ad93a936986900433f303b369f71af1b.ppt
- Количество слайдов: 27
Expression and purification of bacteria membrane proteins for structural studies

To solve a membrane protein structure is very hard for the following reasons: • Hard to over-express large quantity, functional protein; • To get the protein into solution for purification it needs to be solublized with detergent, which might destroy the protein, we need protein that is functional, folded and monodisperse, many detergents need to be screened; • Hard to crystallize: with detergent around the molecule it’s much harder to make contacts for crystal packing; • Crystals often don’t diffract well because of high solvent contents.
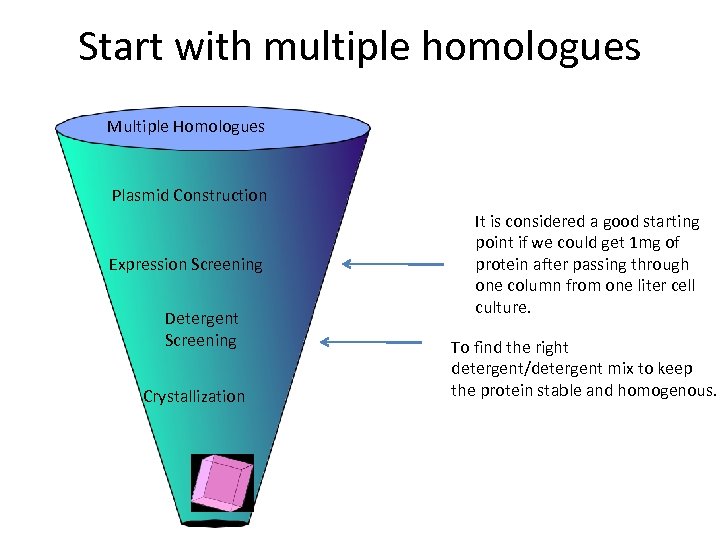
Start with multiple homologues Multiple Homologues Plasmid Construction Expression Screening Detergent Screening Crystallization It is considered a good starting point if we could get 1 mg of protein after passing through one column from one liter cell culture. To find the right detergent/detergent mix to keep the protein stable and homogenous.
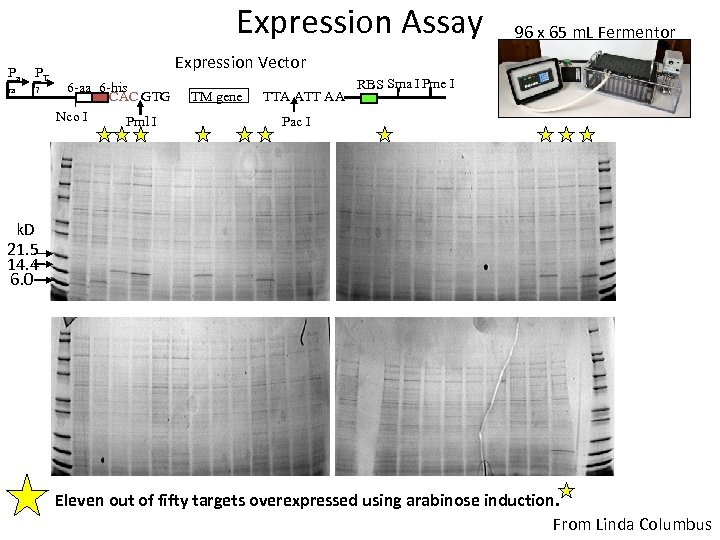
Expression Assay Pa PT ra 7 96 x 65 m. L Fermentor Expression Vector 6 -aa 6 -his CAC GTG Nco I Pml I TM gene TTA ATT AA RBS Sma I Pme I Pac I k. D 21. 5 14. 4 6. 0 Eleven out of fifty targets overexpressed using arabinose induction. From Linda Columbus

Special considerations when overexpressing membrane proteins for crystallographic studies Difficulties Over-expressed protein often toxic to host cells Solutions Choose a tight promoter to prevent leaky expression. Induce at mid-log phase so the cell will keep growing after induction, monitor the cell growth after induction. Problems with misfolded protein Lower growth temperature, in inclusion bodies avoid over-loading translocon and chaparones.

First purification step: isolate the membrane fraction • Disrupt the harvested cells by mechanical force (high pressure) using equipment such as French Press or Xpress; • Remove unbroken cells with a “low speed” centrifugation (10 k xg). This step removes cell debris that are not membrane, as well as some cell organelles such as inclusion bodies; • Collect the membrane fraction by centrifuge the supernatant from the last step with a “high speed” ultra-centrifugation (35 -40 k xg). This step removes all the soluble proteins.

Properties of detergent molecules detergent = very expensive soap From Anatrace. com

Critical micelle concentration (CMC) From Anatrace. com
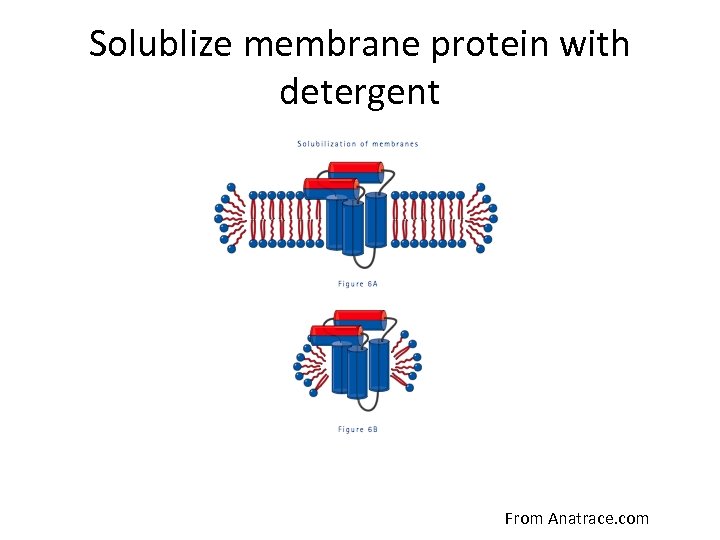
Solublize membrane protein with detergent From Anatrace. com
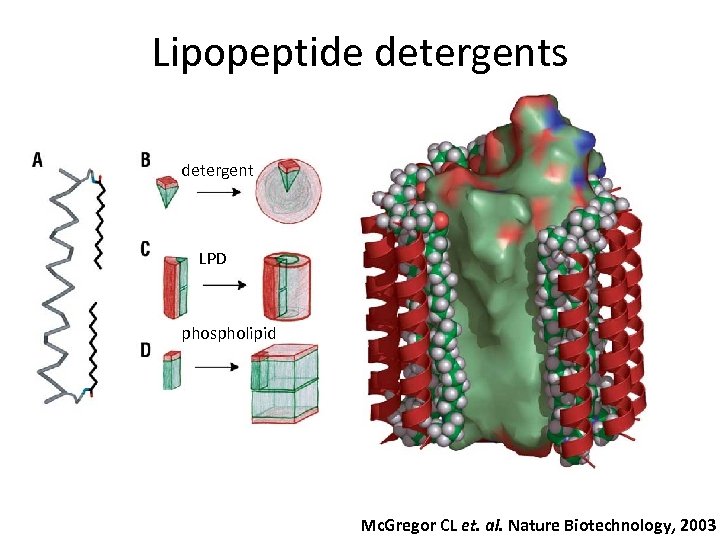
Lipopeptide detergents detergent LPD phospholipid Mc. Gregor CL et. al. Nature Biotechnology, 2003

Special considerations when purifying membrane proteins Difficulties Solutions Extracting protein from its lipid bilayer often result in unstable/inactive proteins Test a few detergent chosen by their reported success and price. Consider detergent exchange. The amount of de-lipidation is often Keep in mind every column the protein important for the protein’s stability and passes is a de-lipidation process as well. homogeneity Sometimes it’s important to add back lipid to keep the protein happy. Because detergent forms micelles, it often concentrates with the protein sample, when the concentration is too high, the empty detergent micelles pack into crystals and making it impossible to get well-diffracting protein crystals. Use 2 -3 times CMC in all the buffers used for purification; control the protein : detergent ratio in the last purification step; Dialyze the sample to lower detergent concentration if necessary.
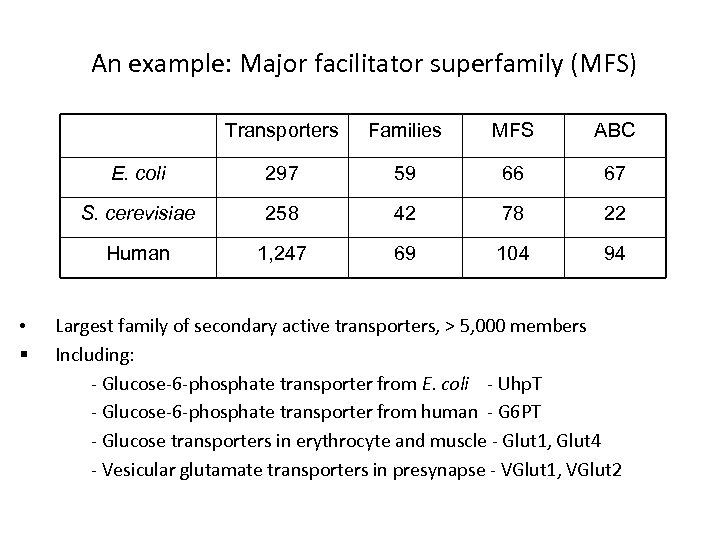
An example: Major facilitator superfamily (MFS) Transporters MFS ABC E. coli 297 59 66 67 S. cerevisiae 258 42 78 22 Human • § Families 1, 247 69 104 94 Largest family of secondary active transporters, > 5, 000 members Including: - Glucose-6 -phosphate transporter from E. coli - Uhp. T - Glucose-6 -phosphate transporter from human - G 6 PT - Glucose transporters in erythrocyte and muscle - Glut 1, Glut 4 - Vesicular glutamate transporters in presynapse - VGlut 1, VGlut 2
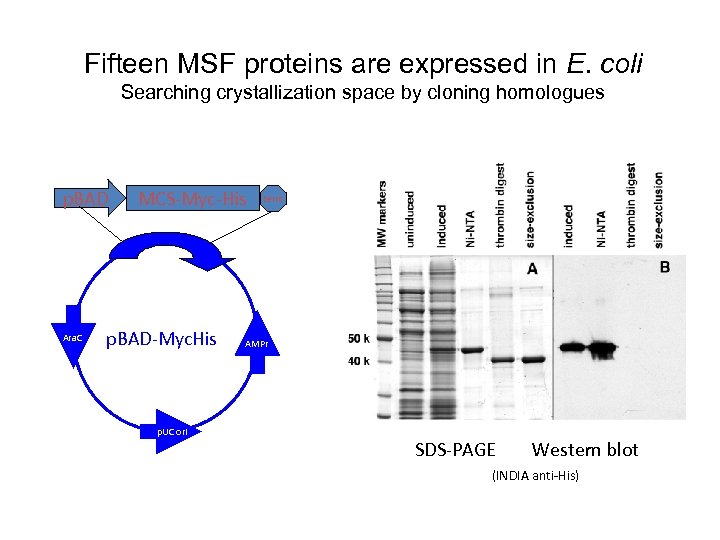
Fifteen MSF proteins are expressed in E. coli Searching crystallization space by cloning homologues p. BAD Ara. C MCS-Myc-His p. BAD-Myc. His p. UC ori term AMPr SDS-PAGE Western blot (INDIA anti-His)
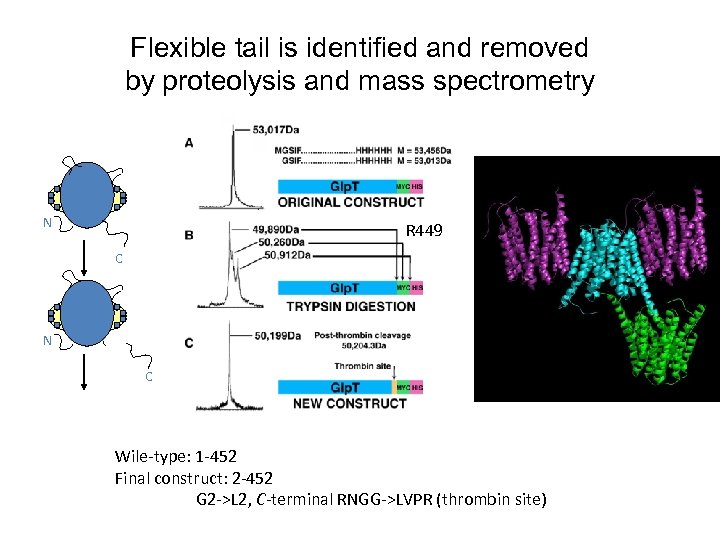
Flexible tail is identified and removed by proteolysis and mass spectrometry N R 449 C N C Wile-type: 1 -452 Final construct: 2 -452 G 2 ->L 2, C-terminal RNGG->LVPR (thrombin site)
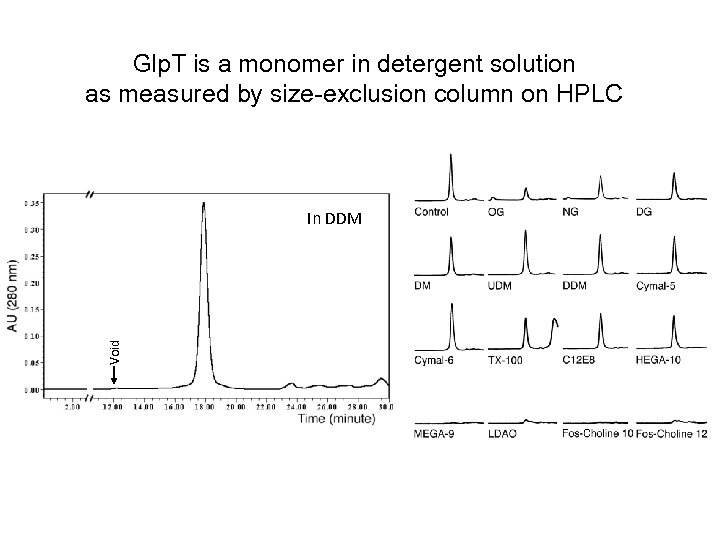
Glp. T is a monomer in detergent solution as measured by size-exclusion column on HPLC Void In DDM

Crystallization Commercially-available MP screens • Memb. Fac (Hampton) • Mem. Start & Memb. Sys, Mem. Gold (Molecular Dimensions) • JBScreen Membrane 1 -3 (Jena Biosciences) • The Mb. Class (Nextal) Statistics show most membrane protein crystalizes in PEG based Conditions, a through PEG/p. H screen should be performed as well.

Web resources Membrane protein structure databases: Topology database http: //blanco. biomol. uci. edu/mptopo/ Known structures http: //blanco. biomol. uci. edu/Membrane_Proteins_xtal. html Known structures with statistics of crystallization conditions http: //www. mpibpfrankfurt. mpg. de/michel/public/memprotstruct. html PDB database for membrane proteins http: //pdbtm. enzim. hu/
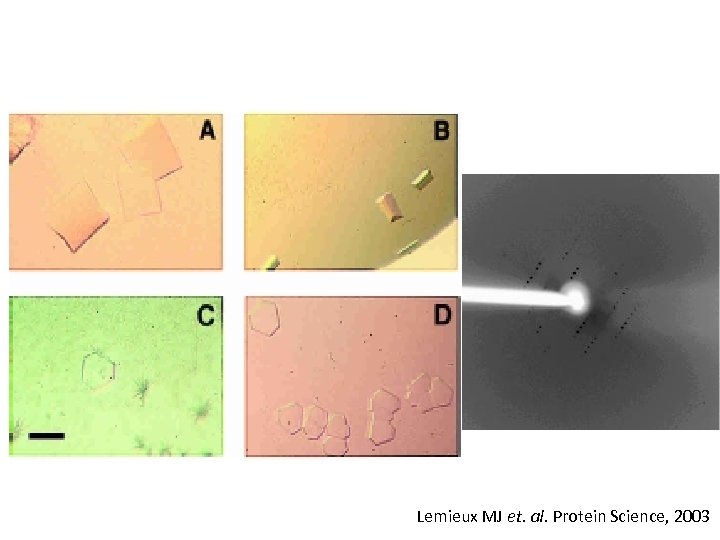
Lemieux MJ et. al. Protein Science, 2003
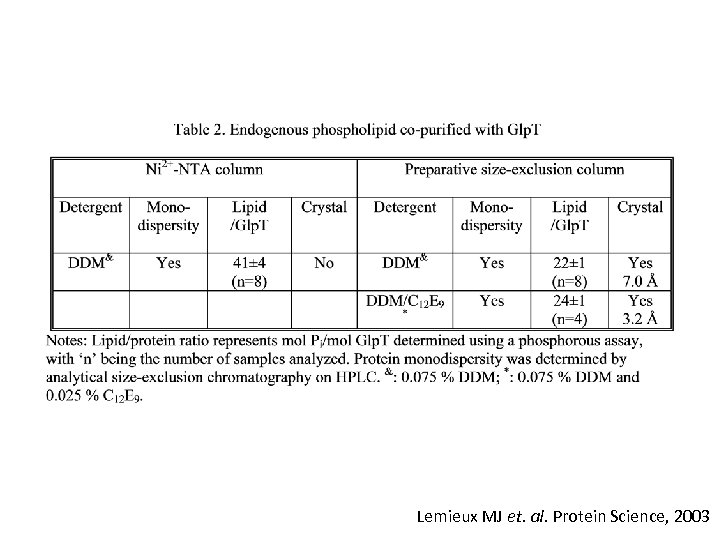
Lemieux MJ et. al. Protein Science, 2003

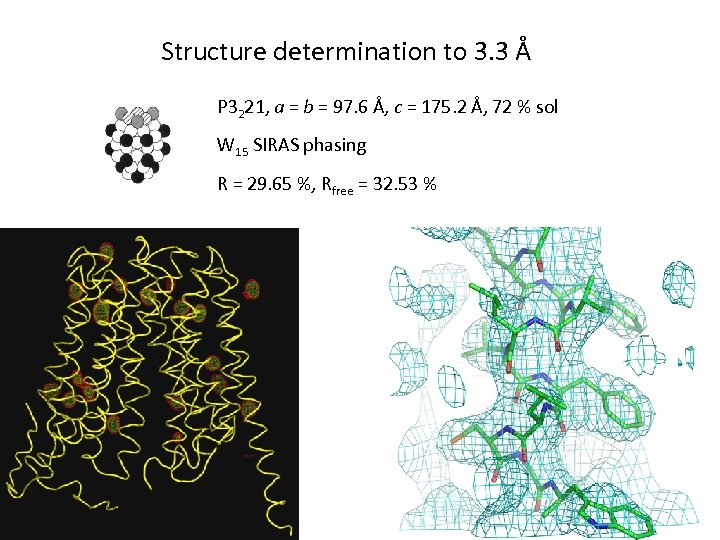
Structure determination to 3. 3 Å P 3221, a = b = 97. 6 Å, c = 175. 2 Å, 72 % sol W 15 SIRAS phasing R = 29. 65 %, Rfree = 32. 53 %

Another example: bacteria amino acid transporter It took 3 years before a well-behaving target was identified in the pipeline: Leucine transporter from thermophilic bacterium Aquifex aeolicus
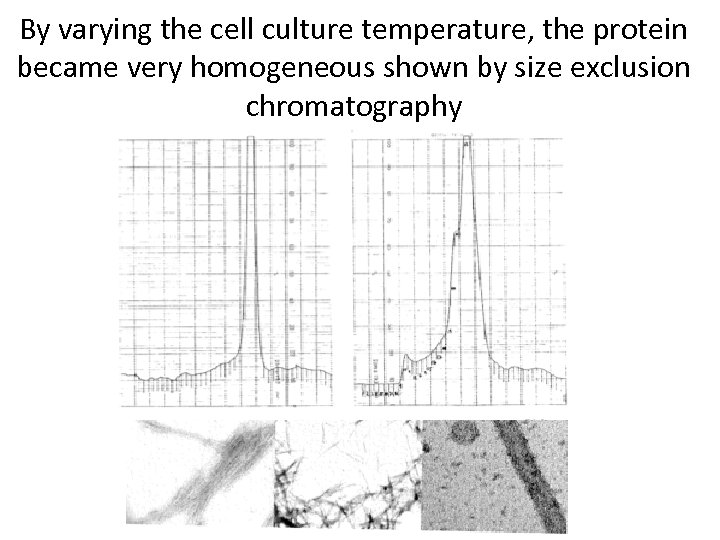
By varying the cell culture temperature, the protein became very homogeneous shown by size exclusion chromatography
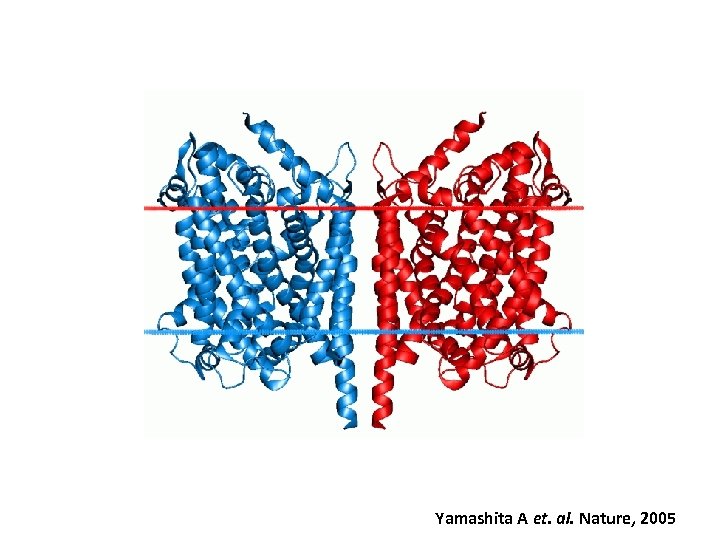
Yamashita A et. al. Nature, 2005

It often takes a long time before a suitable target is identified, Can we speed up this process?
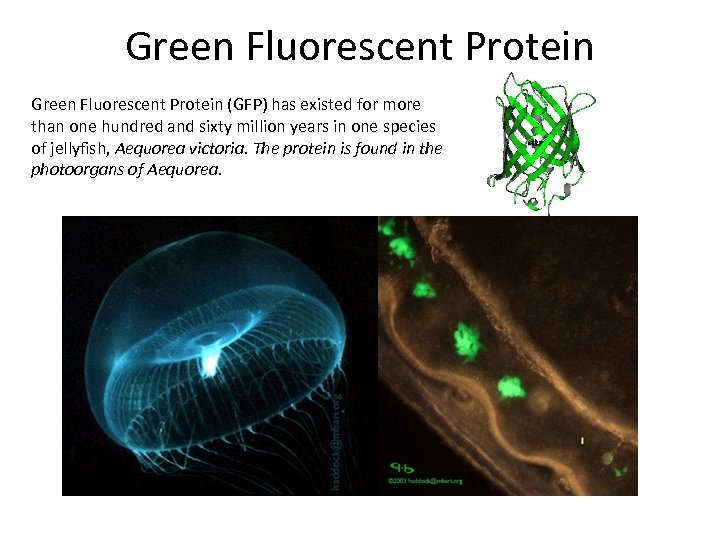
Green Fluorescent Protein (GFP) has existed for more than one hundred and sixty million years in one species of jellyfish, Aequorea victoria. The protein is found in the photoorgans of Aequorea.
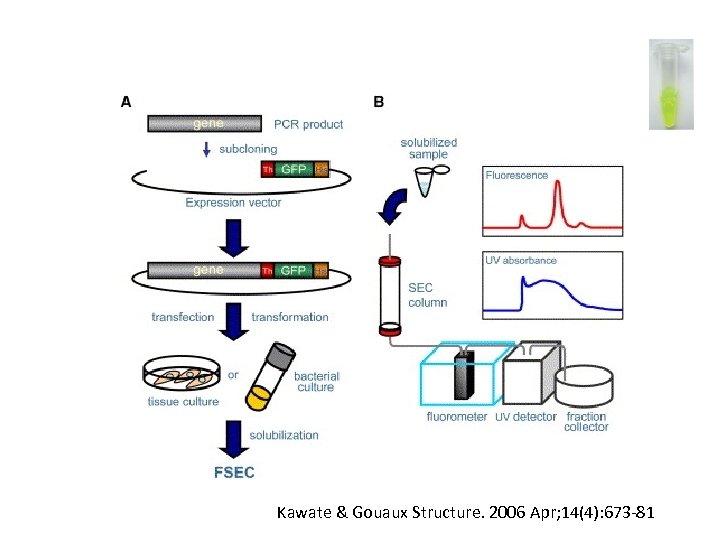
Kawate & Gouaux Structure. 2006 Apr; 14(4): 673 -81
ad93a936986900433f303b369f71af1b.ppt