84e6214a38c414a997011e5dcaa36a67.ppt
- Количество слайдов: 41
 Gastrointestinal Stromal Tumors Twelfth GRW Symposium Surgical Grand Rounds 1 -15 -04
Gastrointestinal Stromal Tumors Twelfth GRW Symposium Surgical Grand Rounds 1 -15 -04
 Gastrointestinal Stromal Tumors Case Presentation HPI: 31 yo male with a recent history of UGI bleed. EUS c/w duodenal mass. EUS bx – GIST tumor. Admitted OUMC 12/122/03 PMHX: Local excision of duodenal GIST in Oct 01 PE: Unremarkable except for well healed abdominal scar
Gastrointestinal Stromal Tumors Case Presentation HPI: 31 yo male with a recent history of UGI bleed. EUS c/w duodenal mass. EUS bx – GIST tumor. Admitted OUMC 12/122/03 PMHX: Local excision of duodenal GIST in Oct 01 PE: Unremarkable except for well healed abdominal scar
 Gastrointestinal Stromal Tumors Hosp course: Underwent pylorus preserving Whipple on 12/12/03 with findings: Mass in second portion of duodenum, no gross tumor elsewhere. Post-op developed RUQ fluid collection, amylase poor, resolved. Patient did well and discharged on 12/23/03 Pathology: 3. 0 cm malignant GIST, c-Kit positive, 1 of 7 nodes positive
Gastrointestinal Stromal Tumors Hosp course: Underwent pylorus preserving Whipple on 12/12/03 with findings: Mass in second portion of duodenum, no gross tumor elsewhere. Post-op developed RUQ fluid collection, amylase poor, resolved. Patient did well and discharged on 12/23/03 Pathology: 3. 0 cm malignant GIST, c-Kit positive, 1 of 7 nodes positive
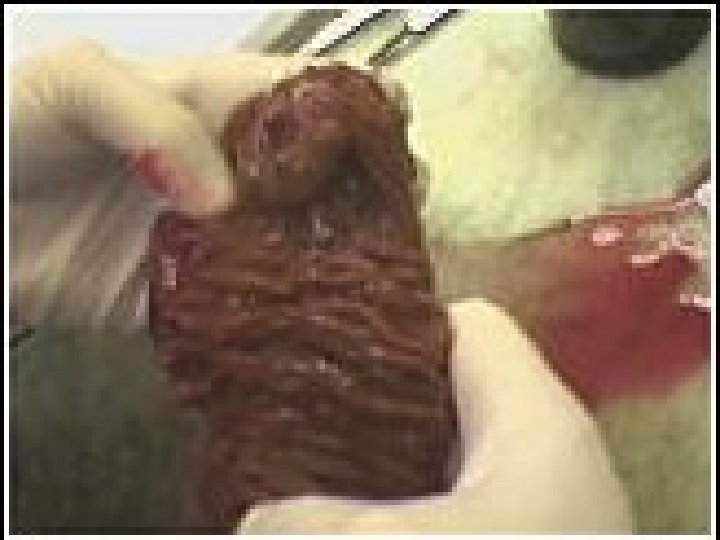
 Gastrointestinal Stromal Tumors Postoperative plans: Referred for consideration of adjuvant Imatinib Mesylate (Gleevec)
Gastrointestinal Stromal Tumors Postoperative plans: Referred for consideration of adjuvant Imatinib Mesylate (Gleevec)
 Gastrointestinal Stromal Tumors Case Presentation 2 HPI: 39 YO F transferred from outlying hospital 5 days S/P lap for UGI bleed with finding of duodenal mass. Bx c/w GIST tumor. Mass not resected. PMHx: Hepatitis C PE: Neg except for healing midline wound
Gastrointestinal Stromal Tumors Case Presentation 2 HPI: 39 YO F transferred from outlying hospital 5 days S/P lap for UGI bleed with finding of duodenal mass. Bx c/w GIST tumor. Mass not resected. PMHx: Hepatitis C PE: Neg except for healing midline wound
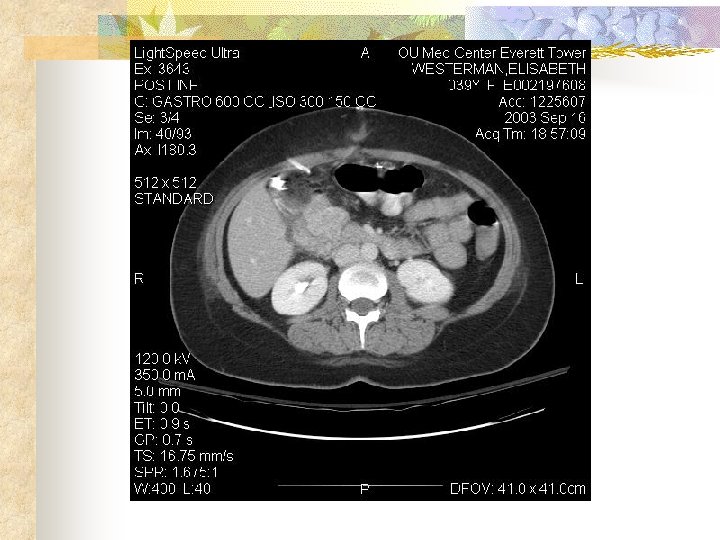
 Gastrointestinal Stromal Tumors Hospital course: Patient had no further UGI bleeding. Was seen in consultation by Med/Onc who recommended D/C until role of neoadjuvant therapy could be defined. Ultimately recommended against neoadjuvant therapy.
Gastrointestinal Stromal Tumors Hospital course: Patient had no further UGI bleeding. Was seen in consultation by Med/Onc who recommended D/C until role of neoadjuvant therapy could be defined. Ultimately recommended against neoadjuvant therapy.
 Gastrointestinal Stromal Tumors Hospital course: Underwent re-exploration and pylorus preserving Whipple on 10/27/03. Findings – mass in 2 nd portion of duodenum without evidence of metastases. Pathology – 4. 5 cm, hypercellular GIST tumor, c-Kit positive, no evidence of nodal metastases, uncertain malignant potential. Patient did well and discharged for followup by surgery and med/onc.
Gastrointestinal Stromal Tumors Hospital course: Underwent re-exploration and pylorus preserving Whipple on 10/27/03. Findings – mass in 2 nd portion of duodenum without evidence of metastases. Pathology – 4. 5 cm, hypercellular GIST tumor, c-Kit positive, no evidence of nodal metastases, uncertain malignant potential. Patient did well and discharged for followup by surgery and med/onc.
 Gastrointestinal Stromal Tumors What are they? 1. Soft tissue sarcomas 2. Arise from interstitial cells of Cajal 3. Represent 0. 1% - 3% of GI cancers 4. Represent 5% of all soft tissue sarcomas 5. 10% - 30% are highly malignant 6. c-kit or PDGFRA positive tumors Int J Cancer 107: 2003
Gastrointestinal Stromal Tumors What are they? 1. Soft tissue sarcomas 2. Arise from interstitial cells of Cajal 3. Represent 0. 1% - 3% of GI cancers 4. Represent 5% of all soft tissue sarcomas 5. 10% - 30% are highly malignant 6. c-kit or PDGFRA positive tumors Int J Cancer 107: 2003
 Gastrointestinal Stromal Tumors n n n Tyrosine Kinase Receptors 1 of 4 classes of cell-surface receptors 1 of 2 types of protein kinases Lack catalytic activity Ligand binding forms dimeric receptor Activates a cytosolic protein-tyrosine kinase
Gastrointestinal Stromal Tumors n n n Tyrosine Kinase Receptors 1 of 4 classes of cell-surface receptors 1 of 2 types of protein kinases Lack catalytic activity Ligand binding forms dimeric receptor Activates a cytosolic protein-tyrosine kinase
 Gastrointestinal Stromal Tumors n n n All are potentially malignant 1 st tumor to benefit from “targeted” therapy Criteria to predict behavior tumor behavior n n n Size Necrosis Mitotic rate
Gastrointestinal Stromal Tumors n n n All are potentially malignant 1 st tumor to benefit from “targeted” therapy Criteria to predict behavior tumor behavior n n n Size Necrosis Mitotic rate
 Gastrointestinal Stromal Tumors Risk group Very low Low Intermediate High Risk of Metastases Size Mitotic count <2 cm <5 per 50 HPFs 2 -5 cm <5 per 50 HPFs <5 cm 6 -10 per 50 HPFs 5 -10 cm <5 per 50 HPFs >5 cm >5 per 50 HPFs >10 cm any mitotic rate any size >10 per HPFs
Gastrointestinal Stromal Tumors Risk group Very low Low Intermediate High Risk of Metastases Size Mitotic count <2 cm <5 per 50 HPFs 2 -5 cm <5 per 50 HPFs <5 cm 6 -10 per 50 HPFs 5 -10 cm <5 per 50 HPFs >5 cm >5 per 50 HPFs >10 cm any mitotic rate any size >10 per HPFs
 Gastrointestinal Stromal Tumors n n Incidence appears to be increasing Mayo clinic study indicates incidence stable but diagnostic criteria changed Median age at diagnosis 58 years (range 40 -80 years ? ) 5 year survival variable (28 – 60%) Ann Surg 231: 2000
Gastrointestinal Stromal Tumors n n Incidence appears to be increasing Mayo clinic study indicates incidence stable but diagnostic criteria changed Median age at diagnosis 58 years (range 40 -80 years ? ) 5 year survival variable (28 – 60%) Ann Surg 231: 2000
 Gastrointestinal Stromal Tumors Location n n Stomach 54% Small bowel 47% Rectum 6% Retroperitoneum 7% Clin Cancer Res 9(9): 2003
Gastrointestinal Stromal Tumors Location n n Stomach 54% Small bowel 47% Rectum 6% Retroperitoneum 7% Clin Cancer Res 9(9): 2003
 Gastrointestinal Stromal Tumors n n n Presentation Generally nonspecific GI bleeding 50% Abdominal pain 20 -50% Obstruction 10 -30% Asymptomatic 20% Int J Cancer 107: 2003, Ann Surg 231: 2000, Ann Chir Gynaecol 87: 1998
Gastrointestinal Stromal Tumors n n n Presentation Generally nonspecific GI bleeding 50% Abdominal pain 20 -50% Obstruction 10 -30% Asymptomatic 20% Int J Cancer 107: 2003, Ann Surg 231: 2000, Ann Chir Gynaecol 87: 1998
 Gastrointestinal Stromal Tumors Genetics c-kit, a 145 k. D transmembrane glycoprotein n n Activating mutation usually at codon 11 Constitutive ligand-independent activation of kinase
Gastrointestinal Stromal Tumors Genetics c-kit, a 145 k. D transmembrane glycoprotein n n Activating mutation usually at codon 11 Constitutive ligand-independent activation of kinase
 Gastrointestinal Stromal Tumors Genetics Platelet derived growth receptor alpha (PDGFRA) n Tyrosine kinase activator n Similar to c-kit n Helps define GIST
Gastrointestinal Stromal Tumors Genetics Platelet derived growth receptor alpha (PDGFRA) n Tyrosine kinase activator n Similar to c-kit n Helps define GIST
 Gastrointestinal Stromal Tumors n n n Treatment Surgical excision is primary treatment option but recurrence rates are high Resistant to standard chemotherapy regimens due to over-expression of efflux pumps (p-glycoprotein & MDR-1) Radiation therapy limited by large tumor sizes and sensitivity of adjacent bowel
Gastrointestinal Stromal Tumors n n n Treatment Surgical excision is primary treatment option but recurrence rates are high Resistant to standard chemotherapy regimens due to over-expression of efflux pumps (p-glycoprotein & MDR-1) Radiation therapy limited by large tumor sizes and sensitivity of adjacent bowel
 Gastrointestinal Stromal Tumors n n Imatinib Mesylate Orally bioactive tyrosine kinase inhibitor Shown to be effective against GIST tumors in two trials in the US and Europe reported in 2001 & 2002 N Engl J Med 347: 2002 Lancet 358: 2001
Gastrointestinal Stromal Tumors n n Imatinib Mesylate Orally bioactive tyrosine kinase inhibitor Shown to be effective against GIST tumors in two trials in the US and Europe reported in 2001 & 2002 N Engl J Med 347: 2002 Lancet 358: 2001
 Gastrointestinal Stromal Tumors n n Studies University of Chicago Oregon University and Health Sciences Washington Hospital Center Johns Hopkins Hospitals
Gastrointestinal Stromal Tumors n n Studies University of Chicago Oregon University and Health Sciences Washington Hospital Center Johns Hopkins Hospitals
 Gastrointestinal Stromal Tumors n n n University of Chicago Retrospective study 1995 -2002 57 patients 96% c-kit positive Curative resection in 50% Treatment of metastatic disease 50% Wu, et al. Surgery. 2003; 134(4): 656.
Gastrointestinal Stromal Tumors n n n University of Chicago Retrospective study 1995 -2002 57 patients 96% c-kit positive Curative resection in 50% Treatment of metastatic disease 50% Wu, et al. Surgery. 2003; 134(4): 656.
 Gastrointestinal Stromal Tumors University of Chicago Imatinib mesylate treatment n 29 patients treated for metastatic disease n 3 received adjuvant therapy after resection n Median follow-up 18 months
Gastrointestinal Stromal Tumors University of Chicago Imatinib mesylate treatment n 29 patients treated for metastatic disease n 3 received adjuvant therapy after resection n Median follow-up 18 months
 Gastrointestinal Stromal Tumors University of Chicago Results n 23 (40%) patients alive and NED n 22 (39%) patients alive with disease n 7 (13%) patients died n 5 (10%) lost to follow-up n Overall survival 87% at 18 months
Gastrointestinal Stromal Tumors University of Chicago Results n 23 (40%) patients alive and NED n 22 (39%) patients alive with disease n 7 (13%) patients died n 5 (10%) lost to follow-up n Overall survival 87% at 18 months
 Gastrointestinal Stromal Tumors University of Chicago Results in 26 patients with metastatic disease n All treated with Imatinib mesylate n 5 (2%) died n 22 (84%) stable disease or regression
Gastrointestinal Stromal Tumors University of Chicago Results in 26 patients with metastatic disease n All treated with Imatinib mesylate n 5 (2%) died n 22 (84%) stable disease or regression
 Gastrointestinal Stromal Tumors University of Chicago Imatinib mesylate adjuvant therapy results n Only 3 patients in adjuvant group n All considered “high risk” due to tumor characteristics n All NED with mean follow-up of 7 months n 1 “high risk” patient who refused adjuvant therapy developed liver mets at 9 months
Gastrointestinal Stromal Tumors University of Chicago Imatinib mesylate adjuvant therapy results n Only 3 patients in adjuvant group n All considered “high risk” due to tumor characteristics n All NED with mean follow-up of 7 months n 1 “high risk” patient who refused adjuvant therapy developed liver mets at 9 months
 Gastrointestinal Stromal Tumors University of Chicago Toxicity of Imatinib mesylate n Generally well tolerated n Mild fatigue n Periorbital edema n Mild diarrhia n 2 deaths not due to disease not relate to therapy
Gastrointestinal Stromal Tumors University of Chicago Toxicity of Imatinib mesylate n Generally well tolerated n Mild fatigue n Periorbital edema n Mild diarrhia n 2 deaths not due to disease not relate to therapy
 Gastrointestinal Stromal Tumors University of Chicago Study summary n No neoadjuvant data n Patients in “low risk”group with R 0 resections did well n Patients treated with metastatic disease did well in short term n Adjuvant data group too small for analysis
Gastrointestinal Stromal Tumors University of Chicago Study summary n No neoadjuvant data n Patients in “low risk”group with R 0 resections did well n Patients treated with metastatic disease did well in short term n Adjuvant data group too small for analysis
 Gastrointestinal Stromal Tumors n n n Oregon University Study Prospective study of 127 patients in phase II multi -center trial of Imatinib mesylate Evaluated relationship between mutation type (ckit & PDGFR) and clinical outcome Further evaluated codon mutation site and outcome Evaluated in-vitro response to Imatinib Evaluated clinical response to Imatinib Heinrich, et al. J Clin Onc, 1(23), 2003: 4342.
Gastrointestinal Stromal Tumors n n n Oregon University Study Prospective study of 127 patients in phase II multi -center trial of Imatinib mesylate Evaluated relationship between mutation type (ckit & PDGFR) and clinical outcome Further evaluated codon mutation site and outcome Evaluated in-vitro response to Imatinib Evaluated clinical response to Imatinib Heinrich, et al. J Clin Onc, 1(23), 2003: 4342.
 Gastrointestinal Stromal Tumors University of Oregon Study Results n Invitro responses differ with mutation and codon site n Clinical responses differ with mutation and codon site n May eventually allow even more directed therapy as newer drugs are developed
Gastrointestinal Stromal Tumors University of Oregon Study Results n Invitro responses differ with mutation and codon site n Clinical responses differ with mutation and codon site n May eventually allow even more directed therapy as newer drugs are developed
 Gastrointestinal Stromal Tumors n n n Washington Hospital Center Study Retrospective 69 patients with compatible histology and c -kit positivity Study purposes n n Develop prognostic indicators Establish the natural history of the tumor
Gastrointestinal Stromal Tumors n n n Washington Hospital Center Study Retrospective 69 patients with compatible histology and c -kit positivity Study purposes n n Develop prognostic indicators Establish the natural history of the tumor
 Gastrointestinal Stromal Tumors Washington Hospital Center Study Results n Tumor site n n Even distribution between stomach and small bowel Prognostic factors n n n Tumor size Peritoneal cancer index Completeness of cytoreduction
Gastrointestinal Stromal Tumors Washington Hospital Center Study Results n Tumor site n n Even distribution between stomach and small bowel Prognostic factors n n n Tumor size Peritoneal cancer index Completeness of cytoreduction
 Gastrointestinal Stromal Tumors n n n Johns Hopkins 38 tumors studied Evaluated tumor suppressor genes Assessed degree of hypermethylation House, et al. J Gastroint Surg, 7(8); 2003: 1004.
Gastrointestinal Stromal Tumors n n n Johns Hopkins 38 tumors studied Evaluated tumor suppressor genes Assessed degree of hypermethylation House, et al. J Gastroint Surg, 7(8); 2003: 1004.
 Gastrointestinal Stromal Tumors n n Johns Hopkins Hypermethylation of Cp. G-rich islands is common in human malignancies Occurs in promotor area of gene Can it be used to predict tumor behavior? Does it explain resistance to Imatinib?
Gastrointestinal Stromal Tumors n n Johns Hopkins Hypermethylation of Cp. G-rich islands is common in human malignancies Occurs in promotor area of gene Can it be used to predict tumor behavior? Does it explain resistance to Imatinib?
 Gastrointestinal Stromal Tumors n n n Johns Hopkins 38 tumors resected from 1989 to 2001 All c-kit positive 11 candidate tumor suppressor genes studied
Gastrointestinal Stromal Tumors n n n Johns Hopkins 38 tumors resected from 1989 to 2001 All c-kit positive 11 candidate tumor suppressor genes studied
 Gastrointestinal Stromal Tumors n n n Johns Hopkins 84% of tumors had hypermethylation of at least 1 gene Multigene methylation seen in 42% of tumors E-cadherin hypermethylation and absence of methylation of h. MLH 1 predicted early recurrence
Gastrointestinal Stromal Tumors n n n Johns Hopkins 84% of tumors had hypermethylation of at least 1 gene Multigene methylation seen in 42% of tumors E-cadherin hypermethylation and absence of methylation of h. MLH 1 predicted early recurrence
 Gastrointestinal Stromal Tumors n n n Summary GIST tumors now well characterized All have malignant potential Complete surgical resection important Metastatic disease responds to Imatinib Role of neoadjuvant therapy unclear Role adjuvant therapy unclear
Gastrointestinal Stromal Tumors n n n Summary GIST tumors now well characterized All have malignant potential Complete surgical resection important Metastatic disease responds to Imatinib Role of neoadjuvant therapy unclear Role adjuvant therapy unclear
 Gastrointestinal Stromal Tumors Currently Available Trials Neoadjuvant study n n n RTOG S-0132/ACRIN 6665 Patients with recurrent or measurable peritoneal disease 8 wks Imatinib followed by resection
Gastrointestinal Stromal Tumors Currently Available Trials Neoadjuvant study n n n RTOG S-0132/ACRIN 6665 Patients with recurrent or measurable peritoneal disease 8 wks Imatinib followed by resection
 Gastrointestinal Stromal Tumors Currently Available Trials Adjuvant study EORTC 64024 n Patients with R 0 resections eligible n Patients stratified according to risk factors n Patients randomized to either n n Imatinib 400 mg/day X 2 years Observation
Gastrointestinal Stromal Tumors Currently Available Trials Adjuvant study EORTC 64024 n Patients with R 0 resections eligible n Patients stratified according to risk factors n Patients randomized to either n n Imatinib 400 mg/day X 2 years Observation
 Gastrointestinal Stromal Tumors Currently Available Trials Adjuvant study ACOSOG Z 9001 n Eligible patients n n n Complete gross resections R 0 or R 1 (microscopic + margins) Randomization arms n n n Imatinib 400 mg/day X 1 year Placebo patients with recurrence receive Imatinib for 2 years
Gastrointestinal Stromal Tumors Currently Available Trials Adjuvant study ACOSOG Z 9001 n Eligible patients n n n Complete gross resections R 0 or R 1 (microscopic + margins) Randomization arms n n n Imatinib 400 mg/day X 1 year Placebo patients with recurrence receive Imatinib for 2 years
 Gastrointestinal Stromal Tumors Thank-you
Gastrointestinal Stromal Tumors Thank-you


