f2409da2670ac5eff443c4a8cec14f71.ppt
- Количество слайдов: 27
 Evaluating and monitoring perinatal health in Europe Jennifer Zeitlin www. europeristat. com
Evaluating and monitoring perinatal health in Europe Jennifer Zeitlin www. europeristat. com
 The EURO-PERISTAT Project l Project aim: to develop valid and reliable indicators which can be used for monitoring and evaluating perinatal health in the EU l Project is part of the European Commission’s Public Health programme (DG-SANCO)
The EURO-PERISTAT Project l Project aim: to develop valid and reliable indicators which can be used for monitoring and evaluating perinatal health in the EU l Project is part of the European Commission’s Public Health programme (DG-SANCO)
 Perinatal Indicators – Why monitor across Europe ? l European countries face common challenges in perinatal health l Approaches to perinatal health differ greatly throughout Europe – – – l Comparing policies and outcomes Monitoring trends Developing European health policies Strength in numbers: attaining critical mass
Perinatal Indicators – Why monitor across Europe ? l European countries face common challenges in perinatal health l Approaches to perinatal health differ greatly throughout Europe – – – l Comparing policies and outcomes Monitoring trends Developing European health policies Strength in numbers: attaining critical mass
 Perinatal Indicators – Routine compilation across Europe l EUROSTAT, the WHO Regional Office for Europe, and the OECD compile a number of indicators related to perinatal health and care l However, many indicators are not compiled on a comparable basis, and key indicators for comparing perinatal health and the quality of health services are not available in all member states.
Perinatal Indicators – Routine compilation across Europe l EUROSTAT, the WHO Regional Office for Europe, and the OECD compile a number of indicators related to perinatal health and care l However, many indicators are not compiled on a comparable basis, and key indicators for comparing perinatal health and the quality of health services are not available in all member states.
 EURO-PERISTAT Network l Phase I: 15 Member states l Phase II & III 15 + 10 new MS + Norway l Phase IV: 27 MS + Norway, Switzerland + ? l Scientific Committee – – Phase I: One clinician (neonatologists, obstetrician, midwife) and epidemiologist from each country Phase II: one representative per country + a Scientific Advisory Group
EURO-PERISTAT Network l Phase I: 15 Member states l Phase II & III 15 + 10 new MS + Norway l Phase IV: 27 MS + Norway, Switzerland + ? l Scientific Committee – – Phase I: One clinician (neonatologists, obstetrician, midwife) and epidemiologist from each country Phase II: one representative per country + a Scientific Advisory Group
 EURO-PERISTAT Scope 3 primary components l Selection of an indicator set and development of new indicators l Collection of data on indicators l Reporting on indicators
EURO-PERISTAT Scope 3 primary components l Selection of an indicator set and development of new indicators l Collection of data on indicators l Reporting on indicators
 EURO-PERISTAT Indicators Based existing national and international indicators l A DELPHI consensus process to select indicators l – – PANEL: European clinicians (obstetrics, midwifery and neonatology) as well as epidemiologists and statisticians Updates: with new MS in 2004, next in 2011
EURO-PERISTAT Indicators Based existing national and international indicators l A DELPHI consensus process to select indicators l – – PANEL: European clinicians (obstetrics, midwifery and neonatology) as well as epidemiologists and statisticians Updates: with new MS in 2004, next in 2011
 EURO-PERISTAT Indicators ü 10 Core Indicators ü 23 Recommended Indicators • • ü 11 for immediate implementation 12 for further development Update in 2004 • 4 new indicators for development
EURO-PERISTAT Indicators ü 10 Core Indicators ü 23 Recommended Indicators • • ü 11 for immediate implementation 12 for further development Update in 2004 • 4 new indicators for development
 EURO-PERISTAT Indicators Core indicators are essential to monitoring perinatal health Recommended indicators are those considered desirable for a more complete picture of perinatal health across the member states and Indicators for further development represent important aspects of perinatal health, but further work is required before they can be operationalised in the member states.
EURO-PERISTAT Indicators Core indicators are essential to monitoring perinatal health Recommended indicators are those considered desirable for a more complete picture of perinatal health across the member states and Indicators for further development represent important aspects of perinatal health, but further work is required before they can be operationalised in the member states.
 EURO-PERISTAT Indicators Development of new indicators w Longer term health consequences of perinatal events for infant/child health w Maternal morbidity w Positive reproductive health outcomes (support provided to women, satisfaction with care) w Social inequalities in reproductive health care and outcomes
EURO-PERISTAT Indicators Development of new indicators w Longer term health consequences of perinatal events for infant/child health w Maternal morbidity w Positive reproductive health outcomes (support provided to women, satisfaction with care) w Social inequalities in reproductive health care and outcomes
 Data Collection l Verification of feasibility – – Data collection on year 2000 in PERISTAT I Data collection on year 2004 ü the European Journal of Obstetrics and Gynecology, Vol 111, Supp 1, 28 November 2003 ü The European Perinatal Health Report
Data Collection l Verification of feasibility – – Data collection on year 2000 in PERISTAT I Data collection on year 2004 ü the European Journal of Obstetrics and Gynecology, Vol 111, Supp 1, 28 November 2003 ü The European Perinatal Health Report
 Download report on www. europeristat. com
Download report on www. europeristat. com
 Data Collection: results l l l PERISTAT indicators available in many countries although not easily accessible Recommendations are REALISTIC Indicators useful NOW for monitoring and evaluating BUT – – – No indicator available for ALL countries Significant work to produce all of PERISTAT indicators Some essential dimensions very weak everywhere
Data Collection: results l l l PERISTAT indicators available in many countries although not easily accessible Recommendations are REALISTIC Indicators useful NOW for monitoring and evaluating BUT – – – No indicator available for ALL countries Significant work to produce all of PERISTAT indicators Some essential dimensions very weak everywhere
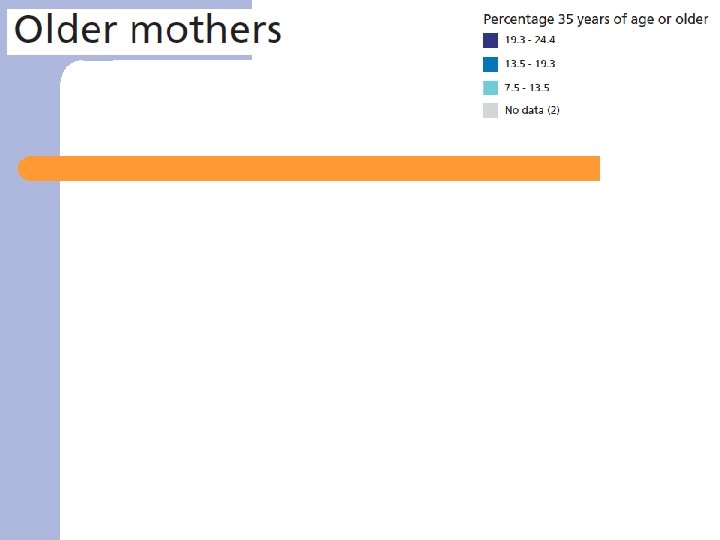
 Results – data from 2004 ü Population characteristics/Risk factors ü Health services ü Fetal/infant/child health ü Maternal health
Results – data from 2004 ü Population characteristics/Risk factors ü Health services ü Fetal/infant/child health ü Maternal health
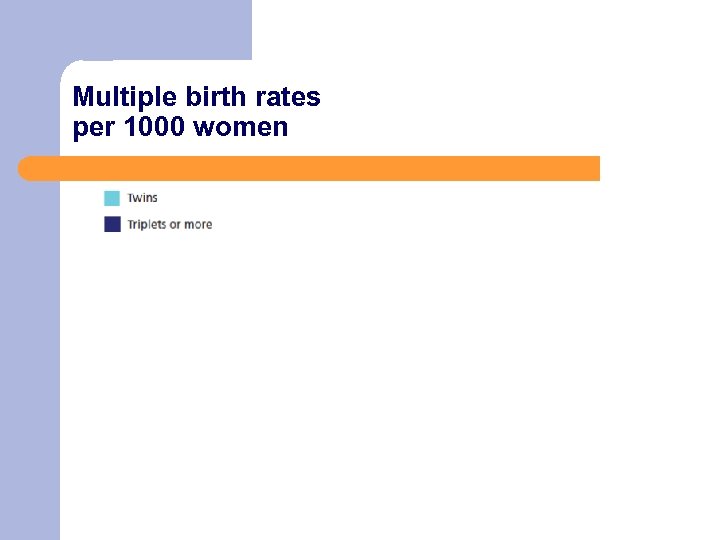 Multiple birth rates per 1000 women
Multiple birth rates per 1000 women
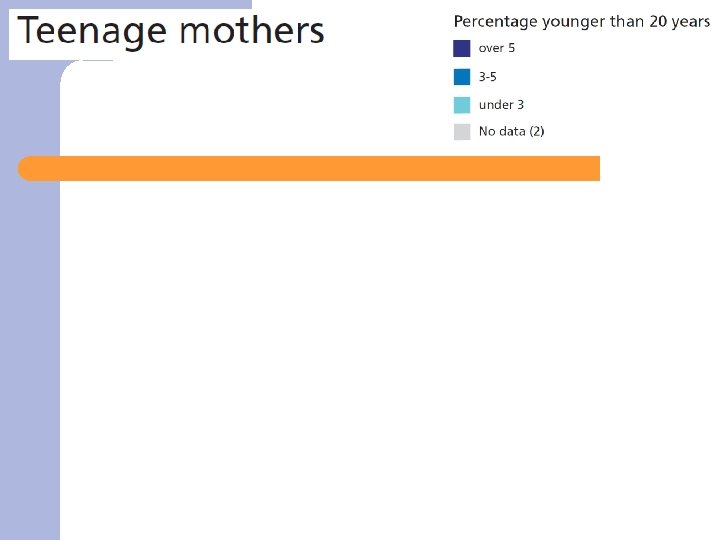
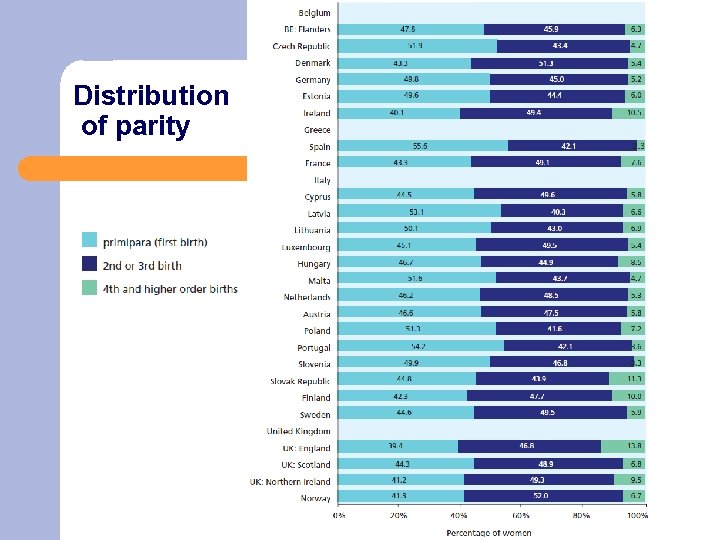 Distribution of parity
Distribution of parity
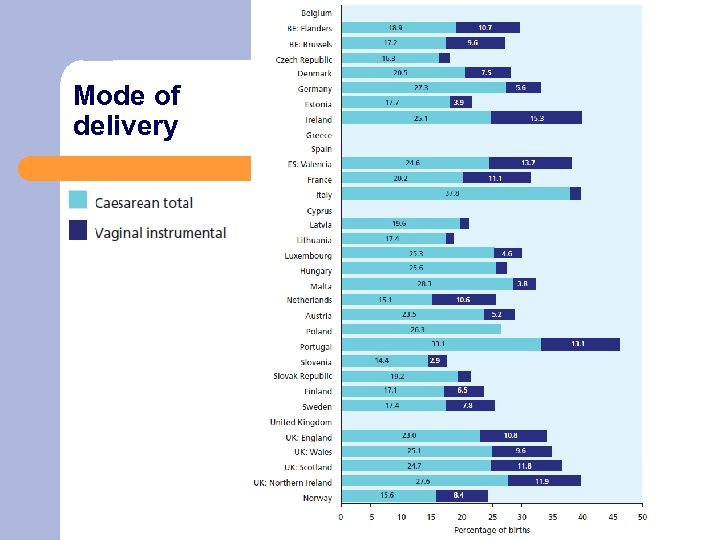 Mode of delivery
Mode of delivery
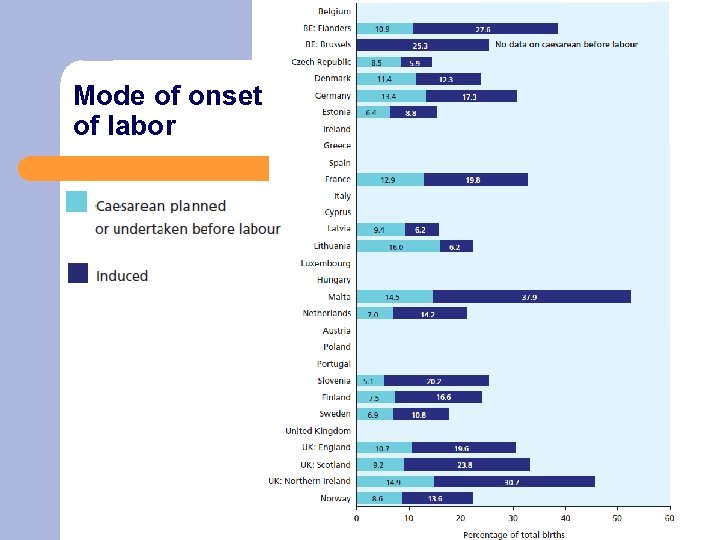 Mode of onset of labor
Mode of onset of labor
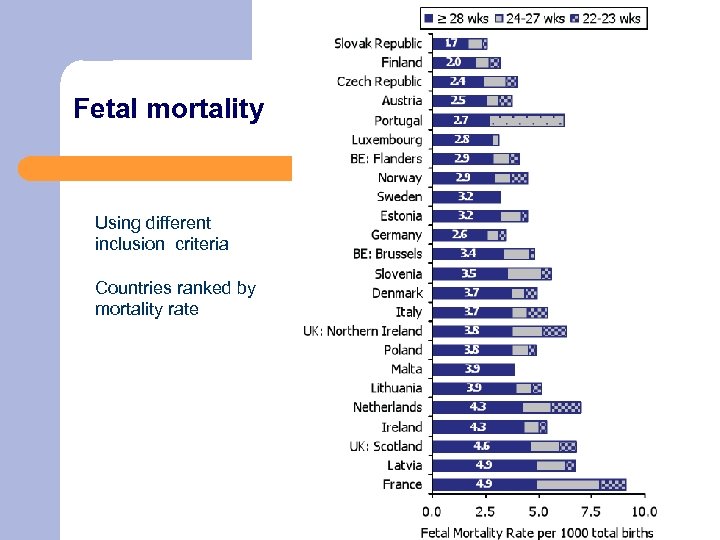 Fetal mortality Using different inclusion criteria Countries ranked by mortality rate
Fetal mortality Using different inclusion criteria Countries ranked by mortality rate
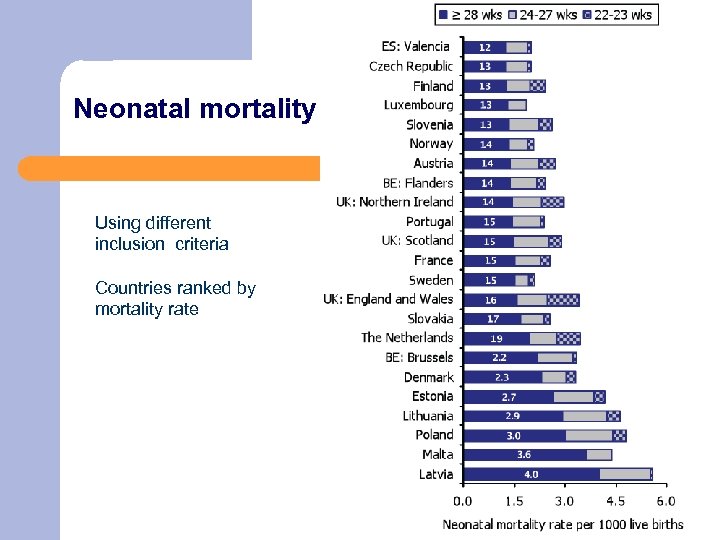 Neonatal mortality Using different inclusion criteria Countries ranked by mortality rate
Neonatal mortality Using different inclusion criteria Countries ranked by mortality rate
 Next steps: Euro-peristat Action l l New project begins in April 2011 Objectives Integrate Euro-peristat indicators into routine databases (EUROSTAT, ECHIM) Create a sustainable network for reporting on health indicators Produce a new report on data from 2010
Next steps: Euro-peristat Action l l New project begins in April 2011 Objectives Integrate Euro-peristat indicators into routine databases (EUROSTAT, ECHIM) Create a sustainable network for reporting on health indicators Produce a new report on data from 2010
 Disseminate and publish our work to policy makers, clinicians & users Health Information is not Health ≠ But good health policies and practices need good evidence
Disseminate and publish our work to policy makers, clinicians & users Health Information is not Health ≠ But good health policies and practices need good evidence
 Executive Board • • • Sophie Alexander, Université Libre de Bruxelles, School of Public Health Henrique Barros, University of Porto Medical School István Berbik, Vaszary Kolos Teaching Hospital Béatrice Blondel, INSERM U 953 Marie-Hélène Bouvier-Colle, INSERM U 953 Simone Buitendijk TNO Institute Prevention and Health Mika Gissler THL National Institute for Health and Welfare Alison Macfarlane City University, Department of Midwifery Ashna Mohangoo TNO Institute Prevention and Health Živa Novak-Antolic University Medical Centre Katarzyna Szamotulska National Research Institute of Mother and Child Jennifer Zeitlin INSERM U 953 (project leader)
Executive Board • • • Sophie Alexander, Université Libre de Bruxelles, School of Public Health Henrique Barros, University of Porto Medical School István Berbik, Vaszary Kolos Teaching Hospital Béatrice Blondel, INSERM U 953 Marie-Hélène Bouvier-Colle, INSERM U 953 Simone Buitendijk TNO Institute Prevention and Health Mika Gissler THL National Institute for Health and Welfare Alison Macfarlane City University, Department of Midwifery Ashna Mohangoo TNO Institute Prevention and Health Živa Novak-Antolic University Medical Centre Katarzyna Szamotulska National Research Institute of Mother and Child Jennifer Zeitlin INSERM U 953 (project leader)
 Scientific committee and data providers l Listed at: http: //www. europeristat. com/Network/Scientific. Committee/index. shtml
Scientific committee and data providers l Listed at: http: //www. europeristat. com/Network/Scientific. Committee/index. shtml
 More information: www. europeristat. com
More information: www. europeristat. com


