3d2f8b968b3816da642a44df688c2238.ppt
- Количество слайдов: 98
 Evolution of Integrated Surveillance in the United States Scott J. N. Mc. Nabb, Ph. D. , M. S. Ruth Ann Jajosky, D. M. D. , M. P. H. Robert Fagan Scott Danos, M. P. H. Program Briefing April 29, 2008 Division of Integrated Surveillance Systems and Services National Center for Public Health Informatics Coordinating Center for Health Information and Service Centers for Disease Control and Prevention The findings and conclusions in this presentation are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention.
Evolution of Integrated Surveillance in the United States Scott J. N. Mc. Nabb, Ph. D. , M. S. Ruth Ann Jajosky, D. M. D. , M. P. H. Robert Fagan Scott Danos, M. P. H. Program Briefing April 29, 2008 Division of Integrated Surveillance Systems and Services National Center for Public Health Informatics Coordinating Center for Health Information and Service Centers for Disease Control and Prevention The findings and conclusions in this presentation are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention.
 Mc. Nabb, S. J. N. , D. Koo, R. W. Pinner, and J. D. Seligman. Informatics and Public Health at CDC. MMWR. Dec. 22, 2006. 55(Sup 02); 25 – 8.
Mc. Nabb, S. J. N. , D. Koo, R. W. Pinner, and J. D. Seligman. Informatics and Public Health at CDC. MMWR. Dec. 22, 2006. 55(Sup 02); 25 – 8.
 Fundamental Theorem of Medical Informatics (Charles Friedman) • Informatics is as much about people as it is technology • In practice, people and technology are in an interactive partnership 3
Fundamental Theorem of Medical Informatics (Charles Friedman) • Informatics is as much about people as it is technology • In practice, people and technology are in an interactive partnership 3
 Perspectives & Points-of-View
Perspectives & Points-of-View
 What is case reporting? Actions taken by providers* to recognize and report a condition of public health significance** to a local, county, or state public health agency * physicians, infection-control, laboratories ** required by law or not
What is case reporting? Actions taken by providers* to recognize and report a condition of public health significance** to a local, county, or state public health agency * physicians, infection-control, laboratories ** required by law or not
 What is case notification? • Actions taken by a U. S. State or Territorial Health Department to recognize a case of public health significance at the federal level and notify the federal public health system (e. g. , through the National Notifiable Diseases Surveillance System [NNDSS]) • Actions taken by a local, state, and national agency to recognize a public health emergency of international concern (PHEIC) and notify the World Health Organization
What is case notification? • Actions taken by a U. S. State or Territorial Health Department to recognize a case of public health significance at the federal level and notify the federal public health system (e. g. , through the National Notifiable Diseases Surveillance System [NNDSS]) • Actions taken by a local, state, and national agency to recognize a public health emergency of international concern (PHEIC) and notify the World Health Organization
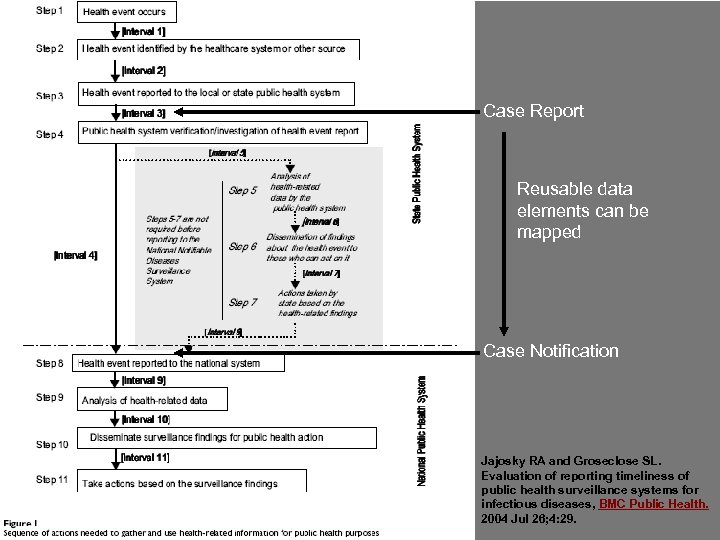 Case Report Reusable data elements can be mapped Case Notification Jajosky RA and Groseclose SL. Evaluation of reporting timeliness of public health surveillance systems for infectious diseases, BMC Public Health. 2004 Jul 26; 4: 29.
Case Report Reusable data elements can be mapped Case Notification Jajosky RA and Groseclose SL. Evaluation of reporting timeliness of public health surveillance systems for infectious diseases, BMC Public Health. 2004 Jul 26; 4: 29.
 Clinical Information Subject Information Clinical Information Reporter/Health Care Provider Information
Clinical Information Subject Information Clinical Information Reporter/Health Care Provider Information
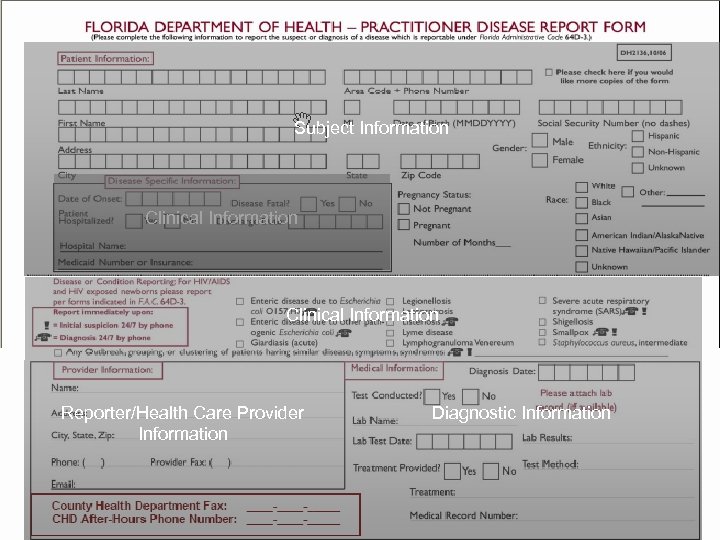 Subject Information Clinical Information Reporter/Health Care Provider Information Diagnostic Information
Subject Information Clinical Information Reporter/Health Care Provider Information Diagnostic Information
 Epidemiologic Information
Epidemiologic Information
 Epidemiologic Information
Epidemiologic Information
 What does Integrated Bio. Surveillance Mean? In the process sense of the word, the term integrating Bio. Surveillance means achieving N – 1 systems through which CDC is notified of outcomes of national public health significance.
What does Integrated Bio. Surveillance Mean? In the process sense of the word, the term integrating Bio. Surveillance means achieving N – 1 systems through which CDC is notified of outcomes of national public health significance.
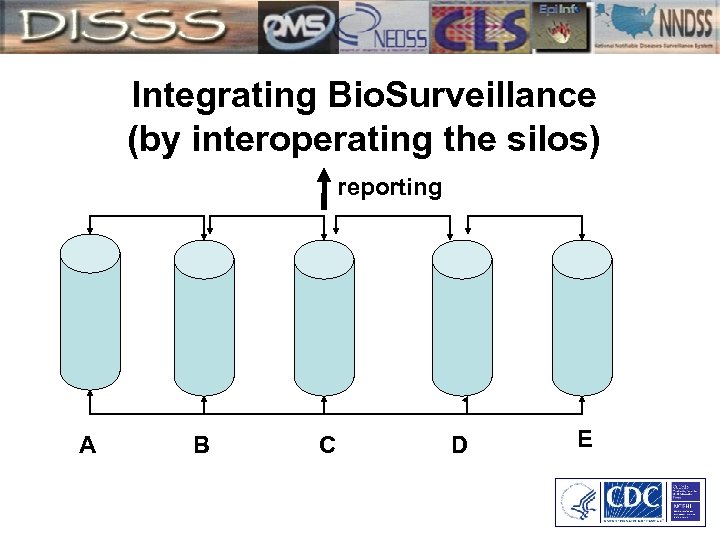 Integrating Bio. Surveillance (by interoperating the silos) reporting A B C D E
Integrating Bio. Surveillance (by interoperating the silos) reporting A B C D E
 What does Integrated Bio. Surveillance Mean? In the process sense of the word, integrating Bio. Surveillance means working to achieve efficient and effective public health work practices that are supported by interoperable information systems to detect, register, confirm, report, and analyze health outcome data while visualizing and reporting out messages that guide [and are guided by] acute and planned responses.
What does Integrated Bio. Surveillance Mean? In the process sense of the word, integrating Bio. Surveillance means working to achieve efficient and effective public health work practices that are supported by interoperable information systems to detect, register, confirm, report, and analyze health outcome data while visualizing and reporting out messages that guide [and are guided by] acute and planned responses.
 What does Integrated Bio. Surveillance Mean? In the end-state sense of the word, integrated Bio. Surveillance means a person-based, longitudinal registry of public health data built from case reports vis-à-vis an event-based one.
What does Integrated Bio. Surveillance Mean? In the end-state sense of the word, integrated Bio. Surveillance means a person-based, longitudinal registry of public health data built from case reports vis-à-vis an event-based one.
 What does Integrated Bio. Surveillance Mean? In the end-state sense of the word, integrated biosurveillance means one set of standards for "bringing together" or interoperating existing or new data streams; one sign-on, data-entry once, and one system that allows individual access from any computer; one source and set of individualized public health tools for customized data views, command sets, and public health management; one set of guidelines for establishing and managing databases; one-stop shopping for information in public health informatics and one source about integration of public health information for all users; one grid with one access to all information, but all information is not stored in one place; one medicine (i. e. , health without regard to species differences); yet one size does NOT fit all.
What does Integrated Bio. Surveillance Mean? In the end-state sense of the word, integrated biosurveillance means one set of standards for "bringing together" or interoperating existing or new data streams; one sign-on, data-entry once, and one system that allows individual access from any computer; one source and set of individualized public health tools for customized data views, command sets, and public health management; one set of guidelines for establishing and managing databases; one-stop shopping for information in public health informatics and one source about integration of public health information for all users; one grid with one access to all information, but all information is not stored in one place; one medicine (i. e. , health without regard to species differences); yet one size does NOT fit all.
 National Notifiable Diseases Surveillance System (NNDSS) Ruth Ann Jajosky, D. M. D. , M. P. H.
National Notifiable Diseases Surveillance System (NNDSS) Ruth Ann Jajosky, D. M. D. , M. P. H.
 What is the NNDSS? (1) • State-based public health surveillance system • Based upon a list of Nationally Notifiable Infectious Diseases (NNID) • There approximately 77 NNID • Unifying principle: regular, frequent, and timely information is necessary for prevention and control of NNID • Council of State and Territorial Epidemiologists (CSTE) • Collaborates with CDC in the administration of the NNDSS • Represents collective voice of epidemiologists in the States and Territories on issues involving public health practice • Through CSTE position statement process, they document policy decisions • Annually approve changes to NNID list and national surveillance case definitions 18
What is the NNDSS? (1) • State-based public health surveillance system • Based upon a list of Nationally Notifiable Infectious Diseases (NNID) • There approximately 77 NNID • Unifying principle: regular, frequent, and timely information is necessary for prevention and control of NNID • Council of State and Territorial Epidemiologists (CSTE) • Collaborates with CDC in the administration of the NNDSS • Represents collective voice of epidemiologists in the States and Territories on issues involving public health practice • Through CSTE position statement process, they document policy decisions • Annually approve changes to NNID list and national surveillance case definitions 18
 What is the NNDSS? (2) • 57 reporting jurisdictions report data to CDC each week • All U. S. states, NYC, Washington DC, 5 U. S. territories • No personal identifiers sent • States report data voluntarily to CDC • The list of NNID can change each year • Disease reporting is mandated only at the local or state level • The list of reportable diseases is different in each state • U. S. Constitution gives the State Health Officer the authority for public health • CDC assumed responsibility for collection and reporting of NNID in 1961 19
What is the NNDSS? (2) • 57 reporting jurisdictions report data to CDC each week • All U. S. states, NYC, Washington DC, 5 U. S. territories • No personal identifiers sent • States report data voluntarily to CDC • The list of NNID can change each year • Disease reporting is mandated only at the local or state level • The list of reportable diseases is different in each state • U. S. Constitution gives the State Health Officer the authority for public health • CDC assumed responsibility for collection and reporting of NNID in 1961 19
 NNDSS Background • At the federal level, the data are used to: • • • Monitoring trends Monitoring the effectiveness of prevention and control activities Program planning and evaluation Policy development Research • At the state level, the data are also used as indicated above but also to implement immediate public health action (disease prevention and control activities) • Some NNID are reported to the World Health Organization (WHO) 20 • Public Health Emergencies of International Concern (PHEIC), under the revised International Health Regulations
NNDSS Background • At the federal level, the data are used to: • • • Monitoring trends Monitoring the effectiveness of prevention and control activities Program planning and evaluation Policy development Research • At the state level, the data are also used as indicated above but also to implement immediate public health action (disease prevention and control activities) • Some NNID are reported to the World Health Organization (WHO) 20 • Public Health Emergencies of International Concern (PHEIC), under the revised International Health Regulations
 Major NNDSS Products • • MMWR Tables I and II MMWR Table IV (HIV/AIDS, TB data) MMWR Figure I MMWR Summary of Notifiable Diseases, U. S. NNDSS Link (AVR tool) Annual reporting requirements assessment Policies and procedures • Data-stewardship agreements, standardized case definitions, residency rules, publication criteria, etc. • NNDSS case definitions web site 21
Major NNDSS Products • • MMWR Tables I and II MMWR Table IV (HIV/AIDS, TB data) MMWR Figure I MMWR Summary of Notifiable Diseases, U. S. NNDSS Link (AVR tool) Annual reporting requirements assessment Policies and procedures • Data-stewardship agreements, standardized case definitions, residency rules, publication criteria, etc. • NNDSS case definitions web site 21
 22
22
 NNDSS Data • Summary data in National Electronic Telecommunications System for Surveillance (NETSS) format (beginning 1951) • Case-specific data in NETSS format (1992 to 2008) • Two data conversion processes • • • 23 National Electronic Disease Surveillance System (NEDSS)- to-NETSS conversion • NEDSS-base system (NBS) data from 16 states (core and diseasespecific data) • Supports current publication and analytical needs NETSS-to-NEDSS conversion • Supports analyses of data in Data Marts Complex transition converting systems and data Quality control and quality assurance Resource intensive Steep learning curve for data analysts
NNDSS Data • Summary data in National Electronic Telecommunications System for Surveillance (NETSS) format (beginning 1951) • Case-specific data in NETSS format (1992 to 2008) • Two data conversion processes • • • 23 National Electronic Disease Surveillance System (NEDSS)- to-NETSS conversion • NEDSS-base system (NBS) data from 16 states (core and diseasespecific data) • Supports current publication and analytical needs NETSS-to-NEDSS conversion • Supports analyses of data in Data Marts Complex transition converting systems and data Quality control and quality assurance Resource intensive Steep learning curve for data analysts
 • NNDSS Terms • State-based public health surveillance system • National Center for Public Health Informatics compiles the data which becomes the official U. S. statistics for this system • NETSS • Legacy message format for data reported to CDC for selected NNDSS conditions • CDC-developed surveillance information system • NEDSS • New message format for data reported to CDC for the NNDSS and other systems • CDC-developed a surveillance information system (NBS) • There are other dimensions (NEDSS discussion later in presentation) 24
• NNDSS Terms • State-based public health surveillance system • National Center for Public Health Informatics compiles the data which becomes the official U. S. statistics for this system • NETSS • Legacy message format for data reported to CDC for selected NNDSS conditions • CDC-developed surveillance information system • NEDSS • New message format for data reported to CDC for the NNDSS and other systems • CDC-developed a surveillance information system (NBS) • There are other dimensions (NEDSS discussion later in presentation) 24
 What’s the difference between NNDSS, NETSS, NEDSS, and NNDSS Link? 25
What’s the difference between NNDSS, NETSS, NEDSS, and NNDSS Link? 25
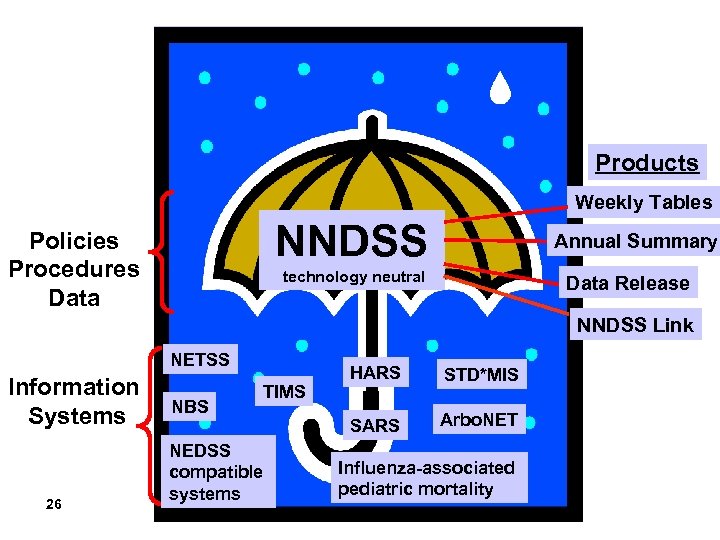 Products Weekly Tables NNDSS Policies Procedures Data Annual Summary technology neutral Data Release NNDSS Link NETSS Information Systems 26 NBS TIMS NEDSS compatible systems HARS STD*MIS SARS Arbo. NET Influenza-associated pediatric mortality
Products Weekly Tables NNDSS Policies Procedures Data Annual Summary technology neutral Data Release NNDSS Link NETSS Information Systems 26 NBS TIMS NEDSS compatible systems HARS STD*MIS SARS Arbo. NET Influenza-associated pediatric mortality
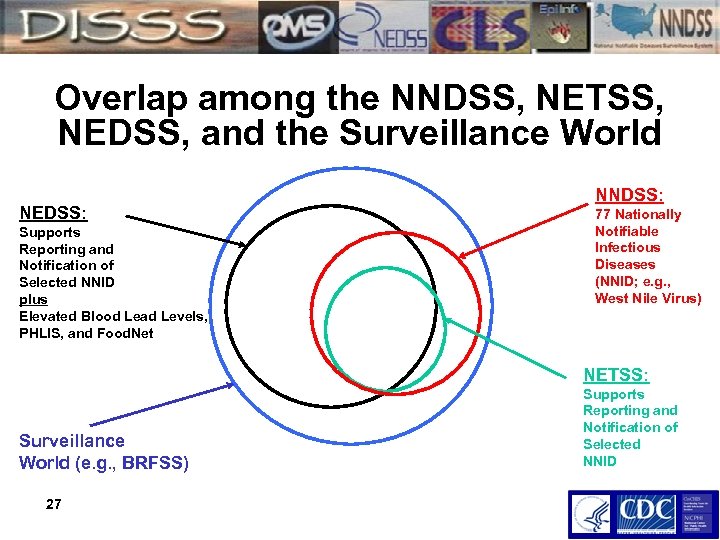 Overlap among the NNDSS, NETSS, NEDSS, and the Surveillance World NEDSS: Supports Reporting and Notification of Selected NNID plus Elevated Blood Lead Levels, PHLIS, and Food. Net NNDSS: 77 Nationally Notifiable Infectious Diseases (NNID; e. g. , West Nile Virus) NETSS: Surveillance World (e. g. , BRFSS) 27 Supports Reporting and Notification of Selected NNID
Overlap among the NNDSS, NETSS, NEDSS, and the Surveillance World NEDSS: Supports Reporting and Notification of Selected NNID plus Elevated Blood Lead Levels, PHLIS, and Food. Net NNDSS: 77 Nationally Notifiable Infectious Diseases (NNID; e. g. , West Nile Virus) NETSS: Surveillance World (e. g. , BRFSS) 27 Supports Reporting and Notification of Selected NNID
 Collaborative Work with CSTE • 2007 CSTE-CDC State Reportable Conditions Assessment (SRCA) • Major change in methodology to solicit and document reporting requirements • All reporting requirements (ID and non-ID) in all NNDSS reporting jurisdictions • Transitional step toward working with Onto. Reason to gather reporting requirements in a Knowledgebase (Kb) at the local level • Objective of Kb: Provide a central up-to-date listing of reportable conditions by jurisdiction to facilitate more complete case-reporting to Public Health and to reduce the burden on Public Health stakeholders to gather this information on their own 28
Collaborative Work with CSTE • 2007 CSTE-CDC State Reportable Conditions Assessment (SRCA) • Major change in methodology to solicit and document reporting requirements • All reporting requirements (ID and non-ID) in all NNDSS reporting jurisdictions • Transitional step toward working with Onto. Reason to gather reporting requirements in a Knowledgebase (Kb) at the local level • Objective of Kb: Provide a central up-to-date listing of reportable conditions by jurisdiction to facilitate more complete case-reporting to Public Health and to reduce the burden on Public Health stakeholders to gather this information on their own 28
 Additional Work with CSTE • 2008 SRCA • Development of algorithms to trigger case reporting from the healthcare sector to local, county, and state public health • Transform human-readable national surveillance case definitions to machine-readable format 29
Additional Work with CSTE • 2008 SRCA • Development of algorithms to trigger case reporting from the healthcare sector to local, county, and state public health • Transform human-readable national surveillance case definitions to machine-readable format 29
 Collaborative Work with CDC Programs on Data Marts • Development of NCIRD Data Mart • Includes but is not limited to bacterial meningitis and invasive respiratory disease pathogens • Vaccine-preventable diseases (VPDs) • Development of NNDSS Data Mart • Develop a plan to transition data storage and analysis to NEDSS formats for processing of all NNDSS data • Must address existing independent Data Marts containing NNDSS data • Automated logical error-checking with feedback and summary reports to data providers • Current and historical data 30
Collaborative Work with CDC Programs on Data Marts • Development of NCIRD Data Mart • Includes but is not limited to bacterial meningitis and invasive respiratory disease pathogens • Vaccine-preventable diseases (VPDs) • Development of NNDSS Data Mart • Develop a plan to transition data storage and analysis to NEDSS formats for processing of all NNDSS data • Must address existing independent Data Marts containing NNDSS data • Automated logical error-checking with feedback and summary reports to data providers • Current and historical data 30

 The History of NETSS-to-NEDSS Robert Fagan
The History of NETSS-to-NEDSS Robert Fagan
 In the Beginning • Before 1985: no direct electronic transmission of public health surveillance data • States did send aggregated counts of 49 National Notifiable Diseases (NNID) to CDC via paper and phone (versus the 77 NNID in 2008) • Data elements: state, MMWR week/year, disease, total count • Slow and difficult to update • The Territory of American Samoa still sends aggregated data by phone or fax • Disease counts sent to CDC based on statedetermined definitions, not nationally standardized case definitions 33
In the Beginning • Before 1985: no direct electronic transmission of public health surveillance data • States did send aggregated counts of 49 National Notifiable Diseases (NNID) to CDC via paper and phone (versus the 77 NNID in 2008) • Data elements: state, MMWR week/year, disease, total count • Slow and difficult to update • The Territory of American Samoa still sends aggregated data by phone or fax • Disease counts sent to CDC based on statedetermined definitions, not nationally standardized case definitions 33
 1985: Beginning of the Electronic Era • Extrapolating data from paper reports difficult • Strong need for detailed case reports • Six States with ample resources and staff volunteered for Electronic Surveillance Project (ESP) 34
1985: Beginning of the Electronic Era • Extrapolating data from paper reports difficult • Strong need for detailed case reports • Six States with ample resources and staff volunteered for Electronic Surveillance Project (ESP) 34
 1985 – 1990: ESP • Electronic Surveillance Project (ESP) was a national 5 year pilot project for electronic notifiable diseases • Each state developed their own version of an electronic health reporting system • Each state included different coding structures, protocols, and diseases • Only commonality was the 40 -byte message standard • CDC developed data-interchange standard to translate received messages • A positive CDC assessment of the pilot supported the development of National Electronic Telecommunications System for Surveillance (NETSS) 35
1985 – 1990: ESP • Electronic Surveillance Project (ESP) was a national 5 year pilot project for electronic notifiable diseases • Each state developed their own version of an electronic health reporting system • Each state included different coding structures, protocols, and diseases • Only commonality was the 40 -byte message standard • CDC developed data-interchange standard to translate received messages • A positive CDC assessment of the pilot supported the development of National Electronic Telecommunications System for Surveillance (NETSS) 35
 1990: NETSS • Developed coding to better reconcile national and state records • Initially intended to expand to all CDC programs • Satisfied states with intuitive, easy-to-use, easy to understand, stable system which needed limited resources…however, • Limited resources restricted the scope of NETSS to event-based PHS • No contact tracing • No case management • Case reporting was event-based, not patient-based 36
1990: NETSS • Developed coding to better reconcile national and state records • Initially intended to expand to all CDC programs • Satisfied states with intuitive, easy-to-use, easy to understand, stable system which needed limited resources…however, • Limited resources restricted the scope of NETSS to event-based PHS • No contact tracing • No case management • Case reporting was event-based, not patient-based 36
 1993 – 1995: EPSVPD • Expanded Program for Surveillance of Vaccine Preventable Diseases: version of NETSS tailored to VPD program • Deployed in two years • Employed by 100% of states • No funding provided to states by CDC • Dedicated, salaried CDC FTE helped states to implement NETSS 37
1993 – 1995: EPSVPD • Expanded Program for Surveillance of Vaccine Preventable Diseases: version of NETSS tailored to VPD program • Deployed in two years • Employed by 100% of states • No funding provided to states by CDC • Dedicated, salaried CDC FTE helped states to implement NETSS 37
 1994: CDC Program Participation • STD program initiated move to NETSS • Meningitis, Lyme disease, Hepatitis, and VPD programs extracted data from and integrated into NETSS by 1994 • Double reporting • TB • Immunizations • HIV did not participate • Fear of confidentiality issues • Reluctant to share resources • NETSS matured as far as possible by 1994 38
1994: CDC Program Participation • STD program initiated move to NETSS • Meningitis, Lyme disease, Hepatitis, and VPD programs extracted data from and integrated into NETSS by 1994 • Double reporting • TB • Immunizations • HIV did not participate • Fear of confidentiality issues • Reluctant to share resources • NETSS matured as far as possible by 1994 38
 Enhancing NETSS • 1995: EPO proposed updating NETSS from DOSbased system • States already moving operating systems to Windows • Push for move to Windows-based system at CDC • Further NETSS updates denied in anticipation of migration to a newer system • Existing candidate systems in use by states were reviewed for possible adoption • CSTE requested an updated, applicationindependent, data interchange system • Health Information Surveillance and Systems Board (HISSB): CDC-wide surveillance committee formed in 39 response to the Katz Report
Enhancing NETSS • 1995: EPO proposed updating NETSS from DOSbased system • States already moving operating systems to Windows • Push for move to Windows-based system at CDC • Further NETSS updates denied in anticipation of migration to a newer system • Existing candidate systems in use by states were reviewed for possible adoption • CSTE requested an updated, applicationindependent, data interchange system • Health Information Surveillance and Systems Board (HISSB): CDC-wide surveillance committee formed in 39 response to the Katz Report
 1998: Integration Project Goals • • • 40 New NETSS data elements were requested by CDC programs Define a common user interface, core data dictionary, and architecture for data model Standardize electronic security protocols
1998: Integration Project Goals • • • 40 New NETSS data elements were requested by CDC programs Define a common user interface, core data dictionary, and architecture for data model Standardize electronic security protocols
 1998: Integration Project • 11 systems involved: NETSS, STD*MIS, HARS, SHAS, ASD, PSD, TIMS, PHLIS, UD, ABC, Food. Net • Dr. Claire Broome worked to establish the title and funds • 1999 – Letter from J. Koplan, CDC Director: “I am requesting that any other surveillance systems development effort be postponed or suspended until you obtain a waiver from HISSB” • 1999: NEDSS Operating Working group (NOW) formed to create National Electronic Disease Surveillance System (NEDSS) 41
1998: Integration Project • 11 systems involved: NETSS, STD*MIS, HARS, SHAS, ASD, PSD, TIMS, PHLIS, UD, ABC, Food. Net • Dr. Claire Broome worked to establish the title and funds • 1999 – Letter from J. Koplan, CDC Director: “I am requesting that any other surveillance systems development effort be postponed or suspended until you obtain a waiver from HISSB” • 1999: NEDSS Operating Working group (NOW) formed to create National Electronic Disease Surveillance System (NEDSS) 41
 NETSS vis-à-vis NEDSS • NETSS • OMB-approved forms • NEDSS • 10 -years elapsed between last update to NETSS and inception of NEDSS, during which major changes in disease epidemiology occurred and new laboratory tests developed • JAD sessions held with federal and state and local public health staff to identify data elements to integrate into NEDSS • OMB-approved forms • Paper-based, supplemental reporting enhancements to NETSS • Surveillance program worksheets 42
NETSS vis-à-vis NEDSS • NETSS • OMB-approved forms • NEDSS • 10 -years elapsed between last update to NETSS and inception of NEDSS, during which major changes in disease epidemiology occurred and new laboratory tests developed • JAD sessions held with federal and state and local public health staff to identify data elements to integrate into NEDSS • OMB-approved forms • Paper-based, supplemental reporting enhancements to NETSS • Surveillance program worksheets 42
 1999 – Present: NEDSS “The National Electronic Disease Surveillance System (NEDSS) is an initiative that promotes the use of data and information system standards to advance the development of efficient, integrated, and interoperable surveillance systems at federal, state and local levels. It is a major component of the Public Health Information Network (PHIN)” http: //www. cdc. gov/nedss/ 43
1999 – Present: NEDSS “The National Electronic Disease Surveillance System (NEDSS) is an initiative that promotes the use of data and information system standards to advance the development of efficient, integrated, and interoperable surveillance systems at federal, state and local levels. It is a major component of the Public Health Information Network (PHIN)” http: //www. cdc. gov/nedss/ 43
 Public Health Information Network (PHIN) • Engaged in improving public health by developing and disseminating best practices in research and processes to achieve meaningful and interoperable public health information systems • PHIN was an outgrowth of NEDSS to embrace broader public health surveillance and IT needs borne from shortcomings during 9/11 and anthrax events 44
Public Health Information Network (PHIN) • Engaged in improving public health by developing and disseminating best practices in research and processes to achieve meaningful and interoperable public health information systems • PHIN was an outgrowth of NEDSS to embrace broader public health surveillance and IT needs borne from shortcomings during 9/11 and anthrax events 44
 ESP & NETSS: Lessons Learned (1) • Use an industry standard approach for message creation, not a proprietary software Create Data Interchange/Message as first step Broadly distribute the message format; this allows the project to leverage state resources to build solutions Develop the CDC side of the system right away after distribution of message format to allow CDC to accept data Develop recommendation for core record part of the application and publish it Offer the CDC developed software v 1. 0 free; use only core information allowing rapid deployment • • • 45
ESP & NETSS: Lessons Learned (1) • Use an industry standard approach for message creation, not a proprietary software Create Data Interchange/Message as first step Broadly distribute the message format; this allows the project to leverage state resources to build solutions Develop the CDC side of the system right away after distribution of message format to allow CDC to accept data Develop recommendation for core record part of the application and publish it Offer the CDC developed software v 1. 0 free; use only core information allowing rapid deployment • • • 45
 ESP & NETSS: Lessons Learned (2) • Work with CDC programs to co-develop disease specific program modules Divide CDC programs into groups by deployment versions as resources allow Emphasize Analysis Visualization and Reports (AVR); never release without strong analysis access to data Start from the very beginning working with user representative partnerships committed to using the developed application Solutions must accommodate the differences among states as the reporting protocols vary from state to state; public health is a state mandate not federal Hire dedicated, salaried CDC employee to help states implement NETSS ESPVPD • • • 46
ESP & NETSS: Lessons Learned (2) • Work with CDC programs to co-develop disease specific program modules Divide CDC programs into groups by deployment versions as resources allow Emphasize Analysis Visualization and Reports (AVR); never release without strong analysis access to data Start from the very beginning working with user representative partnerships committed to using the developed application Solutions must accommodate the differences among states as the reporting protocols vary from state to state; public health is a state mandate not federal Hire dedicated, salaried CDC employee to help states implement NETSS ESPVPD • • • 46
 Where do we find ourselves now?
Where do we find ourselves now?
 NEDSS: Yesterday, Today, and Tomorrow Scott Danos, M. P. H.
NEDSS: Yesterday, Today, and Tomorrow Scott Danos, M. P. H.
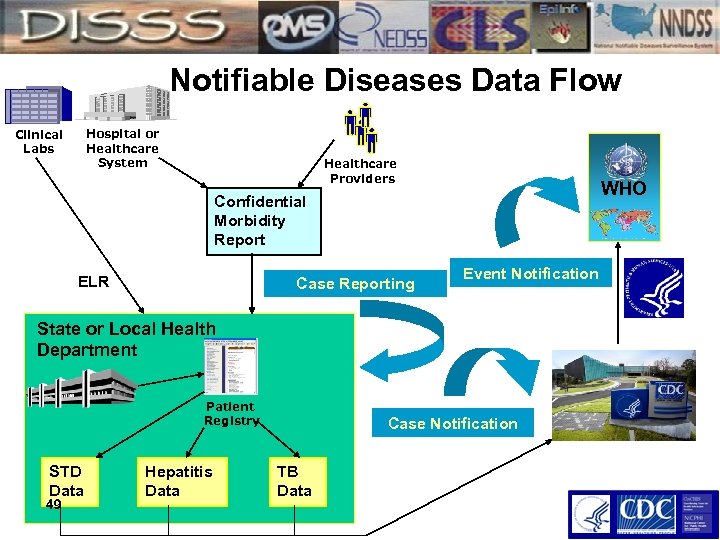 Notifiable Diseases Data Flow Clinical Labs Hospital or Healthcare System Healthcare Providers WHO Confidential Morbidity Report ELR Case Reporting Event Notification State or Local Health Department Patient Registry STD Data 49 Hepatitis Data Case Notification TB Data
Notifiable Diseases Data Flow Clinical Labs Hospital or Healthcare System Healthcare Providers WHO Confidential Morbidity Report ELR Case Reporting Event Notification State or Local Health Department Patient Registry STD Data 49 Hepatitis Data Case Notification TB Data
 NEDSS Characteristics • Patient-centric • Involves highly complex, relational data to support dynamic relationship • NBS: complex physical database • Requires large effort to sustain • Challenges in supporting multiple data formats both standards-based and non-standards-based • Inevitable difficulty in translating data from paper to electronic format 50
NEDSS Characteristics • Patient-centric • Involves highly complex, relational data to support dynamic relationship • NBS: complex physical database • Requires large effort to sustain • Challenges in supporting multiple data formats both standards-based and non-standards-based • Inevitable difficulty in translating data from paper to electronic format 50
 Issues at CDC in NEDSS Implementation (1) • Many CDC silo systems were built or allowed to continue or develop • No mandate to promote the use of a single application • Initial target date for full national implementation was 2000, with deployment in 1998 • Not realized • HISSB authorized to distribute $321, 000/year to various CDC programs through NEDSS • • Standards needed to launch project Funds were a drop-in-the-bucket • Contributions by technical SMEs were considered but not embraced in lieu of exploring a possible explosive growth in Internet capability • 51 Insufficient public health end-user input about what could and should be built
Issues at CDC in NEDSS Implementation (1) • Many CDC silo systems were built or allowed to continue or develop • No mandate to promote the use of a single application • Initial target date for full national implementation was 2000, with deployment in 1998 • Not realized • HISSB authorized to distribute $321, 000/year to various CDC programs through NEDSS • • Standards needed to launch project Funds were a drop-in-the-bucket • Contributions by technical SMEs were considered but not embraced in lieu of exploring a possible explosive growth in Internet capability • 51 Insufficient public health end-user input about what could and should be built
 Issues at CDC in NEDSS Implementation (2) • Data model decisions • Conceptual Data Model, derived from the HL 7 v 2 RIM, was used to develop the physical data model (too literal) • Absence of end-user review and input • Continued challenges in data extraction by SMEs • Software decisions • Silver. Stream selected for the application server • Failure to identify and incorporate appropriate COTS products • De-duplication • AVR • Standards decisions • HL 7 v 3 52
Issues at CDC in NEDSS Implementation (2) • Data model decisions • Conceptual Data Model, derived from the HL 7 v 2 RIM, was used to develop the physical data model (too literal) • Absence of end-user review and input • Continued challenges in data extraction by SMEs • Software decisions • Silver. Stream selected for the application server • Failure to identify and incorporate appropriate COTS products • De-duplication • AVR • Standards decisions • HL 7 v 3 52
 First Epidemiology and Laboratory Capacity Request for Proposal (ELC RFP) – July 20, 2001 • Element Development • Implement an integrated data repository • Accept, route and process electronic HL 7 messages containing laboratory and clinical content • Conduct and support web browser-based data entry and data management • Develop active data translation and exchange (integration broker) functionality • Modern application programming practices - component based, object oriented and cross platform where possible • Formerly - Develop transportable business logic capability • Develop data analysis, visualization and reporting capability • Implement a directory of public health personnel • Implement a security system and appropriate security policies • Charter Site • NEDSS Base System (NBS) 53
First Epidemiology and Laboratory Capacity Request for Proposal (ELC RFP) – July 20, 2001 • Element Development • Implement an integrated data repository • Accept, route and process electronic HL 7 messages containing laboratory and clinical content • Conduct and support web browser-based data entry and data management • Develop active data translation and exchange (integration broker) functionality • Modern application programming practices - component based, object oriented and cross platform where possible • Formerly - Develop transportable business logic capability • Develop data analysis, visualization and reporting capability • Implement a directory of public health personnel • Implement a security system and appropriate security policies • Charter Site • NEDSS Base System (NBS) 53
 NEDSS-Base System (NBS) and NEDSS Status Today 54
NEDSS-Base System (NBS) and NEDSS Status Today 54
 NBS Fact Sheet (1) • Vision • NBS is a platform which many public health surveillance systems, processes, and data can be integrated in a secure environment • Implementation standards are provided for states developing their own NEDSS compatible systems • What is it? • Platform to support state notifiable disease surveillance and analysis activities • Successor of NETSS • A modular system (all or part of the Base System may be used) • NBS is not… • NEDSS • Complete NEDSS solution • The integrated disease surveillance system for all states 55
NBS Fact Sheet (1) • Vision • NBS is a platform which many public health surveillance systems, processes, and data can be integrated in a secure environment • Implementation standards are provided for states developing their own NEDSS compatible systems • What is it? • Platform to support state notifiable disease surveillance and analysis activities • Successor of NETSS • A modular system (all or part of the Base System may be used) • NBS is not… • NEDSS • Complete NEDSS solution • The integrated disease surveillance system for all states 55
 NBS Fact Sheet (2) • Facilitates public health surveillance at the local, state and federal levels • In production by 16 states • 900 + users nationwide • Covers roughly 20% of the US population • Web enabled, built using J 2 EE (Java 2 Enterprise Edition) standards • Product continues to evolve through maintenance releases and incremental functionality 56
NBS Fact Sheet (2) • Facilitates public health surveillance at the local, state and federal levels • In production by 16 states • 900 + users nationwide • Covers roughly 20% of the US population • Web enabled, built using J 2 EE (Java 2 Enterprise Edition) standards • Product continues to evolve through maintenance releases and incremental functionality 56
 NBS Fact Sheet (3) • Entities • • • Patient Provider Organization • Acts • • • Lab Report Morbidity Report Investigations • Generic, Hepatitis, Pertussis, Measles, CRS, BMIRD, Foodborne Treatment Vaccination • Messaging • • 57 • • NND Notifications Electronic Lab Reports (ELR) De-Duplication Reporting module Geocoding framework Administration
NBS Fact Sheet (3) • Entities • • • Patient Provider Organization • Acts • • • Lab Report Morbidity Report Investigations • Generic, Hepatitis, Pertussis, Measles, CRS, BMIRD, Foodborne Treatment Vaccination • Messaging • • 57 • • NND Notifications Electronic Lab Reports (ELR) De-Duplication Reporting module Geocoding framework Administration
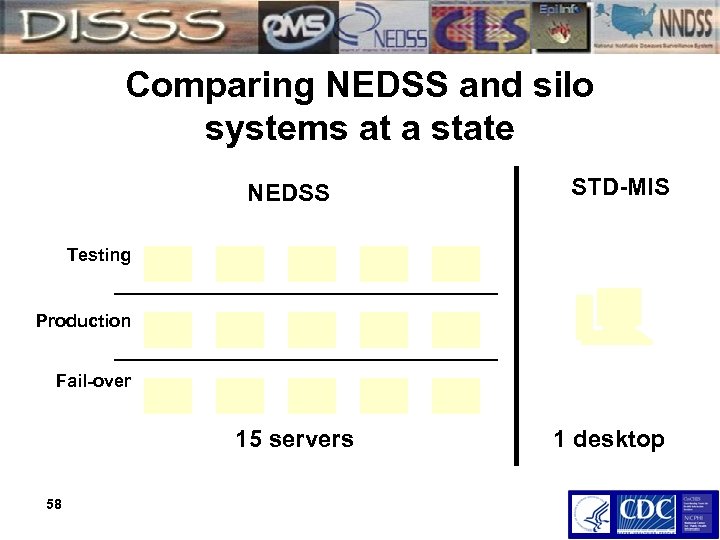 Comparing NEDSS and silo systems at a state NEDSS STD-MIS Testing Production Fail-over 15 servers 58 1 desktop
Comparing NEDSS and silo systems at a state NEDSS STD-MIS Testing Production Fail-over 15 servers 58 1 desktop
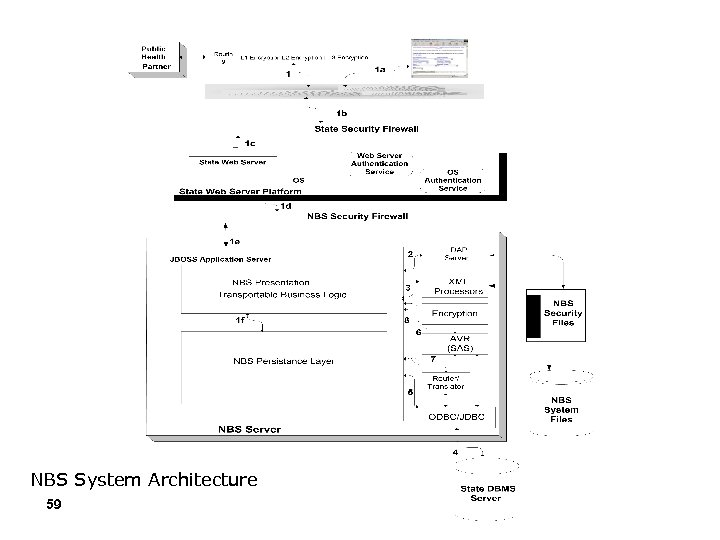 NBS System Architecture 59
NBS System Architecture 59
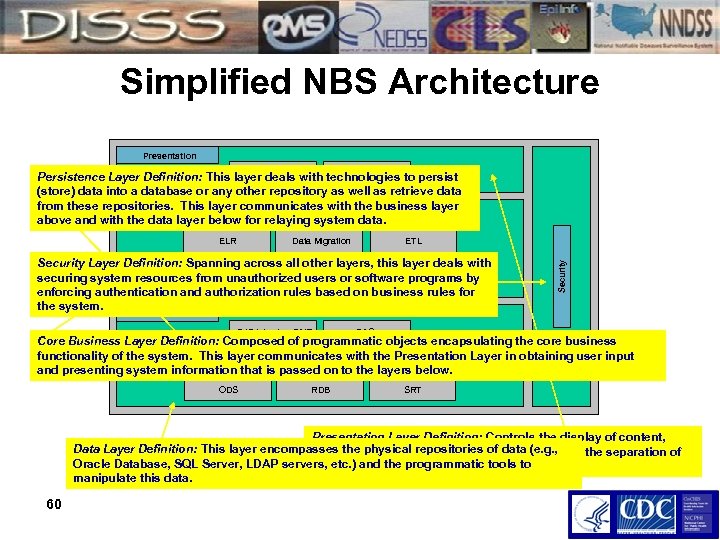 Simplified NBS Architecture Presentation Cocoon Struts Persistence Layer Definition: This layer deals with technologies to persist (store) data into a database or any other repository as well as retrieve data from these repositories. This layer communicates with the business layer Core Business above and with the data layer below for relaying system data. Data Migration ETL Security Layer Definition: Spanning across all other layers, this layer deals with Patient Locator Deduplication Core NBS text securing system resources from unauthorized users or software programs by enforcing authentication and authorization rules based on business rules for the system. Security ELR Persistence EJB 1. 1 using BMP DAOs Core Business Layer Definition: Composed of programmatic objects encapsulating the core business functionality of the system. This layer communicates with the Presentation Layer in obtaining user input Data and presenting system information that is passed on to the layers below. ODS RDB SRT Presentation Layer Definition: Controls the display of content, Data Layer Definition: This layer encompasses the physical repositories of data (e. g. , navigation within the application and allows for the separation of Oracle Database, SQL Server, LDAP servers, etc. ) and the programmatic tools to front-end and back-end functionality. manipulate this data. 60
Simplified NBS Architecture Presentation Cocoon Struts Persistence Layer Definition: This layer deals with technologies to persist (store) data into a database or any other repository as well as retrieve data from these repositories. This layer communicates with the business layer Core Business above and with the data layer below for relaying system data. Data Migration ETL Security Layer Definition: Spanning across all other layers, this layer deals with Patient Locator Deduplication Core NBS text securing system resources from unauthorized users or software programs by enforcing authentication and authorization rules based on business rules for the system. Security ELR Persistence EJB 1. 1 using BMP DAOs Core Business Layer Definition: Composed of programmatic objects encapsulating the core business functionality of the system. This layer communicates with the Presentation Layer in obtaining user input Data and presenting system information that is passed on to the layers below. ODS RDB SRT Presentation Layer Definition: Controls the display of content, Data Layer Definition: This layer encompasses the physical repositories of data (e. g. , navigation within the application and allows for the separation of Oracle Database, SQL Server, LDAP servers, etc. ) and the programmatic tools to front-end and back-end functionality. manipulate this data. 60
 NBS Technical Overview • Based on Java/J 2 EE technologies • Component based architecture represented by: • Cocoon/Struts framework for the front-end • EJB framework at the persistence layer • LDAP based security model with container controlled transactions. • Application Server • Initially deployed on Silver. Stream Application server • Migrated to Weblogic • Now to JBoss Application server. • NBS extensively uses Industry standard components like XML within its framework 61
NBS Technical Overview • Based on Java/J 2 EE technologies • Component based architecture represented by: • Cocoon/Struts framework for the front-end • EJB framework at the persistence layer • LDAP based security model with container controlled transactions. • Application Server • Initially deployed on Silver. Stream Application server • Migrated to Weblogic • Now to JBoss Application server. • NBS extensively uses Industry standard components like XML within its framework 61
 NBS Release History Version Date 1. 0 1. 1 10/01/2002 06/01/2003 1. 1. 1 10/01/2003 1. 1. 2 01/01/2004 1. 1. 3 04/01/2004 1. 1. 3 SP 1 1. 1. 3 SP 2 1. 1. 4 08/01/2004 02/01/2005 07/01/2005 1. 1. 4 SP 1 1. 1. 5 SP 2 1. 1. 6 1. 1. 7 01/01/2006 09/15/2006 12/15/2006 04/24/2007 09/28/2007 12/31/2007 62 Summary First production-ready version of NBS Release 1. 0 Enhancing manual lab, electronic lab, and morbidity reporting, adding data de-duplication, point-in-time demographics, treatment management, and other miscellaneous ERs. Adding reporting, external data warehouse, and the ability to extend data collection through ‘locally defined fields’ Adding a repository for collecting data from state-defined and collaboratively defined fields, and sending the NND message Remaining NETSS functionality, the ability to create custom forms, usability enhancements, enhancements to the BMIRD PAM, and the integration of the Foodborne PAM Enhancing base functionality Implementing end user ERs, implementing Lyme questions Standard Operating Environment (SOE) upgrade, upgrade of Foodborne PAM, ERs Enhancing NBS Program Area Modules (PAMS), addition of new data mart Migration to JBoss Application Server, new data marts, ERs Enhancing reporting module, application performance, ERs Implementing new ERs Enhancing reporting module, application performance, new ERs 2008 form changes, application performance improvements, new ERs
NBS Release History Version Date 1. 0 1. 1 10/01/2002 06/01/2003 1. 1. 1 10/01/2003 1. 1. 2 01/01/2004 1. 1. 3 04/01/2004 1. 1. 3 SP 1 1. 1. 3 SP 2 1. 1. 4 08/01/2004 02/01/2005 07/01/2005 1. 1. 4 SP 1 1. 1. 5 SP 2 1. 1. 6 1. 1. 7 01/01/2006 09/15/2006 12/15/2006 04/24/2007 09/28/2007 12/31/2007 62 Summary First production-ready version of NBS Release 1. 0 Enhancing manual lab, electronic lab, and morbidity reporting, adding data de-duplication, point-in-time demographics, treatment management, and other miscellaneous ERs. Adding reporting, external data warehouse, and the ability to extend data collection through ‘locally defined fields’ Adding a repository for collecting data from state-defined and collaboratively defined fields, and sending the NND message Remaining NETSS functionality, the ability to create custom forms, usability enhancements, enhancements to the BMIRD PAM, and the integration of the Foodborne PAM Enhancing base functionality Implementing end user ERs, implementing Lyme questions Standard Operating Environment (SOE) upgrade, upgrade of Foodborne PAM, ERs Enhancing NBS Program Area Modules (PAMS), addition of new data mart Migration to JBoss Application Server, new data marts, ERs Enhancing reporting module, application performance, ERs Implementing new ERs Enhancing reporting module, application performance, new ERs 2008 form changes, application performance improvements, new ERs
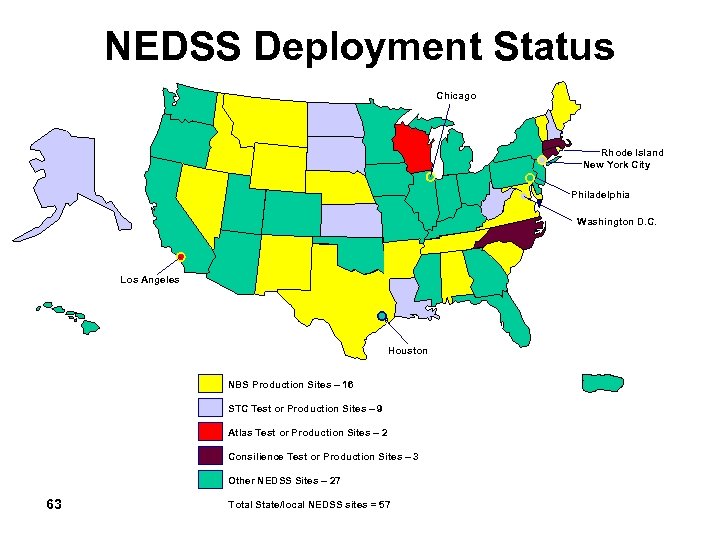 NEDSS Deployment Status Chicago Rhode Island New York City Philadelphia Washington D. C. Los Angeles Houston NBS Production Sites – 16 STC Test or Production Sites – 9 Atlas Test or Production Sites – 2 Consilience Test or Production Sites – 3 Other NEDSS Sites – 27 63 Total State/local NEDSS sites = 57
NEDSS Deployment Status Chicago Rhode Island New York City Philadelphia Washington D. C. Los Angeles Houston NBS Production Sites – 16 STC Test or Production Sites – 9 Atlas Test or Production Sites – 2 Consilience Test or Production Sites – 3 Other NEDSS Sites – 27 63 Total State/local NEDSS sites = 57
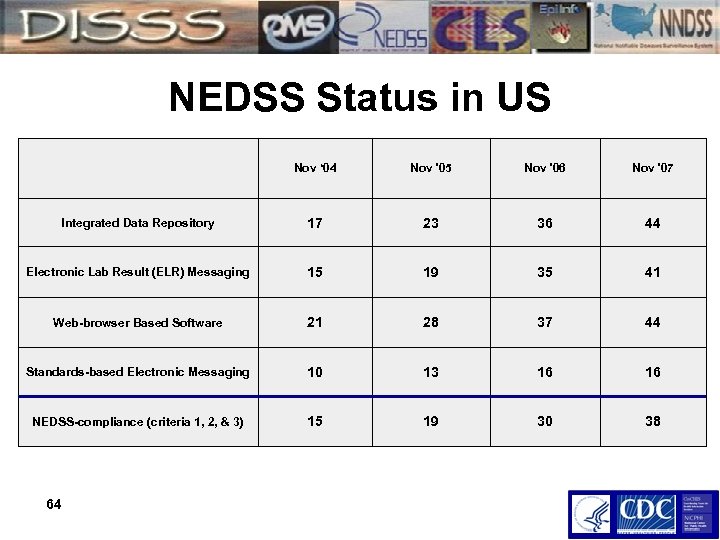 NEDSS Status in US Nov ‘ 04 Nov '05 Nov '06 Nov '07 Integrated Data Repository 17 23 36 44 Electronic Lab Result (ELR) Messaging 15 19 35 41 Web-browser Based Software 21 28 37 44 Standards-based Electronic Messaging 10 13 16 16 NEDSS-compliance (criteria 1, 2, & 3) 15 19 30 38 64
NEDSS Status in US Nov ‘ 04 Nov '05 Nov '06 Nov '07 Integrated Data Repository 17 23 36 44 Electronic Lab Result (ELR) Messaging 15 19 35 41 Web-browser Based Software 21 28 37 44 Standards-based Electronic Messaging 10 13 16 16 NEDSS-compliance (criteria 1, 2, & 3) 15 19 30 38 64
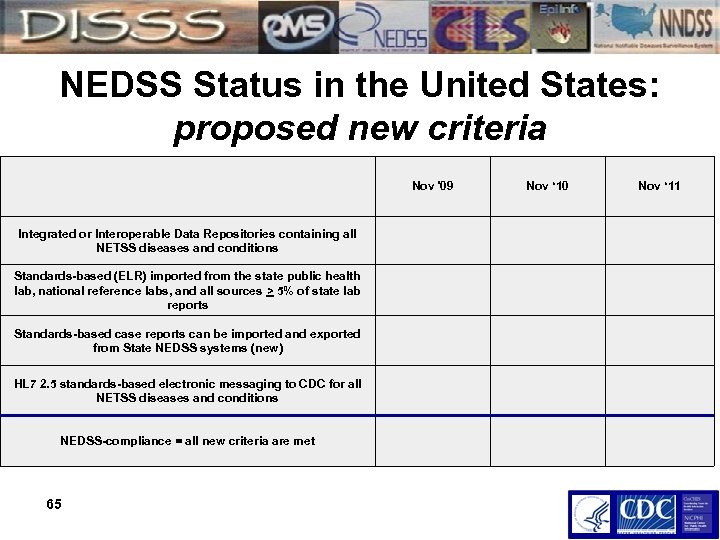 NEDSS Status in the United States: proposed new criteria Integrated or Interoperable Data Repositories containing all NETSS diseases and conditions Standards-based (ELR) imported from the state public health lab, national reference labs, and all sources > 5% of state lab reports Standards-based case reports can be imported and exported from State NEDSS systems (new) HL 7 2. 5 standards-based electronic messaging to CDC for all NETSS diseases and conditions NEDSS-compliance = all new criteria are met 65 Nov '09 Nov ‘ 10 Nov ‘ 11
NEDSS Status in the United States: proposed new criteria Integrated or Interoperable Data Repositories containing all NETSS diseases and conditions Standards-based (ELR) imported from the state public health lab, national reference labs, and all sources > 5% of state lab reports Standards-based case reports can be imported and exported from State NEDSS systems (new) HL 7 2. 5 standards-based electronic messaging to CDC for all NETSS diseases and conditions NEDSS-compliance = all new criteria are met 65 Nov '09 Nov ‘ 10 Nov ‘ 11
 NEDSS Message Mapping Guides and CDC Program Datamarts 66
NEDSS Message Mapping Guides and CDC Program Datamarts 66
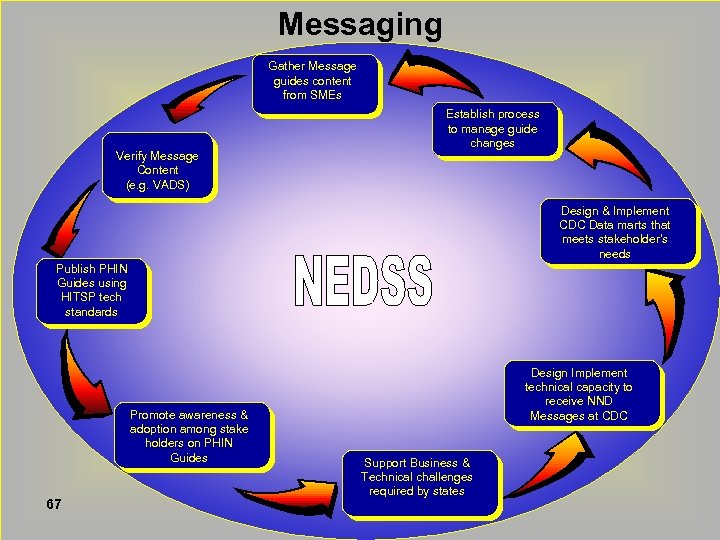 Messaging Gather Message guides content from SMEs Verify Message Content (e. g. VADS) Establish process to manage guide changes Design & Implement CDC Data marts that meets stakeholder’s needs Publish PHIN Guides using HITSP tech standards Promote awareness & adoption among stake holders on PHIN Guides 67 Design Implement technical capacity to receive NND Messages at CDC Support Business & Technical challenges required by states
Messaging Gather Message guides content from SMEs Verify Message Content (e. g. VADS) Establish process to manage guide changes Design & Implement CDC Data marts that meets stakeholder’s needs Publish PHIN Guides using HITSP tech standards Promote awareness & adoption among stake holders on PHIN Guides 67 Design Implement technical capacity to receive NND Messages at CDC Support Business & Technical challenges required by states
 Key Steps to Developing the Message Mapping Guide • Gather message mapping guide data elements from Program and submit to the DISS-Vocabulary and Messaging Team • Initial draft is created by DISS-Vocabulary and Messaging and reviewed by DISSS • After DISSS review the draft guide is passed to the Program for review • Engage with program as needed during the review process to address questions and concerns • Obtain approval from program to post guide to Site. Scape • Notify DISS-Vocabulary and Messaging Team of the Program’s approval to post to Site. Scape • Broadcast Site. Scape posting to CDC programs and external partners 68
Key Steps to Developing the Message Mapping Guide • Gather message mapping guide data elements from Program and submit to the DISS-Vocabulary and Messaging Team • Initial draft is created by DISS-Vocabulary and Messaging and reviewed by DISSS • After DISSS review the draft guide is passed to the Program for review • Engage with program as needed during the review process to address questions and concerns • Obtain approval from program to post guide to Site. Scape • Notify DISS-Vocabulary and Messaging Team of the Program’s approval to post to Site. Scape • Broadcast Site. Scape posting to CDC programs and external partners 68
 Key Steps to Preparing and Publishing the Message Mapping Guide • Engage external partners as needed during the 30 -day Site. Scape review process to address questions and concerns • Triage external partner responses to appropriate teams • Vocabulary and Messaging will meet with the Program, DMB and CDS to determine impact of requested changes • Receive Program final approval of the message mapping guide • DISS-Vocabulary and Messaging will make Program approved changes • DISS-Vocabulary and Messaging submit message mapping guide to DAMC for PHIN web site clearance • The message mapping guide is posted to the PHIN web site for external partners 69
Key Steps to Preparing and Publishing the Message Mapping Guide • Engage external partners as needed during the 30 -day Site. Scape review process to address questions and concerns • Triage external partner responses to appropriate teams • Vocabulary and Messaging will meet with the Program, DMB and CDS to determine impact of requested changes • Receive Program final approval of the message mapping guide • DISS-Vocabulary and Messaging will make Program approved changes • DISS-Vocabulary and Messaging submit message mapping guide to DAMC for PHIN web site clearance • The message mapping guide is posted to the PHIN web site for external partners 69
 Key Steps to Promoting Awareness and Providing Technical Support • Establish a CDC technical support team with the following skills: • • • HL 7 Orion Rhapsody and Symphonia SQL database JMS PHIN-MS and other messaging technologies Public health message mapping guides • Publish NEDSS message mapping guides on the PHIN website • Identify and engage state system developers or vendor developers, alerting them to the availability of these guides • Provide a triage system that can manage technical assistance requests from the states • Prepare documentation that addresses most support issues • Establish a test and staging environment, in collaboration with the DMB team, to evaluate and certify NEDSS messages 70
Key Steps to Promoting Awareness and Providing Technical Support • Establish a CDC technical support team with the following skills: • • • HL 7 Orion Rhapsody and Symphonia SQL database JMS PHIN-MS and other messaging technologies Public health message mapping guides • Publish NEDSS message mapping guides on the PHIN website • Identify and engage state system developers or vendor developers, alerting them to the availability of these guides • Provide a triage system that can manage technical assistance requests from the states • Prepare documentation that addresses most support issues • Establish a test and staging environment, in collaboration with the DMB team, to evaluate and certify NEDSS messages 70
 Key Steps Required for CDC to Receive NEDSS Message Mapping Guide Data Production Environment • Set up PHIN-MS service action pair • Set up a PHIN-MS production receiver • Set up DMB production environment • Establish connectivity between DMB and PHIN-MS • Deploy DMB code in production • Implement reporting and production support procedures for providing feedback to states • Ready to accept production messages from states 71
Key Steps Required for CDC to Receive NEDSS Message Mapping Guide Data Production Environment • Set up PHIN-MS service action pair • Set up a PHIN-MS production receiver • Set up DMB production environment • Establish connectivity between DMB and PHIN-MS • Deploy DMB code in production • Implement reporting and production support procedures for providing feedback to states • Ready to accept production messages from states 71
 Key Steps to Develop and Support NEDSS Data Marts • Gather user requirements • Design Data Mart • Perform NETSS-to-NEDSS conversion, including diseasespecific data • Test conversion, perform quality control checks, load NETSS value sets • Prepare documentation • Data dictionary, document path and translations of data elements for QA/QC processes • Train data analysts in access and use of relational data • Convert existing SAS analysis programs from NETSS to NEDSS • Review Data Documentation, Data Mart, and SAS programs with Users • Perform User acceptance testing • Establish Change Control process • Establish error-checking with feedback to CDC program and data providers 72
Key Steps to Develop and Support NEDSS Data Marts • Gather user requirements • Design Data Mart • Perform NETSS-to-NEDSS conversion, including diseasespecific data • Test conversion, perform quality control checks, load NETSS value sets • Prepare documentation • Data dictionary, document path and translations of data elements for QA/QC processes • Train data analysts in access and use of relational data • Convert existing SAS analysis programs from NETSS to NEDSS • Review Data Documentation, Data Mart, and SAS programs with Users • Perform User acceptance testing • Establish Change Control process • Establish error-checking with feedback to CDC program and data providers 72
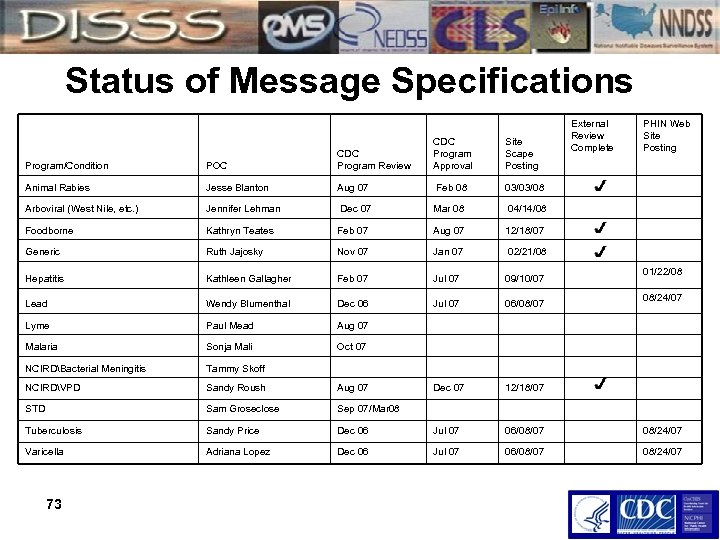 Status of Message Specifications CDC Program Approval Site Scape Posting External Review Complete PHIN Web Site Posting Program/Condition POC CDC Program Review Animal Rabies Jesse Blanton Aug 07 Feb 08 03/03/08 Arboviral (West Nile, etc. ) Jennifer Lehman Dec 07 Mar 08 04/14/08 Foodborne Kathryn Teates Feb 07 Aug 07 12/18/07 Generic Ruth Jajosky Nov 07 Jan 07 02/21/08 Hepatitis Kathleen Gallagher Feb 07 Jul 07 09/10/07 Lead Wendy Blumenthal Dec 06 Jul 07 06/08/07 Lyme Paul Mead Aug 07 Malaria Sonja Mali Oct 07 NCIRDBacterial Meningitis Tammy Skoff NCIRDVPD Sandy Roush Aug 07 Dec 07 12/18/07 STD Sam Groseclose Sep 07/Mar 08 Tuberculosis Sandy Price Dec 06 Jul 07 06/08/07 08/24/07 Varicella Adriana Lopez Dec 06 Jul 07 06/08/07 08/24/07 73 01/22/08 08/24/07
Status of Message Specifications CDC Program Approval Site Scape Posting External Review Complete PHIN Web Site Posting Program/Condition POC CDC Program Review Animal Rabies Jesse Blanton Aug 07 Feb 08 03/03/08 Arboviral (West Nile, etc. ) Jennifer Lehman Dec 07 Mar 08 04/14/08 Foodborne Kathryn Teates Feb 07 Aug 07 12/18/07 Generic Ruth Jajosky Nov 07 Jan 07 02/21/08 Hepatitis Kathleen Gallagher Feb 07 Jul 07 09/10/07 Lead Wendy Blumenthal Dec 06 Jul 07 06/08/07 Lyme Paul Mead Aug 07 Malaria Sonja Mali Oct 07 NCIRDBacterial Meningitis Tammy Skoff NCIRDVPD Sandy Roush Aug 07 Dec 07 12/18/07 STD Sam Groseclose Sep 07/Mar 08 Tuberculosis Sandy Price Dec 06 Jul 07 06/08/07 08/24/07 Varicella Adriana Lopez Dec 06 Jul 07 06/08/07 08/24/07 73 01/22/08 08/24/07
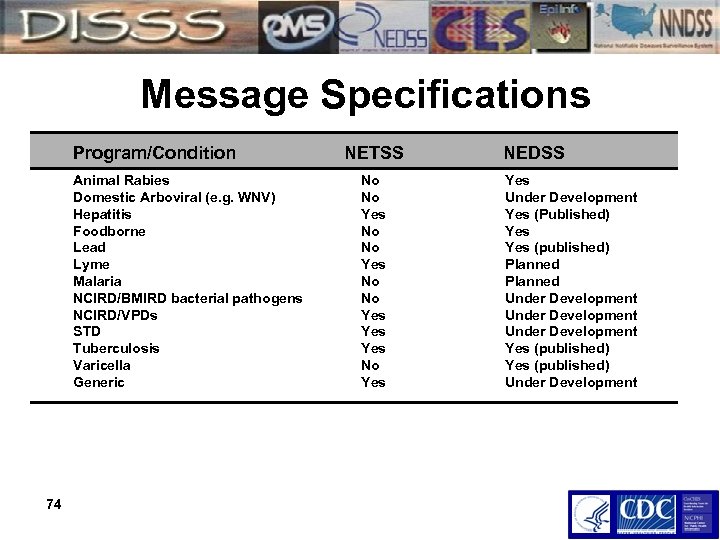 Message Specifications Program/Condition NETSS Animal Rabies Domestic Arboviral (e. g. WNV) Hepatitis Foodborne Lead Lyme Malaria NCIRD/BMIRD bacterial pathogens NCIRD/VPDs STD Tuberculosis Varicella Generic 74 No No Yes Yes No Yes NEDSS Yes Under Development Yes (Published) Yes (published) Planned Under Development Yes (published) Under Development
Message Specifications Program/Condition NETSS Animal Rabies Domestic Arboviral (e. g. WNV) Hepatitis Foodborne Lead Lyme Malaria NCIRD/BMIRD bacterial pathogens NCIRD/VPDs STD Tuberculosis Varicella Generic 74 No No Yes Yes No Yes NEDSS Yes Under Development Yes (Published) Yes (published) Planned Under Development Yes (published) Under Development
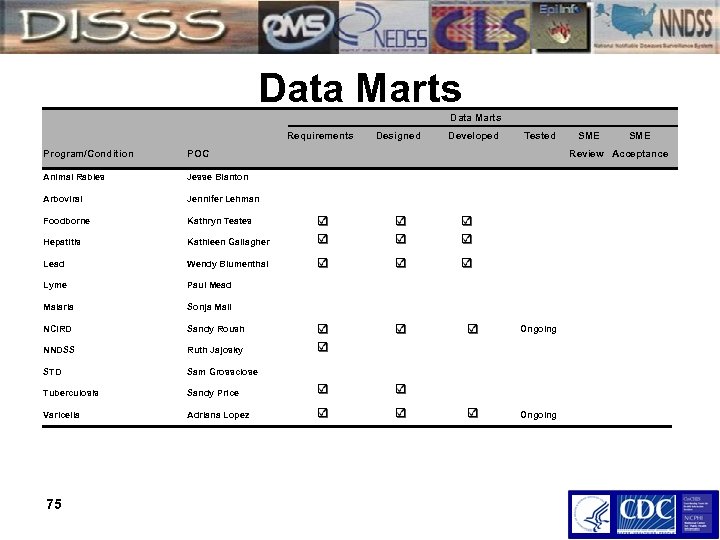 Data Marts Requirements Designed Developed Tested SME Program/Condition POC Animal Rabies Jesse Blanton Arboviral Jennifer Lehman Foodborne Kathryn Teates Hepatitis Kathleen Gallagher Lead Wendy Blumenthal Lyme Paul Mead Malaria Sonja Mali NCIRD Sandy Roush NNDSS Ruth Jajosky STD Sam Grossclose Tuberculosis Sandy Price Varicella Adriana Lopez 75 Review Acceptance Ongoing
Data Marts Requirements Designed Developed Tested SME Program/Condition POC Animal Rabies Jesse Blanton Arboviral Jennifer Lehman Foodborne Kathryn Teates Hepatitis Kathleen Gallagher Lead Wendy Blumenthal Lyme Paul Mead Malaria Sonja Mali NCIRD Sandy Roush NNDSS Ruth Jajosky STD Sam Grossclose Tuberculosis Sandy Price Varicella Adriana Lopez 75 Review Acceptance Ongoing
 NEDSS Major Stakeholders 76
NEDSS Major Stakeholders 76
 HHS, AHIC, OMB, GAO, U. S. Congress • National vision of integrated (interoperable) PHS systems o Ownership Status: CDC, in collaboration with national stakeholders • Standards for meeting NHIN/AHIC requirements o Biosurveillance MDS o Case Reports to PH o Notification Reports to CDC o Treatment messages Status: Widely known by the national community Status: CSTE and CDC working on this Status: Initial message mapping now posted to PHIN web site Status: Little work done, to date, for surveillance system support • Enhance biosurveillance data flow to U. S. agencies o National biosurveillance o IHRs o PAPHA o HSPD-21 Status: NEDSS data indirectly support this initiative Status: NEDSS data are part of the reporting system Status: NEDSS is part of this system Status: NNDSS is being considered to be focused and enhanced • Required government reports o CPIC/OMB 300 o EVM o 508 Compliance o OMB PRA o Audits, CJs, questions 77 Status: The NEDSS project is currently scoring 5 (out of 5) Status: This process is underway but moving slowly Status: A current report to HHS was provided in 2007 Status: Approval obtained, renewal package submitted to OMB Status: NEDSS continues to fare satisfactorily
HHS, AHIC, OMB, GAO, U. S. Congress • National vision of integrated (interoperable) PHS systems o Ownership Status: CDC, in collaboration with national stakeholders • Standards for meeting NHIN/AHIC requirements o Biosurveillance MDS o Case Reports to PH o Notification Reports to CDC o Treatment messages Status: Widely known by the national community Status: CSTE and CDC working on this Status: Initial message mapping now posted to PHIN web site Status: Little work done, to date, for surveillance system support • Enhance biosurveillance data flow to U. S. agencies o National biosurveillance o IHRs o PAPHA o HSPD-21 Status: NEDSS data indirectly support this initiative Status: NEDSS data are part of the reporting system Status: NEDSS is part of this system Status: NNDSS is being considered to be focused and enhanced • Required government reports o CPIC/OMB 300 o EVM o 508 Compliance o OMB PRA o Audits, CJs, questions 77 Status: The NEDSS project is currently scoring 5 (out of 5) Status: This process is underway but moving slowly Status: A current report to HHS was provided in 2007 Status: Approval obtained, renewal package submitted to OMB Status: NEDSS continues to fare satisfactorily
 CDC National Programs • CDC vision of integrated (interoperable) PHS systems o Integrated Surveillance o Who leads this effort at CDC Status: Definition still evolving within the CDC community Status: Tacit recognition of NCPHI role but inadequate collaboration • Access to data submitted in NEDSS format o Data access and analysis Status: Multi-year efforts beginning to show results • Standards and Informatics tools for CDC systems o PHIN-VADS o PHIN-MS o Notification Reports to CDC o MSS o PHIN-Dir o Ontology tools Status: Not widely known or embraced by the CDC community Status: Limited adoption by the CDC community Status: Limited awareness or adoption by the CDC community Status: Some interest by CDC community but limited adoption Status: Limited awareness or adoption by the CDC community Status: Near total unawareness by CDC community • Funding to support systems integration project efforts o System Development 78 Status: No support available
CDC National Programs • CDC vision of integrated (interoperable) PHS systems o Integrated Surveillance o Who leads this effort at CDC Status: Definition still evolving within the CDC community Status: Tacit recognition of NCPHI role but inadequate collaboration • Access to data submitted in NEDSS format o Data access and analysis Status: Multi-year efforts beginning to show results • Standards and Informatics tools for CDC systems o PHIN-VADS o PHIN-MS o Notification Reports to CDC o MSS o PHIN-Dir o Ontology tools Status: Not widely known or embraced by the CDC community Status: Limited adoption by the CDC community Status: Limited awareness or adoption by the CDC community Status: Some interest by CDC community but limited adoption Status: Limited awareness or adoption by the CDC community Status: Near total unawareness by CDC community • Funding to support systems integration project efforts o System Development 78 Status: No support available
 NEDSS States (non-NBS adopters) • National vision of integrated (interoperable) PHS systems o PHIN vision Status: Still evolving o Open source, SOA, Grid Status: Confusing to most stakeholders • Standards for meeting PHIN requirements o ELR o Case Notification to CDC o Case Reports to PH Status: Tech specs nearing completion; major challenges in tech support Status: Initial specs (v 1) completed, much remains; probable challenges in tech support Status: Prototype specs (v 1) completed, much remains; probable challenges in tech support • Funding (ELC, BT, other) assistance o ELC o BT Coop Ag o State IT support Status: Level funding Status: Risk of diminished funding Status: Frequently experiencing reductions and control by PH project managers • Informatics tools and services o MSS o PHIN-MS o PHIN-VADS o PHIN-Dir o Ontology tools 79 Status: Adoption increasing; challenges in tech support Status: Adoption widespread; challenges in tech support Status: Adoption limited; challenges in tech support Status: Pilot project, much remains to be done; challenges in tech support
NEDSS States (non-NBS adopters) • National vision of integrated (interoperable) PHS systems o PHIN vision Status: Still evolving o Open source, SOA, Grid Status: Confusing to most stakeholders • Standards for meeting PHIN requirements o ELR o Case Notification to CDC o Case Reports to PH Status: Tech specs nearing completion; major challenges in tech support Status: Initial specs (v 1) completed, much remains; probable challenges in tech support Status: Prototype specs (v 1) completed, much remains; probable challenges in tech support • Funding (ELC, BT, other) assistance o ELC o BT Coop Ag o State IT support Status: Level funding Status: Risk of diminished funding Status: Frequently experiencing reductions and control by PH project managers • Informatics tools and services o MSS o PHIN-MS o PHIN-VADS o PHIN-Dir o Ontology tools 79 Status: Adoption increasing; challenges in tech support Status: Adoption widespread; challenges in tech support Status: Adoption limited; challenges in tech support Status: Pilot project, much remains to be done; challenges in tech support
 NEDSS States (NBS adopters) • NBS evolves to better meet stakeholder needs o Continued Development Status: Contracts in place but scope will be limited o NBS Performance Status: Currently meeting stakeholder needs • NBS supports PHIN requirements o NBS Is PHIN-Compliant Status: Work underway to meet these requirements • Funding (ELC, BT, other) assistance o ELC o BT Coop Ag o State IT support Status: Level funding Status: Risk of diminished funding Status: Frequently experiencing reductions and control by PH project managers • Informatics tools and services o MSS o PHIN-MS o PHIN-VADS o PHIN-Dir o Ontology tools 80 Status: Supplied in NBS and MSS Status: Adoption limited; challenges in tech support Status: Pilot project, much remains to be done; challenges in tech support
NEDSS States (NBS adopters) • NBS evolves to better meet stakeholder needs o Continued Development Status: Contracts in place but scope will be limited o NBS Performance Status: Currently meeting stakeholder needs • NBS supports PHIN requirements o NBS Is PHIN-Compliant Status: Work underway to meet these requirements • Funding (ELC, BT, other) assistance o ELC o BT Coop Ag o State IT support Status: Level funding Status: Risk of diminished funding Status: Frequently experiencing reductions and control by PH project managers • Informatics tools and services o MSS o PHIN-MS o PHIN-VADS o PHIN-Dir o Ontology tools 80 Status: Supplied in NBS and MSS Status: Adoption limited; challenges in tech support Status: Pilot project, much remains to be done; challenges in tech support
 DISSS, NCPHI, CCHIS, CDC • CDC vision of integrated (interoperable) PHS systems o PHIN Vision o Open source, SOA, Grid Status: Improved, functional version Status: Conceptually clear but inadequately described operationally • Tight integration with other PHIN systems o OMS, Bio. Sense, etc. Status: This is the vision but challenges remain • Complete transition from NETSS to NEDSS o Retire NETSS Status: To be provided • Access to data submitted in NEDSS format o Data access and analysis Status: Multi-year efforts beginning to show results • Informatics tools and services o MSS o PHIN-MS o PHIN-VADS o PHIN-Dir o Ontology tools 81 Status: Project well established Status: A successful example of CDC informatics tools Status: A good model of CDC informatics tools that is not fully developed, yet Status: Pilot promising informatics tool that will have wide-ranging utility
DISSS, NCPHI, CCHIS, CDC • CDC vision of integrated (interoperable) PHS systems o PHIN Vision o Open source, SOA, Grid Status: Improved, functional version Status: Conceptually clear but inadequately described operationally • Tight integration with other PHIN systems o OMS, Bio. Sense, etc. Status: This is the vision but challenges remain • Complete transition from NETSS to NEDSS o Retire NETSS Status: To be provided • Access to data submitted in NEDSS format o Data access and analysis Status: Multi-year efforts beginning to show results • Informatics tools and services o MSS o PHIN-MS o PHIN-VADS o PHIN-Dir o Ontology tools 81 Status: Project well established Status: A successful example of CDC informatics tools Status: A good model of CDC informatics tools that is not fully developed, yet Status: Pilot promising informatics tool that will have wide-ranging utility
 NEDSS Future Vision and Lessons Learned
NEDSS Future Vision and Lessons Learned
 Future NEDSS Priorities • • Support national standards for exchange of electronic messages Provide tools, technical assistance, and support to stakeholders in achieving integrated surveillance system interoperability Develop and support at CDC NEDSS data access that fully meets stakeholder needs Develop and support an evaluation research agenda that focuses on local and state integrated surveillance systems Increase cooperative agreement support to states and major jurisdictions Ensure that the legacy NBS meets emerging messaging standards and other stakeholder requirements for usability and functionality • • 83
Future NEDSS Priorities • • Support national standards for exchange of electronic messages Provide tools, technical assistance, and support to stakeholders in achieving integrated surveillance system interoperability Develop and support at CDC NEDSS data access that fully meets stakeholder needs Develop and support an evaluation research agenda that focuses on local and state integrated surveillance systems Increase cooperative agreement support to states and major jurisdictions Ensure that the legacy NBS meets emerging messaging standards and other stakeholder requirements for usability and functionality • • 83
 Support national standards for exchange of electronic messages • Electronic Lab (result) Reporting (ELR) o o • PHIN Case Notifications (state-to-CDC) o o • CMR project in collaboration with CSTE and AHIC Biosurveillance Use Case (MDS) o 84 5 Guides now posted Multiple guides in final SME review PHIN Case Reports (provider-to-state, state-tostate, system-to-system) o • Lab. Corp, Mayo, ARUP collaboration PHIN ELR standard now published Bio. Sense split-feed pilot project in Texas
Support national standards for exchange of electronic messages • Electronic Lab (result) Reporting (ELR) o o • PHIN Case Notifications (state-to-CDC) o o • CMR project in collaboration with CSTE and AHIC Biosurveillance Use Case (MDS) o 84 5 Guides now posted Multiple guides in final SME review PHIN Case Reports (provider-to-state, state-tostate, system-to-system) o • Lab. Corp, Mayo, ARUP collaboration PHIN ELR standard now published Bio. Sense split-feed pilot project in Texas
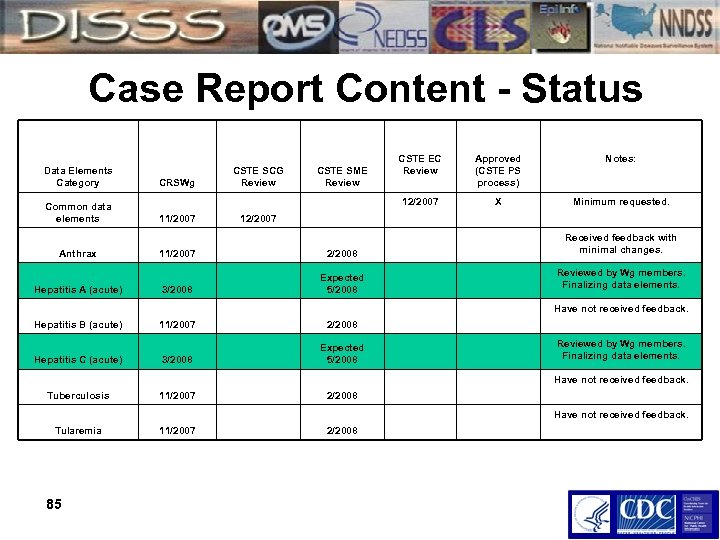 Case Report Content - Status Data Elements Category Common data elements Anthrax Hepatitis A (acute) 11/2007 CSTE SME Review CSTE EC Review Approved (CSTE PS process) Notes: 12/2007 CRSWg CSTE SCG Review X Minimum requested. 12/2007 11/2007 2/2008 Received feedback with minimal changes. 3/2008 Expected 5/2008 Reviewed by Wg members. Finalizing data elements. Have not received feedback. Hepatitis B (acute) Hepatitis C (acute) 11/2007 2/2008 3/2008 Expected 5/2008 Reviewed by Wg members. Finalizing data elements. Have not received feedback. Tuberculosis 11/2007 2/2008 Have not received feedback. Tularemia 85 11/2007 2/2008
Case Report Content - Status Data Elements Category Common data elements Anthrax Hepatitis A (acute) 11/2007 CSTE SME Review CSTE EC Review Approved (CSTE PS process) Notes: 12/2007 CRSWg CSTE SCG Review X Minimum requested. 12/2007 11/2007 2/2008 Received feedback with minimal changes. 3/2008 Expected 5/2008 Reviewed by Wg members. Finalizing data elements. Have not received feedback. Hepatitis B (acute) Hepatitis C (acute) 11/2007 2/2008 3/2008 Expected 5/2008 Reviewed by Wg members. Finalizing data elements. Have not received feedback. Tuberculosis 11/2007 2/2008 Have not received feedback. Tularemia 85 11/2007 2/2008
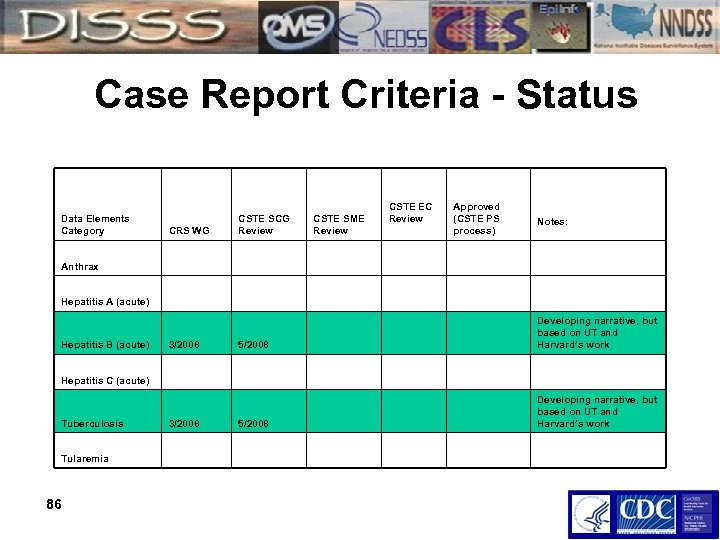 Case Report Criteria - Status Data Elements Category CRS WG CSTE SCG Review CSTE SME Review CSTE EC Review Approved (CSTE PS process) Notes: Anthrax Hepatitis A (acute) Hepatitis B (acute) 3/2008 5/2008 Developing narrative, but based on UT and Harvard’s work Hepatitis C (acute) Tuberculosis Tularemia 86 3/2008
Case Report Criteria - Status Data Elements Category CRS WG CSTE SCG Review CSTE SME Review CSTE EC Review Approved (CSTE PS process) Notes: Anthrax Hepatitis A (acute) Hepatitis B (acute) 3/2008 5/2008 Developing narrative, but based on UT and Harvard’s work Hepatitis C (acute) Tuberculosis Tularemia 86 3/2008
 Provide tools, technical assistance, and support to stakeholders in achieving integrated surveillance system interoperability • NEDSS Message Solution o o • Case Report Standards (CMR Project) o o • • 87 Software Training Collaboration, shared effort Funding Standards for core PH messages Foundation for system interoperability NBS to provide import/export of standard PH core messages Communicate these standards to vendors and other PH system developers
Provide tools, technical assistance, and support to stakeholders in achieving integrated surveillance system interoperability • NEDSS Message Solution o o • Case Report Standards (CMR Project) o o • • 87 Software Training Collaboration, shared effort Funding Standards for core PH messages Foundation for system interoperability NBS to provide import/export of standard PH core messages Communicate these standards to vendors and other PH system developers
 Develop and support at CDC NEDSS data access that fully meets stakeholder needs • • • CDC SME sign-off on data content before publishing new message guides CDC SME sign-off on design of new data marts Additional DMB and CDS resources o o o • 88 New CDC project manager New (very experienced) business analyst (contractor) New SAS programmer dedicated to working with CDC programs (contractor) Commitment from CCID IT Governance Team to focus on a limited number of programs, the publicize successes
Develop and support at CDC NEDSS data access that fully meets stakeholder needs • • • CDC SME sign-off on data content before publishing new message guides CDC SME sign-off on design of new data marts Additional DMB and CDS resources o o o • 88 New CDC project manager New (very experienced) business analyst (contractor) New SAS programmer dedicated to working with CDC programs (contractor) Commitment from CCID IT Governance Team to focus on a limited number of programs, the publicize successes
 Develop and support an evaluation research agenda that focuses on local and state integrated surveillance systems • • • 89 Washington State COE – Kitsap County Washington State – Seattle/King County New Mexico NEDSS project Other Co. E projects Other operational research projects
Develop and support an evaluation research agenda that focuses on local and state integrated surveillance systems • • • 89 Washington State COE – Kitsap County Washington State – Seattle/King County New Mexico NEDSS project Other Co. E projects Other operational research projects
 Increase cooperative agreement support to states and major jurisdictions • • 2006 NEDSS ELC funding: $8, 600, 000 2007 NEDSS ELC funding: $9, 700, 000 2007 NEDSS ELC supplemental funding: $300, 000 2008 NEDSS ELC funding: $10, 941, 082 TOTAL (2000 – 2008) $ 88, 160, 000 90
Increase cooperative agreement support to states and major jurisdictions • • 2006 NEDSS ELC funding: $8, 600, 000 2007 NEDSS ELC funding: $9, 700, 000 2007 NEDSS ELC supplemental funding: $300, 000 2008 NEDSS ELC funding: $10, 941, 082 TOTAL (2000 – 2008) $ 88, 160, 000 90
 NEDSS ELC Impact on States FY 08 • • 91 69. 33 IT staff directly supported 20. 72 Epi staff directly supported Total Award was $10, 941, 082. 33 Average Award for 56 jurisdictions was $195, 376. 47
NEDSS ELC Impact on States FY 08 • • 91 69. 33 IT staff directly supported 20. 72 Epi staff directly supported Total Award was $10, 941, 082. 33 Average Award for 56 jurisdictions was $195, 376. 47
 Ensure that the legacy NEDSS Base System meets emerging messaging standards and other stakeholder requirements for usability and functionality • NBS Development Team (CSC) increases from 16 to 23 • NBS stakeholders submit enhancement requests • NBS stakeholders participate in future release scope • NEDSS management team closely works with all NBS states to address system needs (e. g. performance, reliability, enhancements) 92
Ensure that the legacy NEDSS Base System meets emerging messaging standards and other stakeholder requirements for usability and functionality • NBS Development Team (CSC) increases from 16 to 23 • NBS stakeholders submit enhancement requests • NBS stakeholders participate in future release scope • NEDSS management team closely works with all NBS states to address system needs (e. g. performance, reliability, enhancements) 92
 NEDSS – Lessons Learned • Time to design, develop, implement is typically underestimated • Cost to design, develop, implement, maintain is typically underestimated • Public health technical support infrastructure is often challenged • Executive sponsorship remains an important component of a successful project 93
NEDSS – Lessons Learned • Time to design, develop, implement is typically underestimated • Cost to design, develop, implement, maintain is typically underestimated • Public health technical support infrastructure is often challenged • Executive sponsorship remains an important component of a successful project 93
 NEDSS: Lessons Learned (cont. ) • Verifying data completeness and accuracy is extremely challenging during a period of transition when different surveillance information systems are being used for different conditions in each state and within a state • A long moratorium on changes being made to NETSS legacy, message in addition to the long delay in NEDSS implementation, leaves CDC Programs without the ability to collect the data they need. 94 • History shows that CDC will develop other ways of collecting data, if approved solutions are not adequate. • Interim protocols should always be available
NEDSS: Lessons Learned (cont. ) • Verifying data completeness and accuracy is extremely challenging during a period of transition when different surveillance information systems are being used for different conditions in each state and within a state • A long moratorium on changes being made to NETSS legacy, message in addition to the long delay in NEDSS implementation, leaves CDC Programs without the ability to collect the data they need. 94 • History shows that CDC will develop other ways of collecting data, if approved solutions are not adequate. • Interim protocols should always be available
 NEDSS: Lessons Learned (cont. ) • The complexity of the NEDSS relational multitable database (compared to the flat NETSS single table database) imposes a burden on us in terms of training epidemiologists and data analysts to use NEDSS data • Informaticians and epidemiologists use different vocabularies, thus making communication difficult • States will need technical assistance to transition to NEDSS messaging • CDC programs have a concern about the resources needed for surveillance integration 95 (PAMS, Data Marts)
NEDSS: Lessons Learned (cont. ) • The complexity of the NEDSS relational multitable database (compared to the flat NETSS single table database) imposes a burden on us in terms of training epidemiologists and data analysts to use NEDSS data • Informaticians and epidemiologists use different vocabularies, thus making communication difficult • States will need technical assistance to transition to NEDSS messaging • CDC programs have a concern about the resources needed for surveillance integration 95 (PAMS, Data Marts)
 Intersection of Scientific Disciplines Surveillance Database Registration Reporting Confirmation Analyses Signal Detection Public Health Informatics Decision Support Feedback Acute Response IT Infrastructure Planned Response Action Adapted from: John H. Holmes, Ph. D, Center for Clinical Epidemiology and Biostatistics, University of Pennsylvania School of Medicine Health Informatics Public Health Activities Detection
Intersection of Scientific Disciplines Surveillance Database Registration Reporting Confirmation Analyses Signal Detection Public Health Informatics Decision Support Feedback Acute Response IT Infrastructure Planned Response Action Adapted from: John H. Holmes, Ph. D, Center for Clinical Epidemiology and Biostatistics, University of Pennsylvania School of Medicine Health Informatics Public Health Activities Detection
 Strengths, Weaknesses, Opportunities, and Threats • • Existing data remains siloed at the local and state level • Building systems non collaboratively leads to low adoption rates • 97 Politics of control of data has been the primary obstacle to formation of a national system Getting data directly from medical facilities is challenging; facilities have limited IT resources
Strengths, Weaknesses, Opportunities, and Threats • • Existing data remains siloed at the local and state level • Building systems non collaboratively leads to low adoption rates • 97 Politics of control of data has been the primary obstacle to formation of a national system Getting data directly from medical facilities is challenging; facilities have limited IT resources
 Tipping Point! Integrated Public Health Investments Produce Bio. Surveillance Value High “Do I really need this? ” “I can’t do without this!” Bio. Surveillance Value Bio. Surveillance Transformation Low Integrated PH Investments High
Tipping Point! Integrated Public Health Investments Produce Bio. Surveillance Value High “Do I really need this? ” “I can’t do without this!” Bio. Surveillance Value Bio. Surveillance Transformation Low Integrated PH Investments High


