d90b0e7ef37f44c071ad22f5dad87eb8.ppt
- Количество слайдов: 44

Flagship Biosciences LLC Validation of digital pathology applications in regulated study environments

Digital Pathology in the News CAP 2010 ‘Digital pathology continues to generate industry buzz…. ’ ‘there are over a dozen FDA 510(k) clearances for digital analysis of immunohistochemistry procedures, the waiting game continues for how the agency wants to regulate digitalization of hematoxylin and eosin (H&E) slides using whole slide imaging (WSI) systems’ ‘once these regulatory barriers are negotiated, digital pathology will move ahead at breakneck speed’ www. flagshipbio. com

New Technologies for Health Care • Star Trek technologies – VISOR – Hypospray – Tricorder • The holy grail of medicine • Digital Radiology • Digital Pathology Are new technologies outpacing regulatory guidance? Who are the guiding decision-makers? www. flagshipbio. com

Regulatory Needs in Digital Pathology? • Use of whole slide images in an electronic environment – from acquisition to storage • Systems qualifications (IQ/OQ/PQ validation) • Quantitative image analysis on whole slide and TMA images • Accessioning, viewing, scoring by pathologists, and adjudication • Peer reviews and digital archiving www. flagshipbio. com

Regulatory & Compliance Digital Pathology in Drug Development Novel regulatory problems? Discovery www. flagshipbio. com Clinical Preclinical

Regulatory Guidance • Regulatory requirements for digital pathology present a complex series of processes in the drug development process – – Digital images Storage Annotations Image analysis • www. hhs. gov or www. fda. gov • CFR - Code of Federal Regulations Title 21 (Food and Drugs) – – PART 11 Electronic Records; Electronic Signatures PART 58 Good Laboratory Practice for Nonclinical laboratory Studies 501(K) Premarket Notification In Vitro Diagnostic Multivariate Index Assays (21 CFR 809. 3) • CLIA - Clinical Laboratory Improvement Amendments www. flagshipbio. com

Digital Pathology and IA Discovery – IHC investigations in potential new target organs • Researchers seeking to validate hypothesis • Verification and replication of literature claims • Tissues from commercial tissue banks have unknown demographics, outcomes, unknown pre-analytical variables, etc – Xenograft modeling • In vivo pathobiology studies • Early efficacy studies www. flagshipbio. com

Digital Pathology and IA Preclinical • Toxicology studies – Safety – Efficacy • Pharmacokinetic • Special studies • Peer review – Veterinary toxicological pathologists • North America, Japan, Europe (England, Germany, France, Switzerland) • Few overseas - especially in emerging biotech areas such as India and China • VIPER www. flagshipbio. com

Digital Pathology and IA Clinical • Clinical trials – Inclusion criteria – Retrospective analysis • Companion DX – Selection of biomarkers – Kit development – Pathology scoring • Treatment regimens for personalized medicine – HER 2, ER, PR – breast cancer – EGFR – lung cancer (NSCLC) • Multiplexing multiple biomarkers (IHC-based) www. flagshipbio. com

Multiplexing Multiple Markers on One Slide is Difficult Quantum Dots …ready for the clinic…next year • Tough problem Dual-stained IHC slides • Great research tool, double-staining is generally not high quality enough to run in diagnostic settings • Problems with cross-reactivity between chromogens, avoid DAB • US Labs Tri. View for prostate and breast – for color aid for pathologist, not quantitation – Breast: CK 5/6 (cytoplasmic brown) and p 63 (nuclear/brown) stain myoepithelial cells, while CK 8/18 labels the cytoplasm (cytoplasmic/red) of ductal or lobular epithelium. Dual or triple stained immunofluorescent (IF) slides • Expensive, no anatomical tissue context • IF not used extensively in the clinic www. flagshipbio. com
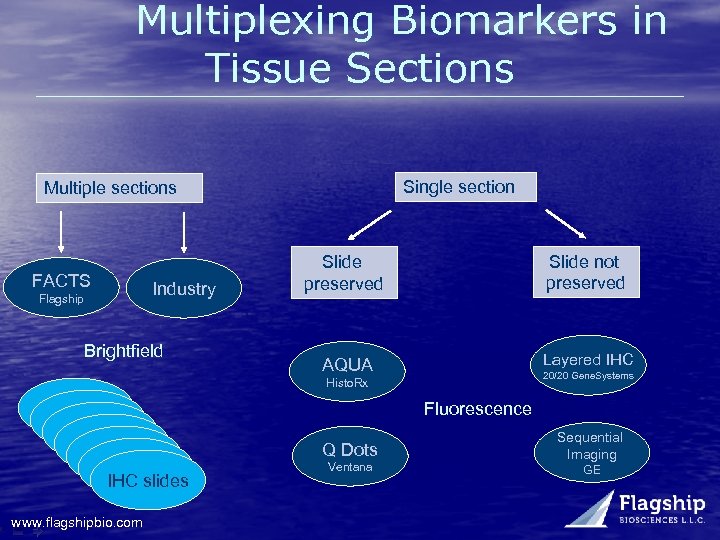
Multiplexing Biomarkers in Tissue Sections Single section Multiple sections FACTS Flagship Industry Brightfield Slide not preserved Slide preserved Layered IHC AQUA 20/20 Gene. Systems Histo. Rx IHC slide IHC slides www. flagshipbio. com Fluorescence Q Dots Ventana Sequential Imaging GE
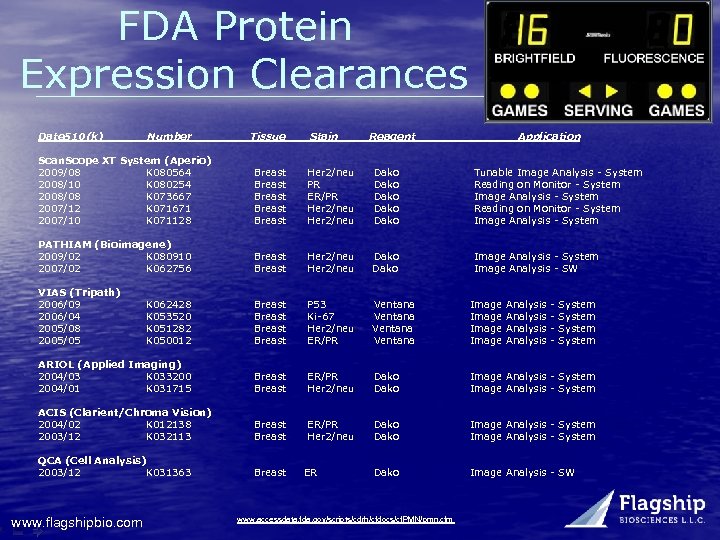
FDA Protein Expression Clearances Date 510(k) Number Tissue Stain Reagent Application Scan. Scope XT System (Aperio) 2009/08 K 080564 2008/10 K 080254 2008/08 K 073667 2007/12 K 071671 2007/10 K 071128 Breast Breast Her 2/neu PR ER/PR Her 2/neu Dako Dako Tunable Image Analysis - System Reading on Monitor - System Image Analysis - System PATHIAM (Bioimagene) 2009/02 K 080910 2007/02 K 062756 Breast Her 2/neu Dako Image Analysis - System Image Analysis - SW VIAS (Tripath) 2006/09 2006/04 2005/08 2005/05 K 062428 K 053520 K 051282 K 050012 Breast P 53 Ki-67 Her 2/neu ER/PR Ventana Image ARIOL (Applied Imaging) 2004/03 K 033200 2004/01 K 031715 Breast ER/PR Her 2/neu Dako Image Analysis - System ACIS (Clarient/Chroma Vision) 2004/02 K 012138 2003/12 K 032113 Breast ER/PR Her 2/neu Dako Image Analysis - System QCA (Cell Analysis) 2003/12 K 031363 Breast Dako Image Analysis - SW www. flagshipbio. com ER www. accessdata. fda. gov/scripts/cdrh/cfdocs/cf. PMN/pmn. cfm Analysis - System
www. flagshipbio. com
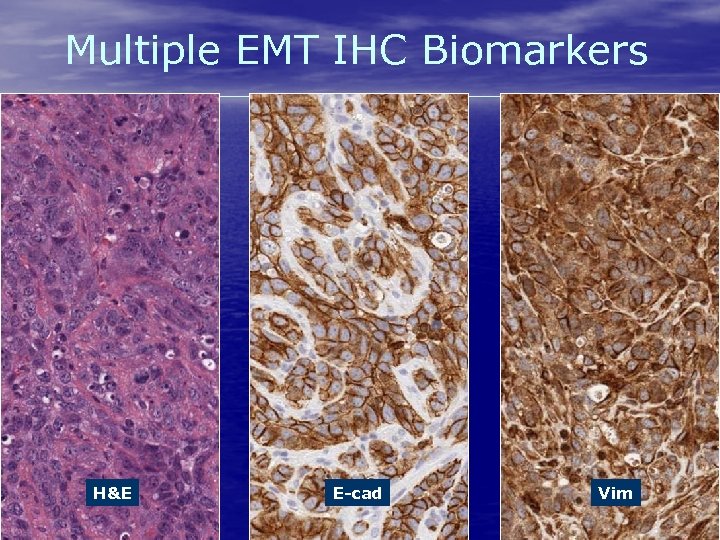
Multiple EMT IHC Biomarkers H&E www. flagshipbio. com E-cad 14 Vim

www. flagshipbio. com

FACTS* Feature Analysis on Consecutive Tissue Sections A multiplexing biomarker approach for analysis www. flagshipbio. com *Patent Pending

Automating quantitative IHC ROI analysis in tissue is a HARD problem… • What works on a few samples doesn’t translate to real-world samples, especially in clinical trials where the ability to control sample acquisition, handling, fixation, IHC, and scanning is limited • IHC histologies simply do not have enough biology information to allow the computer to quickly build a reproducible, reliable system • Tissue variability is difficult – on any computer software www. flagshipbio. com Where is my ROI?

Common IA Needs Neurology Amyloid plaque Neurofibrillary tangles & tau Toxicology Spleen red/white pulp Liver toxicologies Diabetes Beta cell mass in islets with stereology Oncology Xenograft tumor / normal / necrosis Tumor bank samples tumor / normal / necrosis Biomarkers in kidney glomeruli Easy www. flagshipbio. com Robust Difficult Clinical trials samples tumor / normal / necrosis Impossible

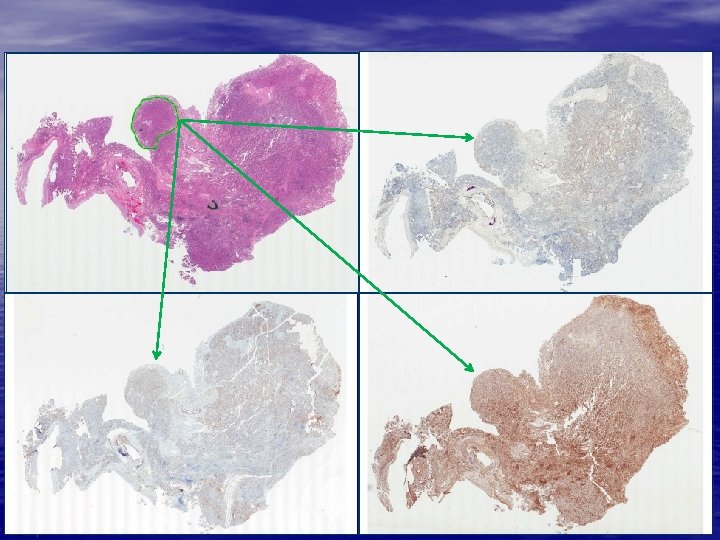
Feature Analysis on Consecutive Tissue Sections (FACTS) 1. Consecutive tissue sectioning www. flagshipbio. com 2. Automated feature recognition 3. Image and ROI registration 4. QC and pathologist review

1. Consecutive tissue sectioning • GOAL: Minimal disruption to histology lab processes – Careful sectioning to get excellent consecutive tissue ribbons – Control pre-analytical factors *All slides for biomarkers must be taken in same session www. flagshipbio. com

Feature Analysis on Consecutive Tissue Sections (FACTS) 1. Consecutive tissue sectioning Biomarker -1 Biomarker -2 1 a. Slide staining H&E Biomarker -3 Biomarker -4 www. flagshipbio. com

1. Consecutive tissue sectioning 2. Automated feature recognition GOAL: Optimal reproducible and scalable whole slide feature analysis • Automatically recognizing features with assist of special stains • Special stain examples: – Oncology: Tumor / stroma / necrosis differentiation • Prostate & Lung substructures – Diabetes / Pancreas: anti-insulin antibody for islets – Kidney / renal tox: glomeruli stains www. flagshipbio. com
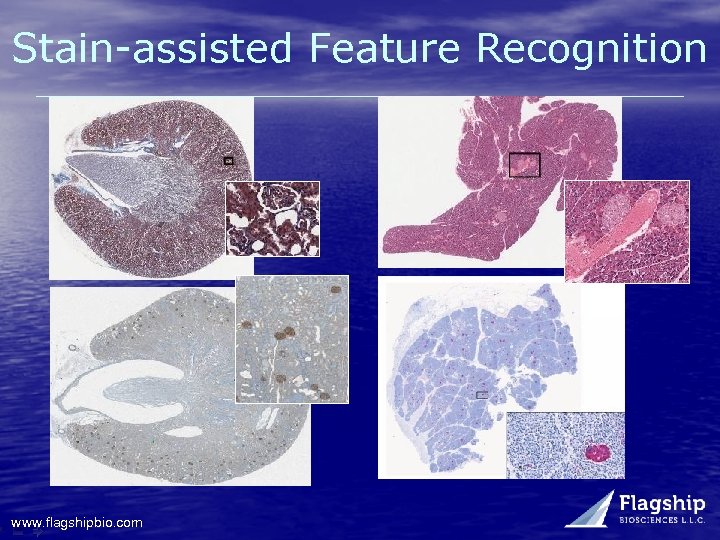
Stain-assisted Feature Recognition www. flagshipbio. com

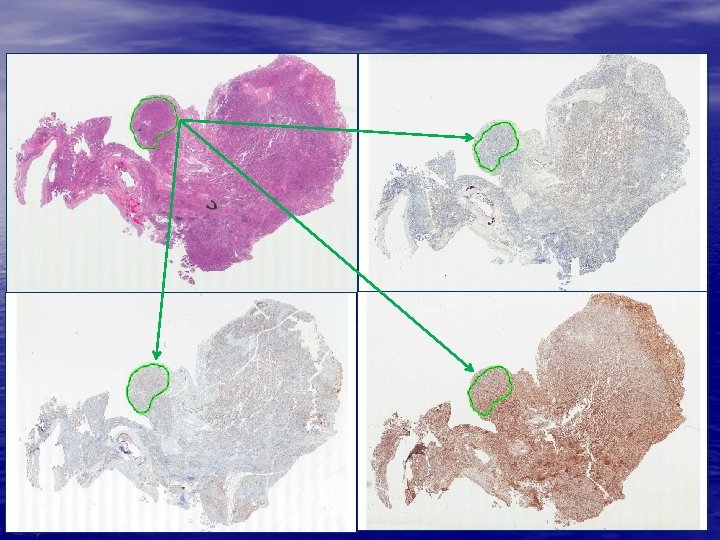
1. Consecutive tissue sectioning 2. Automated feature recognition 3. Image and ROI registration GOAL: Successfully register image with <3% error rate on ROI transfers between consecutive sections • Image registration approaches from radiology • Multi-modal, semi-automatic approach • Requires first rotating, translating, and sizing two whole slide images • Secondary step involves transferred ROI alignment (rotating, translating, sizing approach to near boundaries) www. flagshipbio. com

1. Consecutive tissue sectioning 2. Automated feature recognition 3. Image and ROI registration 4. QC and pathologist review GOAL: Increase analysis accuracy while improving pathologist productivity • Technician review and exclusion of poorly identified features – Features missing in adjacent sections (e. g. end-cut glomeruli or islets) – Non-specific staining impacting feature recognition – Poorly matched features • Pathologist review and sign-out www. flagshipbio. com
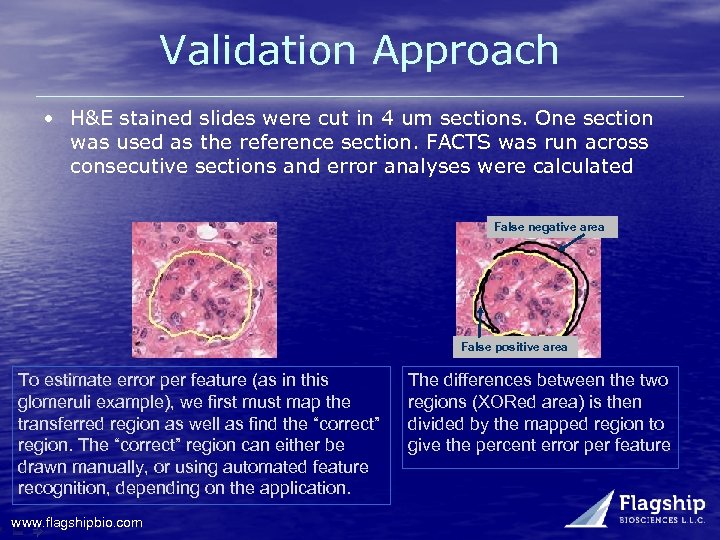
Validation Approach • H&E stained slides were cut in 4 um sections. One section was used as the reference section. FACTS was run across consecutive sections and error analyses were calculated False negative area False positive area To estimate error per feature (as in this glomeruli example), we first must map the transferred region as well as find the “correct” region. The “correct” region can either be drawn manually, or using automated feature recognition, depending on the application. www. flagshipbio. com The differences between the two regions (XORed area) is then divided by the mapped region to give the percent error per feature
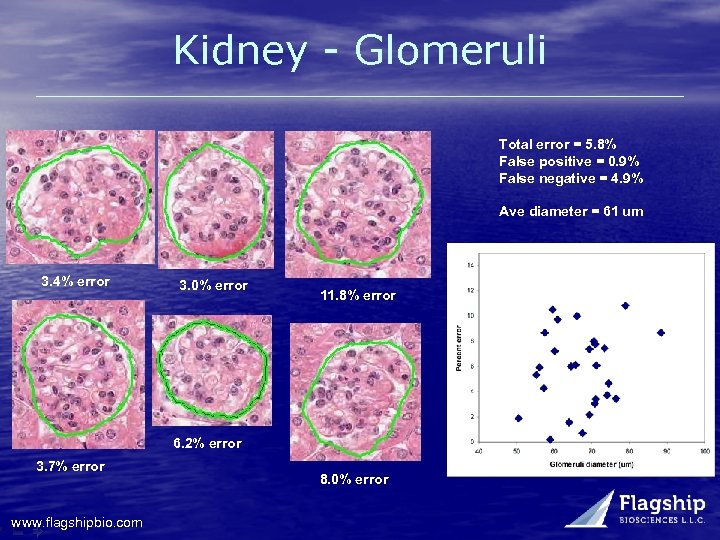
Kidney - Glomeruli Total error = 5. 8% False positive = 0. 9% False negative = 4. 9% Ave diameter = 61 um 3. 4% error 3. 0% error 11. 8% error 6. 2% error 3. 7% error www. flagshipbio. com 8. 0% error
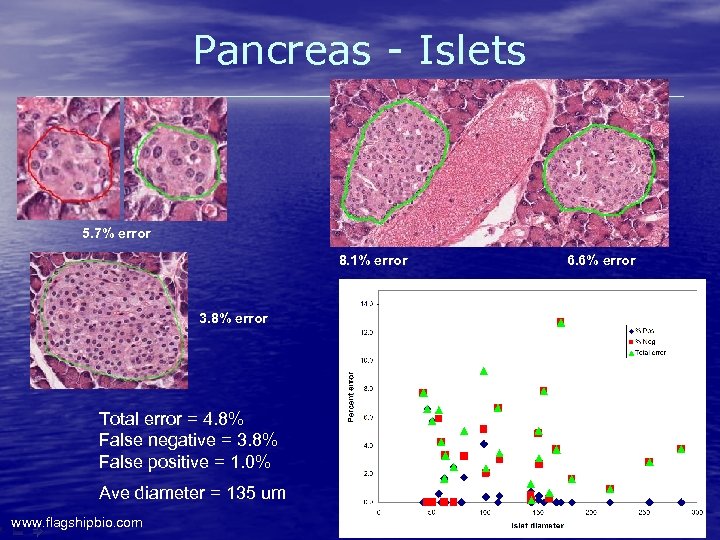
Pancreas - Islets 5. 7% error 8. 1% error 3. 8% error Total error = 4. 8% False negative = 3. 8% False positive = 1. 0% Ave diameter = 135 um www. flagshipbio. com 6. 6% error
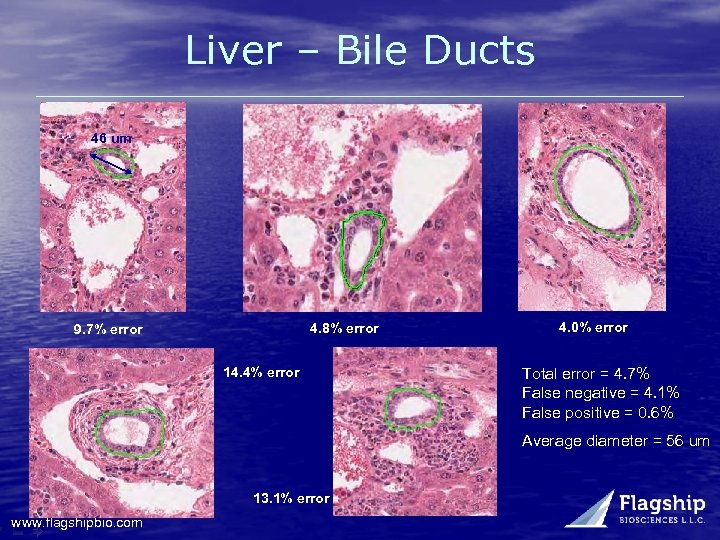
Liver – Bile Ducts 46 um 4. 8% error 9. 7% error 14. 4% error 4. 0% error Total error = 4. 7% False negative = 4. 1% False positive = 0. 6% Average diameter = 56 um 13. 1% error www. flagshipbio. com
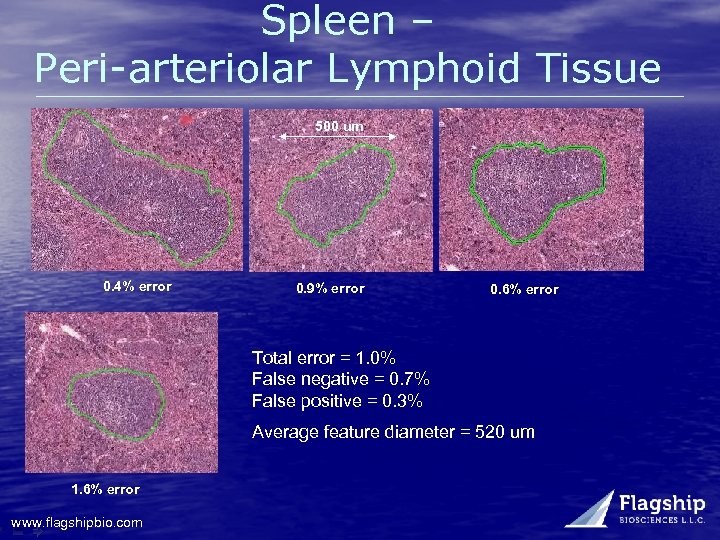
Spleen – Peri-arteriolar Lymphoid Tissue 500 um 0. 4% error 0. 9% error 0. 6% error Total error = 1. 0% False negative = 0. 7% False positive = 0. 3% Average feature diameter = 520 um 1. 6% error www. flagshipbio. com
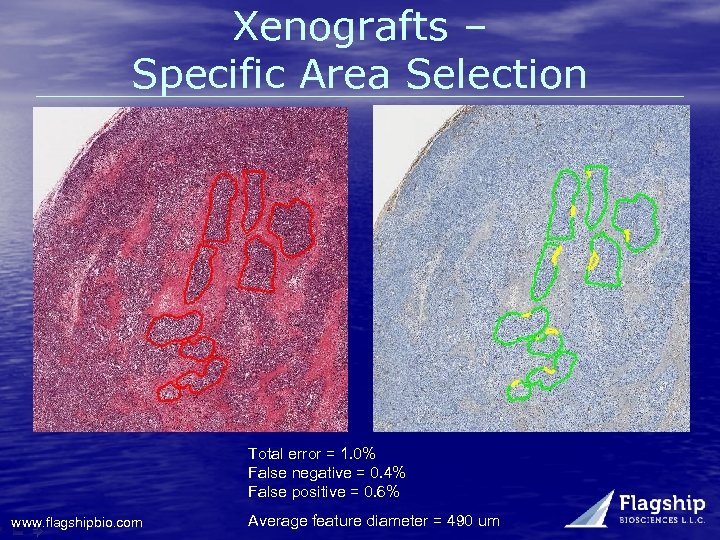
Xenografts – Specific Area Selection Total error = 1. 0% False negative = 0. 4% False positive = 0. 6% www. flagshipbio. com Average feature diameter = 490 um
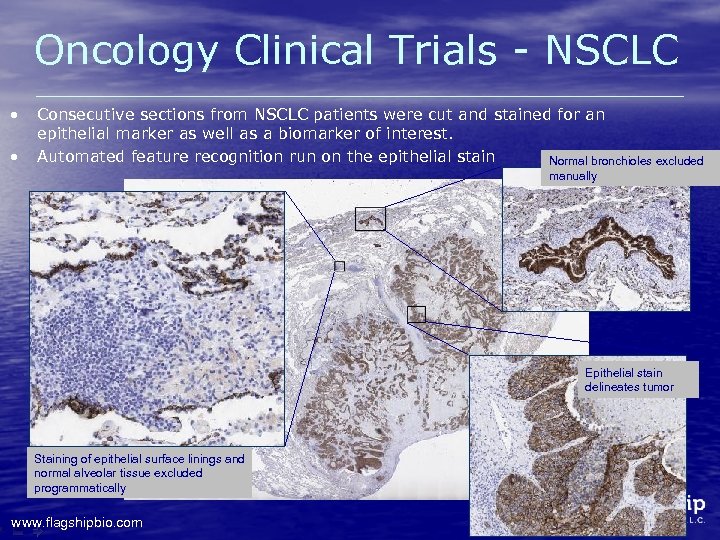
Oncology Clinical Trials - NSCLC • • Consecutive sections from NSCLC patients were cut and stained for an epithelial marker as well as a biomarker of interest. Automated feature recognition run on the epithelial stain Normal bronchioles excluded manually Epithelial stain delineates tumor Staining of epithelial surface linings and normal alveolar tissue excluded programmatically www. flagshipbio. com
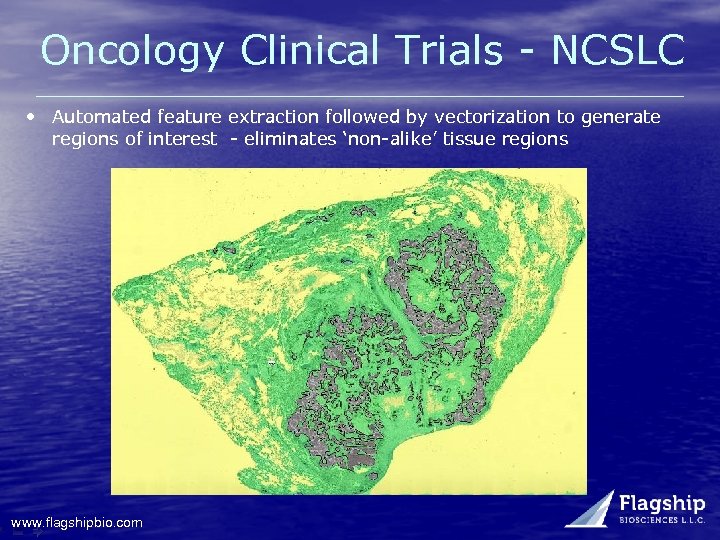
Oncology Clinical Trials - NCSLC • Automated feature extraction followed by vectorization to generate regions of interest - eliminates ‘non-alike’ tissue regions www. flagshipbio. com
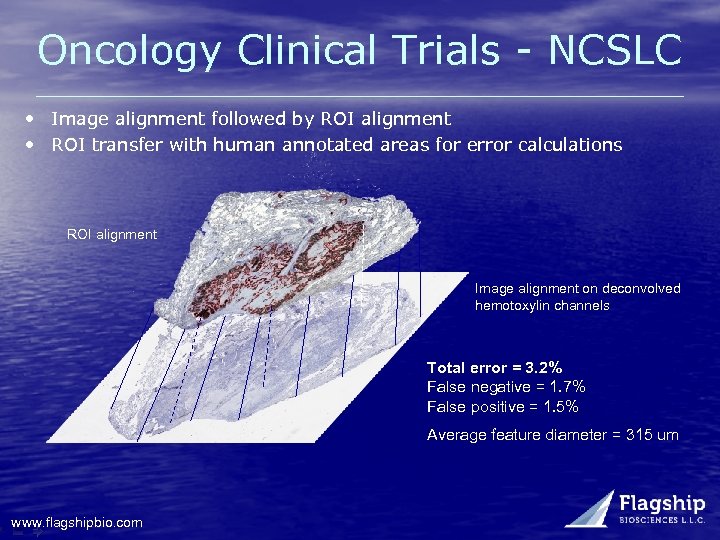
Oncology Clinical Trials - NCSLC • Image alignment followed by ROI alignment • ROI transfer with human annotated areas for error calculations ROI alignment Image alignment on deconvolved hemotoxylin channels Total error = 3. 2% False negative = 1. 7% False positive = 1. 5% Average feature diameter = 315 um www. flagshipbio. com
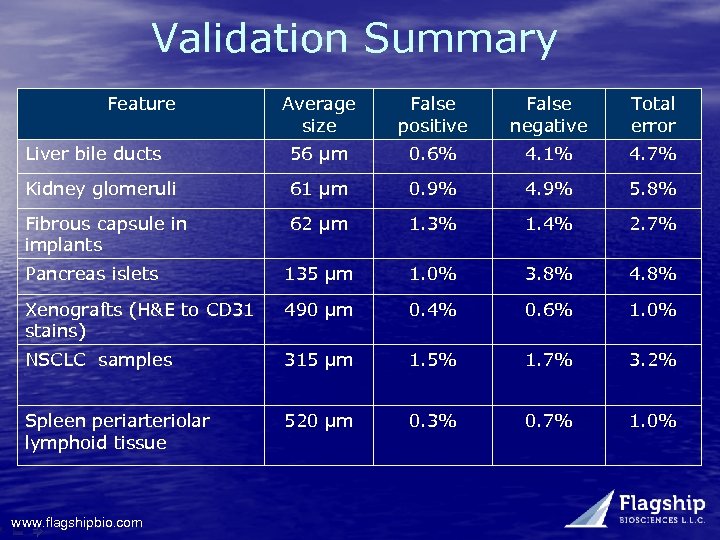
Validation Summary Feature Average size False positive False negative Total error Liver bile ducts 56 µm 0. 6% 4. 1% 4. 7% Kidney glomeruli 61 µm 0. 9% 4. 9% 5. 8% Fibrous capsule in implants 62 µm 1. 3% 1. 4% 2. 7% Pancreas islets 135 µm 1. 0% 3. 8% 4. 8% Xenografts (H&E to CD 31 stains) 490 µm 0. 4% 0. 6% 1. 0% NSCLC samples 315 µm 1. 5% 1. 7% 3. 2% Spleen periarteriolar lymphoid tissue 520 µm 0. 3% 0. 7% 1. 0% www. flagshipbio. com
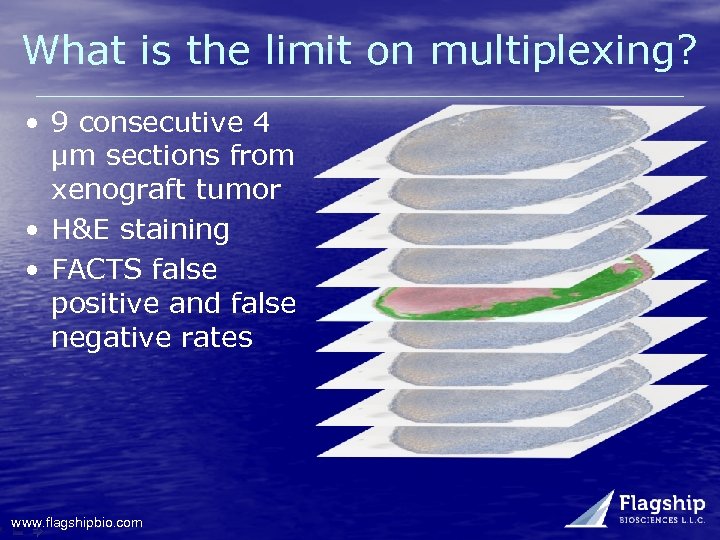
What is the limit on multiplexing? • 9 consecutive 4 µm sections from xenograft tumor • H&E staining • FACTS false positive and false negative rates www. flagshipbio. com

What is the limit on multiplexing? Tissue Section False Negative % False Positive % Total error % +4 2. 1 2. 6 4. 7 +3 1. 8 1. 1 2. 9 +2 1. 8 0. 6 2. 4 +1 1. 4 0. 7 2. 1 Reference section -1 0. 7 1. 9 2. 6 -2 1. 1 2. 2 3. 3 -3 1. 8 3. 9 5. 7 -4 1. 9 3. 6 5. 5 www. flagshipbio. com
www. flagshipbio. com
www. flagshipbio. com

Advantages of FACTS • Multiple IHC biomarkers can be developed into one IVDMIA • More reliable approach for highly variable samples seen in real world situations • Cost-effective and fits well into current GLP and CLIA practice • No novel double/triple stains or biomarker development required • Full audit trail of glass slides • Follows a precedent path with standard brightfield IHC IA digital imaging 510 k approval process www. flagshipbio. com

Regulatory Alignment of FACTS • • • Trackable, reproducible image transfer and registration Similar process as precedent FDA clearances Requires no novel histology processes Review and pathologist sign out is the same Validation through FDA regulations and CLIA compliance www. flagshipbio. com

Ongoing Flagship Projects with FACTS Preclinical Toxicology • Liver – bile ducts • Kidney: glomeruli dysfunction • Pancreas: islets, alpha/beta cell mass • Spleen: red / white pulp, EMH www. flagshipbio. com Discovery & Clinical • Multiple IHC measurements in xenografts • IVDMIA development in lung samples • Stroma / Cancer in ER/PR/HER 2 • TMA multiplexing in discovery and retrospective clinical trials • Prognos. Dx epigenetic markers (5 histone markers)

What will you do FIRST with FACTS? www. flagshipbio. com

Steve Potts Trevor Johnson David Young Scott Watson Frank Voelker Erik Hagendorn Rob Diller Rob Keller Contact us at: pathservices@flagshipbio. com Dave@flagshipbio. com www. flagshipbio. com
d90b0e7ef37f44c071ad22f5dad87eb8.ppt