8f12bb38770715a3e8a275efbe38d29d.ppt
- Количество слайдов: 15

FDG PET/CT

FDG-PET/CT Technical Committee Breakout 081204 Regular meetings of working groups with RSNA administrative support and milestones for delivery during 2009 Regular “SNM cross-over” each TC tcon Communication of recommendations to vendors with reasonable expectation of implementation timeframe Expanded list of co-variates New Profiles • QC • Export standards (RIC, cross-check, Bresolin) • De-identification irrespective of medium (DICOM cross-check) • Trials and service apps (public & private label & access) • Performance elements and standards (Yap)

Monitoring Rx Benefit = Individualized Medicine Convergence of Biomarkers and Diagnostics H/o Commercial Dx Biomarkers become Dx Individuals - thresholds Population means, s Categorical, staging Continuous, quantitative Workflow/# studies Automation/bias Binary – choice of Rx Longitudinal – monitor Rx Quantifying the effects of drugs in development may beg access to biomarkers as diagnostics

Quantitative Imaging Are we there yet?
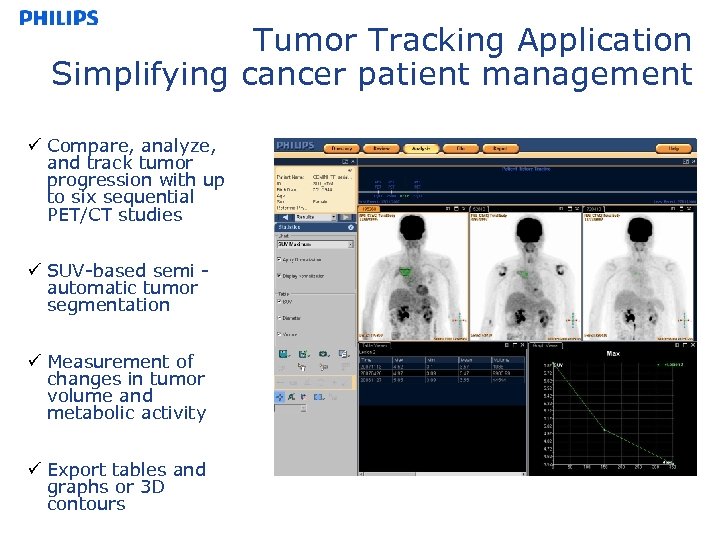
Tumor Tracking Application Simplifying cancer patient management ü Compare, analyze, and track tumor progression with up to six sequential PET/CT studies ü SUV-based semi automatic tumor segmentation ü Measurement of changes in tumor volume and metabolic activity ü Export tables and graphs or 3 D contours

Longitudinal Quantification in Hybrid Imaging Reality Today with Siemens syngo True. D ü Visualization 3 Time Points Display MIP Triangulation Gating Support ü Quantification 2 D/3 D Iso-Contouring 1 -Click SUVmax RT Structure Export ü Trending Charts VOI Propagation Deformable Registration 6 Siemens Medical Solutions Innovation is in our genes. Molecular Imaging
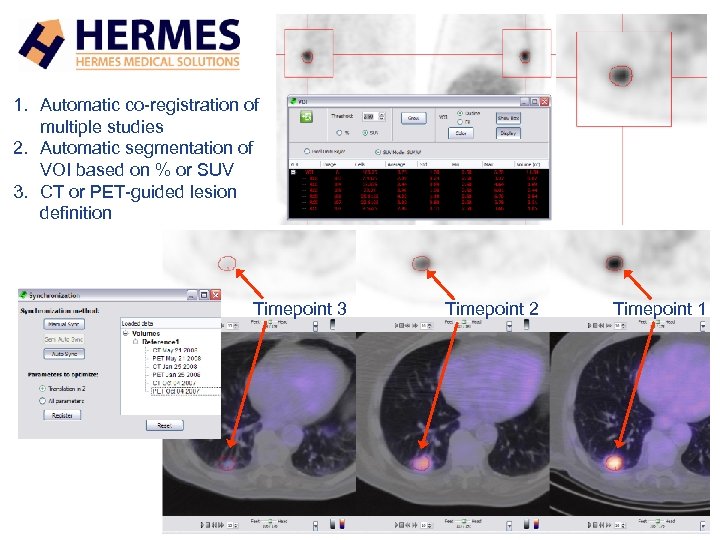
1. Automatic co-registration of multiple studies 2. Automatic segmentation of VOI based on % or SUV 3. CT or PET-guided lesion definition Timepoint 3 Timepoint 2 Timepoint 1
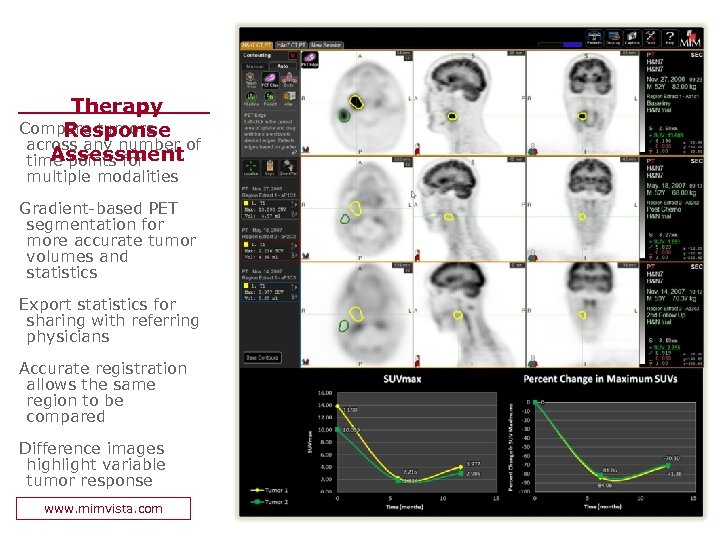
Therapy Compare tumors Response across any number of Assessment time points for multiple modalities Gradient-based PET segmentation for more accurate tumor volumes and statistics Export statistics for sharing with referring physicians Accurate registration allows the same region to be compared Difference images highlight variable tumor response www. mimvista. com

GE PET VCAR…Powerful Longitudinal Analysis ü Automatically register serial studies for longitudinal accuracy ü Automatically segment and propagate ROIs for efficiency and robustness ü Quantify serial changes with a comprehensive set of metrics (SUV, Total Lesion Glycolysis, Functional Volume) and present in tabulated and graphical form ü Export tabulated data for easy utilization in 3 rd party analysis routines

FDG-PET/CT Technical Committee Aim The aim of the QIBA FDG-PET/CT Technical Committee is to foster adoption of • pragmatic and cost-effective standards for • accurate and reproducible quantitation of • tumor metabolism via • longitudinal measures by FDG-PET/CT, with • clinical relevance and known sigma.
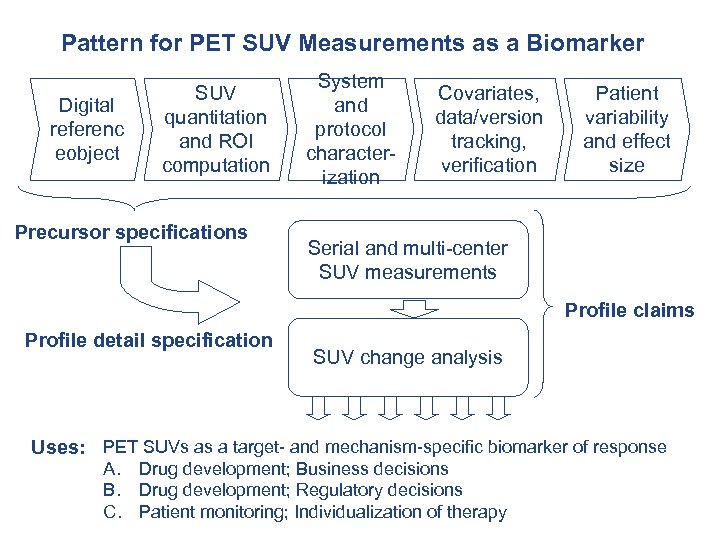
Pattern for PET SUV Measurements as a Biomarker Digital referenc eobject SUV quantitation and ROI computation Precursor specifications System and protocol characterization Covariates, data/version tracking, verification Patient variability and effect size Serial and multi-center SUV measurements Profile claims Profile detail specification SUV change analysis Uses: PET SUVs as a target- and mechanism-specific biomarker of response A. Drug development; Business decisions B. Drug development; Regulatory decisions C. Patient monitoring; Individualization of therapy
![FDG-PETCT Technical Committee e Subcommittee Topics fil[chair] of ke us sa tat or t FDG-PETCT Technical Committee e Subcommittee Topics fil[chair] of ke us sa tat or t](https://present5.com/presentation/8f12bb38770715a3e8a275efbe38d29d/image-12.jpg)
FDG-PETCT Technical Committee e Subcommittee Topics fil[chair] of ke us sa tat or t s f ed rren [David Clunie] Quantitation Computation et u el c d to Digital Reference Objectss– Images [Paul Kinahan] de ting Covariates rationale (Normalization) [Yuying Hwang] sli a eethend. Adoption) Ro. I Definition (and up [Tim Turkington] itt d m e om ne bc ize Su s
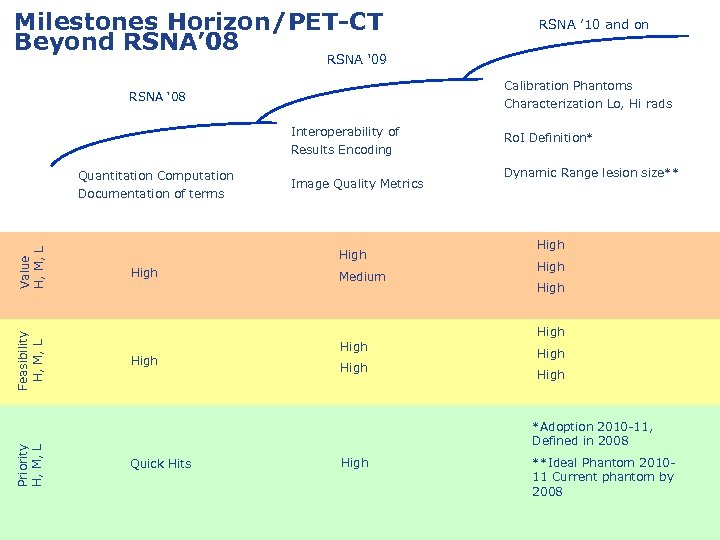
Milestones Horizon/PET-CT Beyond RSNA’ 08 RSNA ’ 10 and on RSNA ‘ 09 Calibration Phantoms Characterization Lo, Hi rads RSNA ‘ 08 Interoperability of Results Encoding Priority H, M, L Feasibility H, M, L Value H, M, L Quantitation Computation Documentation of terms Image Quality Metrics High Medium Ro. I Definition* Dynamic Range lesion size** High High High *Adoption 2010 -11, Defined in 2008 Quick Hits High **Ideal Phantom 201011 Current phantom by 2008

Quantitative Imaging Biomarker Alliance FDGPET/CT Working Group Report Molecular Imaging and Biology 1536 -1632 (Print) 1860 -2002 (Online) RSNA News September 2008, Vol 18, No 9 Hallett WA, Maguire RP, Mc. Carthy TJ, Schmidt ME, Young H. Considerations for generic oncology FDG-PET/CT protocol preparation in drug development. IDrugs, 2007 Nov; 10(11): 791 -6.

FDG PET/CT
8f12bb38770715a3e8a275efbe38d29d.ppt