d33be2a9960898d4fd21a0b1fe92c358.ppt
- Количество слайдов: 104
 “FAMI-QS” European Feed Additives and Pre. MIxtures Quality System Context and insights
“FAMI-QS” European Feed Additives and Pre. MIxtures Quality System Context and insights
 Back Comment l l l 2 Please use the link “back” in the upper right corner to jump to the general content page Please use the links in the various pages starting the different chapters to take you quickly to your point of interest Please use the links to the internet to access some information there quickly The Expert Group of FAMI-QS June 2005 (V 3) End
Back Comment l l l 2 Please use the link “back” in the upper right corner to jump to the general content page Please use the links in the various pages starting the different chapters to take you quickly to your point of interest Please use the links to the internet to access some information there quickly The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Content l l l Regulatory background The FAMI-QS Concept The Association The Certification The Code – – 3 Products Manufacturing HACCP Non-Conformances The Expert Group of FAMI-QS June 2005 (V 3) End
Back Content l l l Regulatory background The FAMI-QS Concept The Association The Certification The Code – – 3 Products Manufacturing HACCP Non-Conformances The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Content l l l 4 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
Back Content l l l 4 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Hygiene in the feed chain l Directive 95/69/EC – l The Feed hygiene Regulation proposal – 5 (present) (finalized 12/04, published January 2005 as 183/2005/EC, effective as of 01/01/06) The Expert Group of FAMI-QS June 2005 (V 3) End
Back Hygiene in the feed chain l Directive 95/69/EC – l The Feed hygiene Regulation proposal – 5 (present) (finalized 12/04, published January 2005 as 183/2005/EC, effective as of 01/01/06) The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Directive 95/69 – “Establishments” l l l 6 Approval for activities which, are considered potentially hazardous to animals, humans or the environment. Registration for products considered less sensitive. In both cases establishments and intermediaries must comply with detailed "quality control" requirements. Compliance is a precondition before approval (inspection by the competent authority) can be given. The obligation to comply with those requirements continues once approval/registration has been obtained. The Expert Group of FAMI-QS June 2005 (V 3) End
Back Directive 95/69 – “Establishments” l l l 6 Approval for activities which, are considered potentially hazardous to animals, humans or the environment. Registration for products considered less sensitive. In both cases establishments and intermediaries must comply with detailed "quality control" requirements. Compliance is a precondition before approval (inspection by the competent authority) can be given. The obligation to comply with those requirements continues once approval/registration has been obtained. The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Feed Hygiene Regulation l l 95/69 'enriched' by the Feed Hygiene Regulation Integrate the manufacturing and distribution of feed additives, premixtures and complete feedingstuffs in a global food chain Integrate, in particular, HACCP principle Scope – Feed Business l – – 7 Feed additives, premixtures, feed materials, feedingstuffs Farmers Imports The Expert Group of FAMI-QS June 2005 (V 3) End
Back Feed Hygiene Regulation l l 95/69 'enriched' by the Feed Hygiene Regulation Integrate the manufacturing and distribution of feed additives, premixtures and complete feedingstuffs in a global food chain Integrate, in particular, HACCP principle Scope – Feed Business l – – 7 Feed additives, premixtures, feed materials, feedingstuffs Farmers Imports The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Feed Hygiene Regulation l Obligations for the chain: – – – l Guides for application of: – – 8 Good Manufacturing Practices (including logistics) Microbiological criteria (specific) Hazard Analysis of Critical Control Points (HACCP) Financial Guarantees Approval / Registration Good Manufacturing Practices HACCP The Expert Group of FAMI-QS June 2005 (V 3) End
Back Feed Hygiene Regulation l Obligations for the chain: – – – l Guides for application of: – – 8 Good Manufacturing Practices (including logistics) Microbiological criteria (specific) Hazard Analysis of Critical Control Points (HACCP) Financial Guarantees Approval / Registration Good Manufacturing Practices HACCP The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Content l l l 9 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
Back Content l l l 9 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Advantages l l l 10 Optimisation of efforts for feed safety by agreed upon measures Auditable harmonized code throughout EU, thus Minimization of need for checks by authorities, users The Expert Group of FAMI-QS June 2005 (V 3) End
Back Advantages l l l 10 Optimisation of efforts for feed safety by agreed upon measures Auditable harmonized code throughout EU, thus Minimization of need for checks by authorities, users The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Compliance l A tool for complying with the Feed Hygiene Regulation, which: – – l 11 encourages the development of systems to good practice for hygiene requires the application of HACCP principles Positive dynamic with chain partners The Expert Group of FAMI-QS June 2005 (V 3) End
Back Compliance l A tool for complying with the Feed Hygiene Regulation, which: – – l 11 encourages the development of systems to good practice for hygiene requires the application of HACCP principles Positive dynamic with chain partners The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Basis l Code is based on established guidelines such as – – 12 Codex Alimentarius (HACCP) EN ISO 9001: 2000 Management systems developed in different countries like GMP+, FEMAS, Q+S, … EU proposal on feed hygiene The Expert Group of FAMI-QS June 2005 (V 3) End
Back Basis l Code is based on established guidelines such as – – 12 Codex Alimentarius (HACCP) EN ISO 9001: 2000 Management systems developed in different countries like GMP+, FEMAS, Q+S, … EU proposal on feed hygiene The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Information l Information about the Code itself, as well as the association and the certification process can be found under www. fami-qs. org 13 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Information l Information about the Code itself, as well as the association and the certification process can be found under www. fami-qs. org 13 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Content l l l 14 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
Back Content l l l 14 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Association l l l 15 'European Feed Additives and Pre. MIxtures Quality System Association' 'Not-for-profit' association (Asbl under Belgian law) Formally created on May 04, 2004 First General Assembly in Brussels in May 2004 Second General Assembly in Brussels in January 2005 The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Association l l l 15 'European Feed Additives and Pre. MIxtures Quality System Association' 'Not-for-profit' association (Asbl under Belgian law) Formally created on May 04, 2004 First General Assembly in Brussels in May 2004 Second General Assembly in Brussels in January 2005 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Purpose and Activities l l 16 Manage and co-ordinate all activities related to the EU Feed Additives and premixture Quality System – FAMI-QS Establish, register and maintain the FAMI-QS system Rule the use of the code Coordinate adequate certification systems The Expert Group of FAMI-QS June 2005 (V 3) End
Back Purpose and Activities l l 16 Manage and co-ordinate all activities related to the EU Feed Additives and premixture Quality System – FAMI-QS Establish, register and maintain the FAMI-QS system Rule the use of the code Coordinate adequate certification systems The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Membership Structure l Full members – – – l Associate members – – 17 Manufacturers or traders of FA or PM Members of FEFANA Min. 5, no max. , full voting rights Involved in the feed additive or premixture business FAMI-QS certified The Expert Group of FAMI-QS June 2005 (V 3) End
Back Membership Structure l Full members – – – l Associate members – – 17 Manufacturers or traders of FA or PM Members of FEFANA Min. 5, no max. , full voting rights Involved in the feed additive or premixture business FAMI-QS certified The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Full Members* l l l Adisseo ADM Ajinomoto Alltech BASF Degussa l l l DSM Finn. Feeds Lohmann Novus Novozymes Phytobiotics *status June 2005 18 The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Full Members* l l l Adisseo ADM Ajinomoto Alltech BASF Degussa l l l DSM Finn. Feeds Lohmann Novus Novozymes Phytobiotics *status June 2005 18 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Board, Secretary General and Quality Manager l l l 19 Rolf-Dieter Reinhardt (BASF) Hermann Roth (Phytobiotics) Matthew Russel (Alltech) Manfred Peisker (ADM) Didier Jans (Secretary General) Ioan Paraian (Quality Manager/Coordinator) The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Board, Secretary General and Quality Manager l l l 19 Rolf-Dieter Reinhardt (BASF) Hermann Roth (Phytobiotics) Matthew Russel (Alltech) Manfred Peisker (ADM) Didier Jans (Secretary General) Ioan Paraian (Quality Manager/Coordinator) The Expert Group of FAMI-QS June 2005 (V 3) End
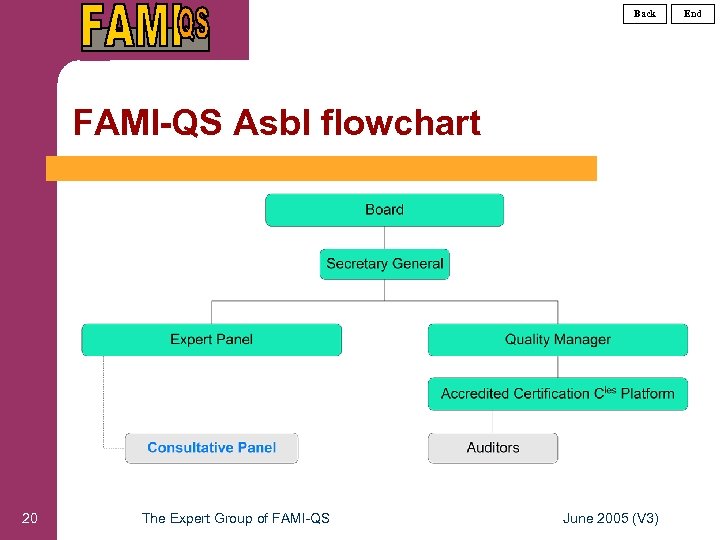 Back FAMI-QS Asbl flowchart 20 The Expert Group of FAMI-QS June 2005 (V 3) End
Back FAMI-QS Asbl flowchart 20 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The History l l l l l 21 Development of the Code of Practice (April ’ 03 – April ’ 04) FAMI-QS founded (May ’ 04) Training of certification bodies (June ‘ 04) Coordination meeting with Partner Platform (national organisations for feed quality systems, June ‘ 04) FAMI-QS internal approval of certification bodies (mid-July) Final versions (ver. 1) of Code of Practice and Description of Certification Process (July ‘ 04) Web site (August ’ 04) Start of certification process, audits (September ‘ 04) Second version of Code published in June 2005 First seven guidance with more detailed information on topics such as transportation, HACCP, hygiene etc published The Expert Group of FAMI-QS June 2005 (V 3) End
Back The History l l l l l 21 Development of the Code of Practice (April ’ 03 – April ’ 04) FAMI-QS founded (May ’ 04) Training of certification bodies (June ‘ 04) Coordination meeting with Partner Platform (national organisations for feed quality systems, June ‘ 04) FAMI-QS internal approval of certification bodies (mid-July) Final versions (ver. 1) of Code of Practice and Description of Certification Process (July ‘ 04) Web site (August ’ 04) Start of certification process, audits (September ‘ 04) Second version of Code published in June 2005 First seven guidance with more detailed information on topics such as transportation, HACCP, hygiene etc published The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Status l l First companies certified (15 companies / 30 sites)* Increasing interest from other companies who plan to become certified, including companies from outside EU (India, China) (another 15 companies / 30 sites)* * Status June 2005 22 The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Status l l First companies certified (15 companies / 30 sites)* Increasing interest from other companies who plan to become certified, including companies from outside EU (India, China) (another 15 companies / 30 sites)* * Status June 2005 22 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Content l l l 23 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
Back Content l l l 23 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Granting a FAMI-QS Certificate l l l Recognised certification bodies only, certification bodies required to achieve accreditation under ISO 45011 Recognition is provided by the FAMIQS organisation Certification body responsibilities: – – – l 24 Planning Assessment of documents Audit reports Certification Use of certificate is conditioned to FAMI-QS membership (associate or full) The Expert Group of FAMI-QS June 2005 (V 3) End
Back Granting a FAMI-QS Certificate l l l Recognised certification bodies only, certification bodies required to achieve accreditation under ISO 45011 Recognition is provided by the FAMIQS organisation Certification body responsibilities: – – – l 24 Planning Assessment of documents Audit reports Certification Use of certificate is conditioned to FAMI-QS membership (associate or full) The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Certification of EU operators l l All EU based operators should be certified by 01 Jan. 2006 Operators buying Feed Additives from another EU based operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of their supplier (in case a supplier is not certified yet). – 25 This case covers the period until January 2006. The Expert Group of FAMI-QS June 2005 (V 3) End
Back Certification of EU operators l l All EU based operators should be certified by 01 Jan. 2006 Operators buying Feed Additives from another EU based operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of their supplier (in case a supplier is not certified yet). – 25 This case covers the period until January 2006. The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Certification of Importers and Manufacturers outside the EU l l Operators buying Feed Additives from a non EU operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of the supplier and the initial manufacturer of the Feed Additive. Deadline for the supplier and initial manufacturer to apply for FAMI-QS certification shall not be later than end 2006, and for getting certification no later than end 2007. – 26 It is the auditor's responsibility and obligation to check that above mentioned deadlines are respected. Failure to do this would create a critical non conformity at the operator's (this means a withdrawal of the certificate). The Expert Group of FAMI-QS June 2005 (V 3) End
Back Certification of Importers and Manufacturers outside the EU l l Operators buying Feed Additives from a non EU operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of the supplier and the initial manufacturer of the Feed Additive. Deadline for the supplier and initial manufacturer to apply for FAMI-QS certification shall not be later than end 2006, and for getting certification no later than end 2007. – 26 It is the auditor's responsibility and obligation to check that above mentioned deadlines are respected. Failure to do this would create a critical non conformity at the operator's (this means a withdrawal of the certificate). The Expert Group of FAMI-QS June 2005 (V 3) End
 Back One certificate for entire EU (. . . ) hereby certifies that the company: (. . . ) has implemented and maintains a Quality Management System in compliance with the FAMIQS Standard (ver. xx, date dd/mm/yy) on the following site(s): (…) for the placing on the market of feed additives belonging to the following category(ies) and functional group(s): (…) Audits, documented in a report, have verified that the Management System fulfils the requirements of this standard. This certificate is valid until: (. . . ) The validity of this certificate can be verified on the FAMI-QS web site (www. fami-qs. org) N. B. : For premixtures, the information about feed additive categories is not relevant 27 The Expert Group of FAMI-QS June 2005 (V 3) End
Back One certificate for entire EU (. . . ) hereby certifies that the company: (. . . ) has implemented and maintains a Quality Management System in compliance with the FAMIQS Standard (ver. xx, date dd/mm/yy) on the following site(s): (…) for the placing on the market of feed additives belonging to the following category(ies) and functional group(s): (…) Audits, documented in a report, have verified that the Management System fulfils the requirements of this standard. This certificate is valid until: (. . . ) The validity of this certificate can be verified on the FAMI-QS web site (www. fami-qs. org) N. B. : For premixtures, the information about feed additive categories is not relevant 27 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Content l l l 28 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
Back Content l l l 28 Regulatory background The FAMI-QS Concept The Association The Certification The Code The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l The Code provides among other items requirements for good manufacturing of – – – l l l 29 Products Handling of ingredients Traceability HACCP Logistics (incl. warehousing, transportation) Its consists of text and questions which are both used to define the requirements Details and explanations can be found in the following slides The Code can be found on the FAMI-QS homepage under documents (or click here) The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Code l The Code provides among other items requirements for good manufacturing of – – – l l l 29 Products Handling of ingredients Traceability HACCP Logistics (incl. warehousing, transportation) Its consists of text and questions which are both used to define the requirements Details and explanations can be found in the following slides The Code can be found on the FAMI-QS homepage under documents (or click here) The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l l Manufacturing l HACCP l 30 Products Non Conformances The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Code l l Manufacturing l HACCP l 30 Products Non Conformances The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l – – – Specification Suppliers Incoming Materials Stability Traceability Manufacturing – 31 – Products Building and Equipment Work Environment Finished Goods The Expert Group of FAMI-QS – – – l l Packaging & labelling Quality Control Verification Blending Warehousing & Storage Transport Cleaning HACCP Non Conformances June 2005 (V 3) End
Back The Code l – – – Specification Suppliers Incoming Materials Stability Traceability Manufacturing – 31 – Products Building and Equipment Work Environment Finished Goods The Expert Group of FAMI-QS – – – l l Packaging & labelling Quality Control Verification Blending Warehousing & Storage Transport Cleaning HACCP Non Conformances June 2005 (V 3) End
 Back Specifications A list of tests, references to analytical procedures, and appropriate acceptance criteria that are numerical limits, ranges, or other criteria for the test described. It establishes the set of criteria to which a material shall conform to be considered acceptable for its intended use. “Compliance to specification” means that the material, when tested according to the listed analytical procedures, meets the listed acceptance criteria. 32 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Specifications A list of tests, references to analytical procedures, and appropriate acceptance criteria that are numerical limits, ranges, or other criteria for the test described. It establishes the set of criteria to which a material shall conform to be considered acceptable for its intended use. “Compliance to specification” means that the material, when tested according to the listed analytical procedures, meets the listed acceptance criteria. 32 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Specifications Principle: The specification ensures that an ingredient used always has a predefined agreed upon quality. 33 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Specifications Principle: The specification ensures that an ingredient used always has a predefined agreed upon quality. 33 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Specifications l Specifications are needed for – – 34 Incoming raw materials Packaging materials Final products Labels The Expert Group of FAMI-QS June 2005 (V 3) End
Back Specifications l Specifications are needed for – – 34 Incoming raw materials Packaging materials Final products Labels The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Specifications l Specifications need to …. – Be adequate to control the quality of the material for its intended use and contain e. g. l l – Reflect possible hazards e. g. l l – – 35 Acceptance limits Reference to the method Microbiology were applicable Undesirable substances were applicable Take into account regulatory requirements Be part of the change control procedure and need to be amended if applicable The Expert Group of FAMI-QS June 2005 (V 3) End
Back Specifications l Specifications need to …. – Be adequate to control the quality of the material for its intended use and contain e. g. l l – Reflect possible hazards e. g. l l – – 35 Acceptance limits Reference to the method Microbiology were applicable Undesirable substances were applicable Take into account regulatory requirements Be part of the change control procedure and need to be amended if applicable The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Suppliers Principle: As the goal is to ensure safety throughout the whole food and feed chain the risks brought in from suppliers have to be considered and controlled equally well as risks brought in by the operator 36 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Suppliers Principle: As the goal is to ensure safety throughout the whole food and feed chain the risks brought in from suppliers have to be considered and controlled equally well as risks brought in by the operator 36 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Suppliers l Suppliers of incoming materials must be – Evaluated l l – Approved l – According to defined criteria including where necessary audit results Listed in an internal list of approved suppliers l l 37 A risk consideration must be made for raw materials and primary packaging Samples tested for at least selected criteria in case of raw materials Agreements made Statements requested The list must be reviewed periodically, at least once a year Evaluated for their continuous performance The Expert Group of FAMI-QS June 2005 (V 3) End
Back Suppliers l Suppliers of incoming materials must be – Evaluated l l – Approved l – According to defined criteria including where necessary audit results Listed in an internal list of approved suppliers l l 37 A risk consideration must be made for raw materials and primary packaging Samples tested for at least selected criteria in case of raw materials Agreements made Statements requested The list must be reviewed periodically, at least once a year Evaluated for their continuous performance The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Suppliers l Targets – All suppliers of Feed additives or premixtures must be approved according to FAMI-QS – For other raw materials appropriate risk analysis must be conducted . 38 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Suppliers l Targets – All suppliers of Feed additives or premixtures must be approved according to FAMI-QS – For other raw materials appropriate risk analysis must be conducted . 38 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Suppliers l Interim measures – – 39 All EU based FA and PM operators should be certified by January 2005. Operators buying feed additives (FA) from another EU based operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of their supplier (in the case a supplier is not certified yet). This case covers the period until January 2005, cf. 1. Operators buying FA from a non EU operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of the supplier and the initial manufacturer of the FA. Deadline for the supplier and initial manufacturer to apply for FAMI-QS certification shall not be later than end 2006, and for getting certification no later than end 2007. It is the auditor's responsibility and obligation to check that above mentioned deadlines are respected. Failure to do this would create a critical non conformity at the operator's (this means a withdrawal of the certificate). The Expert Group of FAMI-QS June 2005 (V 3) End
Back Suppliers l Interim measures – – 39 All EU based FA and PM operators should be certified by January 2005. Operators buying feed additives (FA) from another EU based operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of their supplier (in the case a supplier is not certified yet). This case covers the period until January 2005, cf. 1. Operators buying FA from a non EU operator should provide a verifiable plan with milestones and deadlines for ensuring the certification of the supplier and the initial manufacturer of the FA. Deadline for the supplier and initial manufacturer to apply for FAMI-QS certification shall not be later than end 2006, and for getting certification no later than end 2007. It is the auditor's responsibility and obligation to check that above mentioned deadlines are respected. Failure to do this would create a critical non conformity at the operator's (this means a withdrawal of the certificate). The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Incoming Materials l l 40 Incoming material: A general term used to denote raw materials (starting materials, reagents, solvents), process aids, packaging and labelling materials Raw material: Any material which enters the manufacturing process of the feed additive and/or premixture. The Expert Group of FAMI-QS June 2005 (V 3) End
Back Incoming Materials l l 40 Incoming material: A general term used to denote raw materials (starting materials, reagents, solvents), process aids, packaging and labelling materials Raw material: Any material which enters the manufacturing process of the feed additive and/or premixture. The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Incoming Materials Principle: Incoming materials are one of the critical points to potentially include hazards for humans or environment into the product. Thus they must be strictly controlled in order to avoid such hazards. 41 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Incoming Materials Principle: Incoming materials are one of the critical points to potentially include hazards for humans or environment into the product. Thus they must be strictly controlled in order to avoid such hazards. 41 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Incoming Materials l Requirements for approval of incoming materials must be established. They are – – – 42 Risk considerations Sourcing only from suppliers listed internally as approved Specifications, including test methods The Expert Group of FAMI-QS June 2005 (V 3) End
Back Incoming Materials l Requirements for approval of incoming materials must be established. They are – – – 42 Risk considerations Sourcing only from suppliers listed internally as approved Specifications, including test methods The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Incoming Materials It is important to define – – Transport Storage Processing Handling before usage of incoming materials in order to ensure that no adulteration or additional risk arises 43 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Incoming Materials It is important to define – – Transport Storage Processing Handling before usage of incoming materials in order to ensure that no adulteration or additional risk arises 43 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Incoming Materials l When the incoming material is received the operator has to verify – – – 44 supplier’s lot number full supplier’s name of product date of receipt quantity received any findings of deviations on agreed upon quality. These are to be reported to the quality control unit The Expert Group of FAMI-QS June 2005 (V 3) End
Back Incoming Materials l When the incoming material is received the operator has to verify – – – 44 supplier’s lot number full supplier’s name of product date of receipt quantity received any findings of deviations on agreed upon quality. These are to be reported to the quality control unit The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Incoming Materials l Retained sample – l Rejected material – 45 To be kept for the period of shelf life, either at the supplier or the operator Must be strictly controlled by proper identification/storage to avoid unauthorized usage The Expert Group of FAMI-QS June 2005 (V 3) End
Back Incoming Materials l Retained sample – l Rejected material – 45 To be kept for the period of shelf life, either at the supplier or the operator Must be strictly controlled by proper identification/storage to avoid unauthorized usage The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Stability l A proper shelf life has to be established in order to indicate for the customers, how long the guaranteed specification can be expected. – – 46 Stability has to be defined, preferably by measurements, or if applicable (e. g. in case of premixtures) theoretical considerations can be used Indications about storage conditions which ensure specifications throughout the shelf life period must be given The Expert Group of FAMI-QS June 2005 (V 3) End
Back Stability l A proper shelf life has to be established in order to indicate for the customers, how long the guaranteed specification can be expected. – – 46 Stability has to be defined, preferably by measurements, or if applicable (e. g. in case of premixtures) theoretical considerations can be used Indications about storage conditions which ensure specifications throughout the shelf life period must be given The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Traceability Principle: ‘Traceability’ means the ability to trace and follow a food, feed, food-producing animal or substance intended to be, or expected to be incorporated into a food or feed, through all stages of production, processing and distribution; products (178/2002/EC) 47 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Traceability Principle: ‘Traceability’ means the ability to trace and follow a food, feed, food-producing animal or substance intended to be, or expected to be incorporated into a food or feed, through all stages of production, processing and distribution; products (178/2002/EC) 47 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Traceability Incoming materials l l 48 Maintain a register, that contains the names and addresses of manufacturers of incoming materials, additives or of intermediaries Confirmation by the supplier to have a traceability system established or verification through an audit (future requirement) The Expert Group of FAMI-QS June 2005 (V 3) End
Back Traceability Incoming materials l l 48 Maintain a register, that contains the names and addresses of manufacturers of incoming materials, additives or of intermediaries Confirmation by the supplier to have a traceability system established or verification through an audit (future requirement) The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Traceability Product traceability during manufacturing l l 49 Identify and record the product by suitable means throughout product realisation The nature and quantity of the additives and premixes produced, the respective dates of manufacture and, where appropriate, the number of the batch or of the specific portion of production in the case of continuous manufacture The Expert Group of FAMI-QS June 2005 (V 3) End
Back Traceability Product traceability during manufacturing l l 49 Identify and record the product by suitable means throughout product realisation The nature and quantity of the additives and premixes produced, the respective dates of manufacture and, where appropriate, the number of the batch or of the specific portion of production in the case of continuous manufacture The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Traceability Deliveries l l 50 the name and addresses of the intermediaries or manufacturers or users to whom the additives or premixes have been delivered must be recorded Indicate the nature and quantity of the additive or premix delivered and, where appropriate, the number of the batch or of the specific portion of production in the case of continuous manufacture The Expert Group of FAMI-QS June 2005 (V 3) End
Back Traceability Deliveries l l 50 the name and addresses of the intermediaries or manufacturers or users to whom the additives or premixes have been delivered must be recorded Indicate the nature and quantity of the additive or premix delivered and, where appropriate, the number of the batch or of the specific portion of production in the case of continuous manufacture The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l Products – – – l – Specification Suppliers Incoming Materials Stability Traceability – – – Manufacturing – – – 51 – Building and Equipment Work Environment Finished Goods The Expert Group of FAMI-QS l l Packaging & labelling Quality Control Verification Blending Warehousing & Storage Transport Cleaning HACCP Non Conformances June 2005 (V 3) End
Back The Code l Products – – – l – Specification Suppliers Incoming Materials Stability Traceability – – – Manufacturing – – – 51 – Building and Equipment Work Environment Finished Goods The Expert Group of FAMI-QS l l Packaging & labelling Quality Control Verification Blending Warehousing & Storage Transport Cleaning HACCP Non Conformances June 2005 (V 3) End
 Back Suitable Building & Equipment Principle: Buildings, facilities and equipment should be maintained in a good state of repair and should be of suitable size, construction, and location to facilitate cleaning, maintenance, and properation ý ý Adequate space for orderly placement of equipment and materials to prevent mix-ups ý Adequate workspace - minimizing mix-ups, avoiding cross-contamination ý 52 Adequate building/equipment - open or closed, different level of protection, handling of product at the same time, minimizing crosscontamination, minimizing the risk of errors, hygienic design of plants and equipment permit effective cleaning and maintenance, material of construction, capable of operation Defined areas for the following activities: storage, production, control and laboratory operations (separate rooms for different kind of tests, microbiological tests, powder handling, weighing rooms) The Expert Group of FAMI-QS June 2005 (V 3) End
Back Suitable Building & Equipment Principle: Buildings, facilities and equipment should be maintained in a good state of repair and should be of suitable size, construction, and location to facilitate cleaning, maintenance, and properation ý ý Adequate space for orderly placement of equipment and materials to prevent mix-ups ý Adequate workspace - minimizing mix-ups, avoiding cross-contamination ý 52 Adequate building/equipment - open or closed, different level of protection, handling of product at the same time, minimizing crosscontamination, minimizing the risk of errors, hygienic design of plants and equipment permit effective cleaning and maintenance, material of construction, capable of operation Defined areas for the following activities: storage, production, control and laboratory operations (separate rooms for different kind of tests, microbiological tests, powder handling, weighing rooms) The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Suitable Building & Equipment cont’d ý ý 53 Adequate utilities - direct contact with the product i. e. compressed air (oil free), water quality should be adequate Waste material - clearly identified and disposed in accordance with local regulations The Expert Group of FAMI-QS June 2005 (V 3) End
Back Suitable Building & Equipment cont’d ý ý 53 Adequate utilities - direct contact with the product i. e. compressed air (oil free), water quality should be adequate Waste material - clearly identified and disposed in accordance with local regulations The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Work Environment ý ý adequate control of humidity ý adequate control of temperature ý adequate lighting - to enable all operations to be carried out accurately and safely, according to National regulations (Health & Safety) ý 54 adequate ventilation and exhaust systems - prevent contamination of the product, attention to re-circulating systems adequate clothing requirements should apply to all personnel, facilities for changing cloth, showering (special hygiene practice should be applied) The Expert Group of FAMI-QS June 2005 (V 3) End
Back Work Environment ý ý adequate control of humidity ý adequate control of temperature ý adequate lighting - to enable all operations to be carried out accurately and safely, according to National regulations (Health & Safety) ý 54 adequate ventilation and exhaust systems - prevent contamination of the product, attention to re-circulating systems adequate clothing requirements should apply to all personnel, facilities for changing cloth, showering (special hygiene practice should be applied) The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Production of Finished Goods ý ý Work instructions - product specific procedures for all stages of production ensuring re-produceability, mastering the critical points, details of necessary precaution to be taken to avoid cross -contamination and errors, sampling instructions, weights and measures of components used, persons performing the operation ý 55 Finished product - written specification, unique name or code to avoid mix-ups Records - confirmation that procedures are followed, identify deviations The Expert Group of FAMI-QS June 2005 (V 3) End
Back Production of Finished Goods ý ý Work instructions - product specific procedures for all stages of production ensuring re-produceability, mastering the critical points, details of necessary precaution to be taken to avoid cross -contamination and errors, sampling instructions, weights and measures of components used, persons performing the operation ý 55 Finished product - written specification, unique name or code to avoid mix-ups Records - confirmation that procedures are followed, identify deviations The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Production - Packaging & Labeling Principle: Where products are packaged, care must be taken to avoid contamination during the packaging process, and to ensure that packaged products are correctly identified and labelled in compliance with the provisions of feed regulations in force. Packaging must be appropriate to product type and to maintain contents for their intended shelf life ý ý Each package must be labelled - name of the product, batch/lot number, shelf life (traceability) ý Label control - identification, examination of identity and conformity with a master, release, reconciliation (numbers of labels printed vs. number of labels used) ý 56 Packaging material is appropriate - no detrimental effect on the product (primary packaging material) One labeling operation at the same time - to avoid mix-ups The Expert Group of FAMI-QS June 2005 (V 3) End
Back Production - Packaging & Labeling Principle: Where products are packaged, care must be taken to avoid contamination during the packaging process, and to ensure that packaged products are correctly identified and labelled in compliance with the provisions of feed regulations in force. Packaging must be appropriate to product type and to maintain contents for their intended shelf life ý ý Each package must be labelled - name of the product, batch/lot number, shelf life (traceability) ý Label control - identification, examination of identity and conformity with a master, release, reconciliation (numbers of labels printed vs. number of labels used) ý 56 Packaging material is appropriate - no detrimental effect on the product (primary packaging material) One labeling operation at the same time - to avoid mix-ups The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Production - Quality Controls Principle: Quality control ensures that the necessary tests are actually carried out and that finished products are not released for use until their quality has been judged to be satisfactory ý ý In-Process testing - monitoring the process ý Analytical methods and test procedures - level of detail that analyst are able to understand how to proceed ý 57 All finished product should be inspected prior to dispatch - ensure that the customer receives material which is released; product is meeting written specifications Measuring and test equipment - properly calibrated and maintained The Expert Group of FAMI-QS June 2005 (V 3) End
Back Production - Quality Controls Principle: Quality control ensures that the necessary tests are actually carried out and that finished products are not released for use until their quality has been judged to be satisfactory ý ý In-Process testing - monitoring the process ý Analytical methods and test procedures - level of detail that analyst are able to understand how to proceed ý 57 All finished product should be inspected prior to dispatch - ensure that the customer receives material which is released; product is meeting written specifications Measuring and test equipment - properly calibrated and maintained The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Production - Verification of the Process Principle: Any process where the resulting output cannot be controlled by subsequent monitoring or measurement. This includes any processes where deficiencies become apparent only after the product is in use or has been delivered ý Verification shall demonstrate the ability of a process to achieve planned results - focus on deemed critical operations i. e. blending (homogeneity), to built in quality into the process ý The operator shall establish arrangements for these processes including, ý ý ý 58 Defined criteria for review and approval of the manufacturing processes - monitoring of CCPs Approval of equipment - equipment including instrumentation is qualified (qualified operating range) Qualification of personnel - proper training of personnel, adequate number, appropriate education, experience Use of specific methods and procedures - additional testing Requirements for records - record keeping The Expert Group of FAMI-QS June 2005 (V 3) End
Back Production - Verification of the Process Principle: Any process where the resulting output cannot be controlled by subsequent monitoring or measurement. This includes any processes where deficiencies become apparent only after the product is in use or has been delivered ý Verification shall demonstrate the ability of a process to achieve planned results - focus on deemed critical operations i. e. blending (homogeneity), to built in quality into the process ý The operator shall establish arrangements for these processes including, ý ý ý 58 Defined criteria for review and approval of the manufacturing processes - monitoring of CCPs Approval of equipment - equipment including instrumentation is qualified (qualified operating range) Qualification of personnel - proper training of personnel, adequate number, appropriate education, experience Use of specific methods and procedures - additional testing Requirements for records - record keeping The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Production - Blending of Finished Products Principle: Manufacturer should not rely on blending - mixing adultered feed with good feed to bring the contaminant to a suitable low level - instead exercising maximum effort to avoid contamination in the first place ý ý 59 Before starting a blending operation an assessment of the risks has to be done Blending process should be controlled to ensure homogeneity with respect to critical product attributes The Expert Group of FAMI-QS June 2005 (V 3) End
Back Production - Blending of Finished Products Principle: Manufacturer should not rely on blending - mixing adultered feed with good feed to bring the contaminant to a suitable low level - instead exercising maximum effort to avoid contamination in the first place ý ý 59 Before starting a blending operation an assessment of the risks has to be done Blending process should be controlled to ensure homogeneity with respect to critical product attributes The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Warehousing – Storage Principle: The finished product, intermediates and raw materials should be handled and stored under appropriate conditions so that their quality and purity are not affected ý ý A stock rotation system should be in place - FIFO ý Materials should be stored in a manner to prevent degradation, contamination, and cross-contamination ý 60 Storage areas should be of sufficient capacity - to allow orderly storage of various categories: raw materials, packaging materials, intermediates, finished product, products in quarantine, released, rejected, returned or recalled products, hazardous/unstable chemical Storage areas should be clean, dry and maintained - to minimise the risk of damage of packaging and spillage of material The Expert Group of FAMI-QS June 2005 (V 3) End
Back Warehousing – Storage Principle: The finished product, intermediates and raw materials should be handled and stored under appropriate conditions so that their quality and purity are not affected ý ý A stock rotation system should be in place - FIFO ý Materials should be stored in a manner to prevent degradation, contamination, and cross-contamination ý 60 Storage areas should be of sufficient capacity - to allow orderly storage of various categories: raw materials, packaging materials, intermediates, finished product, products in quarantine, released, rejected, returned or recalled products, hazardous/unstable chemical Storage areas should be clean, dry and maintained - to minimise the risk of damage of packaging and spillage of material The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Warehousing - Storage cont’d ý ý Storage conditions based on label claim - where special storage conditions are required on the label (T, % r. H) monitoring and recording is required, retention time for records is at least shelf life ý All returned goods should be placed in quarantine and returned to saleable stock only after this has been approved by nominated person following a satisfactory quality re-evaluation ý 61 All containers should be clearly marked with unique identifier(s) which reflect at least name of the material, lot number, shelf life/best use before date, storage conditions Pallets must be serviceable, e. g. depending on risk considerations clean and dry. All pallets, which are returned, must be inspected and if necessary cleaned before re-use The Expert Group of FAMI-QS June 2005 (V 3) End
Back Warehousing - Storage cont’d ý ý Storage conditions based on label claim - where special storage conditions are required on the label (T, % r. H) monitoring and recording is required, retention time for records is at least shelf life ý All returned goods should be placed in quarantine and returned to saleable stock only after this has been approved by nominated person following a satisfactory quality re-evaluation ý 61 All containers should be clearly marked with unique identifier(s) which reflect at least name of the material, lot number, shelf life/best use before date, storage conditions Pallets must be serviceable, e. g. depending on risk considerations clean and dry. All pallets, which are returned, must be inspected and if necessary cleaned before re-use The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Warehousing - Pest Control Principle: Buildings should be free of infestation by rodents, birds, insects and other vermin ý ý ý 62 There should be a written program - location, frequency, agents Pest-control agents - safe, approved, MSDS available to the operator Record keeping - date, name, results, Included into HACCP plan - risk of cross-contamination due to infestation or use of pesticides If done by an outside contractor all requirements must be laid down in a contract The Expert Group of FAMI-QS June 2005 (V 3) End
Back Warehousing - Pest Control Principle: Buildings should be free of infestation by rodents, birds, insects and other vermin ý ý ý 62 There should be a written program - location, frequency, agents Pest-control agents - safe, approved, MSDS available to the operator Record keeping - date, name, results, Included into HACCP plan - risk of cross-contamination due to infestation or use of pesticides If done by an outside contractor all requirements must be laid down in a contract The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Transport Principle: Products should be transported in such a way that their integrity is not impaired and that storage conditions are maintained ý ý Special attention should be paid for vehicle hygiene and cleanliness, correct loading and avoidance of contamination and cross-contamination which must be verified by visual inspection prior to loading. ý 63 The outside container should offer adequate protection from all external influences and should be clearly labeled Dispatch records should be established - date, customer’s name and address, name of the product, lot number, quantity, transport and storage conditions The Expert Group of FAMI-QS June 2005 (V 3) End
Back Transport Principle: Products should be transported in such a way that their integrity is not impaired and that storage conditions are maintained ý ý Special attention should be paid for vehicle hygiene and cleanliness, correct loading and avoidance of contamination and cross-contamination which must be verified by visual inspection prior to loading. ý 63 The outside container should offer adequate protection from all external influences and should be clearly labeled Dispatch records should be established - date, customer’s name and address, name of the product, lot number, quantity, transport and storage conditions The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Transport cont’d ý ý 64 For bulk deliveries the transportation agent shall provide information about the two previous loads In case the two previous loads consisted of products which may compromise the safety of the final product or are not allowed to be used in feedingstuff according to existing regulations - cleaning certificate, information about the means of cleaning and drying and guarantee that a clean, empty, dry and odourless cargo compartment and discharge equipment is made available The Expert Group of FAMI-QS June 2005 (V 3) End
Back Transport cont’d ý ý 64 For bulk deliveries the transportation agent shall provide information about the two previous loads In case the two previous loads consisted of products which may compromise the safety of the final product or are not allowed to be used in feedingstuff according to existing regulations - cleaning certificate, information about the means of cleaning and drying and guarantee that a clean, empty, dry and odourless cargo compartment and discharge equipment is made available The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Cleaning ý ý Cleaning activities should be documented. This should beaddressed as part of the Hazard analysis ý Containers and equipment used for internal transport, storage, conveying handling and weighing shall be kept clean ý Adequate resources to ensure good state of cleanliness ý 65 A written cleaning program (building, warehouse, equipment, multi use packaging material) available defining responsibility, schedule, methods and materials; for equipment: instruction for dissembling and reassembling, protection after cleaning If there is a risk - potential for carry-over should be determined The Expert Group of FAMI-QS June 2005 (V 3) End
Back Cleaning ý ý Cleaning activities should be documented. This should beaddressed as part of the Hazard analysis ý Containers and equipment used for internal transport, storage, conveying handling and weighing shall be kept clean ý Adequate resources to ensure good state of cleanliness ý 65 A written cleaning program (building, warehouse, equipment, multi use packaging material) available defining responsibility, schedule, methods and materials; for equipment: instruction for dissembling and reassembling, protection after cleaning If there is a risk - potential for carry-over should be determined The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l l Manufacturing l HACCP l 66 Products Non Conformances The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Code l l Manufacturing l HACCP l 66 Products Non Conformances The Expert Group of FAMI-QS June 2005 (V 3) End
 Back HACCP Principle: Reference is made to the guideline published in Codex Alimentarius l l 67 www. codexalimentarius. net CAC/CRP 1 General principles on food hygiene Annex on Hazard Analysis and Critical Control Point and Guidelines for its Application The Expert Group of FAMI-QS June 2005 (V 3) End
Back HACCP Principle: Reference is made to the guideline published in Codex Alimentarius l l 67 www. codexalimentarius. net CAC/CRP 1 General principles on food hygiene Annex on Hazard Analysis and Critical Control Point and Guidelines for its Application The Expert Group of FAMI-QS June 2005 (V 3) End
 Back HACCP Principle: Prior to application of HACCP to any operator in the feed chain that operator should be operating according to a quality system (prerequisite program) l l 68 It is important that the HACCP plan is integrated with the operator’s quality system, prerequisite system Hazard analysis should be conducted after all prerequisite programs are designed and implemented The Expert Group of FAMI-QS June 2005 (V 3) End
Back HACCP Principle: Prior to application of HACCP to any operator in the feed chain that operator should be operating according to a quality system (prerequisite program) l l 68 It is important that the HACCP plan is integrated with the operator’s quality system, prerequisite system Hazard analysis should be conducted after all prerequisite programs are designed and implemented The Expert Group of FAMI-QS June 2005 (V 3) End
 Back HACCP l Two principles – specific to one product and defined process line – general to a group of products following the same unit operations l 69 unit operations are individual stages of manufacturing process, from incoming ingredients to packaging the final product, e. g. drying, extruding and blending, etc. The Expert Group of FAMI-QS June 2005 (V 3) End
Back HACCP l Two principles – specific to one product and defined process line – general to a group of products following the same unit operations l 69 unit operations are individual stages of manufacturing process, from incoming ingredients to packaging the final product, e. g. drying, extruding and blending, etc. The Expert Group of FAMI-QS June 2005 (V 3) End
 Back HACCP Plan Important: The fundamental principles have to be addressed l l l l 70 Conduct a hazard analysis – on each product – on a group of products Determine CCP’s Establish critical limits Establish monitoring procedures Establish corrective actions Establish verification procedures Establish record-keeping and documentation procedures The Expert Group of FAMI-QS June 2005 (V 3) End
Back HACCP Plan Important: The fundamental principles have to be addressed l l l l 70 Conduct a hazard analysis – on each product – on a group of products Determine CCP’s Establish critical limits Establish monitoring procedures Establish corrective actions Establish verification procedures Establish record-keeping and documentation procedures The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Hazard Analysis The process of collecting and evaluating information on hazards and conditions leading to their presence to decide which are significant for feed/food safety and therefore should be addressed in the HACCP plan l Conditions or incidents most likely to happen, because – – l l l 71 it historically has occurred there is a reasonable possibility that it might occur in the absence of such control Identify the true feed safety hazards Develop a list of hazards which are of such significance that they are reasonably likely to cause injury or illness if not effectively controlled Each identified potential hazard should be evaluated, giving consideration to its severity and likely occurrence The Expert Group of FAMI-QS June 2005 (V 3) End
Back Hazard Analysis The process of collecting and evaluating information on hazards and conditions leading to their presence to decide which are significant for feed/food safety and therefore should be addressed in the HACCP plan l Conditions or incidents most likely to happen, because – – l l l 71 it historically has occurred there is a reasonable possibility that it might occur in the absence of such control Identify the true feed safety hazards Develop a list of hazards which are of such significance that they are reasonably likely to cause injury or illness if not effectively controlled Each identified potential hazard should be evaluated, giving consideration to its severity and likely occurrence The Expert Group of FAMI-QS June 2005 (V 3) End
 Back CCP definition CCP: Critical Control Point is a point, step or procedure at which control can be applied and a safety hazard can be prevented, eliminated or reduced to acceptable levels. l l 72 Measurement giving a ”GO” or ”NO-GO” situation Identify the point/points in the process where the identified hazards best can be controlled The Expert Group of FAMI-QS June 2005 (V 3) End
Back CCP definition CCP: Critical Control Point is a point, step or procedure at which control can be applied and a safety hazard can be prevented, eliminated or reduced to acceptable levels. l l 72 Measurement giving a ”GO” or ”NO-GO” situation Identify the point/points in the process where the identified hazards best can be controlled The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Hazard analysis – Ex. 1 73 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Hazard analysis – Ex. 1 73 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Hazard analysis – EX. 2 74 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Hazard analysis – EX. 2 74 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Hazard analysis – EX. 2 cont. 75 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Hazard analysis – EX. 2 cont. 75 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Hazard analysis – EX. 3 76 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Hazard analysis – EX. 3 76 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back HACCP Summary l l l 77 HACCP is the best way to control safety hazards that are likely to occur Every HACCP plan can be different and still be effective Allow flexibility in designing the optimal HACCP system to control safety hazards The Expert Group of FAMI-QS June 2005 (V 3) End
Back HACCP Summary l l l 77 HACCP is the best way to control safety hazards that are likely to occur Every HACCP plan can be different and still be effective Allow flexibility in designing the optimal HACCP system to control safety hazards The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l l Manufacturing l HACCP l 78 Products Non Conformances – Products – Audits The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Code l l Manufacturing l HACCP l 78 Products Non Conformances – Products – Audits The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l l Manufacturing l HACCP l 79 Products Non Conformances – Products – Audits The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Code l l Manufacturing l HACCP l 79 Products Non Conformances – Products – Audits The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Non-conforming products Principle: The operator shall ensure that products which do not conform to expected requirements are identified and controlled to prevent its unintended use or delivery. – General requirements l Inspection and release of product (internal) l Ensure no mix-up with approved batches 80 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Non-conforming products Principle: The operator shall ensure that products which do not conform to expected requirements are identified and controlled to prevent its unintended use or delivery. – General requirements l Inspection and release of product (internal) l Ensure no mix-up with approved batches 80 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Non-conforming products Principle: When non-conforming products are detected after delivery or use has started, the operator shall take action appropriate to the effects, or potential effects, of the non-conformance Complaint handling system – Recall system – 81 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Non-conforming products Principle: When non-conforming products are detected after delivery or use has started, the operator shall take action appropriate to the effects, or potential effects, of the non-conformance Complaint handling system – Recall system – 81 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Compliant handling system l Formalized system – – – Definition of responsibilities Traceability Evaluation of cause of complaint l l – – – 82 Categories (product, packaging or transport) Seriousness (health & safety) Feed-back to customer Measurement & recurrence Corrective actions The Expert Group of FAMI-QS June 2005 (V 3) End
Back Compliant handling system l Formalized system – – – Definition of responsibilities Traceability Evaluation of cause of complaint l l – – – 82 Categories (product, packaging or transport) Seriousness (health & safety) Feed-back to customer Measurement & recurrence Corrective actions The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Recall l l 83 Formal procedure – Responsibility – Flow description, competencies, step-by-step – Documentation & archiving Implementation of mock recalls & documentation Evaluation & corrective actions Information to authorities The Expert Group of FAMI-QS June 2005 (V 3) End
Back Recall l l 83 Formal procedure – Responsibility – Flow description, competencies, step-by-step – Documentation & archiving Implementation of mock recalls & documentation Evaluation & corrective actions Information to authorities The Expert Group of FAMI-QS June 2005 (V 3) End
 Back The Code l l Manufacturing l HACCP l 84 Products Non conformances – Products – Audits The Expert Group of FAMI-QS June 2005 (V 3) End
Back The Code l l Manufacturing l HACCP l 84 Products Non conformances – Products – Audits The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Non-conformance Principle: A non-conformance, deviation or deficiency to Co. P noted by an inspector during an inspection of an establishment that is conformed in writing to the company in the exit notice. l l 85 Non-conformances are classified according to the degree of severeness: Critical Major Minor The risk assigned will be in relation to the nature of the nonconformances as well as the number of occurrences The Expert Group of FAMI-QS June 2005 (V 3) End
Back Non-conformance Principle: A non-conformance, deviation or deficiency to Co. P noted by an inspector during an inspection of an establishment that is conformed in writing to the company in the exit notice. l l 85 Non-conformances are classified according to the degree of severeness: Critical Major Minor The risk assigned will be in relation to the nature of the nonconformances as well as the number of occurrences The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Critical non-conformance Principle: Observation describing a situation that is likely to result in a noncompliant product or a situation that may result in an immediate or latent safety risk or any observation that involves fraud, misrepresentation or falsification of data l 86 When the auditor observes a regulatory violation or a feed safety failure which may require that the operator: – Immediately interrupts production – Holds products in quarantine – Discontinues shipping to customers – Recalls of products The Expert Group of FAMI-QS June 2005 (V 3) End
Back Critical non-conformance Principle: Observation describing a situation that is likely to result in a noncompliant product or a situation that may result in an immediate or latent safety risk or any observation that involves fraud, misrepresentation or falsification of data l 86 When the auditor observes a regulatory violation or a feed safety failure which may require that the operator: – Immediately interrupts production – Holds products in quarantine – Discontinues shipping to customers – Recalls of products The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Critical non-conformance Example 1: Intentional violations of European and/or national legislation, e. g. l No 87 traceability system in place The Expert Group of FAMI-QS June 2005 (V 3) End
Back Critical non-conformance Example 1: Intentional violations of European and/or national legislation, e. g. l No 87 traceability system in place The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Critical non-conformance Example 2: Direct observation of products being produced, packed or held in a manner which poses a clear threat to animal and/or human health, e. g. l safety of raw material/product cannot be assured due to cross contamination, infestation, or unsanitary conditions l use of bovine material in products used for cattle 88 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Critical non-conformance Example 2: Direct observation of products being produced, packed or held in a manner which poses a clear threat to animal and/or human health, e. g. l safety of raw material/product cannot be assured due to cross contamination, infestation, or unsanitary conditions l use of bovine material in products used for cattle 88 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Critical non-conformance Example 3: Discovery of records showing that products are being or have been produced in a manner which poses a clear threat to animal and/or human health, e. g. l 89 Recorded mixing time is below minimum range and thereby violating product safety The Expert Group of FAMI-QS June 2005 (V 3) End
Back Critical non-conformance Example 3: Discovery of records showing that products are being or have been produced in a manner which poses a clear threat to animal and/or human health, e. g. l 89 Recorded mixing time is below minimum range and thereby violating product safety The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Critical non-conformance Example 4: The product is adulterated such that it contains an added poisonous or deleterious substance, e. g. l pesticides for pest control are being used inconsistently with the labelled directions 90 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Critical non-conformance Example 4: The product is adulterated such that it contains an added poisonous or deleterious substance, e. g. l pesticides for pest control are being used inconsistently with the labelled directions 90 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Principle: A complete failure to implement a requirement of the FAMIQS Code of Practice l l 91 Observation that may result in the production of a product not consistently meeting its market authorization A minor non-conformance from a previous audit which has not been corrected The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Principle: A complete failure to implement a requirement of the FAMIQS Code of Practice l l 91 Observation that may result in the production of a product not consistently meeting its market authorization A minor non-conformance from a previous audit which has not been corrected The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Example 1: – Complete failure to implement a requirement, e. g. : l Some of the HACCP principles are not implemented l Recall procedure is missing or incomplete to a extent where it becomes evident that the recall process is not going to work 92 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Example 1: – Complete failure to implement a requirement, e. g. : l Some of the HACCP principles are not implemented l Recall procedure is missing or incomplete to a extent where it becomes evident that the recall process is not going to work 92 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Example 2: – No calibration program for mechanical, electronic or other critical measuring equipment, or no records maintained – Feed safety programs are deficient in such a manner that they do not comply with the FAMI-QS Code of Practice 93 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Example 2: – No calibration program for mechanical, electronic or other critical measuring equipment, or no records maintained – Feed safety programs are deficient in such a manner that they do not comply with the FAMI-QS Code of Practice 93 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Example 3: - Delegation of responsibilities for persons to insufficiently qualified persons, and no training program is available - Insufficient training for personnel involved in production and QC resulting in related GMP deviations 94 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Example 3: - Delegation of responsibilities for persons to insufficiently qualified persons, and no training program is available - Insufficient training for personnel involved in production and QC resulting in related GMP deviations 94 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Example 4: - No requirement for cleaning in the quality system even though the premises are in an acceptable state of cleanliness 95 The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Example 4: - No requirement for cleaning in the quality system even though the premises are in an acceptable state of cleanliness 95 The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Example 5: - 96 Retained samples not kept for RM or not ensured to be kept by supplier Retained samples not kept for finished products The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Example 5: - 96 Retained samples not kept for RM or not ensured to be kept by supplier Retained samples not kept for finished products The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Major non-conformance Example 6: – 97 An imminent feed/food safety hazard exists, e. g. l birds residing in processing areas or warehouses, l evidence of rodent excreta or gnawing on raw materials or finished products The Expert Group of FAMI-QS June 2005 (V 3) End
Back Major non-conformance Example 6: – 97 An imminent feed/food safety hazard exists, e. g. l birds residing in processing areas or warehouses, l evidence of rodent excreta or gnawing on raw materials or finished products The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Minor non-conformance Principle: A requirement of the FAMI-QS Code of Practice has been addressed, but there is insufficient evidence to demonstrate it has been properly controlled or implemented l 98 Observation that is neither critical nor major but a deviation from the FAMI-QS Code of Practice The Expert Group of FAMI-QS June 2005 (V 3) End
Back Minor non-conformance Principle: A requirement of the FAMI-QS Code of Practice has been addressed, but there is insufficient evidence to demonstrate it has been properly controlled or implemented l 98 Observation that is neither critical nor major but a deviation from the FAMI-QS Code of Practice The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Minor non-conformance, cont. Example 1: – 99 Cleaning is said to be done, but documentation is incomplete The Expert Group of FAMI-QS June 2005 (V 3) End
Back Minor non-conformance, cont. Example 1: – 99 Cleaning is said to be done, but documentation is incomplete The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Minor non-conformance, cont. Example 2: – 100 Incomplete SOPs for handling of materials and products and thereby risk of safety The Expert Group of FAMI-QS June 2005 (V 3) End
Back Minor non-conformance, cont. Example 2: – 100 Incomplete SOPs for handling of materials and products and thereby risk of safety The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Minor non-conformance, cont. Example 3: – 101 Inadequate specifications, and consequently the documentation is incomplete The Expert Group of FAMI-QS June 2005 (V 3) End
Back Minor non-conformance, cont. Example 3: – 101 Inadequate specifications, and consequently the documentation is incomplete The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Minor non-conformance, cont. Example 4: – 102 No organization chart The Expert Group of FAMI-QS June 2005 (V 3) End
Back Minor non-conformance, cont. Example 4: – 102 No organization chart The Expert Group of FAMI-QS June 2005 (V 3) End
 Back Minor non-conformance, cont. Example 5: – 103 Some documents not updated, but the QMS system is working The Expert Group of FAMI-QS June 2005 (V 3) End
Back Minor non-conformance, cont. Example 5: – 103 Some documents not updated, but the QMS system is working The Expert Group of FAMI-QS June 2005 (V 3) End
 Back …the European Quality Standard for Feed Additives and Premixtures Thank you for your attention 104 The Expert Group of FAMI-QS June 2005 (V 3) End
Back …the European Quality Standard for Feed Additives and Premixtures Thank you for your attention 104 The Expert Group of FAMI-QS June 2005 (V 3) End


