a3c8fc38db65f2029f313e0bbf7641a0.ppt
- Количество слайдов: 23

Food and Drug Administration Center for Biologics Evaluation and Research The Office of Cellular, Tissue, and Gene Therapies Web Seminar Series presents: The chemistry, manufacturing and controls (CMC) section of a gene therapy IND Andrew P. Byrnes, Ph. D. Chief, Gene Transfer and Immunogenicity Branch Division of Cellular and Gene Therapies

What are gene therapy products? • Gene therapy products: – Are administered as nucleic acids, viruses or genetically-engineered microorganisms, and – Mediate effects via: • Transcription or translation of transferred genetic material, or • Integration into the genome • How are gene therapy products used? – To modify cells directly in patients, or – To modify cells in vitro that are then administered to patients

Examples of gene therapy products 1. Plasmid expressing an enzyme 2. AAV expressing a coagulation factor 3. T cells modified with a retrovirus to express a novel receptor 4. Bacterium expressing a tumor antigen 5. Oncolytic adenovirus expressing a cytokine
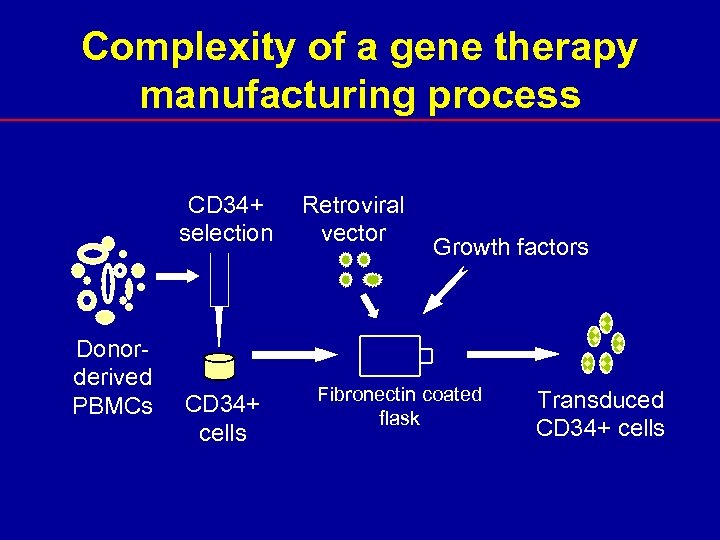
Complexity of a gene therapy manufacturing process CD 34+ selection Donorderived PBMCs CD 34+ cells Retroviral vector Growth factors Fibronectin coated flask Transduced CD 34+ cells

Before you begin manufacturing… 2008 Guidance for FDA Reviewers and Sponsors: Content and Review of Chemistry, Manufacturing, and Control (CMC) Information for Human Gene Therapy Investigational New Drug Applications (INDs)

Presentation outline • Components used in product manufacture • Final product testing and characterization • Good manufacturing practices (GMPs)

Components used to manufacture the product • Vector • Cells • Banking system – Master cell bank (MCB) – Master viral bank (MVB) • Reagents

Vector • Description, history and details on derivation of construct • Vector diagram • Sequence analysis (from MVB) – Full sequence for vectors <40 kb – Vectors >40 kb: sequence inserts, flanking regions, modified regions – Description of unexpected sequences • Raw sequence data is not sufficient

Cells • Cell substrate for production of vector – History, source, general characteristics • Cells used as cell therapies – Source • tissue and cell type – Collection procedure • mobilization, surgery, leukapheresis, devices used – Donor Eligibility • infectious disease screening & testing, 21 CFR 1271

MCB and MVB safety testing • Sterility • Mycoplasma • Adventitious Virus – In vitro and in vivo adventitious virus assays – Bovine and porcine viruses • Not needed if reagents tested – For human cell lines: • Typically EBV, HCV, CMV, HIV 1&2, HTLV 1 & 2, B 19 – For murine cell lines: • Typically mouse antibody production test, retroviruses – Replication-competent virus (for MVB)

Master cell bank characterization • Identity – Examples: • Isoenzyme • Karyotype • Short tandem repeat (STR) profiling • Viability • Stability

Reagents used in manufacturing • Tabulation of reagents – – Final concentration Vendor Source (human, bovine, etc. ) Grade • e. g. licensed product, clinical grade, reagent grade • May need to provide details on reagent manufacturing • Certificates of Analysis • Cross reference letters • Qualification program – Safety testing and quality assessment

Product Manufacturing • Vector production / purification – Describe all steps • e. g. cell growth, infection, harvest, purification, formulation, vialing, storage – Flowchart • Describe the formulation – – Buffer components Excipients Product concentration Storage

Final Product Testing • Goals: – Safety – Product characterization – Product lot consistency • List all of your: – Release tests – Test methods – Acceptance criteria (specifications)

Final product testing: safety • Sterility • Mycoplasma • Endotoxin • Adventitious Virus – In vitro virus – Replication competent virus

Final product testing: characterization • Final product lot release testing – Concentration – Purity • e. g. residual cellular DNA, empty viral particles – Identity • e. g. restriction digest – Activity • e. g. infectious titer – Potency • e. g. transgene-specific protein expression – Cell viability (if a cell-based product) • Stability – Storage – Shipping – Compatibility with delivery devices

Product characterization: why? • To demonstrate lot-to-lot consistency • To generate solid clinical data – For pivotal trials, characterization assays will need to be established with appropriate release limits • To show comparability after manufacturing changes

Current Good Manufacturing Practices (c. GMP) • Goals: – A product with defined and reproducible quality – Increased control of the manufacturing process as clinical trials advance • 2008 Guidance for Industry: CGMP for Phase 1 Investigational Drugs

c. GMPs Quality control • Quality (QC) Program – QC independent of production unit – Authority to accept or reject materials, lots, procedures and specifications – Prevent, detect, and correct deviations and failures

c. GMPs Questions for phase I • Is the manufacturing process reproducible? • Do you have appropriate testing at critical steps? • Is there adequate control of the quality of the raw and source materials? • Are the records and record keeping systems adequate?

c. GMPs Examples for early development • Procedures to prevent contamination & cross-contamination – Aseptic processing – Facility and equipment cleaning and changeover – Tracking and segregation of patient-specific products • Methods qualification – Appropriate method specificity, sensitivity, reproducibility – Lack of interference • Process qualification of safety related processes – Removal of potentially dangerous impurities

Summary • For a phase I submission, product safety is the focus of the CMC assessment – Freedom from microbes and adventitious agents – Safety-related characterization – Appropriate GMPs • Gene therapies and other complex biologics can be a challenge to characterize, and often require unique assays • Product control and process control should increase with clinical development

Further information CBER guidance documents: http: //www. fda. gov/Biologics. Blood. Vaccines/Guidance. Complian ce. Regulatory. Information/default. htm If the webinar series and referenced websites leave you with unanswered questions: CBEROCTGTRMS@fda. hhs. gov or 301 -827 -5102
a3c8fc38db65f2029f313e0bbf7641a0.ppt