1269765ce5bc24312d82f89362fc548e.ppt
- Количество слайдов: 36

FDA Thailand By HIV Module/Marketing Group Mr. Manaswee Arayasiri 1

FDA Thailand • Introduction • Drugs registration • The effect of FDA on Marketing 2

Introduction • • • About FDA Thailand Role and Responsibility Relating Acts Administration of Acts Organization of FDA for controlling 3

About FDA Thailand • Consumer protection activities on food and drugs have begun in Thailand since 1909 • Established in 1922 as a Narcotic division • In 1937, the agency renamed to Food and Drug division 4

About FDA Thailand • In 1953, the agency renamed to Division of Food and Drug Control. • In 1974, Division of Food and Drug Control was promoted to be the Office of Food and Drug Administration, having the status as department of Ministry of Public Health. 5

Roles and Responsibilities • Major is to ensure that health products(i. e. food, drug etc. ) available to consumers are of standard quality, efficacy, and safety. 6

Roles and Responsibilities • Main tasks are to control and monitor both pre- and post-marketing phases of manufacture, import, transport, storage and sale 7

Relating Act • Drug Act 1987 (5 th revision) • Food Act 1979 • Cosmetic Act 1992 • Narcotic Act 1987 (3 rd revision) 8

Relating Act • Psychotropic Substances Act 1992(3 rd revision) • Volatile Substances Act 1990 • Medical Devices Act 1988 9

Administration of Acts • Committees of drug, food, narcotics, cosmetics etc. follow Acts • Committees to develop policies and promote technological development on food, drugs and chemical safety 10

Organization of FDA for controlling • • Drug control division Food control division Toxic substance control division Narcotic control division Cosmetic control division Medical device control division Public relation and Advertisement control division 11

Drug Registration • How to register drugs • Drugs registration procedure • Other registration 12

How to register drug • Contact Drug control division FDA Thailand Ministry of Public Health Nonthaburi Thailand • E-mail address: www. fda. moph. go. th 13

Drug registration procedure • In this HIV module, Assume use procedure of new drugs registration in term of Generic drug • Generic drug is an imitation of an original brand-name drug, when the patent or trademark protection on original brand-name drug expires 14

Drug registration procedure • Step 1 : Asking permission for Bioequivalence study • Step 2 : Asking permission for produce/order sample drug for study • Step 3 : Asking permission for register Pharmacopoeia 15

Definition • Pharmacopoeia is a book described drugs; one issued by an officially recognized authority and serving as standard • Bioequivalence study is study drugs in term of therapeutic response compare between innovator product and original product 16

Drug registration procedure • Step 1 : Asking permission for Bioequivalence study 17

Document for application • • • Label every size of container Accompany literature Clinical trial report Certificate of free sale Detail of protocol for Bioequivalence study 18

Procedure 19
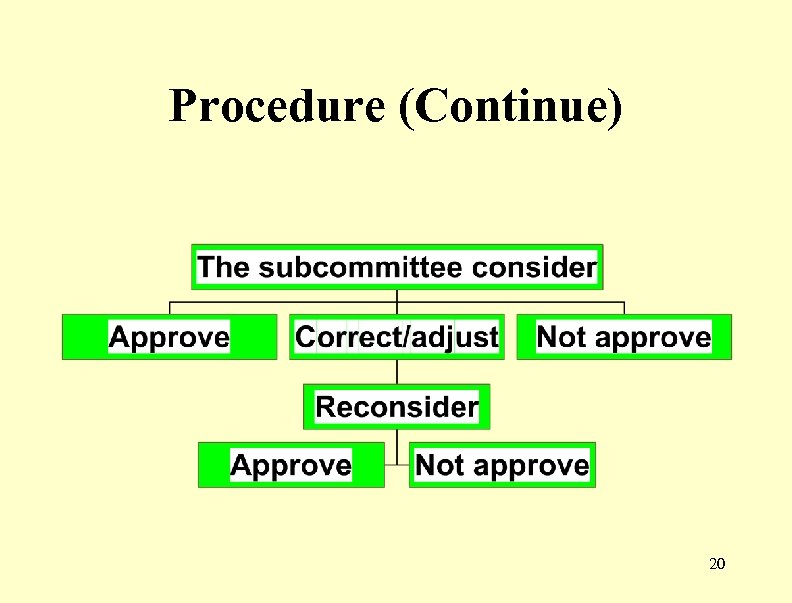
Procedure (Continue) 20

Drug registration procedure • Step 2 : Asking permission for produce/order sample drug for study 21

Document for application • • • Label every size of container Accompany literature Certificate of GMP Certificate of free sale Certificate of Analysis 22

Definition • Certificate of GMP is certificate for factory about Manufacturing practice follow WHO practice • Certificate of Free Sale is document present that drug is legally sold in the country of origin 23

Definition • Certificate of Analysis is report present result of pharmaceutical product standard checking 24

Procedure 25

Procedure (Continue) 26

Drug registration procedure • Step 3 : Asking permission for register pharmacopoeia 27

Document for application • • Sample drugs Label every size of container Accompany literature Result of Bioequivalence study Finished product Specification Certificate of free sale Certificate of import drug (Step 2) 28

Procedure 29
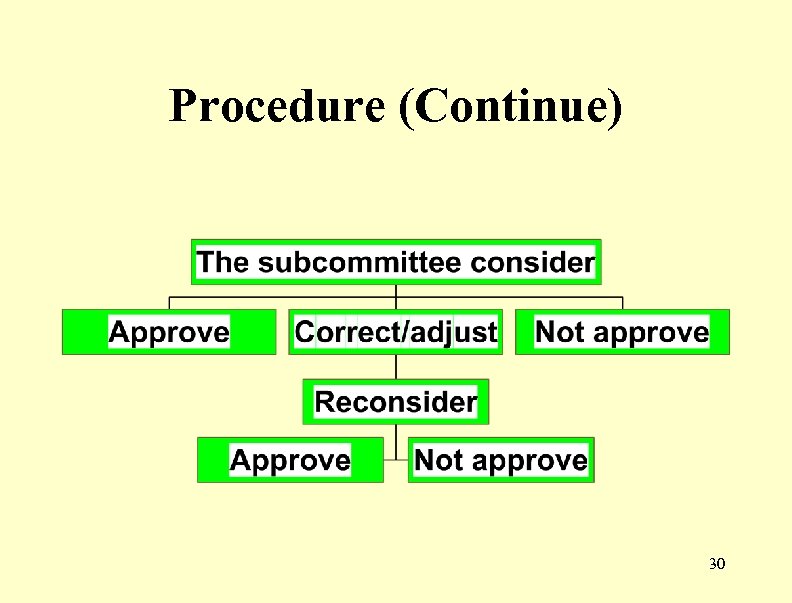
Procedure (Continue) 30

Other registration • License to produce drugs • License to sell drugs 31

The effect of FDA on Marketing • Cost of product • Regulation in Selling 32

Cost of Product • Bioequivalence Study • License for register, production, selling drug • Quality control in production and environment 33

Regulation in Selling • Producer must have license for sell drug • Amount of selling must follow license • Advertisement ; clearly, include side effect and no over/false the real properties 34

Thank you very much • • Dr. Suvit Dr. Maria Mrs. Podjana all listeners 35

Thank you for pay attention 36
1269765ce5bc24312d82f89362fc548e.ppt