35796551fa40041ca25279fea4d0d18d.ppt
- Количество слайдов: 42

Focused: LAB International TSX: LAB Frankfurt: LD 9. F XETRA: LD 9. DE March 2007

Safe-Harbor Statement Except for historical information, the statements made in this presentation are forward-looking statements involving significant risks and uncertainties. These risks and uncertainties, including those related to the timing or successful completion of the Company’s product commercialization activities and dependence on collaborators, are detailed in the Company’s regulatory filings.

LAB INTERNATIONAL Platform Based Integrated Product Development Organisation for Pain and CNS

Evolution…. ▪ 1998 3 rd incarnation as pre-clinical CRO ▪ 2002 IPO to start product development ▪ 2003 Inhalation becomes primary platform ▪ 2004 European formulation and manufacturing ▪ 2006 CRO sale to focus on products ▪ 2006 US formulation and manufacturing ▪ 2006 Broadening of platforms

… and Strategy ▪ Increase focus on pain and CNS as core ▪ Accelerate product development (3 new / year) ▪ Maximize use of 3 proprietary platforms ▪ Complete manufacturing ▪ Establish R&D Group (Low cost environment) ▪ US license of lead product ▪ List on U. S. Stock Exchange
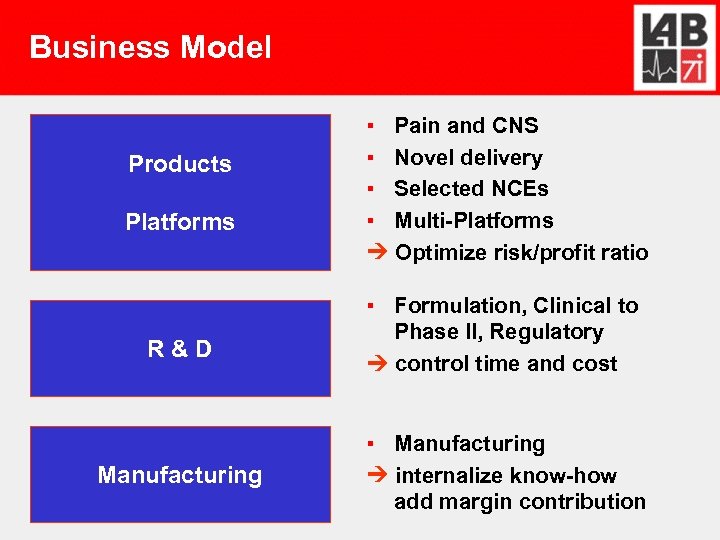
Business Model Products Platforms R&D Manufacturing ▪ Pain and CNS ▪ Novel delivery ▪ Selected NCEs ▪ Multi-Platforms Optimize risk/profit ratio ▪ Formulation, Clinical to Phase II, Regulatory control time and cost ▪ Manufacturing internalize know-how add margin contribution

LAB International Sites

Austin, TX H. O. , Formulation and Manufacturing

Highlights ▪ ▪ Founded 1996 as Pharma. Form 40, 000 sq. ft. / 90 employees IP-related revenues 10 MM/2 MM EBITDA Proprietary platforms - Oral (EDACS) - Transmucosal (Pharma. Film) ▪ Manufacturing processes - Hotmelt extrusion Granulation, Blending, Compression Coating, Encapsulation Bottling, Blistering ▪ FDA inspected (last 2005, no 483) ▪ DEA registered - Manufacturer, Researcher, Exporter

Turku/Finland Dry Powder Formulation

Highlights ▪ ▪ ▪ Founded 2000 as Focus Oy Schering spin-off with 20 years inhalation expertise 26 MM VC funded 35, 000 sq. ft. (130, 000 sq. ft. ) / 40 employees Processes - Device design - Dry powder formulation - Class C clean room facility ▪ Proprietary platforms - Taifun - LURUX Wet Suspension

Products and Platforms I: Inhalation

Unique Market – Limited Competition Established: Pulmonary treatment ▪ $ 15 B growing New: Systemic delivery via inhalation ▪ Peptides and Proteins (Exubera) ▪ Fast delivery for pain & emergency drugs
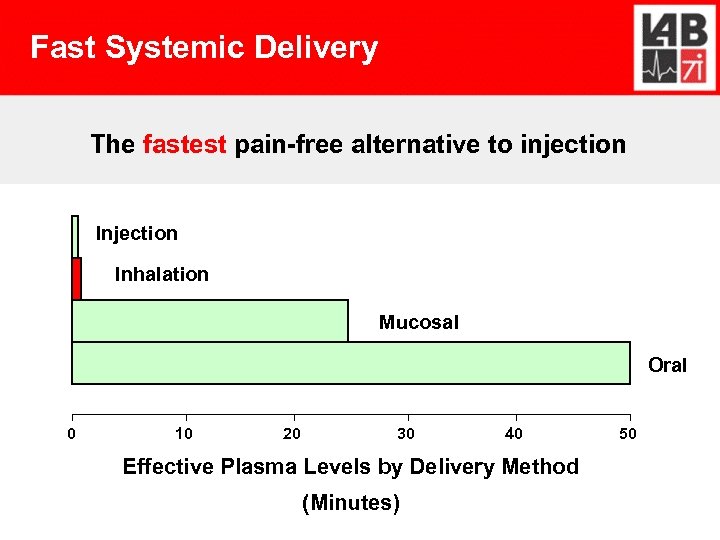
Fast Systemic Delivery The fastest pain-free alternative to injection Inhalation Mucosal Oral 0 10 20 30 40 Effective Plasma Levels by Delivery Method (Minutes) 50

Fentanyl
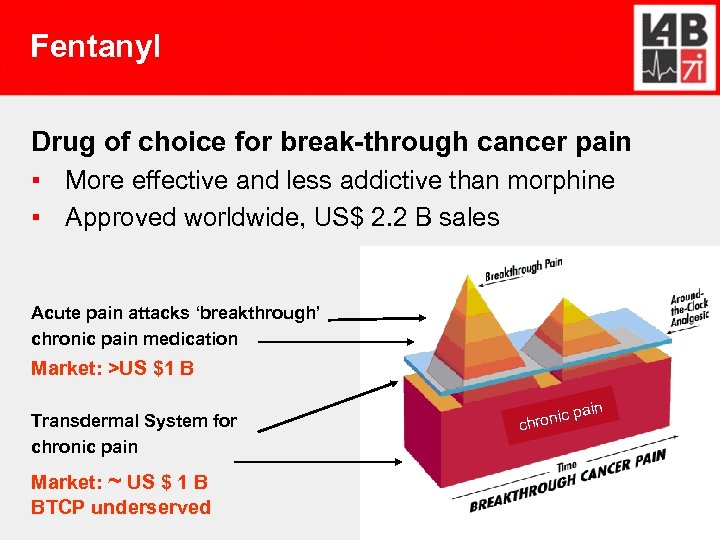
Fentanyl Drug of choice for break-through cancer pain ▪ More effective and less addictive than morphine ▪ Approved worldwide, US$ 2. 2 B sales Acute pain attacks ‘breakthrough’ chronic pain medication Market: >US $1 B Transdermal System for chronic pain Market: ~ US $ 1 B BTCP underserved ic chron pain
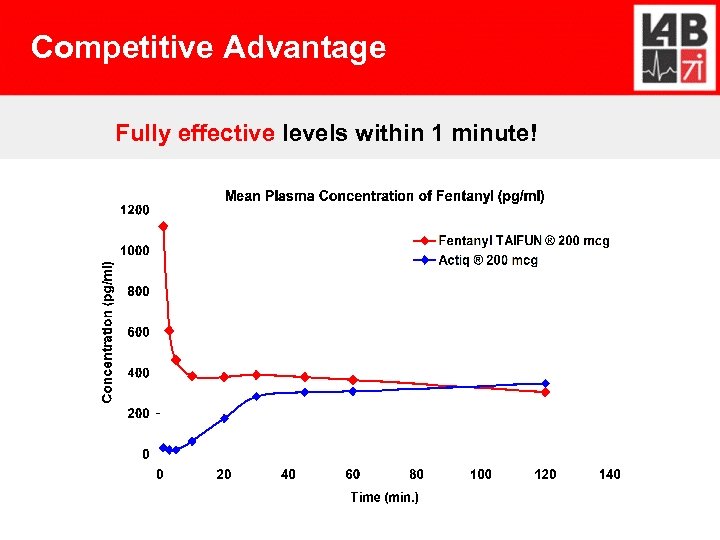
Competitive Advantage Fully effective levels within 1 minute!

Competitive Advantage Fentanyl TAIFUN®: Tmax: 1 min. Actiq®: Tmax: 20 + 35 -45 Minutes Fentora®: Tmax: 35 -45 Minutes Result Fentanyl TAIFUN®: Highest BA, lowest dose

Phase IIa - Main conclusions ▪ ▪ ▪ ▪ Clinical efficacy versus Placebo demonstrated Effect statistically significant after 8 -9 minutes Frequent effect already in 5 min 100 µg already efficacious Good starting dose for titration Dose response demonstrated to lesser extent Safety not different from Placebo

Phase IIb § Open-label results: - 100% successful titration (first 24 patients) to a 400µg dose or less (9 patients with 100µg, 10 with 200µg and 5 only with 400µg) - Significant pain relief in 95% of the pain episodes treated - Same quick onset of efficacy – Median of 7 minutes - All doses well tolerated - New patent application submitted § Final double-blind, placebo controlled extension arm results available in Q 2/07 (Phase III: Q 4/07 -Q 1/09)

CGRP

CGRP: Ideal Anti-Asthmatic Calcitonin Gene Related Peptide ▪ Natural peptide secreted in the lung post asthma attack ▪ Hybrid Drug - Bronchodilatory & Anti-inflammatory - Counteracts airway hyper-reactivity - Blocks acute & late-phase bronchial response ▪ US $15 B Market (Asthma/COPD)

CGRP Clinical Program Phase I: Completed ▪ Safe and well tolerated ▪ No serious adverse events reported Phase IIa - bronchodilative effect : Completed ▪ Statistically significant broncho-protective effects ▪ Similar safety profile to placebo Phase IIb for anti-inflammatory effect ▪ Dose optimization and anti-inflammatory properties

GHRH

GHRH Analogue Rationale ▪ GHRH improves therapy over GH ▪ 8000 fold higher affinity to receptor ▪ US $ 1 B market (350, 000 + patients U. S. only) Lead Indication ▪ Wasting in pre-dialytic Chronic Renal Failure (CRF)

GHRH Phase I/II Findings ▪ Immediate and substantial GH secretion post dosing ▪ 25 -40 fold increase over baseline/placebo at all dose levels (AUC 0 -4 and 0 -12) ▪ All levels safe; no change of circadian rhythm Conclusions ▪ Highly potent ▪ Likely clear improvement over other GHRH products

GHRH Pilot Phase II Effects of GHRH on malnutrition in pre-dialytic CRF ▪ Thesis: Reversal of weight loss improves morbidity ▪ Design: Placebo-controlled, 32 pre-dialytic patients ▪ Duration: 28 days ▪ Primary endpoints: GH, IGF-1, Protein turnover ▪ Timing: Enrollment completed - Results in Q 2/07

Inhalative Platform

TAIFUN – Approved ® Best in class ▪ Small, portable and stylish ▪ Easy to operate (breath actuated) ▪ Superior flow-rate ▪ Superior performance ▪ Regulatory approval in Europe ▪ Proprietary formulation technology

TAIFUN Patents ® Dose metering system ▪ High dose uniformity meets FDA standards Vortex chamber ▪ High lung deposition, independent of flow rate Integrated desiccant system ▪ Reliable performance in extreme humidity “Wet suspension” method ▪ Reliable dose-metering ▪ Homogeneous powder for high chemical and aerodynamic stability
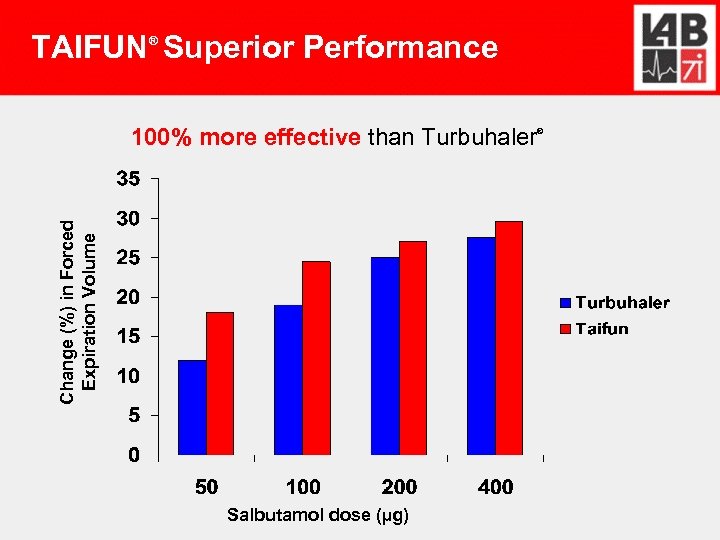
TAIFUN Superior Performance ® Change (%) in Forced Expiration Volume 100% more effective than Turbuhaler Salbutamol dose (µg) ®

TAIFUN Best In Class ® LAB International 2003 industry review: TAIFUN still # 1 Stephen P. Newman: ®

Products and Platforms II: EDACS

EDACS® ▪ Solid oral dosage form to deter abuse of controlled substances - NO crushing and immediate release - NO alcohol induced dose-dumping - NO dissolving and injection ▪ Sustained release opiate formulations ▪ Special advantages for US market ▪ Wide range of release profiles

EDACS Product Portfolio ▪ 3 Products under development to be disclosed in clinical stage (Q 1/08)

Products and Platforms III: Pharma. Film
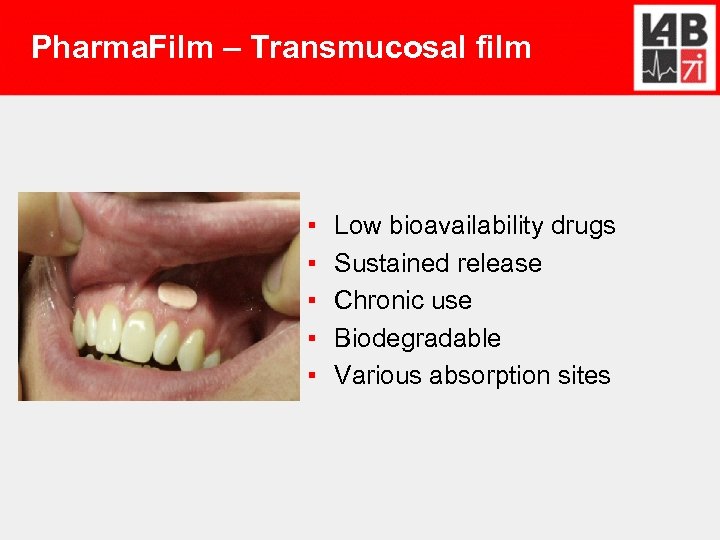
Pharma. Film – Transmucosal film ▪ ▪ ▪ Low bioavailability drugs Sustained release Chronic use Biodegradable Various absorption sites

Financials & Personnel
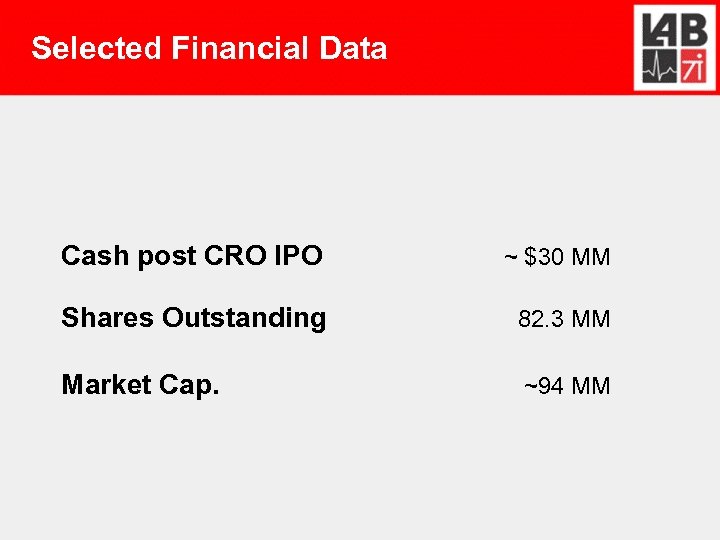
Selected Financial Data Cash post CRO IPO ~ $30 MM Shares Outstanding 82. 3 MM Market Cap. ~94 MM

Experienced Management ▪ ▪ ▪ ▪ Dr. H. Jaeger, CEO, 25+ A. Reiter, CFO, 15+ S. Lermer, President Pharma. Form, 25+ Dr Josef Bossart, SVP, Business Development, 20+ Dr. Roman Denk, VP Corporate Support, 15+ Dr. A. Jekunen, Senior VP, Clinical and Reg. , 15+ Dr. T. Jouhikainen, VP Corp. Development, 10+ Dr. John Koleng, COO, Pharma. Form

LAB International ▪ PDO based on novel platforms ▪ Focused on pain and CNS ▪ High profit potential, low developmental risk ▪ 2007 four products in Phase II, 1 -2 in Phase III ▪ Broad platform coverage ▪ Extensive IP protection ▪ LAB International: From CRO to Specialty Pharma

Focused: LAB International TSX: LAB
35796551fa40041ca25279fea4d0d18d.ppt