b0cadf26b286734568e6978f73bab38a.ppt
- Количество слайдов: 25

FOR DISTRIBUTION BY MEDTRONIC OFFICE OF MEDICAL AFFAIRS ONLY. The information in this document is provided by Medtronic in response to an unsolicited request. It contains information on unapproved uses that have not been approved by the FDA. Medtronic provided financial support related to the information included in this document. Insurers may or may not cover all uses described, it is advisable to confirm coverage with carriers before filing claims. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12

Resolute in Bifurcation Lesions: Data from the RESOLUTE Clinical Program Prof. Ran Kornowski, MD, FESC, FACC Director – Division of Interventional Cardiology Rabin Medical Center and Tel Aviv University, Israel On behalf of J. Belardi, M. Ferenc, P. W. Serruys, S. Silber , P. Widimský, S. Windecker and F-J. Neumann Euro. PCR 2012 For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12

RESOLUTE Pooled – Bifurcation Subset Background • Bifurcation lesions remain a therapeutic challenge and a focus of great interest among interventional cardiologists. • The impact of the Resolute DES platform, with its open-cell design and unique Bio. Linx polymer and Zotarolimus anti-proliferative drug, on the technical and clinical outcomes in bifurcation lesions has been studied in RESOLUTE All Comers and RESOLUTE International and followed up to 2 years. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
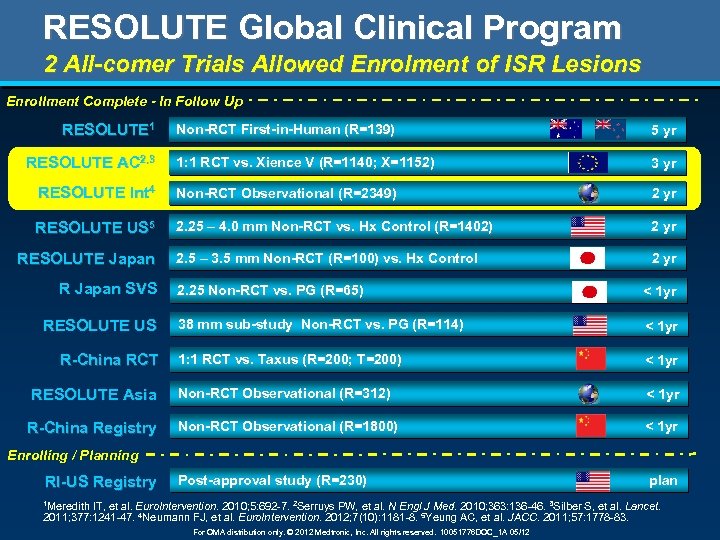
RESOLUTE Global Clinical Program 2 All-comer Trials Allowed Enrolment of ISR Lesions Enrollment Complete - In Follow Up RESOLUTE 1 Non-RCT First-in-Human (R=139) 5 yr 1: 1 RCT vs. Xience V (R=1140; X=1152) 3 yr RESOLUTE Int 4 Non-RCT Observational (R=2349) 2 yr RESOLUTE US 5 2. 25 – 4. 0 mm Non-RCT vs. Hx Control (R=1402) 2 yr 2. 5 – 3. 5 mm Non-RCT (R=100) vs. Hx Control 2 yr RESOLUTE AC 2, 3 RESOLUTE Japan R Japan SVS 2. 25 Non-RCT vs. PG (R=65) < 1 yr 38 mm sub-study Non-RCT vs. PG (R=114) < 1 yr 1: 1 RCT vs. Taxus (R=200; T=200) < 1 yr RESOLUTE Asia Non-RCT Observational (R=312) < 1 yr R-China Registry Non-RCT Observational (R=1800) < 1 yr RESOLUTE US R-China RCT Enrolling / Planning RI-US Registry Post-approval study (R=230) 1 Meredith plan IT, et al. Euro. Intervention. 2010; 5: 692 -7. 2 Serruys PW, et al. N Engl J Med. 2010; 363: 136 -46. 3 Silber S, et al. Lancet. 2011; 377: 1241 -47. 4 Neumann FJ, et al. Euro. Intervention. 2012; 7(10): 1181 -8. 5 Yeung AC, et al. JACC. 2011; 57: 1778 -83. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
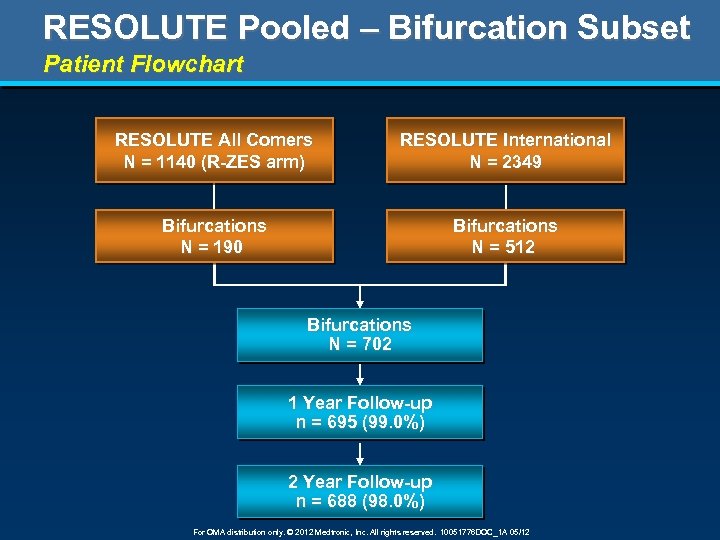
RESOLUTE Pooled – Bifurcation Subset Patient Flowchart RESOLUTE All Comers N = 1140 (R-ZES arm) RESOLUTE International N = 2349 Bifurcations N = 190 Bifurcations N = 512 Bifurcations N = 702 1 Year Follow-up n = 695 (99. 0%) 2 Year Follow-up n = 688 (98. 0%) For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
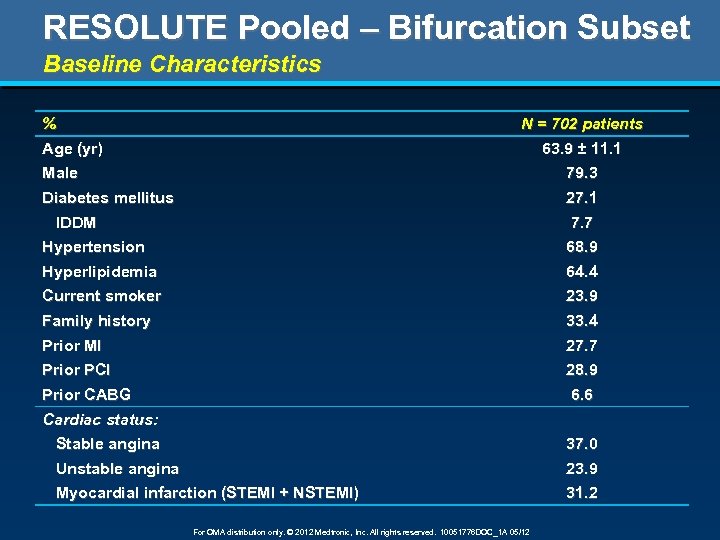
RESOLUTE Pooled – Bifurcation Subset Baseline Characteristics % Age (yr) N = 702 patients 63. 9 ± 11. 1 Male Diabetes mellitus 79. 3 27. 1 IDDM Hypertension 7. 7 68. 9 Hyperlipidemia Current smoker 64. 4 23. 9 Family history Prior MI 33. 4 27. 7 Prior PCI Prior CABG 28. 9 6. 6 Cardiac status: Stable angina Unstable angina Myocardial infarction (STEMI + NSTEMI) For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12 37. 0 23. 9 31. 2
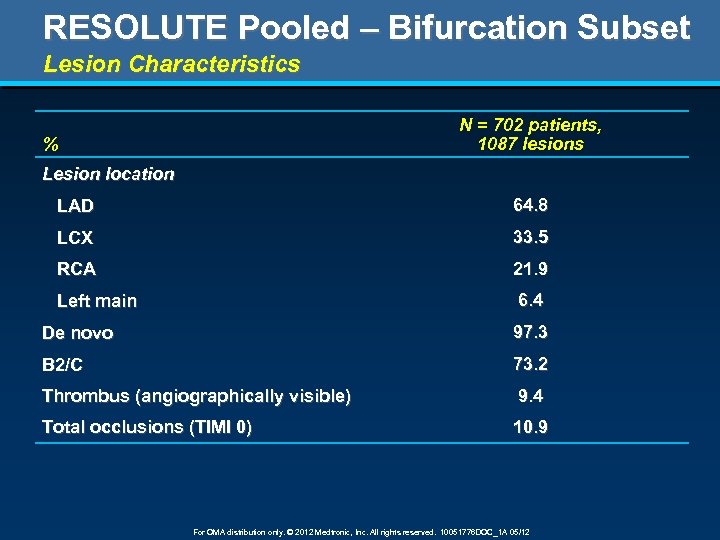
RESOLUTE Pooled – Bifurcation Subset Lesion Characteristics N = 702 patients, 1087 lesions % Lesion location LAD 64. 8 LCX 33. 5 RCA 21. 9 Left main 6. 4 De novo 97. 3 B 2/C 73. 2 Thrombus (angiographically visible) 9. 4 Total occlusions (TIMI 0) 10. 9 For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
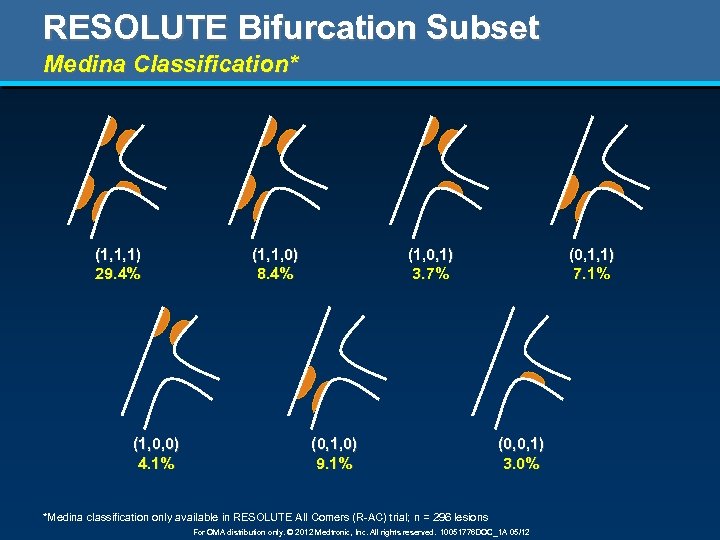
RESOLUTE Bifurcation Subset Medina Classification* (1, 1, 1) 29. 4% (1, 0, 0) 4. 1% (1, 1, 0) 8. 4% (1, 0, 1) 3. 7% (0, 1, 0) 9. 1% (0, 1, 1) 7. 1% (0, 0, 1) 3. 0% *Medina classification only available in RESOLUTE All Comers (R-AC) trial; n = 296 lesions For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
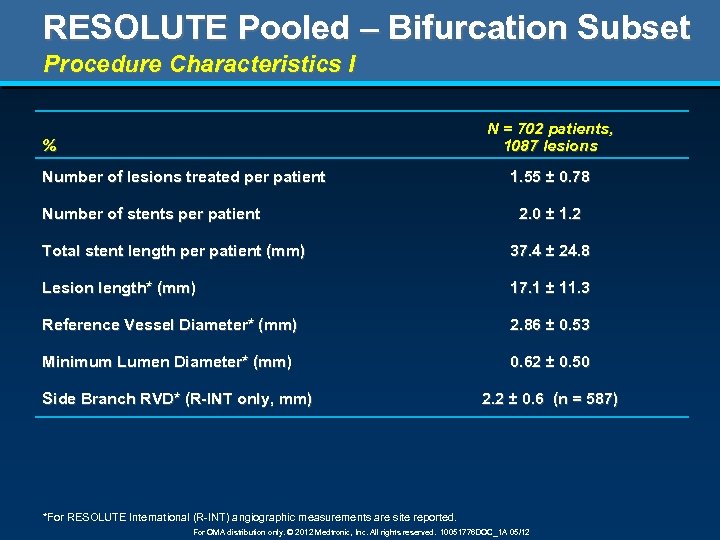
RESOLUTE Pooled – Bifurcation Subset Procedure Characteristics I N = 702 patients, 1087 lesions % Number of lesions treated per patient Number of stents per patient 1. 55 ± 0. 78 2. 0 ± 1. 2 Total stent length per patient (mm) 37. 4 ± 24. 8 Lesion length* (mm) 17. 1 ± 11. 3 Reference Vessel Diameter* (mm) 2. 86 ± 0. 53 Minimum Lumen Diameter* (mm) 0. 62 ± 0. 50 Side Branch RVD* (R-INT only, mm) 2. 2 ± 0. 6 (n = 587) *For RESOLUTE International (R-INT) angiographic measurements are site reported. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
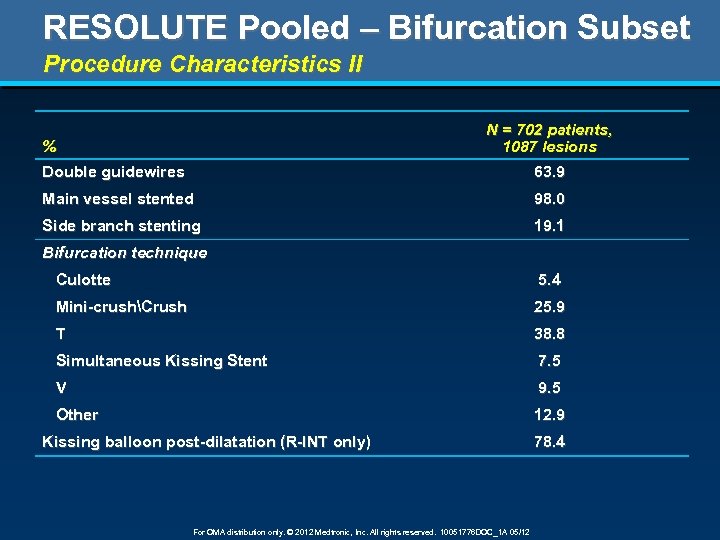
RESOLUTE Pooled – Bifurcation Subset Procedure Characteristics II N = 702 patients, 1087 lesions % Double guidewires 63. 9 Main vessel stented 98. 0 Side branch stenting 19. 1 Bifurcation technique Culotte 5. 4 Mini-crushCrush 25. 9 T 38. 8 Simultaneous Kissing Stent 7. 5 V 9. 5 Other 12. 9 Kissing balloon post-dilatation (R-INT only) For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12 78. 4

RESOLUTE Pooled – Bifurcation Subset Acute Success Rates N = 702 patients, 1087 lesions % Lesion success (the attainment of <50% residual stenosis of the target lesion using any percutaneous method) 99. 6% Device success (the attainment of <50% residual stenosis of the target lesion using only the assigned device) 94. 7% Procedure success (the attainment of <50% residual stenosis of the target lesion and no in-hospital MACE) 93. 9% For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
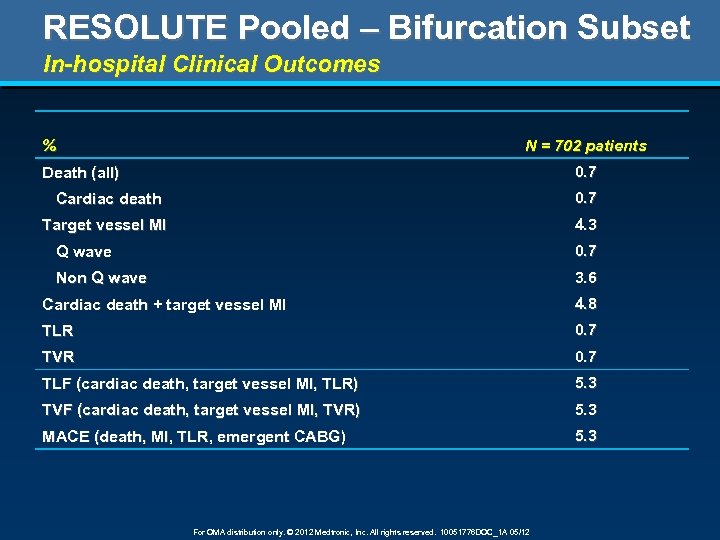
RESOLUTE Pooled – Bifurcation Subset In-hospital Clinical Outcomes % N = 702 patients 0. 7 Death (all) Cardiac death 0. 7 Target vessel MI 4. 3 Q wave 0. 7 Non Q wave 3. 6 Cardiac death + target vessel MI 4. 8 TLR 0. 7 TVR 0. 7 TLF (cardiac death, target vessel MI, TLR) 5. 3 TVF (cardiac death, target vessel MI, TVR) 5. 3 MACE (death, MI, TLR, emergent CABG) 5. 3 For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
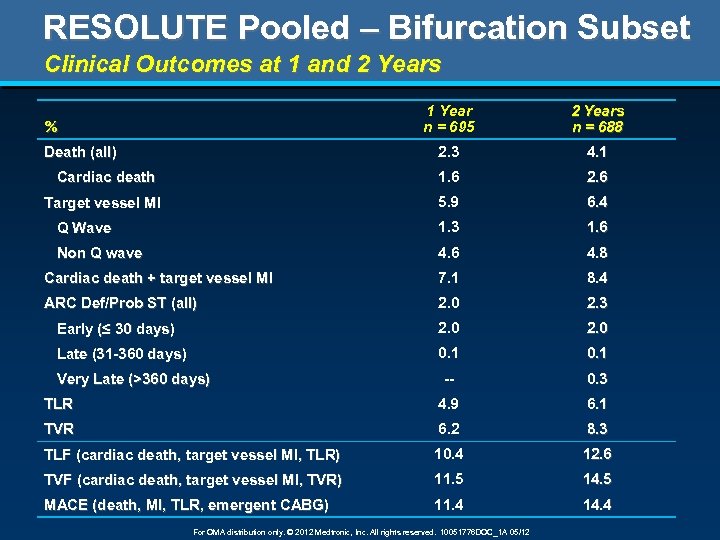
RESOLUTE Pooled – Bifurcation Subset Clinical Outcomes at 1 and 2 Years 1 Year n = 695 2 Years n = 688 2. 3 4. 1 Cardiac death 1. 6 2. 6 Target vessel MI 5. 9 6. 4 Q Wave 1. 3 1. 6 Non Q wave 4. 6 4. 8 Cardiac death + target vessel MI 7. 1 8. 4 ARC Def/Prob ST (all) 2. 0 2. 3 Early (≤ 30 days) 2. 0 Late (31 -360 days) 0. 1 -- 0. 3 TLR 4. 9 6. 1 TVR 6. 2 8. 3 TLF (cardiac death, target vessel MI, TLR) 10. 4 12. 6 TVF (cardiac death, target vessel MI, TVR) 11. 5 14. 5 MACE (death, MI, TLR, emergent CABG) 11. 4 14. 4 % Death (all) Very Late (>360 days) For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
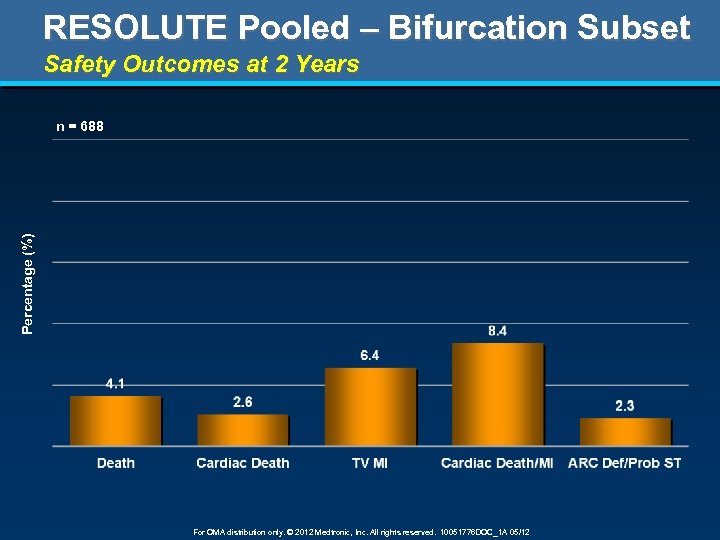
RESOLUTE Pooled – Bifurcation Subset Safety Outcomes at 2 Years Percentage (%) n = 688 For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
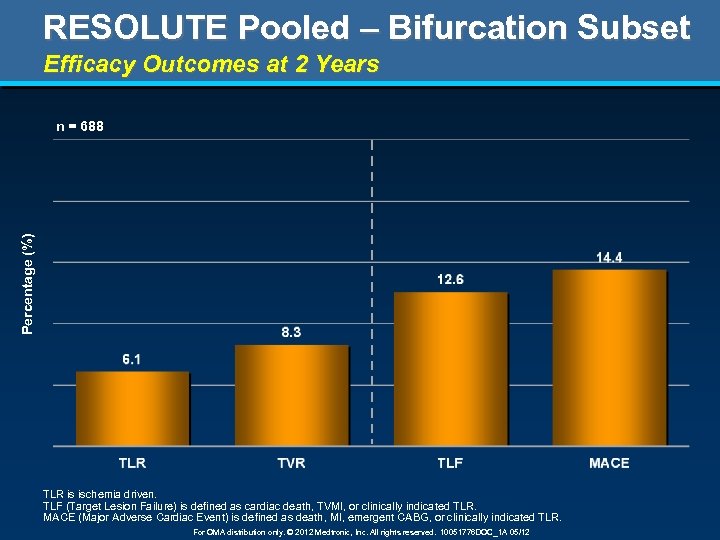
RESOLUTE Pooled – Bifurcation Subset Efficacy Outcomes at 2 Years Percentage (%) n = 688 TLR is ischemia driven. TLF (Target Lesion Failure) is defined as cardiac death, TVMI, or clinically indicated TLR. MACE (Major Adverse Cardiac Event) is defined as death, MI, emergent CABG, or clinically indicated TLR. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
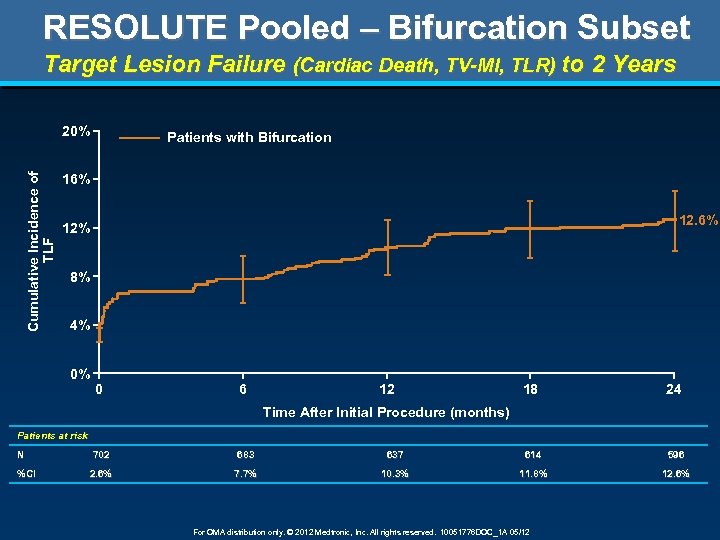
RESOLUTE Pooled – Bifurcation Subset Target Lesion Failure (Cardiac Death, TV-MI, TLR) to 2 Years Cumulative Incidence of TLF 20% Patients with Bifurcation 16% 12% 8% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk N %CI 702 683 637 614 596 2. 6% 7. 7% 10. 3% 11. 8% 12. 6% For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
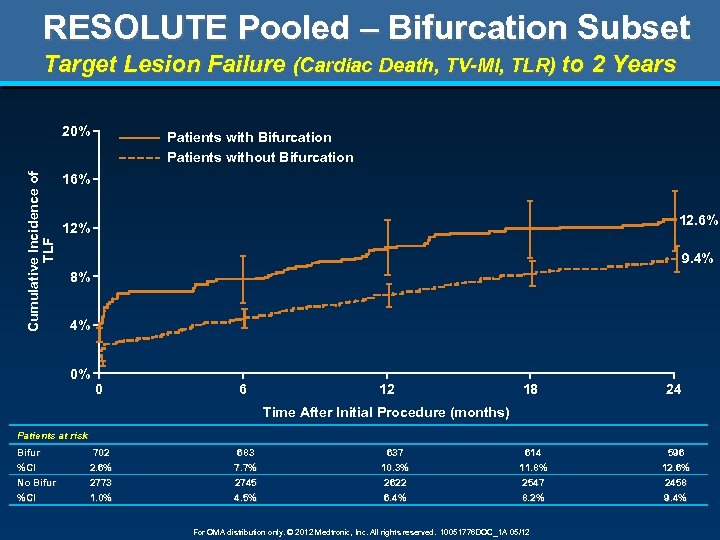
RESOLUTE Pooled – Bifurcation Subset Target Lesion Failure (Cardiac Death, TV-MI, TLR) to 2 Years Cumulative Incidence of TLF 20% Patients with Bifurcation Patients without Bifurcation 16% 12% 9. 4% 8% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk Bifur %CI 702 2. 6% 683 7. 7% 637 10. 3% 614 11. 8% 596 12. 6% No Bifur %CI 2773 1. 0% 2745 4. 5% 2622 6. 4% 2547 8. 2% 2458 9. 4% For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
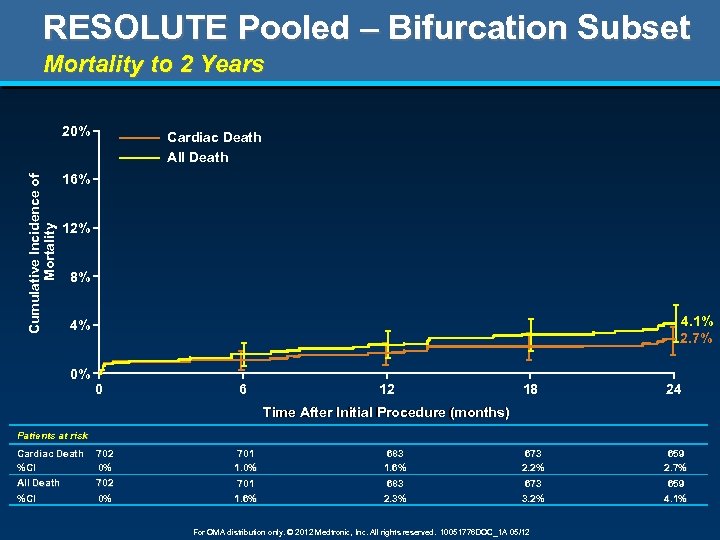
RESOLUTE Pooled – Bifurcation Subset Mortality to 2 Years Cumulative Incidence of Mortality 20% Cardiac Death All Death 16% 12% 8% 4. 1% 2. 7% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) after Patients at risk Cardiac Death %CI All Death 702 0% 702 %CI 0% 701 1. 0% 683 1. 6% 673 2. 2% 659 2. 7% 701 1. 6% 683 2. 3% 673 3. 2% 659 4. 1% For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
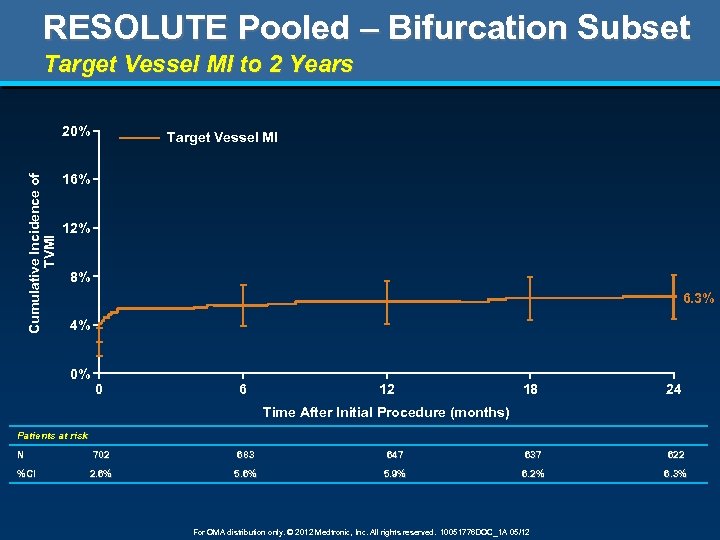
RESOLUTE Pooled – Bifurcation Subset Target Vessel MI to 2 Years Cumulative Incidence of TVMI 20% Target Vessel MI 16% 12% 8% 6. 3% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk N %CI 702 683 647 637 622 2. 6% 5. 9% 6. 2% 6. 3% For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
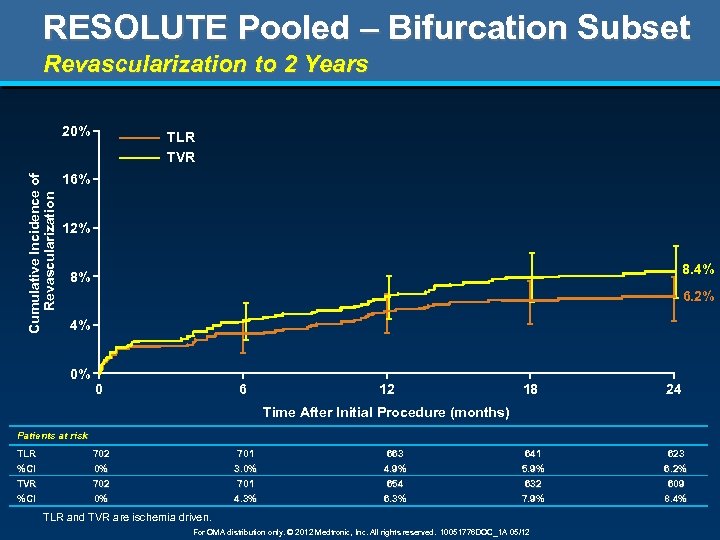
RESOLUTE Pooled – Bifurcation Subset Revascularization to 2 Years Cumulative Incidence of Revascularization 20% TLR TVR 16% 12% 8. 4% 8% 6. 2% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk TLR 702 701 663 641 623 %CI TVR %CI 0% 702 0% 3. 0% 701 4. 3% 4. 9% 654 6. 3% 5. 9% 632 7. 9% 6. 2% 609 8. 4% TLR and TVR are ischemia driven. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
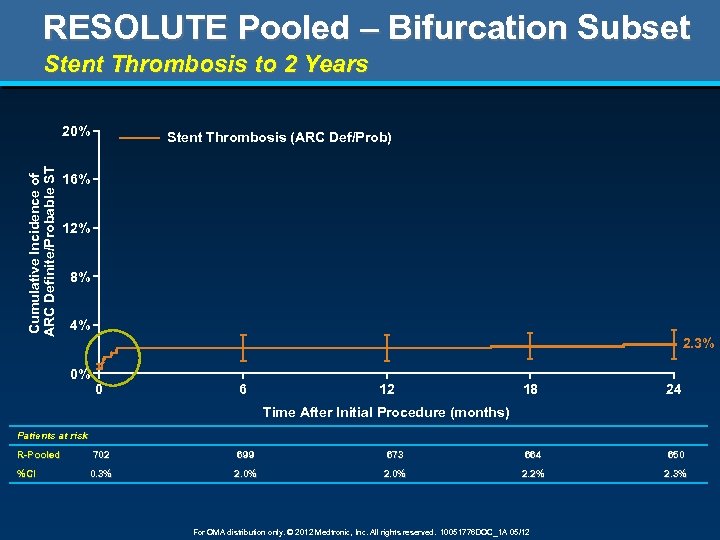
RESOLUTE Pooled – Bifurcation Subset Stent Thrombosis to 2 Years Cumulative Incidence of ARC Definite/Probable ST 20% Stent Thrombosis (ARC Def/Prob) 16% 12% 8% 4% 2. 3% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk R-Pooled %CI 702 699 673 664 650 0. 3% 2. 0% 2. 2% 2. 3% For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
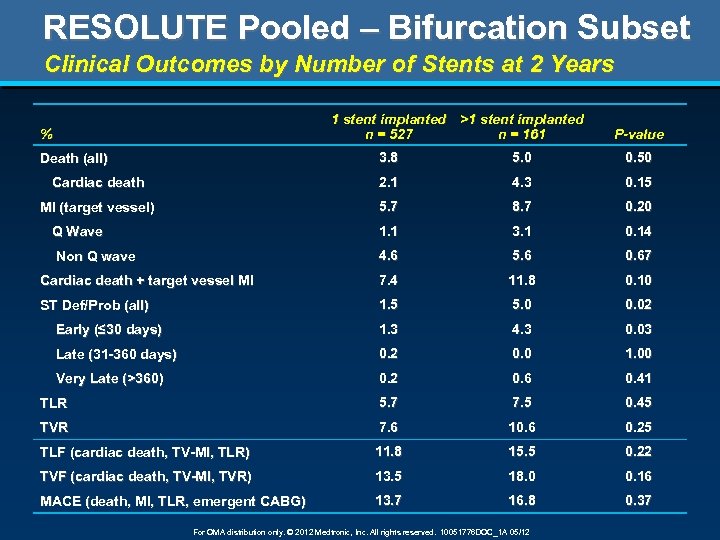
RESOLUTE Pooled – Bifurcation Subset Clinical Outcomes by Number of Stents at 2 Years 1 stent implanted >1 stent implanted n = 527 n = 161 % P-value 3. 8 5. 0 0. 50 Cardiac death 2. 1 4. 3 0. 15 MI (target vessel) 5. 7 8. 7 0. 20 Q Wave 1. 1 3. 1 0. 14 Non Q wave 4. 6 5. 6 0. 67 Cardiac death + target vessel MI 7. 4 11. 8 0. 10 ST Def/Prob (all) 1. 5 5. 0 0. 02 Early (≤ 30 days) 1. 3 4. 3 0. 03 Late (31 -360 days) 0. 2 0. 0 1. 00 Very Late (>360) 0. 2 0. 6 0. 41 TLR 5. 7 7. 5 0. 45 TVR 7. 6 10. 6 0. 25 TLF (cardiac death, TV-MI, TLR) 11. 8 15. 5 0. 22 TVF (cardiac death, TV-MI, TVR) 13. 5 18. 0 0. 16 MACE (death, MI, TLR, emergent CABG) 13. 7 16. 8 0. 37 Death (all) For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
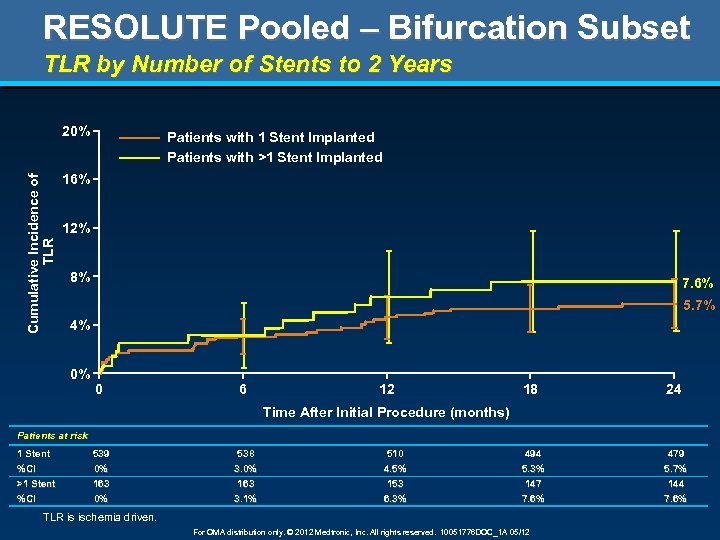
RESOLUTE Pooled – Bifurcation Subset TLR by Number of Stents to 2 Years Cumulative Incidence of TLR 20% Patients with 1 Stent Implanted Patients with >1 Stent Implanted 16% 12% 8% 7. 6% 5. 7% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk 1 Stent 539 538 510 494 479 %CI >1 Stent %CI 0% 163 0% 3. 0% 163 3. 1% 4. 5% 153 6. 3% 5. 3% 147 7. 6% 5. 7% 144 7. 6% TLR is ischemia driven. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
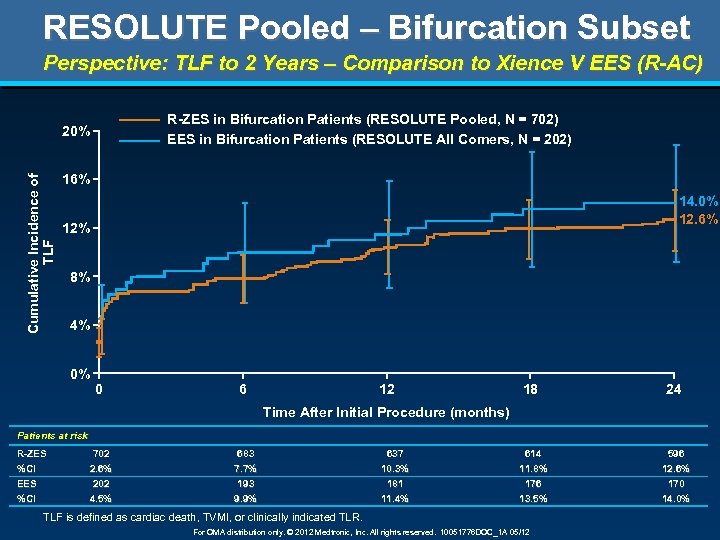
RESOLUTE Pooled – Bifurcation Subset Perspective: TLF to 2 Years – Comparison to Xience V EES (R-AC) R-ZES in Bifurcation Patients (RESOLUTE Pooled, N = 702) EES in Bifurcation Patients (RESOLUTE All Comers, N = 202) Cumulative Incidence of TLF 20% 16% 14. 0% 12. 6% 12% 8% 4% 0% 0 6 12 18 24 Time After Initial Procedure (months) Patients at risk R-ZES %CI EES %CI 702 683 637 614 596 2. 6% 202 4. 5% 7. 7% 193 9. 9% 10. 3% 181 11. 4% 11. 8% 176 13. 5% 12. 6% 170 14. 0% TLF is defined as cardiac death, TVMI, or clinically indicated TLR. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12

RESOLUTE Pooled – Bifurcation Subset Conclusion In the pooled dataset with Resolute bifurcation lesions we observed: • Excellent technical/angiographic success rate with 19. 1% need for SB stenting in the treated cohort. • Sustainable clinical efficacy out to two years, with very low rates of TLR (6. 1%) and a favorable TLF rate of 12. 6%. • Sustained safety with low stent thrombosis (2. 3%, mostly early) up to two years. • Thus, the Resolute DES platform is a viable treatment option for bifurcation lesion management. For OMA distribution only. © 2012 Medtronic, Inc. All rights reserved. 10051776 DOC_1 A 05/12
b0cadf26b286734568e6978f73bab38a.ppt