD boys with mutations in the dystrophin gene that can potentially benefit from skipping of exon 51 • 12 weekly infusion (0· 5, 1, 2, 4, 10, 20 mg/kg) • Participants tolerated all doses well • Muscle biopsies were performed at baseline and at 14 weeks • Patients were followed for 26 weeks Cirak et al Lancet 2011 Aug; 378(9791): 595 -605
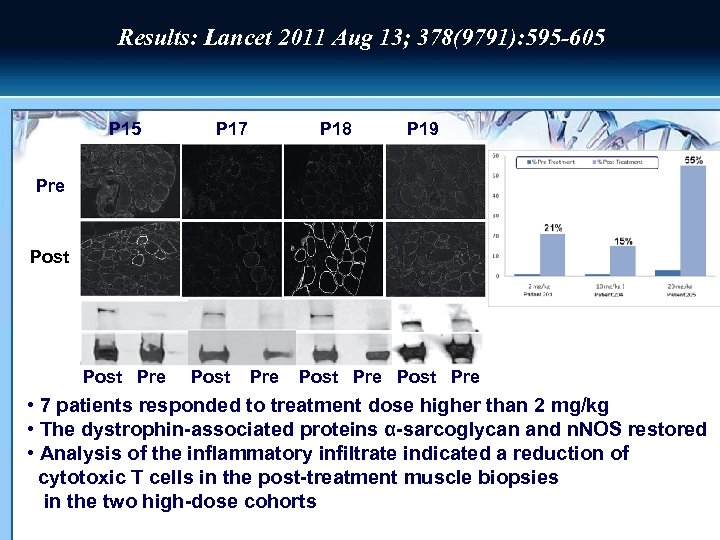 Results: Lancet 2011 Aug 13; 378(9791): 595 -605 P 15 Pre P 15 P 17 P 18 P 17 P 19 P 18 Post Pre Post Pre • 7 patients responded to treatment dose higher than 2 mg/kg • The dystrophin-associated proteins α-sarcoglycan and n. NOS restored • Analysis of the inflammatory infiltrate indicated a reduction of cytotoxic T cells in the post-treatment muscle biopsies in the two high-dose cohorts
Results: Lancet 2011 Aug 13; 378(9791): 595 -605 P 15 Pre P 15 P 17 P 18 P 17 P 19 P 18 Post Pre Post Pre • 7 patients responded to treatment dose higher than 2 mg/kg • The dystrophin-associated proteins α-sarcoglycan and n. NOS restored • Analysis of the inflammatory infiltrate indicated a reduction of cytotoxic T cells in the post-treatment muscle biopsies in the two high-dose cohorts
 Molecular therapies DNA modulating therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides § Mutant RNA removal § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
Molecular therapies DNA modulating therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides § Mutant RNA removal § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
 si. RNA therapy - Phase I trial – ALN-TTR 01 iv. Transthyretrin mediated amyloidosis § § § § ALN-TTR blocks pathogenic accumulations of mutant TTR in peripheral tissues (95% reduction of V 30 M h. TTR deposition) Delivery next generation liponanoparticle (LNP) 2010 -2012 Indication: FAP, FAC Safety study 26 patients or carriers Monitoring serum TTR level
si. RNA therapy - Phase I trial – ALN-TTR 01 iv. Transthyretrin mediated amyloidosis § § § § ALN-TTR blocks pathogenic accumulations of mutant TTR in peripheral tissues (95% reduction of V 30 M h. TTR deposition) Delivery next generation liponanoparticle (LNP) 2010 -2012 Indication: FAP, FAC Safety study 26 patients or carriers Monitoring serum TTR level
 Molecular therapies DNA modulated therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides § Mutant RNA removal § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
Molecular therapies DNA modulated therapies § Gene replacement – to replace the defected gene § Gene transfer – to upregulate therapeutic protein § Gene editing – to correct the defected gene § Gene shifting – to upregulate healthy DNA molecules, change ratio of heteroplasmy RNA modulating therapies § Exon skipping by antisense oligonucleotides § Exon inclusion by antisense oligonucletotides § Mutant RNA removal § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
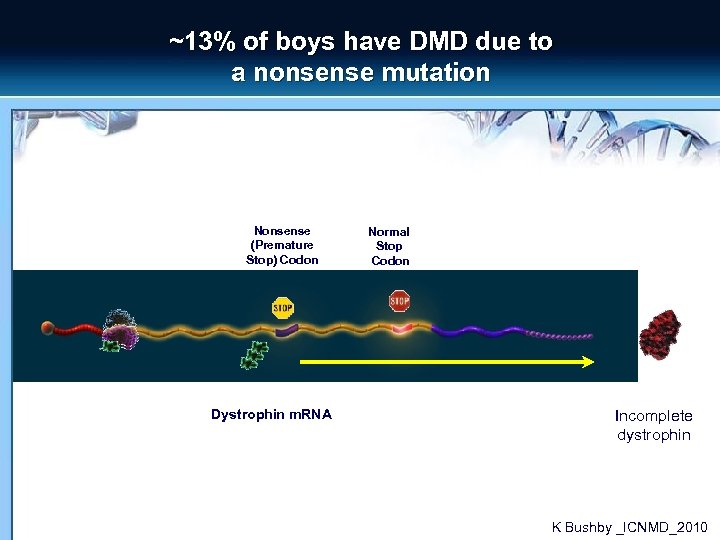 ~13% of boys have DMD due to a nonsense mutation Nonsense (Premature Stop) Codon Dystrophin m. RNA Normal Stop Codon Incomplete dystrophin K Bushby _ICNMD_2010
~13% of boys have DMD due to a nonsense mutation Nonsense (Premature Stop) Codon Dystrophin m. RNA Normal Stop Codon Incomplete dystrophin K Bushby _ICNMD_2010
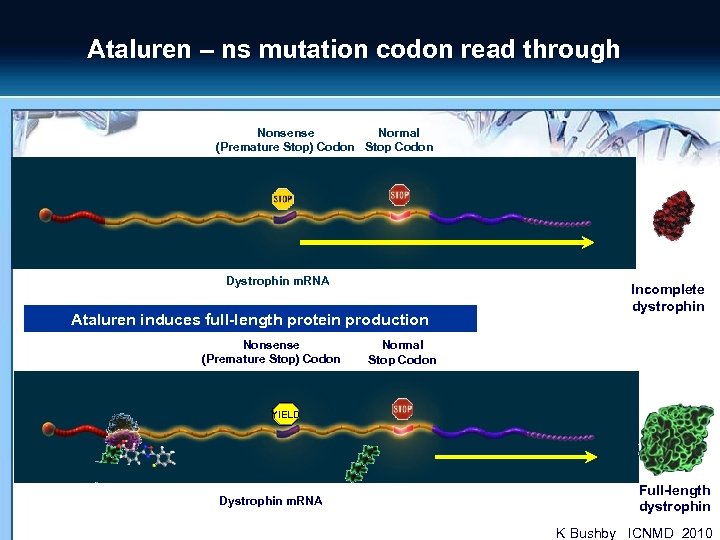 Ataluren – ns mutation codon read through Nonsense Normal (Premature Stop) Codon Stop Codon Dystrophin m. RNA Ataluren induces full-length protein production Nonsense (Premature Stop) Codon Incomplete dystrophin Normal Stop Codon YIELD Dystrophin m. RNA Full-length dystrophin K Bushby _ICNMD_2010
Ataluren – ns mutation codon read through Nonsense Normal (Premature Stop) Codon Stop Codon Dystrophin m. RNA Ataluren induces full-length protein production Nonsense (Premature Stop) Codon Incomplete dystrophin Normal Stop Codon YIELD Dystrophin m. RNA Full-length dystrophin K Bushby _ICNMD_2010
 PTC – Genzyme ATALUREN – Phase IIb § 2010 March § A PTC Therapeutics failed in the Phase IIb trial Tested patients: DMD with nonsense dystrophin mutation Primary endpoint: 6 Min Walking Test – did not increased significantly during the 48 weeks treatment period § § BUT! § In CF the Phase II trial was successfull, Phase III trial is ongoing § Nonsense mutation hemophilia A & B (nm. HA/B) and nonsense mutation methylmalonic acidemia (nm. MMA) Phase II trials are ongoing as well.
PTC – Genzyme ATALUREN – Phase IIb § 2010 March § A PTC Therapeutics failed in the Phase IIb trial Tested patients: DMD with nonsense dystrophin mutation Primary endpoint: 6 Min Walking Test – did not increased significantly during the 48 weeks treatment period § § BUT! § In CF the Phase II trial was successfull, Phase III trial is ongoing § Nonsense mutation hemophilia A & B (nm. HA/B) and nonsense mutation methylmalonic acidemia (nm. MMA) Phase II trials are ongoing as well.
 Molecular therapies DNA modulating therapies § Gene replacement § Gene editing § Gene shifting RNA modulating therapies § Exon skipping by antisense oligonucleotides § Mutant RNA removal § Block RNA expression § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
Molecular therapies DNA modulating therapies § Gene replacement § Gene editing § Gene shifting RNA modulating therapies § Exon skipping by antisense oligonucleotides § Mutant RNA removal § Block RNA expression § Inactivation of the mutant m. RNA by RNAi § Destruction of the mutant m. RNA by ribozymes § Translational manipulation Protein modulating therapies § Neutralization of the mutant protein § Regulating the level of the haploinsufficient protein § Upregulating compansatory molecule
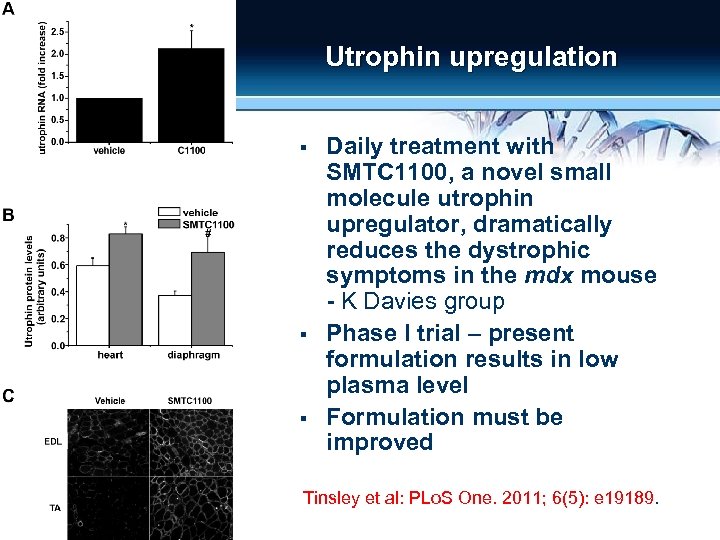 Utrophin upregulation § § § Daily treatment with SMTC 1100, a novel small molecule utrophin upregulator, dramatically reduces the dystrophic symptoms in the mdx mouse - K Davies group Phase I trial – present formulation results in low plasma level Formulation must be improved Tinsley et al: PLo. S One. 2011; 6(5): e 19189.
Utrophin upregulation § § § Daily treatment with SMTC 1100, a novel small molecule utrophin upregulator, dramatically reduces the dystrophic symptoms in the mdx mouse - K Davies group Phase I trial – present formulation results in low plasma level Formulation must be improved Tinsley et al: PLo. S One. 2011; 6(5): e 19189.
 General applications of therapeutic gene transfer 1. Molecular therapy for genetic diseases 2. Establishment of a stable gene reservoir as a source of therapeutic proteins in non - genetic diseases 3. To increase the tumor cells sensitivity to drugs 4. Destruction of malignant cells in neoplasias 5. DNA vaccines
General applications of therapeutic gene transfer 1. Molecular therapy for genetic diseases 2. Establishment of a stable gene reservoir as a source of therapeutic proteins in non - genetic diseases 3. To increase the tumor cells sensitivity to drugs 4. Destruction of malignant cells in neoplasias 5. DNA vaccines
 Neurodegenerative Disorders § Alzheimer Disease A Phase II Study to Assess efficacy of CERE-110 [Adeno-Associated Virus based, Vector. Mediated Delivery of Beta-Nerve Growth Factor (NGF)] in– Ceregene § Parkinson disease - Restoration the dopamin level of the basal ganglia - To prevent or modify the secondary changes due to the dopamin deficiency (Kaplitt procedure)
Neurodegenerative Disorders § Alzheimer Disease A Phase II Study to Assess efficacy of CERE-110 [Adeno-Associated Virus based, Vector. Mediated Delivery of Beta-Nerve Growth Factor (NGF)] in– Ceregene § Parkinson disease - Restoration the dopamin level of the basal ganglia - To prevent or modify the secondary changes due to the dopamin deficiency (Kaplitt procedure)
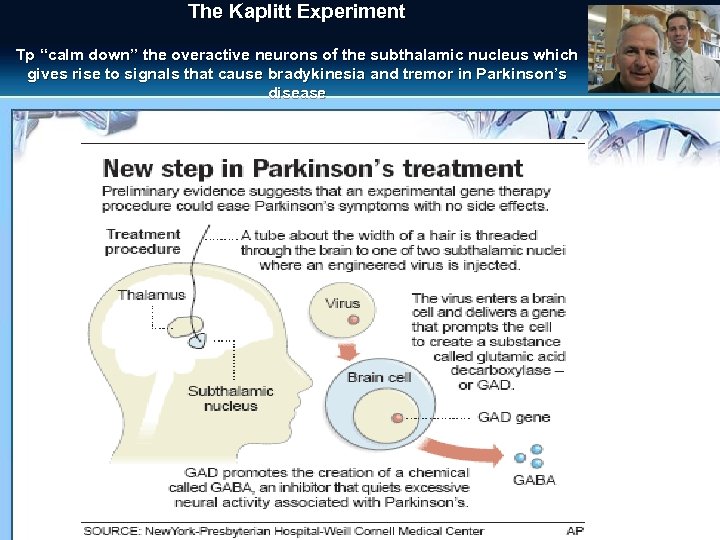 The Kaplitt Experiment Tp “calm down” the overactive neurons of the subthalamic nucleus which gives rise to signals that cause bradykinesia and tremor in Parkinson’s disease
The Kaplitt Experiment Tp “calm down” the overactive neurons of the subthalamic nucleus which gives rise to signals that cause bradykinesia and tremor in Parkinson’s disease
 AAV 2 -GAD gene therapy for advanced Parkinson's disease: a double-blind, randomised trial § 55 patients were randomised § Significant improvements in UPDRS score were noted in the experimental group compared to sham in the offmedication motor scores § At the 6 -month endpoint, UPDRS score for the AAV 2 -GAD group decreased by 8. 1 points ( p<0· 0001) and by 4. 7 points in the sham group (p=0· 003). § The AAV 2 -GAD group showed a significantly greater improvement from baseline in UPDRS scores compared with the sham group over the 6 -month § Side effects: mild or moderate, likely related to surgery and resolved. The most common were headache (7: 2) and nausea (6: 2) Le. Witt Lancet Neurol. 2011 Apr; 10(4): 309 -19
AAV 2 -GAD gene therapy for advanced Parkinson's disease: a double-blind, randomised trial § 55 patients were randomised § Significant improvements in UPDRS score were noted in the experimental group compared to sham in the offmedication motor scores § At the 6 -month endpoint, UPDRS score for the AAV 2 -GAD group decreased by 8. 1 points ( p<0· 0001) and by 4. 7 points in the sham group (p=0· 003). § The AAV 2 -GAD group showed a significantly greater improvement from baseline in UPDRS scores compared with the sham group over the 6 -month § Side effects: mild or moderate, likely related to surgery and resolved. The most common were headache (7: 2) and nausea (6: 2) Le. Witt Lancet Neurol. 2011 Apr; 10(4): 309 -19
 Phase I. Clinical Trial Gene Therapy for Pain § 10 patients with intractable focal pain § Vector: replication-defective HSV-based vector expressing human preproenkephalin (PENK) § Delivery: intradermal injection corresponding to the distribution of the pain § Subjects receiving the low dose of NP 2 reported no substantive change in pain § Subjects in the midle- and high-dose cohorts reported pain relief as assessed by numeric rating scale and SF-MPQ Fink et al. Ann Neurol 2011 Aug; 70(2): 207 -12.
Phase I. Clinical Trial Gene Therapy for Pain § 10 patients with intractable focal pain § Vector: replication-defective HSV-based vector expressing human preproenkephalin (PENK) § Delivery: intradermal injection corresponding to the distribution of the pain § Subjects receiving the low dose of NP 2 reported no substantive change in pain § Subjects in the midle- and high-dose cohorts reported pain relief as assessed by numeric rating scale and SF-MPQ Fink et al. Ann Neurol 2011 Aug; 70(2): 207 -12.
 General applications of therapeutic gene transfer 1. Molecular therapy for genetic diseases 2. Establishment of a stable gene reservoir as a source of therapeutic proteins in non - genetic diseases 3. To increase the tumor cells sensitivity to drugs 4. Destruction of malignant cells in neoplasias 5. DNA vaccines
General applications of therapeutic gene transfer 1. Molecular therapy for genetic diseases 2. Establishment of a stable gene reservoir as a source of therapeutic proteins in non - genetic diseases 3. To increase the tumor cells sensitivity to drugs 4. Destruction of malignant cells in neoplasias 5. DNA vaccines
 Malignant high grade glioma Phase III Trial § Antisense oligonucleotide trabedersen § Designed for the specific inhibition of TGF-β 2 synthesis § TGF-β 2 is overexpressed in more than 90% of highgrade gliomas, its levels are closely related to tumor progression § Inhibition of TGF-β 2 in tumor tissue leads to reversal of tumor-induced immune suppression, inhibition of tumor growth and invasion Curr Pharm Biotechnol 2011 May 27. [Epub ahead of print
Malignant high grade glioma Phase III Trial § Antisense oligonucleotide trabedersen § Designed for the specific inhibition of TGF-β 2 synthesis § TGF-β 2 is overexpressed in more than 90% of highgrade gliomas, its levels are closely related to tumor progression § Inhibition of TGF-β 2 in tumor tissue leads to reversal of tumor-induced immune suppression, inhibition of tumor growth and invasion Curr Pharm Biotechnol 2011 May 27. [Epub ahead of print
 Gene Therapy Is Still a Dream Or Reality? 2011 Gene therapy is not a failure — it is simply too immature to deliver yet on its promises Theodore Friedman Nature Med 1996; 2: 144
Gene Therapy Is Still a Dream Or Reality? 2011 Gene therapy is not a failure — it is simply too immature to deliver yet on its promises Theodore Friedman Nature Med 1996; 2: 144
 THANK YOU FOR YOUR ATTENTION!
THANK YOU FOR YOUR ATTENTION!


