0d4c28812a8d7cd3235923e978b4447c.ppt
- Количество слайдов: 93

Genomics in the Treatment of Early Stage Breast Cancer Oncotype DX™ Breast Cancer Assay ROSEMARY LEEMING, MD, FACS April 11, 2008

Agenda • • • Introduction Development of Oncotype DX Clinical Studies – Validation studies – Hormonal therapy benefit study (B-14) – Chemotherapy benefit study (B-20) • ASCO Data (6/07) – Clinical Utility Studies • Exploratory Studies – RS and local recurrence (SABCS 12 -06) – Node Positive Data (ASCO 6/07) • Other Gene Profiles – Mamma. Print – “Invasiveness” gene signature • • TAILORx Clinical Summary 2

Breast Cancer Treatment Planning: History • Treatment planning for N–, ER+ disease is based on: – Traditional prognostic factors with limited predictive power (tumor size, patient age) or poor reproducibility (tumor grade) – IHC markers (eg, Ki-67) lacking standardization and validation – Limited insight into relative benefits of chemotherapy for different individuals Bundred. Cancer Treat Rev. 2001; 27: 137 -142. 3

Breast Cancer Treatment Planning: Not Optimized • Chemotherapy treatment for N–, ER+ disease – Many women are offered chemotherapy, knowing that few benefit – Guidelines assume all patients benefit equally – Some patients are under-treated, many others are over-treated 4

Oncotype DX™: Unmet Clinical Need for Better Markers Biopsy or Resection Optimize High risk/ Large chemo benefit chemotherapy + local therapy + hormonal therapy Robust markers Low risk/ Little chemo benefit Optimize local therapy and hormonal therapy 5

Development and Validation of a 21 -Gene Assay for N–, ER+, Tam+ Patients YEAR Develop real-time RT-PCR method for paraffin block 2001 Select candidate genes (250 genes) 2002 Model building studies (N = 447, including 233 from NSABP B-20) 2002 Commit to a single 21 -gene assay 2003 Validation studies in NSABP B-14 and Kaiser Permanente 2003 Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 6
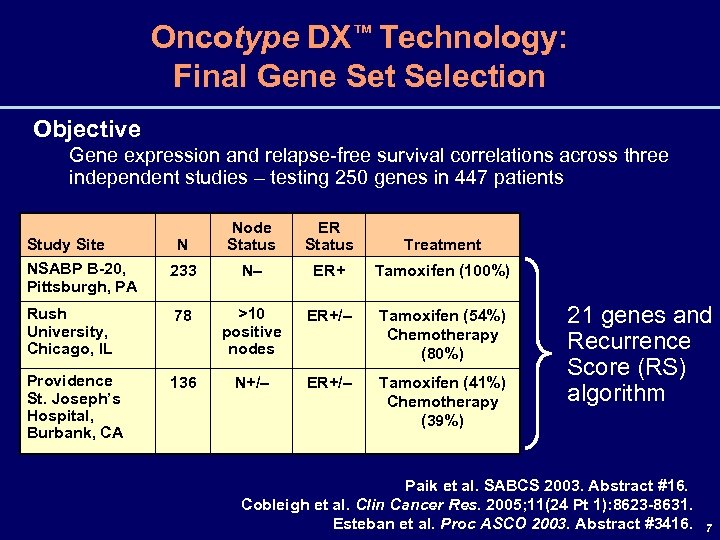
Oncotype DX™ Technology: Final Gene Set Selection Objective Gene expression and relapse-free survival correlations across three independent studies – testing 250 genes in 447 patients N Node Status ER Status Treatment NSABP B-20, Pittsburgh, PA 233 N– ER+ Tamoxifen (100%) Rush University, Chicago, IL 78 >10 positive nodes ER+/– Tamoxifen (54%) Chemotherapy (80%) Providence St. Joseph’s Hospital, Burbank, CA 136 N+/– ER+/– Tamoxifen (41%) Chemotherapy (39%) Study Site 21 genes and Recurrence Score (RS) algorithm Paik et al. SABCS 2003. Abstract #16. Cobleigh et al. Clin Cancer Res. 2005; 11(24 Pt 1): 8623 -8631. Esteban et al. Proc ASCO 2003. Abstract #3416. 7
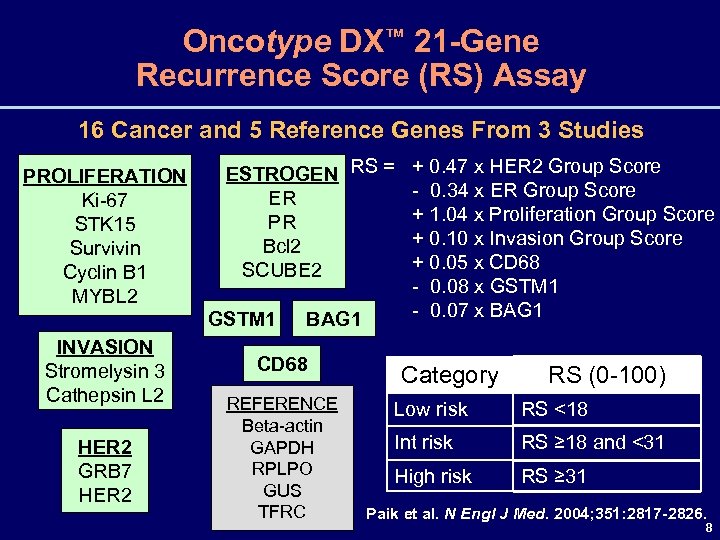
Oncotype DX™ 21 -Gene Recurrence Score (RS) Assay 16 Cancer and 5 Reference Genes From 3 Studies PROLIFERATION Ki-67 STK 15 Survivin Cyclin B 1 MYBL 2 INVASION Stromelysin 3 Cathepsin L 2 HER 2 GRB 7 HER 2 ESTROGEN RS = + 0. 47 x HER 2 Group Score - 0. 34 x ER Group Score ER + 1. 04 x Proliferation Group Score PR + 0. 10 x Invasion Group Score Bcl 2 + 0. 05 x CD 68 SCUBE 2 - 0. 08 x GSTM 1 - 0. 07 x BAG 1 GSTM 1 BAG 1 CD 68 REFERENCE Beta-actin GAPDH RPLPO GUS TFRC Category RS (0 -100) Low risk RS <18 Int risk RS ≥ 18 and <31 High risk RS ≥ 31 Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 8

Oncotype DX™ Clinical Validation: The NSABP B-14 Study* *Paik et al. N Engl J Med. 2004; 351: 2817 -2826.
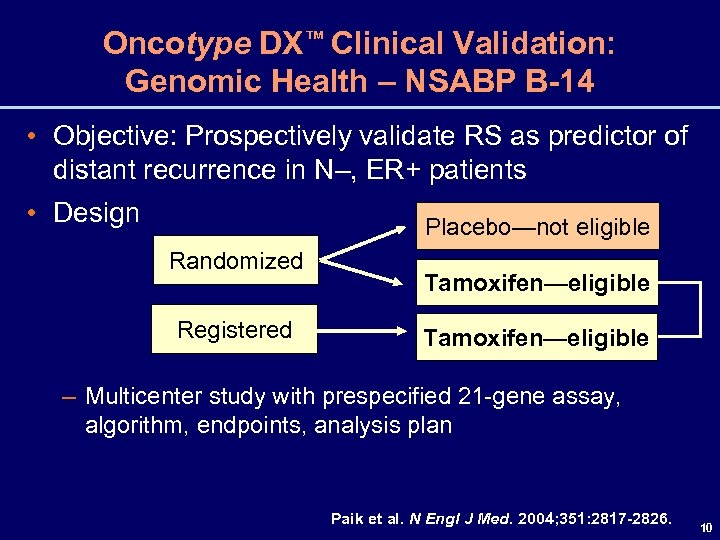
Oncotype DX™ Clinical Validation: Genomic Health – NSABP B-14 • Objective: Prospectively validate RS as predictor of distant recurrence in N–, ER+ patients • Design Placebo—not eligible Randomized Registered Tamoxifen—eligible – Multicenter study with prespecified 21 -gene assay, algorithm, endpoints, analysis plan Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 10
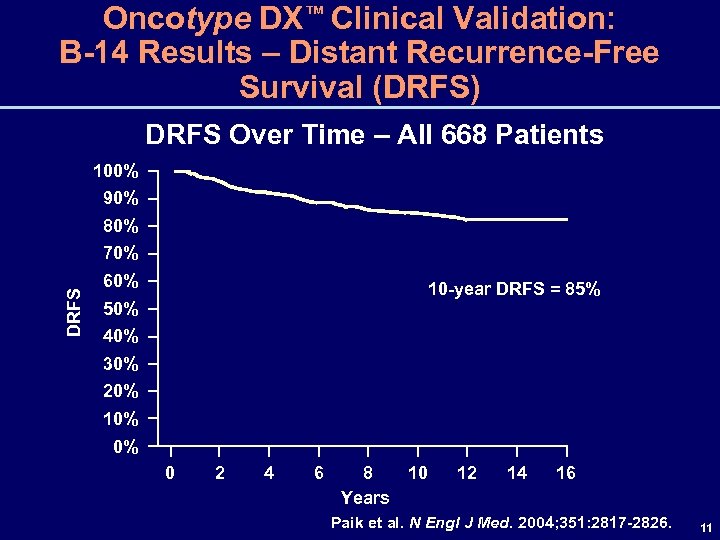
Oncotype DX™ Clinical Validation: B-14 Results – Distant Recurrence-Free Survival (DRFS) DRFS Over Time – All 668 Patients 100% 90% 80% DRFS 70% 60% 10 -year DRFS = 85% 50% 40% 30% 20% 10% 0% 0 2 4 6 8 10 Years 12 14 16 Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 11

Oncotype DX™ Clinical Validation: B-14 Study Objectives • First primary objective – Validate that 10 -year DRFS in the low-risk group (RS <18) is larger than 10 -year DRFS in the high-risk group (RS ≥ 31) • Second primary objective – Determine whether Recurrence Score as a predictor of DRFS is more significant than age and tumor size Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 12
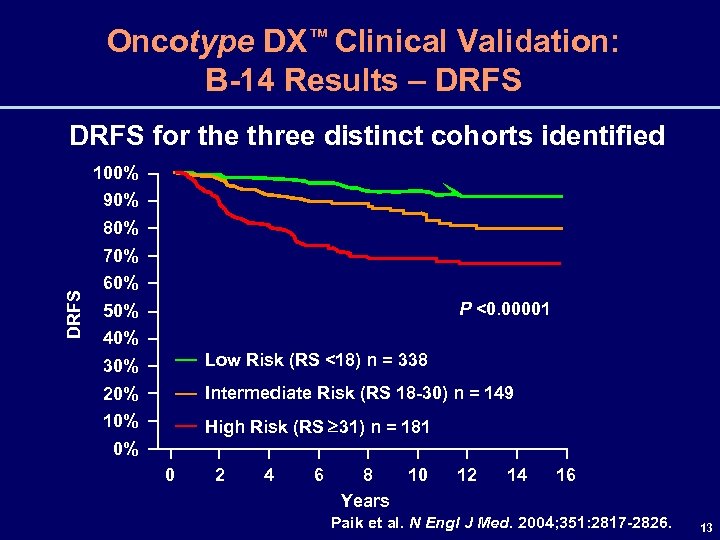
Oncotype DX™ Clinical Validation: B-14 Results – DRFS for the three distinct cohorts identified 100% 90% 80% DRFS 70% 60% P <0. 00001 50% 40% 30% Low Risk (RS <18) n = 338 20% Intermediate Risk (RS 18 -30) n = 149 10% High Risk (RS 31) n = 181 0% 0 2 4 6 8 10 Years 12 14 16 Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 13

Oncotype DX™ Clinical Validation: B-14 Results – DRFS (cont. ) Risk Group Low (RS <18) % of 10 -yr Rate of Patients Recurrence 51% 6. 8% 95% CI 4. 0%, 9. 6% Intermediate (RS 18 -30) 22% 14. 3% 8. 3%, 20. 3% High (RS ≥ 31) 30. 5% 23. 6%, 37. 4% 27% Test for the 10 -year DRFS comparison between the low-and high-risk groups: P <0. 00001 Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 14
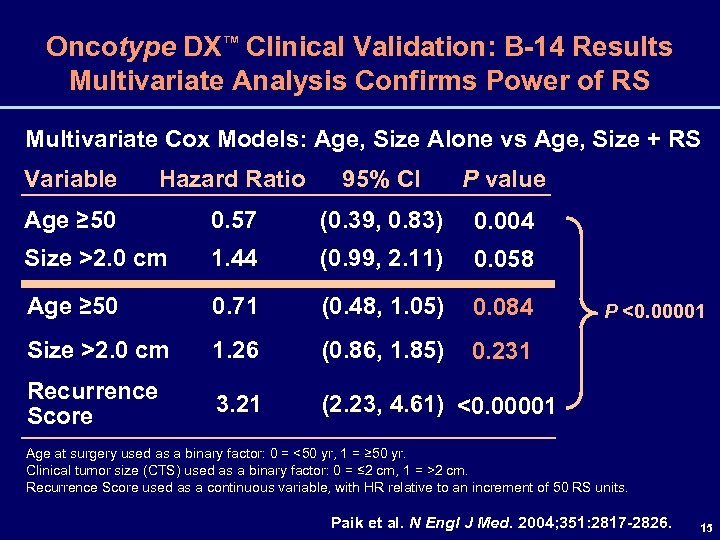
Oncotype DX™ Clinical Validation: B-14 Results Multivariate Analysis Confirms Power of RS Multivariate Cox Models: Age, Size Alone vs Age, Size + RS Variable Hazard Ratio 95% CI P value Age ≥ 50 0. 57 (0. 39, 0. 83) 0. 004 Size >2. 0 cm 1. 44 (0. 99, 2. 11) 0. 058 Age ≥ 50 0. 71 (0. 48, 1. 05) 0. 084 Size >2. 0 cm 1. 26 (0. 86, 1. 85) 0. 231 Recurrence Score 3. 21 (2. 23, 4. 61) <0. 00001 P <0. 00001 Age at surgery used as a binary factor: 0 = <50 yr, 1 = ≥ 50 yr. Clinical tumor size (CTS) used as a binary factor: 0 = ≤ 2 cm, 1 = >2 cm. Recurrence Score used as a continuous variable, with HR relative to an increment of 50 RS units. Paik et al. N Engl J Med. 2004; 351: 2817 -2826. 15

Oncotype DX™ Clinical Validation: Conclusions – NSABP B-14 • RS validated as predictor of recurrence in N–, ER+ patients • RS performance exceeds standard measures (age, size) • 50% of patients are reclassified by RS when compared to NCCN criteria • RS (based on tumor gene expression) more accurately quantifies the risk of distant recurrence than do the NCCN guidelines (based on age, tumor size, and tumor grade) 16

Oncotype DX™ Clinical Validation: The Kaiser Permanente Study Habel et al. Breast Cancer Res. 2006; May 31; 8(3): R 25.

The Kaiser Permanente Study: Methods Study Design Case-control Study Population Kaiser Permanente patients <75 yr in 14 Northern California hospitals diagnosed with node-negative BC 1985 -94, no chemotherapy (N = 4964) Cases: Deaths from BC (n = 220) Controls: Randomly selected, matched on age, race, diagnosis year, KP facility, tamoxifen (n = 570) Data Sources Cancer registry, medical records, archived diagnostic slides, and tumor blocks Habel et al. Breast Cancer Res. May 2006. 18

The Kaiser Permanente Study: Risk of BC Death at 10 Years: ER+, Tam+ Patients Risk Classifier Recurrence Score Low (<18) Intermediate (18 -30) High (>31) 1 Based % of Cases (n = 55) % of Controls (n = 150) 10 -yr Absolute Risk 1 29% 40% 31% 63% 23% 13% 2. 8%2 10. 7% 15. 5% on methods by Langholz and Borgan, Biometrics 1997; 53: 767 -774. 2 Consistent with 3. 0% absolute risk of breast cancer death in similar population of tamoxifen-treated patients in NSABP B-14. Habel et al. Breast Cancer Res. May 2006. 19

The Kaiser Permanente Study: Conclusions • “The RS has now been shown to be strongly associated with risk of breast cancer-specific mortality among LN–, ER+, tam-treated patients participating in a clinical trial and among similar patients from the community setting. ” • “Combining Recurrence Score, tumor grade, and tumor size provides better risk classification than any one of these factors alone. ” Habel et al. Breast Cancer Res. May 2006. 20

Oncotype DX™ Prediction of Tam Benefit: NSABP B-14 Placebo and Tamoxifen Arms* *Paik et al. ASCO 2004. Abstract #510.

Tamoxifen Benefit and Oncotype DX™ NSABP B-14 Tam Benefit Study in N–, ER+ Patients Design Randomized Placebo-Eligible Tam-Eligible Objective: Determine whether the 21 -gene RS assay provides information on: 1) Prognosis (likelihood of recurrence) 2) Response to tamoxifen (change in likelihood of recurrence with tamoxifen) 3) Both Paik et al. ASCO 2004. Abstract #510. 22
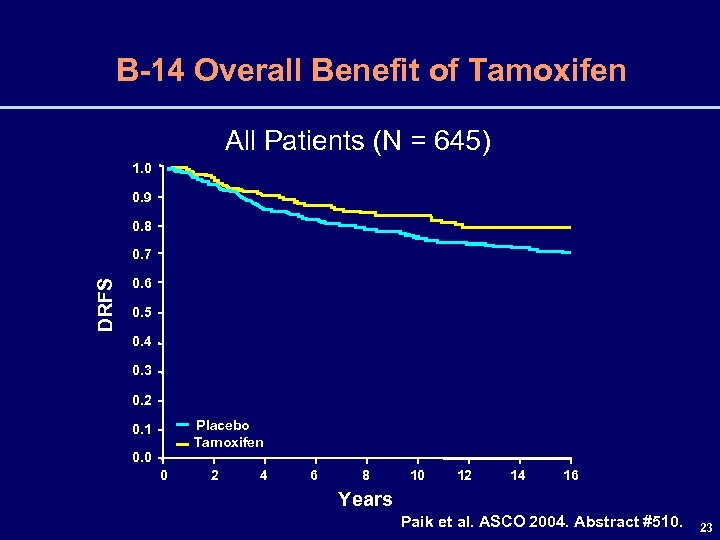
B-14 Overall Benefit of Tamoxifen All Patients (N = 645) 1. 0 0. 9 0. 8 DRFS 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 Placebo Tamoxifen 0. 1 0. 0 0 2 4 6 8 10 12 14 16 Years Paik et al. ASCO 2004. Abstract #510. 23
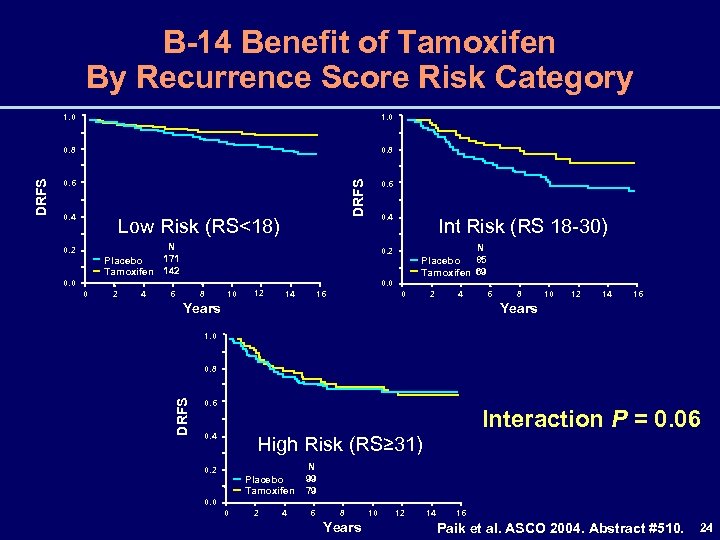
B-14 Benefit of Tamoxifen By Recurrence Score Risk Category 0. 8 0. 6 0. 4 DRFS 1. 0 0. 8 Low Risk (RS<18) 0. 4 N 171 Placebo Tamoxifen 142 0. 0 0 2 4 Int Risk (RS 18 -30) N 85 Placebo Tamoxifen 69 0. 2 6 8 10 12 0. 0 14 16 0 2 4 Years 6 8 10 12 14 16 Years 1. 0 0. 8 DRFS 1. 0 0. 6 Interaction P = 0. 06 0. 4 High Risk (RS≥ 31) 0. 2 Placebo Tamoxifen N 99 79 0. 0 0 2 4 6 8 Years 10 12 14 16 Paik et al. ASCO 2004. Abstract #510. 24

Analysis of Placebo and Tam-Treated Patients in NSABP B-14 • Conclusions – RS combines prognostic and predictive factors into one assay report – RS performance is derived from measurement of expression of each of the 21 genes on a continuous scale with high precision and reproducibility Paik et al. ASCO 2004. Abstract #510. 25

Oncotype DX™ Prediction of Chemo Benefit: NSABP B-20 Study* *Paik et al. J Clin Oncol. 2006; 24: 3726 -3734
Chemotherapy Benefit and Oncotype DX™ NSABP B-20 Chemo Benefit Study in N–, ER+ Pts Design Tam + MF Randomized Tam + CMF Tam Objective: Determine the magnitude of the chemo benefit as a function of the 21 -gene RS assay Paik et al. J Clin Oncol. 2006; 24: 3726 -3734. 27
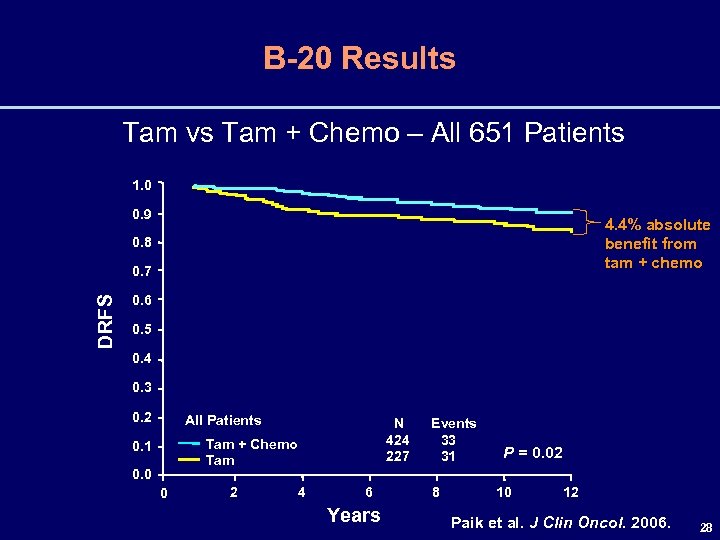
B-20 Results Tam vs Tam + Chemo – All 651 Patients 1. 0 0. 9 4. 4% absolute benefit from tam + chemo 0. 8 DRFS 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 All Patients N 424 227 Tam + Chemo Tam 0. 1 0. 0 0 2 4 6 Years Events 33 31 8 P = 0. 02 10 12 Paik et al. J Clin Oncol. 2006. 28
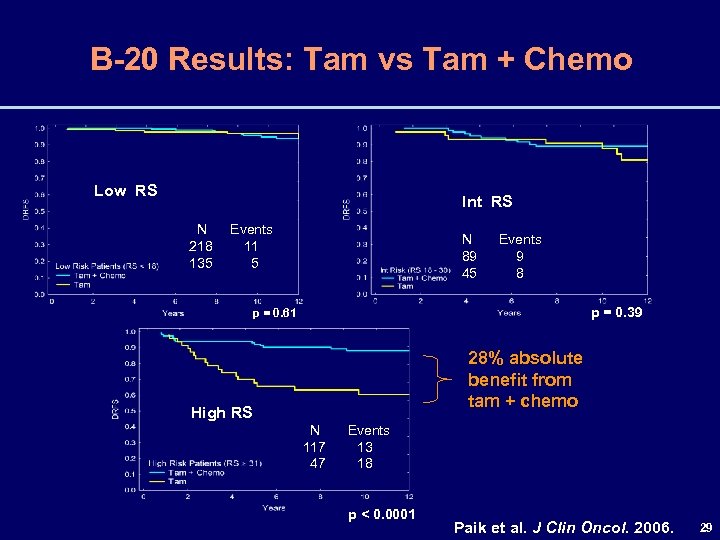
B-20 Results: Tam vs Tam + Chemo Low RS Int RS N Events 218 11 135 5 N Events 89 9 45 8 p = 0. 39 p = 0. 61 28% absolute benefit from tam + chemo High RS N Events 117 13 47 18 p < 0. 0001 Paik et al. J Clin Oncol. 2006. 29
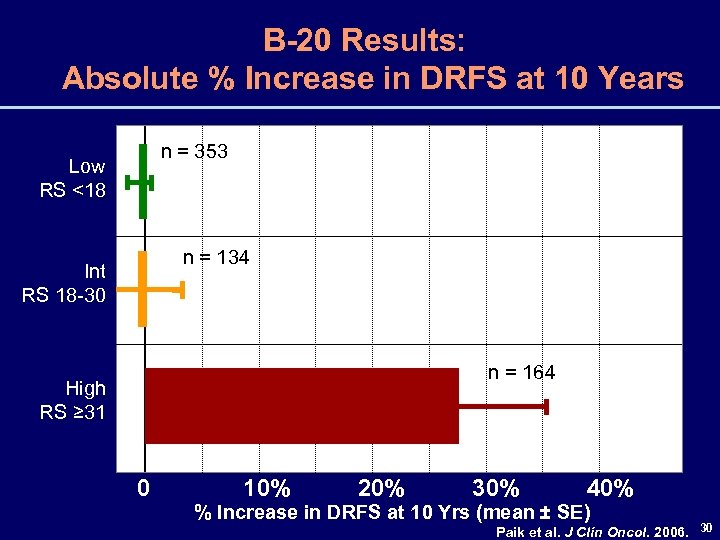
B-20 Results: Absolute % Increase in DRFS at 10 Years n = 353 Low RS <18 n = 134 Int RS 18 -30 n = 164 High RS ≥ 31 0 10% 20% 30% 40% % Increase in DRFS at 10 Yrs (mean ± SE) Paik et al. J Clin Oncol. 2006. 30

RS and Breast Cancer Death in NSABP B-14 and B-20 • Benefits of adjuvant treatment differ by Recurrence Score risk category – Benefits of tamoxifen are greater in patients with low-risk or intermediate-risk tumors – Benefits of chemotherapy are greater in patients with high-risk tumors 31

ASCO ABSTRACTS JUNE 2007 • CLINICAL UTILITY OF ONCOTYPE DX – ABSTRACT #577: LOYOLA UNIVERSITY – ABSTRACT #576: MAYO CLINIC 32

Prospective multi-center study of the impact of the 21 -gene Recurrence Score (RS) assay on medical oncologist (MO) and patient (pt) adjuvant breast cancer (BC) treatment selection. Shelly S. Lo 1, John Norton 1, Patricia B. Mumby 1, Jeffrey Smerage 2, Joseph Kash 3, Helen K. Chew 4, Daniel Hayes 2, Andrew Epstein 5, Kathy S. Albain 1 1 Loyola University, Maywood IL, 2 University of Michigan, Ann. Arbor MI, 3 Edward Hospital, Naperville IL, 4 UC Davis, Sacramento CA, 5 Mount Sinai Medical Center, New York NY

Background • The 21 -gene Recurrence Score (RS) assay (Oncotype DXTM) has been validated to quantify the risk of distant recurrence in tamoxifen treated patients in lymph node negative, estrogen receptor (ER) positive breast cancer. The RS also predicts benefit from adjuvant chemotherapy. • There is little data regarding the impact of the RS on medical oncologist (MO) and patient (pt) decision making. A multi-center study was designed to prospectively examine whether the RS affects MO and pt adjuvant treatment selection. ASCO 2007, Abstract #577 34

Methods • 17 MOs at 1 community and 3 academic practices participated in this study. Each participating MO consecutively offered enrollment to eligible women with node negative, ER positive breast cancer. • Each participating MO and consenting patient completed pre- and post-RS assay questionnaires specifically developed for the study. • MOs stated their adjuvant treatment recommendation and confidence in it pre and post RS assay. • Pts indicated treatment choice pre and post RS assay. In addition, patients completed measures for quality of life, anxiety, and decisional conflict pre and post assay. • RS assay results were returned to MO and shared with pt for routine clinical care. • Frequency distributions and co-frequency tables are used to display categorical distributions of nominal variables; means and standard deviations are used to summarize continuous variables. 35

Medical Oncologist Characteristics • 17 medical oncologists from 4 institutions – University of Michigan: 6 – LUMC: 6 – Edward: 3 – UC Davis: 2 • Length of time in practice <5 yrs: 29% • Length of time in practice>5 yrs: 71% ASCO 2007, Abstract #577 36
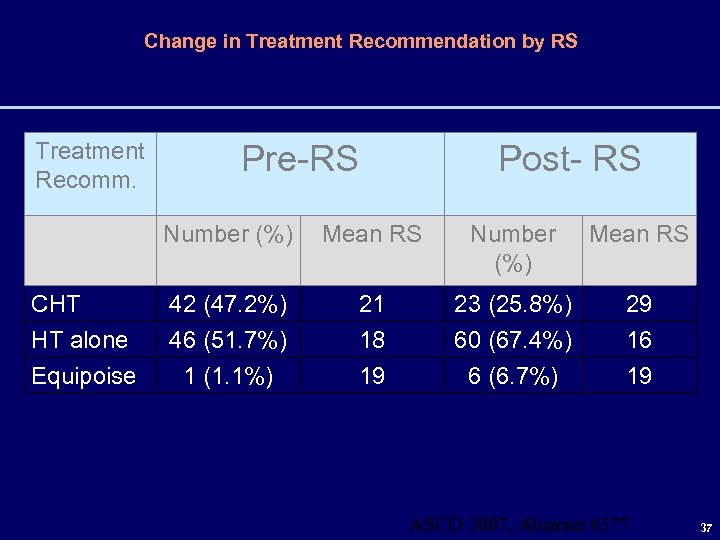
Change in Treatment Recommendation by RS Treatment Recomm. Pre-RS Post- RS Number (%) CHT HT alone Equipoise Mean RS Number (%) Mean RS 42 (47. 2%) 46 (51. 7%) 1 (1. 1%) 21 18 19 23 (25. 8%) 60 (67. 4%) 6 (6. 7%) 29 16 19 ASCO 2007, Abstract #577 37

MO Treatment Recommendations Changed 31. 5% of the Time MO Pre to Post-RS Assay Treatment Recommendation Number of Cases(%) CHT to HT 20 (22. 5) HT to CHT 3 (3. 4) CHT or HT to Equipoise 5 (5. 6) Treatment plan did not change 61 (68. 5) Total 89 (100) Treatment recommendation changed in 28 (31. 5%) after results of RS Assay known. The largest change was from a recommendation of CHT to HT in 22. 5% of cases. 38

Conclusions • RS Assay changed physician adjuvant treatment recommendations in 31. 5% of the cases • RS Assay is used by patients in their adjuvant treatment decisions • Results from the RS assay was associated with less adjuvant chemotherapy administration – The largest treatment recommendation change for MO was changing recommendation from CHT to HT 39

How Well Do Standard Prognostic Criteria Predict Oncotype DX Scores? Kamal AH, Loprinzi CL, Reynolds C, Dueck AC, Geiger XJ, Ingle JN, Carlson RW, Hobday TJ, Winer EP, Perez EA, Goetz MP Mayo Clinic, Rochester, MN; Mayo Clinic, Jacksonville, FL; Stanford Comprehensive Cancer Center, Palo Alto, CA; Dana-Farber Cancer Institute, Boston, MA 40

Mayo Clinic Study Methods: 31 patients with Oncotype DX scores available Slides reviewed for path, receptors and her-2 Cases presented to 6 “academic” oncologists, blinded to RS asked to predict RS and to recommend chemo given RS and asked chemo question again 42

Mayo Clinic Study Results: RS: low – 18, int – 10, high – 3 Concordance between predicted and actual RS low/int vs. high >87% Easier to pick out high risk More important to identify low risk than low/int 43

Mayo Clinic Study Results: Most frequent discrepancies: actual low RS predicted as intermed (31/80 discordant 39%) actual intermed RS predicted as low (29/80 discordant 36%) Treatment recommendations following Oncotype DX changed about 18. 2% most frequent change from CHT to HT 44

Mayo Clinic Study Conclusions: “Proper evaluation and interpretation of traditional prognostic criteria will identify most node negative, ER+ patients at high risk of recurrence (as predicted by RS), but poorly discriminate low vs. intermediate risk. ” Recommendation for treatment was changed in about 20% of cases This study used expert pathologists and nationally known breast oncologists. 45

Conclusions of clinical utility studies • Oncotype DXTM directly changes treatment recommendations approx. 20 – 30+% of the time • Physicians and patients alike find the RS useful • Even when treatment decisions are not altered, the RS can increase confidence in the decision 46

ONCOTYPE DX EXPLORATORY STUDIES: ONCOTYPE DX AND LOCAL RECURRENCE ONCOTYPE DX AND NODE POSITIVE PATIENTS ONCOTYPE DX AND NEO-ADJUVANT CHEMOTHERAPY 47

Association Between Recurrence Score and Risk of Local-regional Failure in N─, ER+ Breast Cancer: Results from NSABP B-14 and NSABP B-20 E. Mamounas, G. Tang, J. Bryant, S Paik, S Shak, J Costantino, D Watson, D. L Wickerham, and N Wolmark 48 48

Study Objectives • To examine the relationship between RS and Risk of Loco-Regional Failure (LRF) in Node-Negative, ER-Positive, Patients from NSABP B-14 and NSABP B-20: –Placebo treated patients from B-14 –Tamoxifen treated patients from B-14, B-20 –Chemo-TAM treated patients from B-20 49

Study Population • Patients with RS Assay from NSABP nodenegative, ER-positive adjuvant trials: – B-14 Placebo: 355 pts – B-14/B-20 Tamoxifen: 895 pts • B-14: 668 • B-20: 227 – B-20 Chemo + Tamoxifen: 424 pts 50

Patient Characteristics Surgery Type Lumpectomy Mastectomy B-14 B-20 Placebo TAM TAM+Chem (n=355) (n=668) (n=227) 32% 68% 42% 58% 48% 52% (n=424) 39% 61% • All lumpectomy patients received breast XRT • Post-mastectomy chest wall XRT was not allowed • Regional nodal XRT was not allowed irrespective of surgical procedure 51
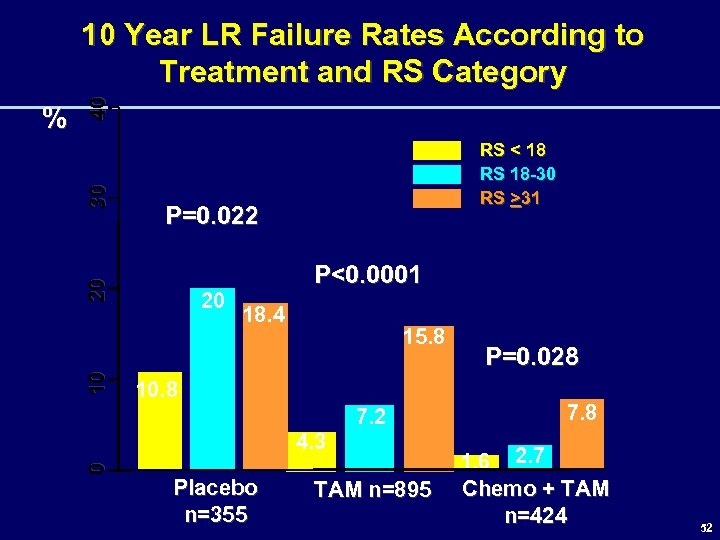
30 P=0. 022 20 10 RS < 18 RS 18 -30 RS >31 20 P<0. 0001 18. 4 15. 8 10. 8 7. 2 4. 3 0 % 40 10 Year LR Failure Rates According to Treatment and RS Category Placebo n=355 TAM n=895 P=0. 028 7. 8 1. 6 2. 7 Chemo + TAM n=424 52
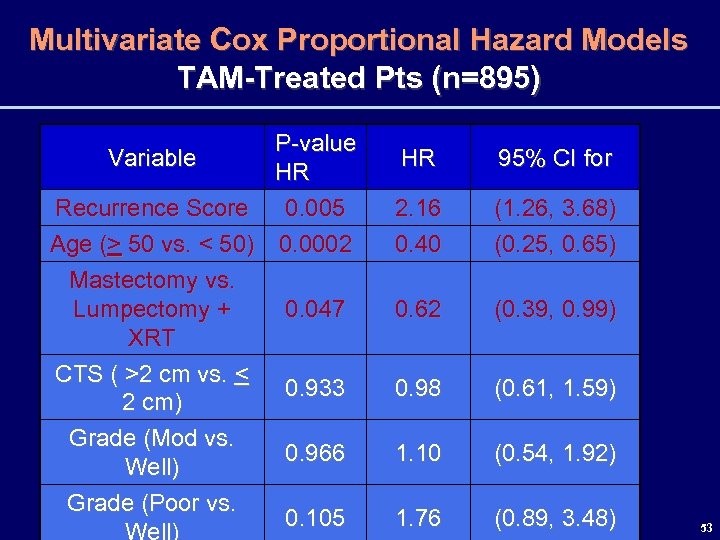
Multivariate Cox Proportional Hazard Models TAM-Treated Pts (n=895) P-value Variable HR Recurrence Score 0. 005 Age (> 50 vs. < 50) 0. 0002 Mastectomy vs. Lumpectomy + 0. 047 XRT CTS ( >2 cm vs. < 0. 933 2 cm) Grade (Mod vs. 0. 966 Well) Grade (Poor vs. 0. 105 Well) HR 95% CI for 2. 16 0. 40 (1. 26, 3. 68) (0. 25, 0. 65) 0. 62 (0. 39, 0. 99) 0. 98 (0. 61, 1. 59) 1. 10 (0. 54, 1. 92) 1. 76 (0. 89, 3. 48) 53
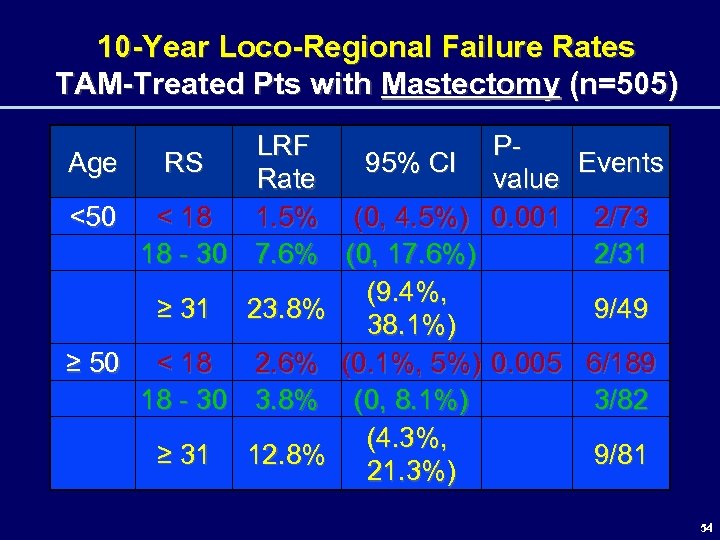
10 -Year Loco-Regional Failure Rates TAM-Treated Pts with Mastectomy (n=505) Age <50 ≥ 50 LRF RS 95% CI Rate < 18 1. 5% (0, 4. 5%) 18 - 30 7. 6% (0, 17. 6%) (9. 4%, ≥ 31 23. 8% 38. 1%) < 18 2. 6% (0. 1%, 5%) 18 - 30 3. 8% (0, 8. 1%) (4. 3%, ≥ 31 12. 8% 21. 3%) PEvents value 0. 001 2/73 2/31 9/49 0. 005 6/189 3/82 9/81 54

Exploratory Analysis 10 -Year Loco-Regional Failure Rates TAM-Treated Pts with Mastectomy (n=505) • RS correlated with 10 -year LRF rates in patients < 50 and > 50 – High (23. 8%) LRF rate in pts <50 with a high RS – However, small sample size of pts <50 who had high RS and mastectomy (49 pts) • Subset analysis - hypothesis-generating ONLY 55 55
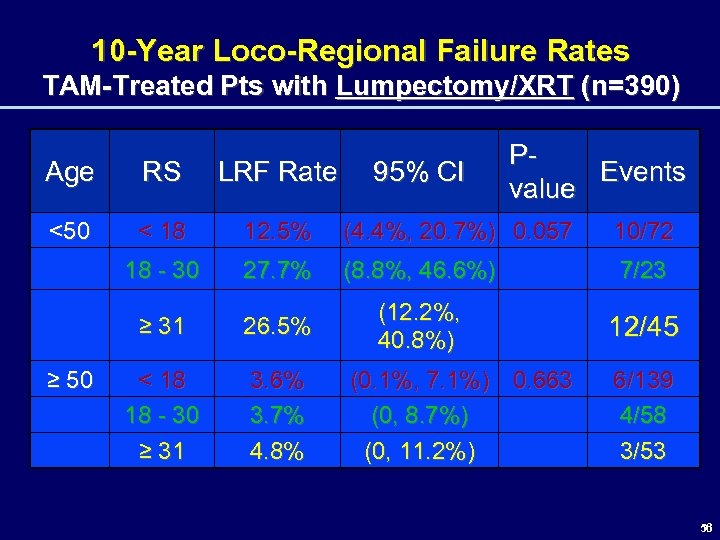
10 -Year Loco-Regional Failure Rates TAM-Treated Pts with Lumpectomy/XRT (n=390) Age RS LRF Rate <50 < 18 12. 5% (4. 4%, 20. 7%) 0. 057 10/72 18 - 30 27. 7% (8. 8%, 46. 6%) 7/23 ≥ 31 26. 5% (12. 2%, 40. 8%) 12/45 < 18 18 - 30 ≥ 31 3. 6% 3. 7% 4. 8% ≥ 50 95% CI PEvents value (0. 1%, 7. 1%) 0. 663 (0, 8. 7%) (0, 11. 2%) 6/139 4/58 3/53 56

Exploratory Analysis Summary 10 -Year Loco-Regional Failure Rates TAM-Treated Pts with Lumpectomy/XRT (n=390) • Women < 50: RS associated with LRF (p = 0. 057) – LRF is high with intermediate/high RS • Women > 50: RS NOT associated with LRF (p = 0. 663) • However, as a subset analysis these findings should be hypothesis-generating ONLY 57 57

Summary • The 21 -Gene Recurrence Score was found to predict risk of LRF in node-negative, ER-positive, pts treated with tamoxifen. • In this group of pts, RS was an independent predictor of LRF along with age and surgery type. • RS also predicted risk of LRF in node-negative, ER-positive pts treated with chemotherapy + tamoxifen and to a lesser extent in those treated without adjuvant therapy 58

Conclusions • The present study demonstrates a similar association between RS and risk for LRF as the one previously shown for RS and risk of distant failure. • This information has biologic implications and may have clinical implications relative to loco-regional therapy decisions in patients with node-negative, ER-positive breast cancer. 59

Can Oncotype DX be used for node positive patients? 60 60

Prognostic Utility of the 21 -Gene Assay in Hormone Receptor (HR) Positive Operable Breast Cancer and 0 -3 Positive Axillary Nodes Treated with Adjuvant Chemohormonal Therapy (CHT): An Analysis of Intergroup Trial E 2197 Goldstein LJ 1, Gray R 1, Childs B 2, Watson D 3, Yoshizawa C 3, Rowley S 2, Shak S 3, Badve S 1, Davidson NE 1, Sledge GW 1, Sparano JA 1 From the Eastern Cooperative Oncology Group 1, sanofi aventis 2, & Genomic Health, Inc. 3
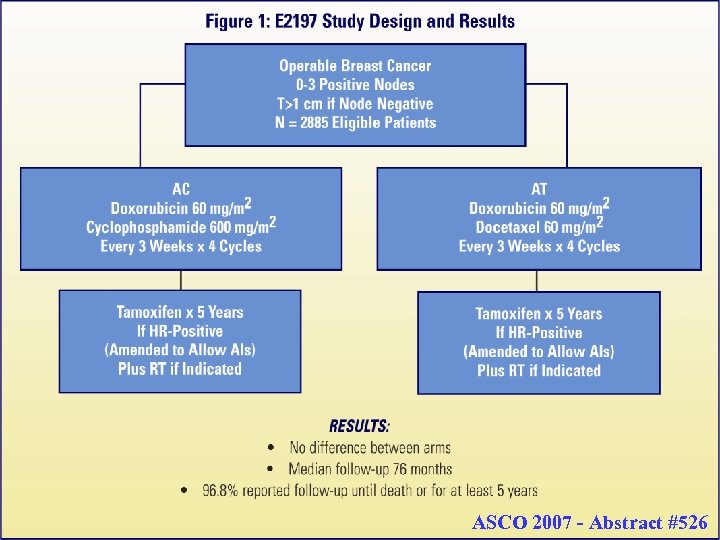
62 ASCO 2007 - Abstract #526 62

Objectives Specific: 1. To evaluate the prognostic utility of Oncotype DX Recurrence Score (RS) in patients with HR-Pos treated with adjuvant chemotherapy 2. To perform an exploratory analysis for individual genes associated with prognosis in patients with HR-Pos and HRNeg disease treated with adjuvant chemotherapy (analysis ongoing) 3. To perform an exploratory analysis to identify individual genes associated with differential sensitivity to AC versus AT (analysis ongoing) 63
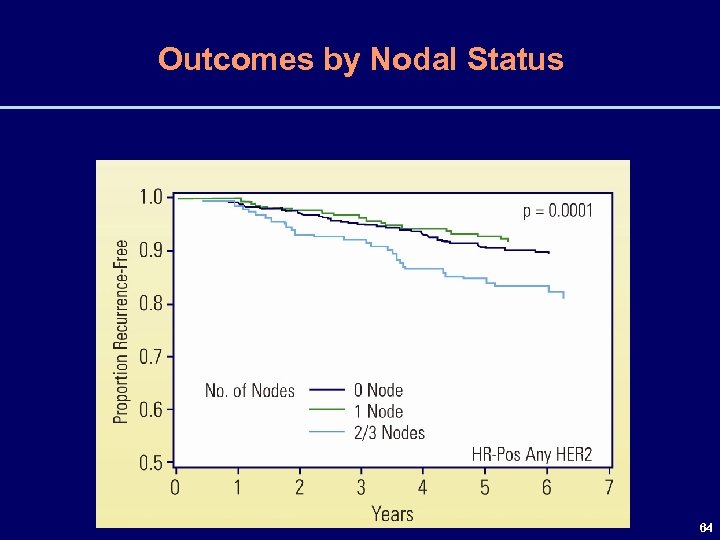
Outcomes by Nodal Status 64
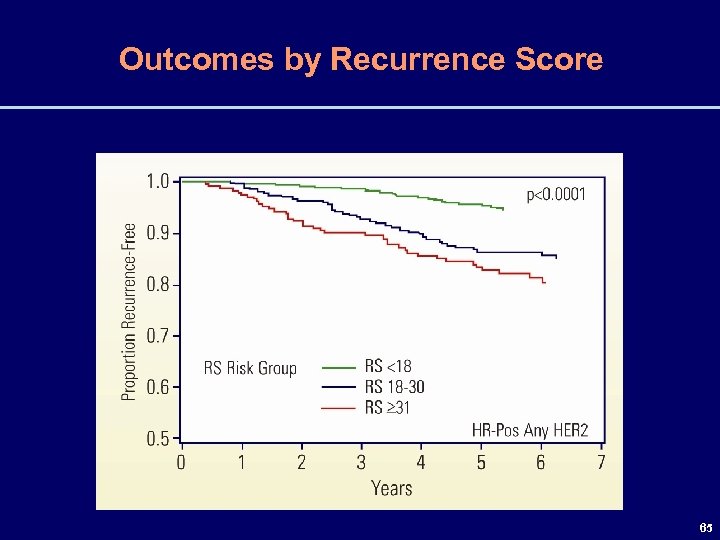
Outcomes by Recurrence Score 65
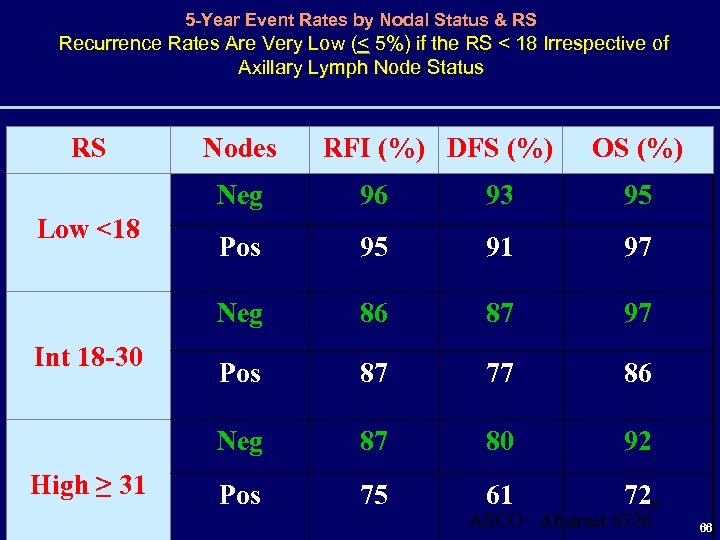
5 -Year Event Rates by Nodal Status & RS Recurrence Rates Are Very Low (< 5%) if the RS < 18 Irrespective of Axillary Lymph Node Status RS Nodes RFI (%) DFS (%) OS (%) Neg High ≥ 31 95 Pos 95 91 97 86 87 97 Pos 87 77 86 Neg Int 18 -30 93 Neg Low <18 96 87 80 92 Pos 75 61 72 66 ASCO - Abstract #526 66

Conclusions For HR-Pos Disease with 0 -3 Pos Axillary Lymph Nodes • Low RS (< 18) predictive of excellent outcomes at 5 yrs • including patients with 1 -3 positive axillary lymph nodes • when adjusted for centrally determined tumor grade • Low RS (< 18) common • ~ 46% of patients with HR-positive disease in this study, of whom 49% had 1 -3 positive axillary lymph nodes 67 ASCO - Abstract #526 67

Prognostic and Predictive Value of the 21 -Gene Recurrence Score Assay in Postmenopausal, Node-Positive (N+), ERPositive (ER+) Breast Cancer SWOG 8814, TBCI 0100 Oral Presentation #10 K. Albain, W. Barlow, S. Shak, G. Hortobagyi, R. Livingston, I. Yeh, P. Ravdin, C. Yoshizawa, F. Baehner, N. Davidson, G. Sledge, E. Winer, C. Hudis, J. Ingle, E. Perez, K. Pritchard, L. Shepherd, C. Allred, K. Osborne, and D. Hayes for The Breast Cancer Intergroup of North America 68

SWOG 8814/TBCI 0100 Correlative Science Rationale • The 21 -gene Recurrence Score assay (RS) is prognostic for women with node(-), ER+ breast cancer on 5 years of tamoxifen* • A high RS predicts large benefit from chemotherapy in node(-) disease, but no improvement if the RS is low** • There are no RS data in a N+ population with a tamoxifen-alone control • SWOG 8814 is an ideal trial to explore this question Albain, SABCS 2007, Abstract #10 *Paik, et al. NEJM, 2004 **Paik, et al. J Clin Oncol, 2006 69
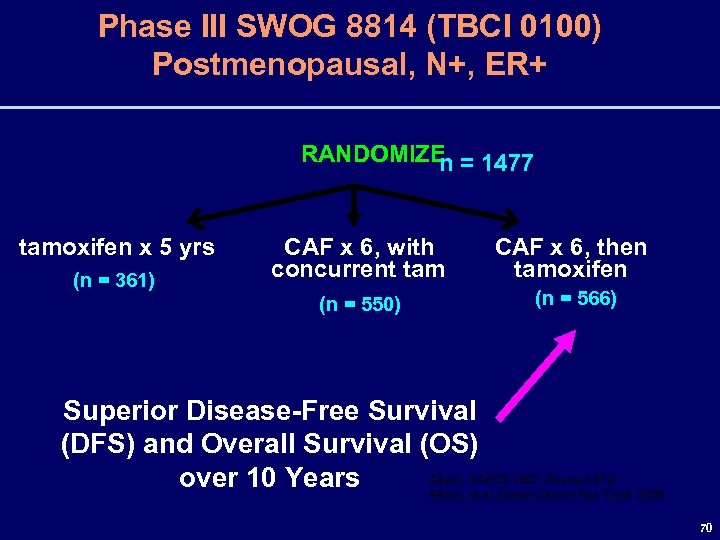
Phase III SWOG 8814 (TBCI 0100) Postmenopausal, N+, ER+ RANDOMIZE = 1477 n tamoxifen x 5 yrs CAF x 6, then tamoxifen (n = 550) (n = 361) CAF x 6, with concurrent tam (n = 566) Superior Disease-Free Survival (DFS) and Overall Survival (OS) Albain, SABCS 2007, Abstract #10 over 10 Years Albain, et al. Breast Cancer Res Treat 2005 70

SWOG 8814/TBCI 0100 Correlative Science Study Two co-primary objectives were to determine if the RS: 1) Provides prognostic information for women with N+ disease treated only with tamoxifen, and 2) Allows prediction of a N+ group that does not derive benefit from chemotherapy Albain, SABCS 2007, Abstract #10 71
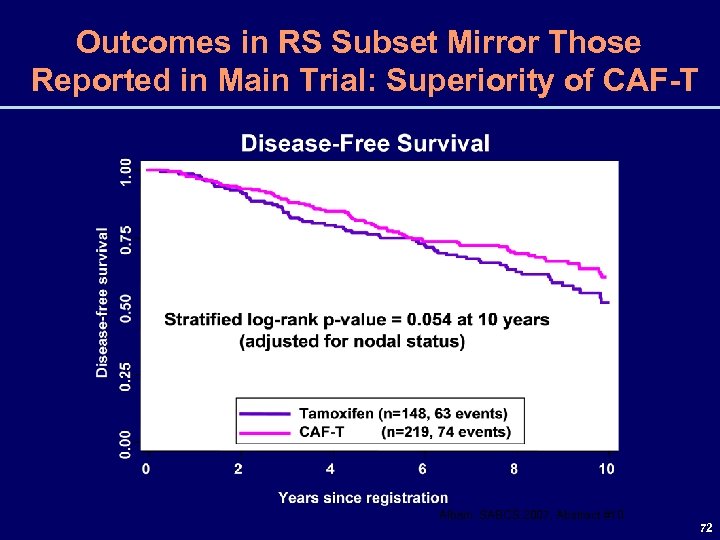
Outcomes in RS Subset Mirror Those Reported in Main Trial: Superiority of CAF-T Albain, SABCS 2007, Abstract #10 72
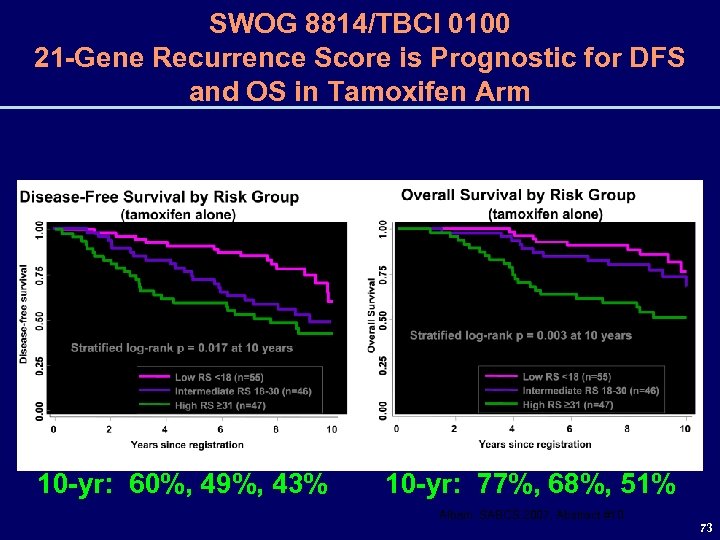
SWOG 8814/TBCI 0100 21 -Gene Recurrence Score is Prognostic for DFS and OS in Tamoxifen Arm 10 -yr: 60%, 49%, 43% 10 -yr: 77%, 68%, 51% Albain, SABCS 2007, Abstract #10 73
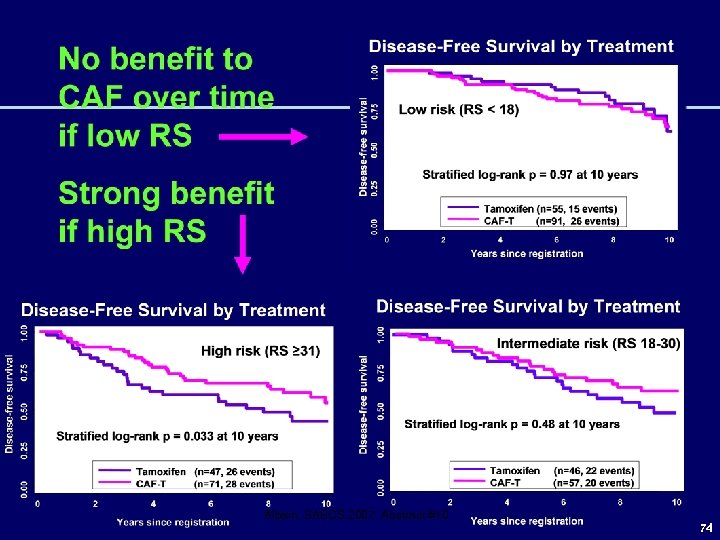
Albain, SABCS 2007, Abstract #10 74

CAF Benefit Greatest in Higher RS for Both Nodal Subsets, with No Benefit in Lower RS Albain, SABCS 2007, Abstract #10 75

NEOADJUVANT DATA Milan and Baylor Studies 76
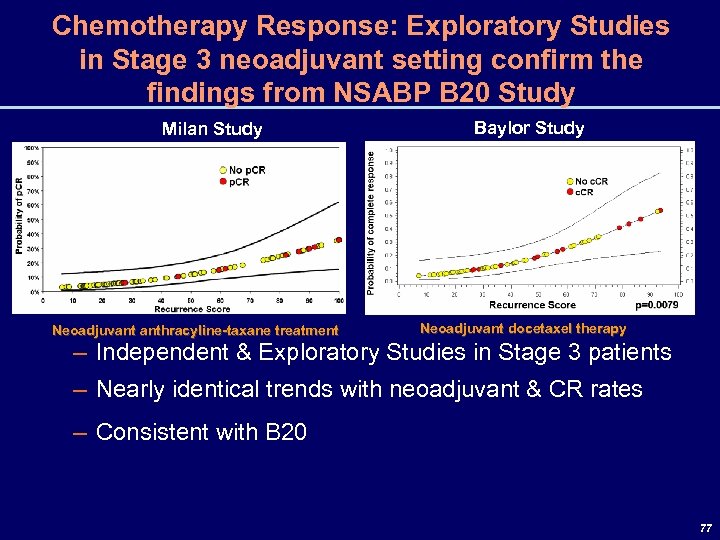
Chemotherapy Response: Exploratory Studies in Stage 3 neoadjuvant setting confirm the findings from NSABP B 20 Study Milan Study Neoadjuvant anthracyline-taxane treatment Baylor Study Neoadjuvant docetaxel therapy – Independent & Exploratory Studies in Stage 3 patients – Nearly identical trends with neoadjuvant & CR rates – Consistent with B 20 77

Gene Expression Profiles in Paraffin-Embedded Core Biopsy Tissue Predict Response to Chemotherapy in Women With Locally Advanced Breast Cancer L. Gianni, M. Zambetti, K. Clark, J. Baker, M. Cronin, J. Wu, G. Mariani, J. Rodriguez, M. Carcangiu, D. Watson, P. Valagussa, R. Rouzier, W. Symmans, J. Ross, G. Hortobagyi, L. Pusztai, and S. Shak Gianni et al. J Clin Oncol. 2005; 23(29): 7265 -7277.
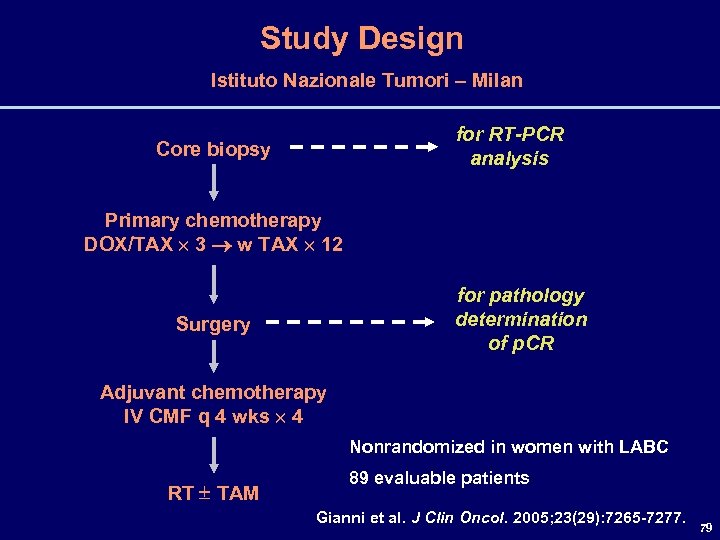
Study Design Istituto Nazionale Tumori – Milan for RT-PCR analysis Core biopsy Primary chemotherapy DOX/TAX 3 w TAX 12 for pathology determination of p. CR Surgery Adjuvant chemotherapy IV CMF q 4 wks 4 Nonrandomized in women with LABC RT TAM 89 evaluable patients Gianni et al. J Clin Oncol. 2005; 23(29): 7265 -7277. 79

Results – Chemotherapy Response • 89 evaluable patients – 11 patients with pathologic complete response (p. CR) • 4 pts ER+ by IHC – p. CR rate in ER+ pts = 8% (95% CI 1%-15%) • 7 pts ER– by IHC – p. CR rate in ER– pts = 23% (95% CI 8%-37%) – Overall p. CR rate = 12% Gianni et al. J Clin Oncol. 2005; 23(29): 7265 -7277. 80
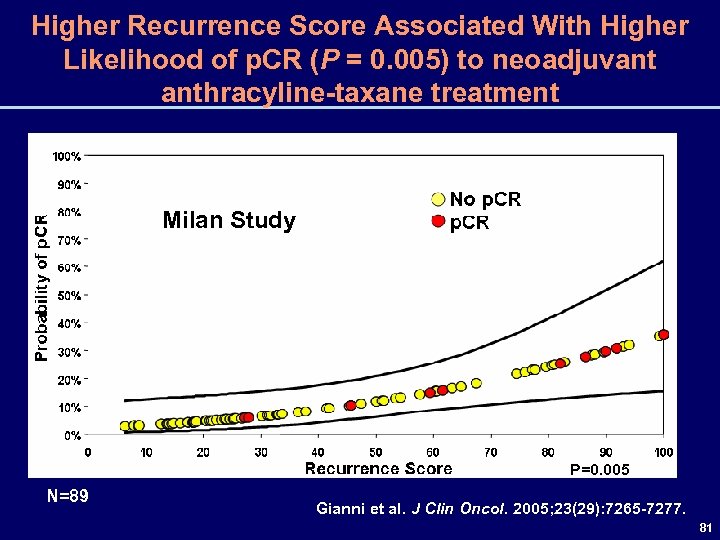
Higher Recurrence Score Associated With Higher Likelihood of p. CR (P = 0. 005) to neoadjuvant anthracyline-taxane treatment Milan Study P=0. 005 N=89 Gianni et al. J Clin Oncol. 2005; 23(29): 7265 -7277. 81

Gene expression profiles in paraffin -embedded core biopsies predict docetaxel chemosensitivity J. Chang, A. Makris, SG. Hilsenbeck, J. Hackett, J Jeong, M Liu, J Baker, K Sexton, K Osborne, S Shak

Methods Patients identified from 3 phase II clinical trials who received neoadjuvant docetaxel (100 mg/m 2 q 3 wks) for 4 cycles before surgery Diagnostic biopsies: 10 -micron sections x 3 98 clinically eligible patients 81 (83%) of 98 with adequate tumor tissue (≥ 5% tumor) 80 (99%) of 81 with adequate RNA and expression signal 72 (90%) of 80 with response data (RECIST) Chang, ASCO 2006, Abstract #538 83
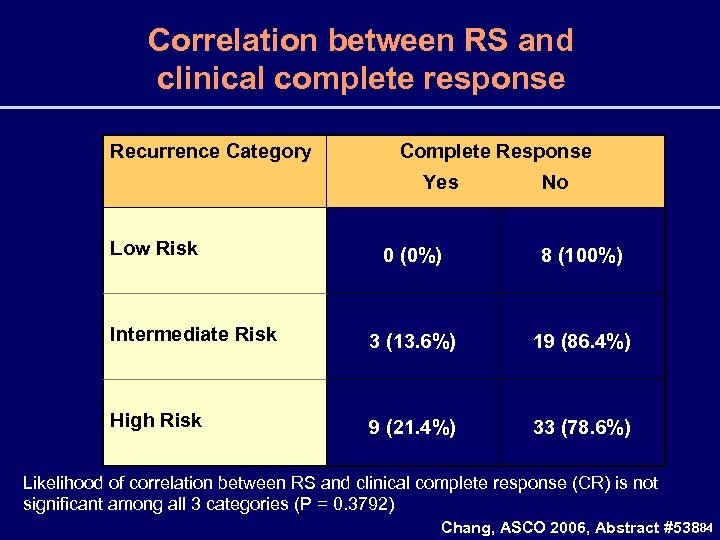
Correlation between RS and clinical complete response Recurrence Category Complete Response Yes Low Risk No 0 (0%) 8 (100%) Intermediate Risk 3 (13. 6%) 19 (86. 4%) High Risk 9 (21. 4%) 33 (78. 6%) Likelihood of correlation between RS and clinical complete response (CR) is not significant among all 3 categories (P = 0. 3792) Chang, ASCO 2006, Abstract #53884
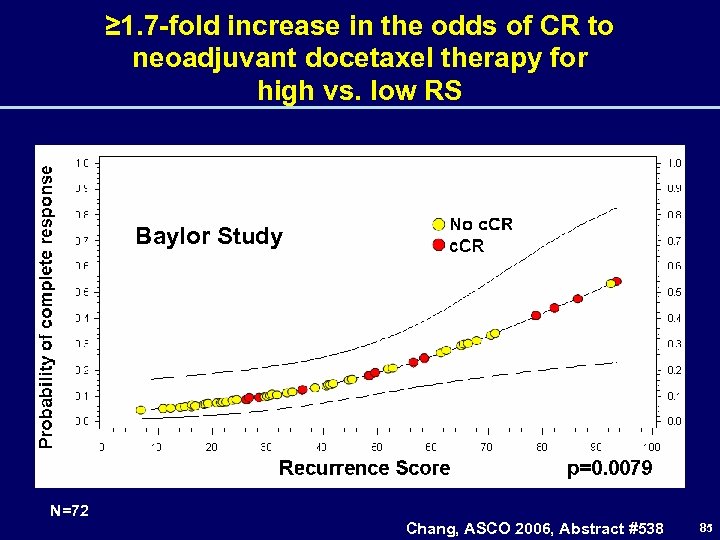
≥ 1. 7 -fold increase in the odds of CR to neoadjuvant docetaxel therapy for high vs. low RS Baylor Study N=72 Chang, ASCO 2006, Abstract #538 85

Exploratory Studies Summary • RS is associated with LRF • Associated with response to modern Neoadjuvant Chemotherapy • Prognostic in Node Positive Disease 86 86

Other Gene Profiles • Mamma. Print – 70 -gene signature – Netherlands–based Agendia • “Invasiveness” Gene Signature – 186 -gene signature – University of Michigan, Ann Arbor • Wound Response Signature 87

Mamma. Print and Oncotype DX Distinctions Mamma. Print Oncotype DX • Fresh Tissue by Microarray • 81% Microarray success rate for RNA extraction • Binary result (low risk vs. high risk) • Patients all <61 years old • FPET by RT-PCR • 99% FPET success rate for RNA extraction • Continuous Predictor Curve (Individual Recurrence Score) • 302 untreated patients (LN-, ER+, ER-) • Clinical Utility Not Yet Established –Patients not representative of those diagnosed and treated today • Insurance Coverage TBD • Multiple, reproducible studies in >2, 600 treated patients with ER+, LN- disease • Provides Clinical Utility for adjuvant treatment decisions –>20, 000 patients, 4, 500 physicians • Over 100 million lives insured • No age barriers 88

TAILORx (PACCT-1 Trial) Sponsored by NCI Administered by ECOG Participating cooperative groups include ECOG, SWOG, NCCTG, CALGB, NCIC, ACOSOG, and NSABP

Trial Assigning Individua. Lized Options for Treatment (Rx) (TAILORx) • Premise – Integration of a molecular profiling test (Oncotype DX™) into the clinical decision-making process • Implications – Reduce chemotherapy overtreatment in those likely to be optimally treated with hormonal therapy alone – Reduce inadequate treatment by identifying individuals who derive great benefit from chemotherapy – Evaluate benefit of chemotherapy where uncertainty still exists about its utility 90
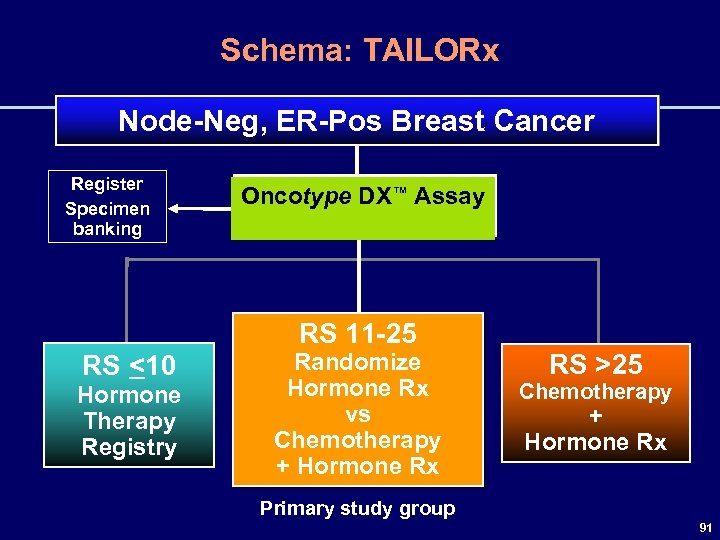
Schema: TAILORx Node-Neg, ER-Pos Breast Cancer Register Specimen banking Oncotype DX™ Assay RS 11 -25 RS <10 Hormone Therapy Registry Randomize Hormone Rx vs Chemotherapy + Hormone Rx RS >25 Chemotherapy + Hormone Rx Primary study group 91

Study Design: Primary Objectives • To determine whether adjuvant hormonal therapy (ie, experimental arm) is not inferior to adjuvant chemohormonal (standard arm) for patients in the “primary study group” (Oncotype DX™ RS 11 -25) • To create a tissue and specimen bank for patients enrolled in this trial to learn more about breast cancer 92

Study Design: Secondary Objectives • Low RS group (<11) – To determine risk of recurrence with hormonal therapy prospectively • Comparison with clinical models (Adjuvant! Online) • Refine estimates of Oncotype DX™ RS in contemporary practice • Evaluate individual gene groups – – – Proliferation gene group HER 2 gene group ER gene group Invasion gene group Other genes 93
94
0d4c28812a8d7cd3235923e978b4447c.ppt