85ddbfb9f7017ea837071450aa672380.ppt
- Количество слайдов: 32

Future directions for agricultural biotechnology Dr. Kirstin Carroll Outreach in Resource Biotechnology Program Oregon State University

Lecture Outline • What is molecular farming in plants? • Why use plants? • What are the risks and concerns? • Current and evolving regulation

What is 'molecular farming in plants'? The use of agricultural plants for the production of useful molecules for non food, feed or fiber applications. Plants are already grown to produce valuable molecules, including many drugs. Molecular farming is different because the plants are genetically engineered (GE) to produce the molecules we want them to.

What is GE? Create recombinant DNA with gene from same or different organism Transfer DNA to plant cell (use either Agrobacterium or ‘ballistic’ transformation) Confirm introduced DNA and expression of foregin protein in plant What is included in the recombinant DNA? On/Off switch Gene of interest Marker gene Environment contaminantion via gene flow Contamination of food supply Secondary metabolite – inctroduct allerginiicty or toxicity

Plant Products 1. Plant derived pharmaceuticals (non-GE) • Over 120 pharmaceutical products currently in use are derived from plants. Mainly from tropical forest species

Plant Products 1. Plant-derived pharmaceuticals (non-GE) 2. Plant-made pharmaceuticals and industrial products (GE) Industrial products proteins enzymes modified starches fats oils waxes plastics Pharmaceuticals recombinant human proteins Therapeutic proteins enzymes Antibodies (plantibodies) vaccines

Strategies for ‘Molecular Farming’ 1. Plant gene expression strategies • Transient transformation • Stable transformation • Chloroplast transformation
Strategies for ‘Molecular Farming’ 1. Plant gene expression strategies 2. Location of trans-gene expression? Protein quantity and preservation • Whole plant • Target specific tissues (e. g. seed, root)
Strategies for ‘Molecular Farming’ 1. Plant gene expression system 2. Location of trans-gene expression? 3. Selection of plant species and characteristics • Mode of reproduction – self/outcrossing • Yield, harvest, production, processing

Why use plants? Advantages Disadvantages Cost reduction Environment contamination Stability Food supply contamination Safety Health safety concerns

Examples of Industrial PMPs Cellulase for production of alcohols Avidin – medical diagnostics b-glycoprotein – biomedical diagnostics Plant-derived plastic: Production of polyhydroxyalkanoate (PHA) To date, more costly than fuel-based plastic

Examples of Industrial PMPs High wax esters Jojoba seeds - gene has been isolated and expressed in Arabidopsis (49 -70% oil present as wax) Astaxanthin red pigment in shell-fish. used in aquaculture Compounds to increase flavor and fragrances

Plant-made Vaccines Edible vaccines Advantages: Administered Directly no purification required no hazards assoc. w/injections Production may be grown locally, where needed most no transportation costs Naturally stored

Plant-made Vaccines Examples of edible vaccines ; pig vaccine in corn, HIV-suppressing protein in spinach, human vaccine for hepatitus B in potato.

Plantibodies - Plants can be used to produce monoclonal antibodies - Tobacco, corn, potatoes, soy, alfalfa, rice - Free from potential contamination of mammalian viruses - Examples: cancer, dental caries, herpes simplex virus, respiratory syncytial virus **GE Corn can produce up to 1 kg antibody/acre and can be stored at RT for up to 5 years! Humphreys DP et al. Curr Opin Drug Discover Dev 2001; 4: 172 -85.

Plant made Pharmaceuticals Therapeutic proteins Blood substitutes – human hemoglobin Proteins to treat diseases CF, HIV, Hypertension, Hepatitis B…. . many others **To date, no plant-produced pharmaceuticals are commercially available.

Current ‘Pharm’ Companies Planet Biotechnology Dental Caries: Caro. Rx™ Colds due to Rhinovirus: Rhino. Rx™ Drug-induced Alopecia: Doxo. Rx™ Biomass biorefinery based on switchgrass. Produce PHAs in green tissue plants for fuel generation. LEX System™ Lemna, (duckweed) Rhizosecretion • Monoclonal antibodies (Drake et al. , 2003) • Recombinant proetins (Gaume et al, 2003)

Current ‘Pharm’ Companies Kentucky Tobacco Research and Development Center Trangenic tobacco Gene. Ware® PMPs and non-protein substances (flavors and fragrances, medicinals, Controlled Pharming and natural insecticides) Ventures In collaboration w/Purdue Transgenic corn Converted limestone mine facility

Current ‘Pharm’ Companies Ventria Bioscience Prodigene Transgenic corn Trypsin and Aprotinin Transgenic rice Lactoferrin Lysozyme

Examples of Current Research • Genetically engineered Arabidopsis plants can sequester arsenic from the soil. (Dhankher et al. 2002 Nature Biotechnology) • Immunogenicity in human of an edible vaccine for hepatitis B (Thanavala et al. , 2005. PNAS) • Expression of single-chain antibodies in transgenic plants. (Galeffi et al. , 2005 Vaccine) • Plant based HIV-1 vaccine candidate: Tat protein produced in spinach. (Karasev et al. 2005 Vaccine) • Plant-derived vaccines against diarrheal diseases. (Tacket. 2005 Vaccine)

Risks and Concerns Environment contamination Gene flow via pollen Non-target species near field sites e. g. butterflies, bees, etc Food supply contamination Accident, intentional, gene flow Health safety concerns Non-target organ responses Side-effects Allergenicity

U. S. Regulatory System (existing regulations) USDA Field Testing -permits -notifications Determination of non-regulated status FDA Food safety Feed safety EPA Pesticide and herbicide registration

Breakdown of Regulatory System: Prodigene Incident 2002 2001 : Field trails of GE corn producing pig vaccine were planted in IA and NB. 2002: USDA discovered “volunteer” corn plants in fields in both IA and NE. Soy was already planted in NE site. $500, 000 fine + $3 million to buy/destroy contaminated soy

USDA Response to Incident Revised regulations so that they were distinct from commodity crops: • Designated equipment must be used. • At least 5 inspections/yr. • Pharm crops must be grown at least 1 mile away from any other fields and planted 28 days before/after surrounding crops

Current Evolving Regulations FDA/USDA Guidance for Industry on Plant-Made Pharmaceuticals Regulations November 2004: Draft Document Other challenges: Industrial hygiene and safety programs
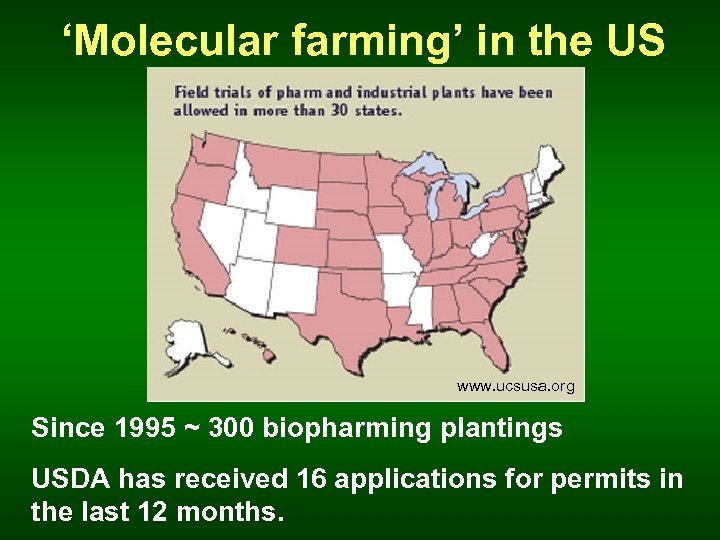
‘Molecular farming’ in the US www. ucsusa. org Since 1995 ~ 300 biopharming plantings USDA has received 16 applications for permits in the last 12 months.

‘Molecular farming’ opposition Concerns: • CONTAINMENT – opponents want a guarantee of 0% contamination of the food supply. • Full disclosure of field trials, crop, gene, location, etc. • Extensive regulatory framework

Suggested Safeguards for ‘molecular farming’ 1. Physical differences E. g. “purple” maize, GFP 2. Sterility Use male sterile plants Terminator technology? 3. Easily detectable by addition of 'reporter genes' PCR markers (avoid antibiotic resistance markers)

Suggested Safeguards for ‘molecular farming’ 4. Chloroplast expression system Increase yield Eliminates potential gene flow Technically difficult (Chlorogen Company) 5. Complete disclosure of DNA sequences 6. Legislate for administration.

Alternatives to ‘molecular farming’? Use only traditional drug production systems microbial, yeast and fungi mammalian cell culture Use only fully contained production systems: Plant cell cultures Hydroponics (rhizosecretion) Greenhouses Use non-food crops Tobacco, Hemp/Cannabis

Economics The expectation is for lower production costs however there is no evidence that pharming will produce cheaper, safe drugs. There are unknown costs associated with containment, litigation and liability, production…. . others?

85ddbfb9f7017ea837071450aa672380.ppt