679028c9e7e71e5075f451464ed558e4.ppt
- Количество слайдов: 25
 Functional Foods and Nutrition Research Mary E Penny Instituto de Investigacion Nutricional 14 th October 2014
Functional Foods and Nutrition Research Mary E Penny Instituto de Investigacion Nutricional 14 th October 2014
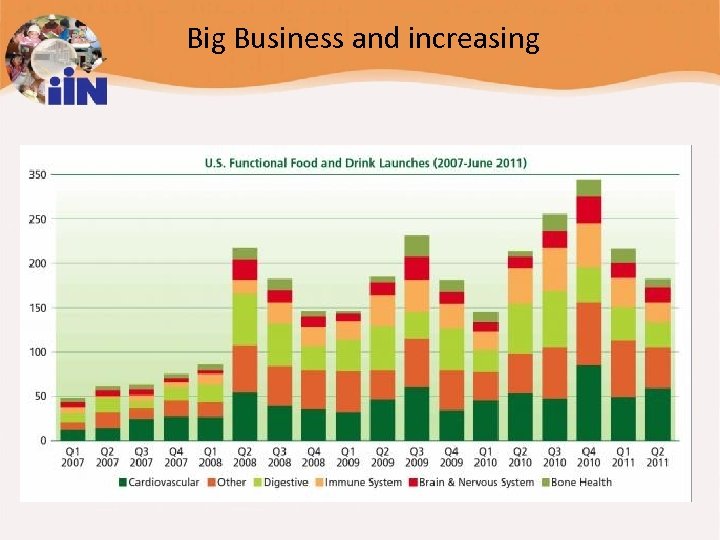 Big Business and increasing
Big Business and increasing
 Definition of Functional Foods Food with added health benefits beyond the usual nutritional value No single agreed definition Usually excludes “supplements”
Definition of Functional Foods Food with added health benefits beyond the usual nutritional value No single agreed definition Usually excludes “supplements”
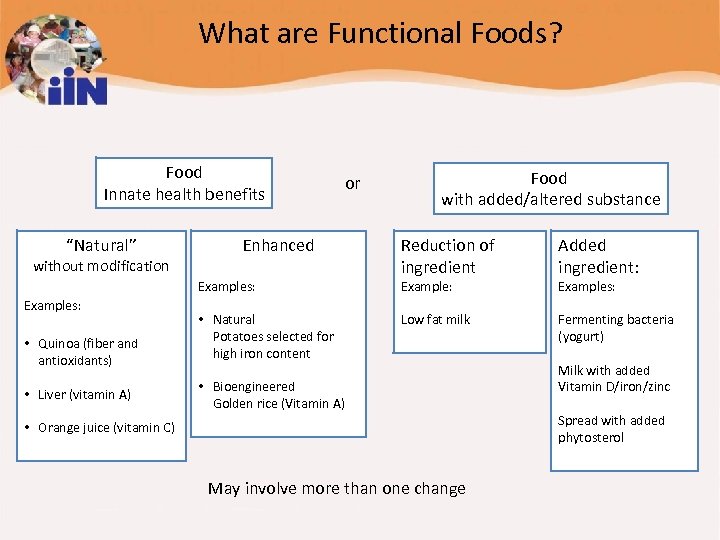 What are Functional Foods? Food Innate health benefits “Natural” without modification or Enhanced Examples: • Quinoa (fiber and antioxidants) • Liver (vitamin A) • Natural Potatoes selected for high iron content Food with added/altered substance Reduction of ingredient Added ingredient: Low fat milk Fermenting bacteria (yogurt) Example: • Bioengineered Golden rice (Vitamin A) • Orange juice (vitamin C) May involve more than one change Examples: Milk with added Vitamin D/iron/zinc Spread with added phytosterol
What are Functional Foods? Food Innate health benefits “Natural” without modification or Enhanced Examples: • Quinoa (fiber and antioxidants) • Liver (vitamin A) • Natural Potatoes selected for high iron content Food with added/altered substance Reduction of ingredient Added ingredient: Low fat milk Fermenting bacteria (yogurt) Example: • Bioengineered Golden rice (Vitamin A) • Orange juice (vitamin C) May involve more than one change Examples: Milk with added Vitamin D/iron/zinc Spread with added phytosterol
 Foods and Food components “Functional” maybe a property of a whole type of food Fruits and vegetables Whole Grains Property of a specific “ingredient “ of the food Fiber, Vitamin A Zinc Antioxidant Protein With increasing interest in “Health” benefits of food there came an increasing tendency to make health claims for foods and so a need for definitions and regulations especially for Foods that have been modified
Foods and Food components “Functional” maybe a property of a whole type of food Fruits and vegetables Whole Grains Property of a specific “ingredient “ of the food Fiber, Vitamin A Zinc Antioxidant Protein With increasing interest in “Health” benefits of food there came an increasing tendency to make health claims for foods and so a need for definitions and regulations especially for Foods that have been modified
 Examples of Categories: Institute of Food Technologists
Examples of Categories: Institute of Food Technologists
 Japan leader in Functional foods 1980 s “Foods for specialized health use” Functional Food Task Force of International Life Sciences Institute (Europe) ILSI Europe Initiated a “concerted action project” in 2001 FUFUSE (Functional food science in Europe) EU - Foods with scientifically substantiated benefits for health and/or performance above normal nutritional functions Purpose: Food products eligible for Health Claims Process for the assessment of Scientific support for claims on food (PASSCLAIM – 2007) Beyond PASSCLAIM – New Guidance (2009) ILSI Europe: Beyond PASSCLAIM 2010
Japan leader in Functional foods 1980 s “Foods for specialized health use” Functional Food Task Force of International Life Sciences Institute (Europe) ILSI Europe Initiated a “concerted action project” in 2001 FUFUSE (Functional food science in Europe) EU - Foods with scientifically substantiated benefits for health and/or performance above normal nutritional functions Purpose: Food products eligible for Health Claims Process for the assessment of Scientific support for claims on food (PASSCLAIM – 2007) Beyond PASSCLAIM – New Guidance (2009) ILSI Europe: Beyond PASSCLAIM 2010
 The EU Concerted Action - Two types of health claims relevant to functional foods, must always be valid in the context of the whole diet and must relate to the amounts of foods normally consumed. These are: 1. TYPE A: "Enhanced function"claims that refer to specific physiological, psychological functions and biological activities beyond their established role in growth, development and other normal functions of the body. This type of claim makes no reference to a disease or a pathological state, e. g. certain non -digestible oligosaccharides improve the growth of a specific bacterial flora in the gut; caffeine can improve cognitive performance. 2. TYPE B "Reduction of disease-risk "claims that relate to the consumption of a food or food component that might help reduce the risk of a specific disease or condition because of specific nutrients or non-nutrients contained within it (e. g. folate can reduce a woman's risk of having a child with neural tube defects, and sufficient calcium intake may help to reduce the risk of osteoporosis in later life).
The EU Concerted Action - Two types of health claims relevant to functional foods, must always be valid in the context of the whole diet and must relate to the amounts of foods normally consumed. These are: 1. TYPE A: "Enhanced function"claims that refer to specific physiological, psychological functions and biological activities beyond their established role in growth, development and other normal functions of the body. This type of claim makes no reference to a disease or a pathological state, e. g. certain non -digestible oligosaccharides improve the growth of a specific bacterial flora in the gut; caffeine can improve cognitive performance. 2. TYPE B "Reduction of disease-risk "claims that relate to the consumption of a food or food component that might help reduce the risk of a specific disease or condition because of specific nutrients or non-nutrients contained within it (e. g. folate can reduce a woman's risk of having a child with neural tube defects, and sufficient calcium intake may help to reduce the risk of osteoporosis in later life).
 Possible health benefits Maintenance of health (Enhanced function) Reduction of specific illness or deficiency – disease risk Treatment or management of illness Gastrointestinal health Calcium - osteoporosis Low fat – overweight The gut microbiome Vitamin A – night blindness Gluten free – celiacs Linear growth in infancy N-3 PUFA – Cardiovascular risk Low glycemic index – Diabetes M Antioxidants – effects on ageing Folic acid – Spina bifida Improved sports performance Mental health – memory European commision: Functional foods 2010
Possible health benefits Maintenance of health (Enhanced function) Reduction of specific illness or deficiency – disease risk Treatment or management of illness Gastrointestinal health Calcium - osteoporosis Low fat – overweight The gut microbiome Vitamin A – night blindness Gluten free – celiacs Linear growth in infancy N-3 PUFA – Cardiovascular risk Low glycemic index – Diabetes M Antioxidants – effects on ageing Folic acid – Spina bifida Improved sports performance Mental health – memory European commision: Functional foods 2010
 2009 ILSI workshop Objective of the workshop was to provide guidance to substantiate health claims on foods Food producers, manufacturers, regulators, public needed to know the evidence on which claims could be made, the basis for claims and the process of regulating claims – note that the concern was to regulate “claims” not “food” Not an easy task: • • • Risk assessment – safety Changing priorities in health – epidemic of non communicable diseases Context of the rest of the diet Variability across consumers Links between diet and health not always known CODEX – Alimentaria provides international regulations and standards with regard to food Considers safety and scientific validity of claims about foods – doesn´t specify “Functional foods” Same framework as conventional foods Lupton JR. Scientific substantiation of claims in the USA: focus on functional foods. Eur J Nutr 2009; 48 (suppl 1): S 27 -S 31
2009 ILSI workshop Objective of the workshop was to provide guidance to substantiate health claims on foods Food producers, manufacturers, regulators, public needed to know the evidence on which claims could be made, the basis for claims and the process of regulating claims – note that the concern was to regulate “claims” not “food” Not an easy task: • • • Risk assessment – safety Changing priorities in health – epidemic of non communicable diseases Context of the rest of the diet Variability across consumers Links between diet and health not always known CODEX – Alimentaria provides international regulations and standards with regard to food Considers safety and scientific validity of claims about foods – doesn´t specify “Functional foods” Same framework as conventional foods Lupton JR. Scientific substantiation of claims in the USA: focus on functional foods. Eur J Nutr 2009; 48 (suppl 1): S 27 -S 31
 How can research help define health claims?
How can research help define health claims?
 Study Design of a study to assess Functional Foods and their components Functional food Consumption/Intake (Bio) markers Food or food component should be characterized. Minumal dose defined Mechanism of action useful but not essential Should comply with existing regulation Safety Stability, Interactions For instance a food with added mineral might be eaten with high fiber or phytate foods lowering bioavailability. Breastmilk How will cooking affect it – example DHA and high temperature cooking How will storage affect it? Organoleptic properties Multimicronutrients, Fish Health change (Health outcome)
Study Design of a study to assess Functional Foods and their components Functional food Consumption/Intake (Bio) markers Food or food component should be characterized. Minumal dose defined Mechanism of action useful but not essential Should comply with existing regulation Safety Stability, Interactions For instance a food with added mineral might be eaten with high fiber or phytate foods lowering bioavailability. Breastmilk How will cooking affect it – example DHA and high temperature cooking How will storage affect it? Organoleptic properties Multimicronutrients, Fish Health change (Health outcome)
 Study Design Target population : who do we want to benefit Study group must be representative of target group Age Cultural considerations/socio-economic Dietary practices/mores Functional food Consumption/Intake (Bio) markers Health change (Health outcome) Healthy vs “not healthy” Low, normal and High responders Physical activity level Appropriate controls Example : Cereal with Probiotic to reduce diarrhea would need to be targetted at children 0 -2 years, population with high diarrhea rates, eating cereal paps,
Study Design Target population : who do we want to benefit Study group must be representative of target group Age Cultural considerations/socio-economic Dietary practices/mores Functional food Consumption/Intake (Bio) markers Health change (Health outcome) Healthy vs “not healthy” Low, normal and High responders Physical activity level Appropriate controls Example : Cereal with Probiotic to reduce diarrhea would need to be targetted at children 0 -2 years, population with high diarrhea rates, eating cereal paps,
 Study Design of a study to assess a Functional Food Functional food Consumption/Intake (Bio) markers Health change (Health outcome) Dose/amount should be consistent with normal consumption Adequate duration and follow-up Food matrix and dietary context Monitoring of compliance
Study Design of a study to assess a Functional Food Functional food Consumption/Intake (Bio) markers Health change (Health outcome) Dose/amount should be consistent with normal consumption Adequate duration and follow-up Food matrix and dietary context Monitoring of compliance
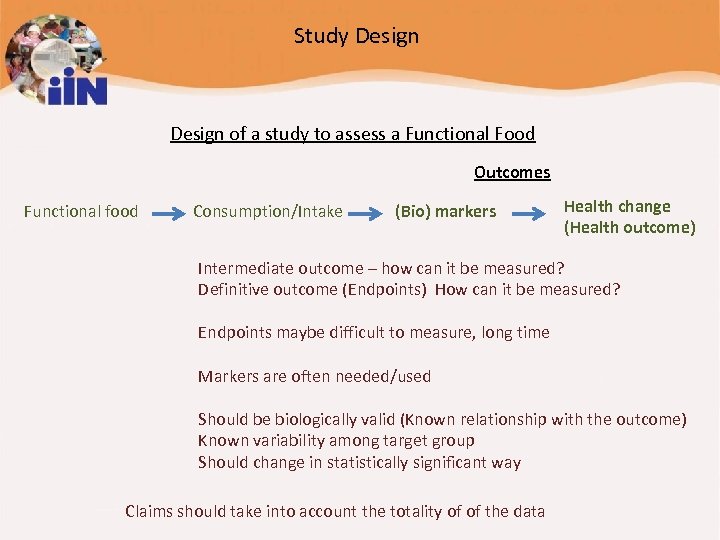 Study Design of a study to assess a Functional Food Outcomes Functional food Consumption/Intake (Bio) markers Health change (Health outcome) Intermediate outcome – how can it be measured? Definitive outcome (Endpoints) How can it be measured? Endpoints maybe difficult to measure, long time Markers are often needed/used Should be biologically valid (Known relationship with the outcome) Known variability among target group Should change in statistically significant way Claims should take into account the totality of of the data
Study Design of a study to assess a Functional Food Outcomes Functional food Consumption/Intake (Bio) markers Health change (Health outcome) Intermediate outcome – how can it be measured? Definitive outcome (Endpoints) How can it be measured? Endpoints maybe difficult to measure, long time Markers are often needed/used Should be biologically valid (Known relationship with the outcome) Known variability among target group Should change in statistically significant way Claims should take into account the totality of of the data
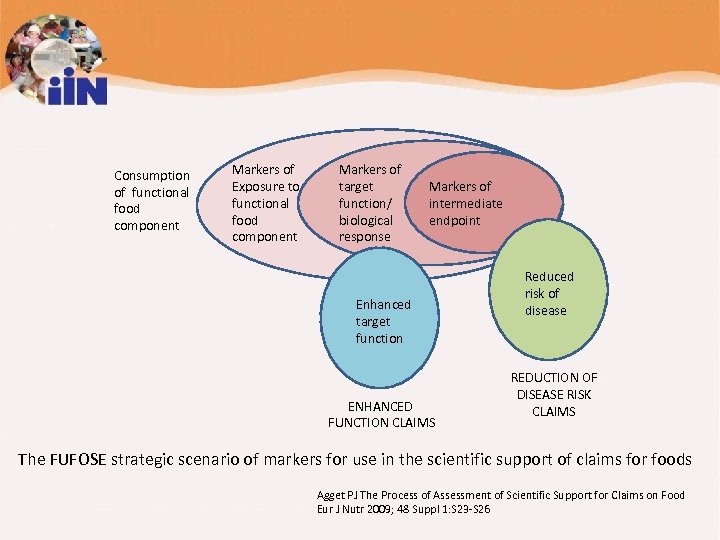 Consumption of functional food component Markers of Exposure to functional food component Markers of target function/ biological response Markers of intermediate endpoint Enhanced target function ENHANCED FUNCTION CLAIMS Reduced risk of disease REDUCTION OF DISEASE RISK CLAIMS The FUFOSE strategic scenario of markers for use in the scientific support of claims for foods Agget PJ The Process of Assessment of Scientific Support for Claims on Food Eur J Nutr 2009; 48 Suppl 1: S 23 -S 26
Consumption of functional food component Markers of Exposure to functional food component Markers of target function/ biological response Markers of intermediate endpoint Enhanced target function ENHANCED FUNCTION CLAIMS Reduced risk of disease REDUCTION OF DISEASE RISK CLAIMS The FUFOSE strategic scenario of markers for use in the scientific support of claims for foods Agget PJ The Process of Assessment of Scientific Support for Claims on Food Eur J Nutr 2009; 48 Suppl 1: S 23 -S 26
 Markers Intermediate measurements: Markers Biological function: Bacterial populations in the gut (probiotics) Key stage in disease development: Bone density Flow mediated dilatation for CVD Antioxidant measure Glycemic index Hemoglobin Satiety-consumption Muscle strength Lipid profile Nutrigenomics: Changes in gene expression as a result of dietary nutrients
Markers Intermediate measurements: Markers Biological function: Bacterial populations in the gut (probiotics) Key stage in disease development: Bone density Flow mediated dilatation for CVD Antioxidant measure Glycemic index Hemoglobin Satiety-consumption Muscle strength Lipid profile Nutrigenomics: Changes in gene expression as a result of dietary nutrients
 Adverse events Adverse Event reporting Other names: Co-effects, Co-events, side effects Events will always occur, complaints cannot be avoided But important to record, for safety monitoring, ethics, and may be expected for publication Registration of trials: Pros and Cons Allows meta-analysis and prevents publication bias – Patenting issues
Adverse events Adverse Event reporting Other names: Co-effects, Co-events, side effects Events will always occur, complaints cannot be avoided But important to record, for safety monitoring, ethics, and may be expected for publication Registration of trials: Pros and Cons Allows meta-analysis and prevents publication bias – Patenting issues
 Types of studies Animal studies provide information on mechanisms of action, biomarkers, potential benefits Longitudinal epidemiological observation studies in humans including detailed dietary studies provide initial evidence eg Fruits and vegetables – but is it enough? But trails will be needed for health claims v Single arm studies are not acceptable Randomized control studies (Gold standard) v Cross over and parrallel designs v Longitudinal RCTs - depends on outcome (prevention of anemia, increased sports performance relatively quick, prevention of dementia long time) Discussion about Pharmacological approach to Food trials - but evidence is needed!
Types of studies Animal studies provide information on mechanisms of action, biomarkers, potential benefits Longitudinal epidemiological observation studies in humans including detailed dietary studies provide initial evidence eg Fruits and vegetables – but is it enough? But trails will be needed for health claims v Single arm studies are not acceptable Randomized control studies (Gold standard) v Cross over and parrallel designs v Longitudinal RCTs - depends on outcome (prevention of anemia, increased sports performance relatively quick, prevention of dementia long time) Discussion about Pharmacological approach to Food trials - but evidence is needed!
 Type of Studies Randomized controlled Trials v Population characteristics, numbers/sample size v Consent process v Inclusion and exclusion criteria/screening v Randomization v Delivery of food, direct observation, packaging, amounts, measurement; surveillance v Adverse effects surveillance v Loss to follow-up v Outcomes: Measurement of outcomes, duration
Type of Studies Randomized controlled Trials v Population characteristics, numbers/sample size v Consent process v Inclusion and exclusion criteria/screening v Randomization v Delivery of food, direct observation, packaging, amounts, measurement; surveillance v Adverse effects surveillance v Loss to follow-up v Outcomes: Measurement of outcomes, duration
 Other considerations Discussion about “Qualified health claims: “may be beneficial” Differences between countries USA, Japan, EU Political/regulatory aspects in the country What are the local regulations about food additives? , Human studies and clinical trial regulations? Dietary Reference Intakes needed for a wider range of nutrients
Other considerations Discussion about “Qualified health claims: “may be beneficial” Differences between countries USA, Japan, EU Political/regulatory aspects in the country What are the local regulations about food additives? , Human studies and clinical trial regulations? Dietary Reference Intakes needed for a wider range of nutrients
 Mind-Map Of Study Design Hendriks H and Welch R. Beyond PASSCLAIM. 2010 – ILSI Europe report series
Mind-Map Of Study Design Hendriks H and Welch R. Beyond PASSCLAIM. 2010 – ILSI Europe report series
 Summary Mind-map - Preparations
Summary Mind-map - Preparations
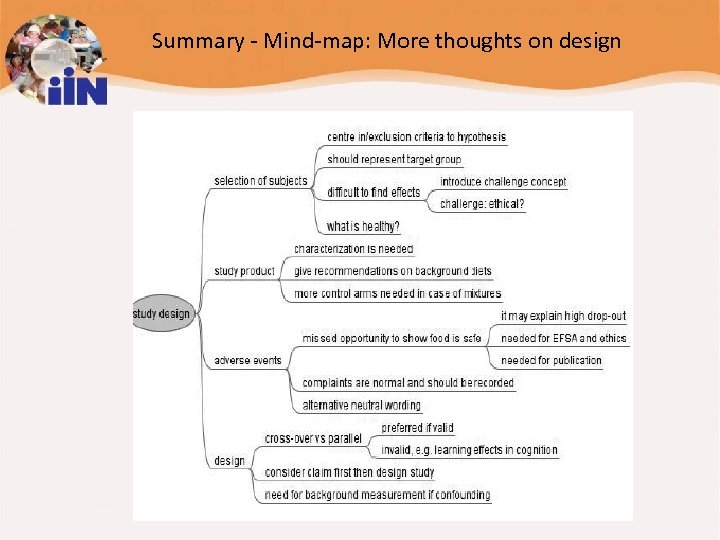 Summary - Mind-map: More thoughts on design
Summary - Mind-map: More thoughts on design
 Gracias
Gracias


