697dfd0f03644e598eb4dd728e8d0b88.ppt
- Количество слайдов: 97
 Eva Rentschler Universität Mainz The „metal-radical approach“ toward magnetic materials workshop synthetic strategies towards. . Kaiserslautern, 23. -25. 10. 2005
Eva Rentschler Universität Mainz The „metal-radical approach“ toward magnetic materials workshop synthetic strategies towards. . Kaiserslautern, 23. -25. 10. 2005
 “Toward Molecular Magnets: The metal-radical approach“ Caneschi, D. Gatteschi, R. Sessoli, P. Rey, Acc. Chem. Res. 22, 392 (1989). The strategy is simple: - Strong direct metal-ligand magnetic exchange interactions are achieved from the coordination of stable free radicals to paramagnetic transition-metal ions, - and if these interactions are extended in one, two, or three spatial directions, cooperative magnetic behavior is obtainable in these molecule-based systems. Lemaire, Pure Appl. Chem. , Vol. 76, No. 2, pp. 277– 293, 2004.
“Toward Molecular Magnets: The metal-radical approach“ Caneschi, D. Gatteschi, R. Sessoli, P. Rey, Acc. Chem. Res. 22, 392 (1989). The strategy is simple: - Strong direct metal-ligand magnetic exchange interactions are achieved from the coordination of stable free radicals to paramagnetic transition-metal ions, - and if these interactions are extended in one, two, or three spatial directions, cooperative magnetic behavior is obtainable in these molecule-based systems. Lemaire, Pure Appl. Chem. , Vol. 76, No. 2, pp. 277– 293, 2004.
 “Toward Molecular Magnets: The metal-radical approach“ Caneschi, D. Gatteschi, R. Sessoli, P. Rey, Acc. Chem. Res. 22, 392 (1989). - Since (and prior to) 1989, literally hundreds of metal-radical complexes have been reported, including a number of magnetically ordered materials. - A wealth of knowledge about the structure and magnetic properties of coordination complexes containing stable radical ligands has been unearthed, and as a result, the metal-radical approach is recognized as one of the more fruitful efforts toward molecular magnetic materials. Lemaire, Pure Appl. Chem. , Vol. 76, No. 2, pp. 277– 293, 2004.
“Toward Molecular Magnets: The metal-radical approach“ Caneschi, D. Gatteschi, R. Sessoli, P. Rey, Acc. Chem. Res. 22, 392 (1989). - Since (and prior to) 1989, literally hundreds of metal-radical complexes have been reported, including a number of magnetically ordered materials. - A wealth of knowledge about the structure and magnetic properties of coordination complexes containing stable radical ligands has been unearthed, and as a result, the metal-radical approach is recognized as one of the more fruitful efforts toward molecular magnetic materials. Lemaire, Pure Appl. Chem. , Vol. 76, No. 2, pp. 277– 293, 2004.
 The families of radicals to be discussed are limited to stable, isolable free-radical species i. e. , radicals that can be prepared and stored under ambient conditions.
The families of radicals to be discussed are limited to stable, isolable free-radical species i. e. , radicals that can be prepared and stored under ambient conditions.
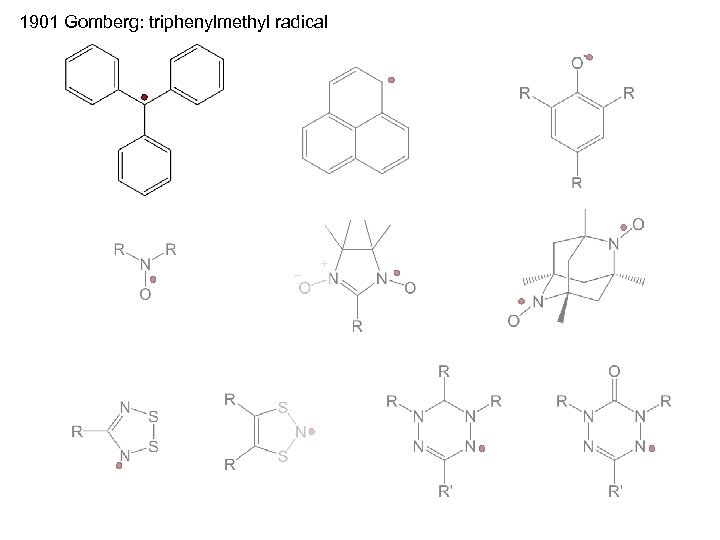 1901 Gomberg: triphenylmethyl radical
1901 Gomberg: triphenylmethyl radical
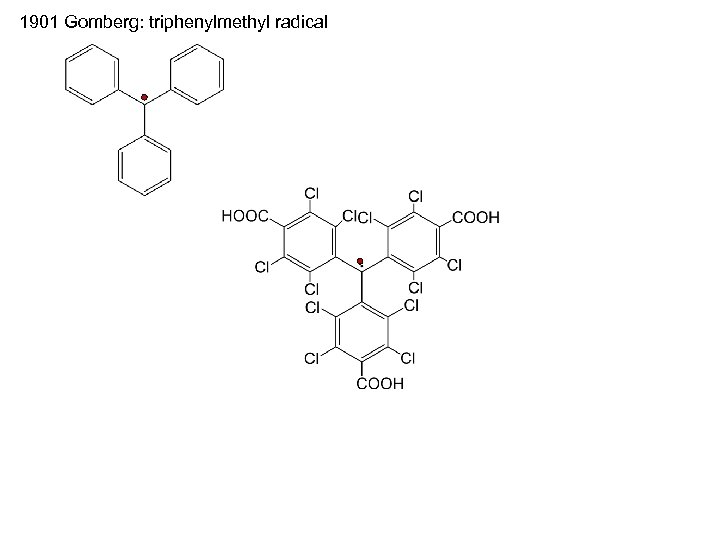 1901 Gomberg: triphenylmethyl radical
1901 Gomberg: triphenylmethyl radical
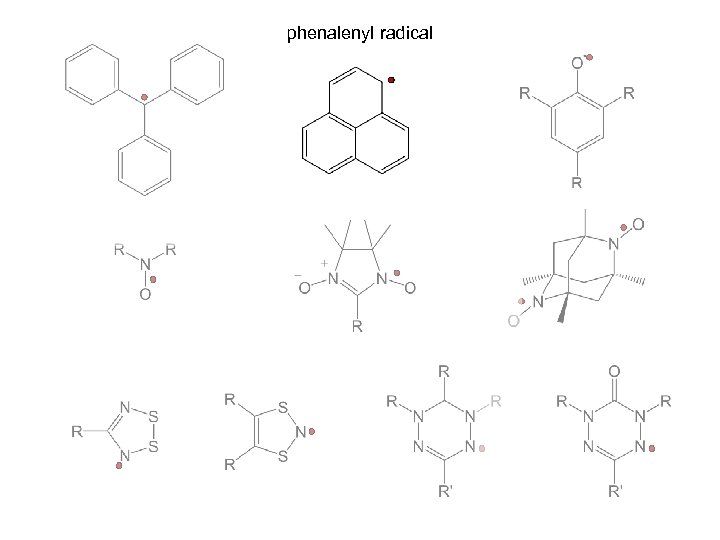 phenalenyl radical
phenalenyl radical
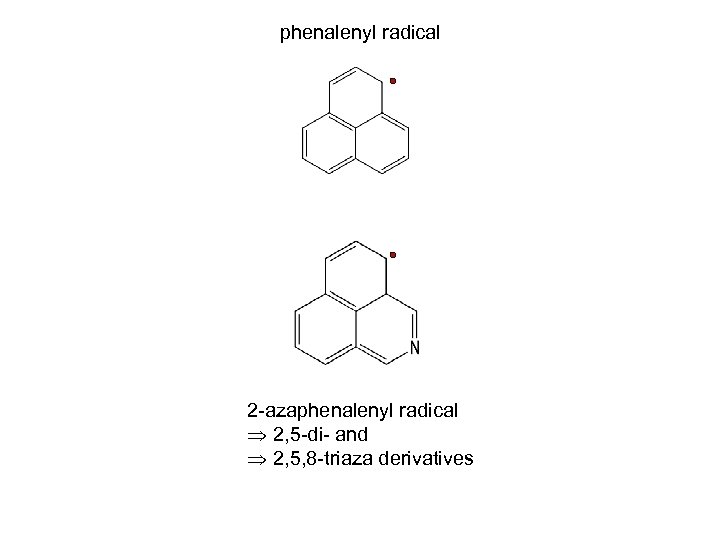 phenalenyl radical 2 -azaphenalenyl radical 2, 5 -di- and 2, 5, 8 -triaza derivatives
phenalenyl radical 2 -azaphenalenyl radical 2, 5 -di- and 2, 5, 8 -triaza derivatives
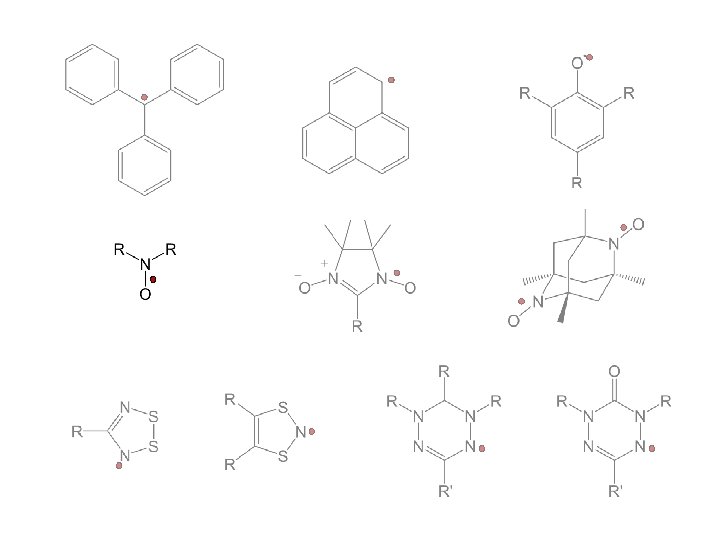
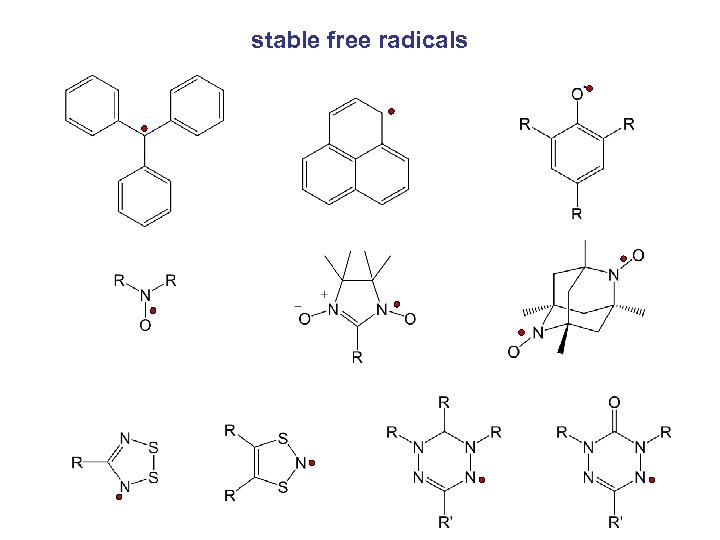 stable free radicals
stable free radicals
![Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ = Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ =](https://present5.com/presentation/697dfd0f03644e598eb4dd728e8d0b88/image-12.jpg) Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ = 7, 7, 8, 8 -tetracyano-p-quinodimethane, TCNE = tetracyanoethene
Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ = 7, 7, 8, 8 -tetracyano-p-quinodimethane, TCNE = tetracyanoethene
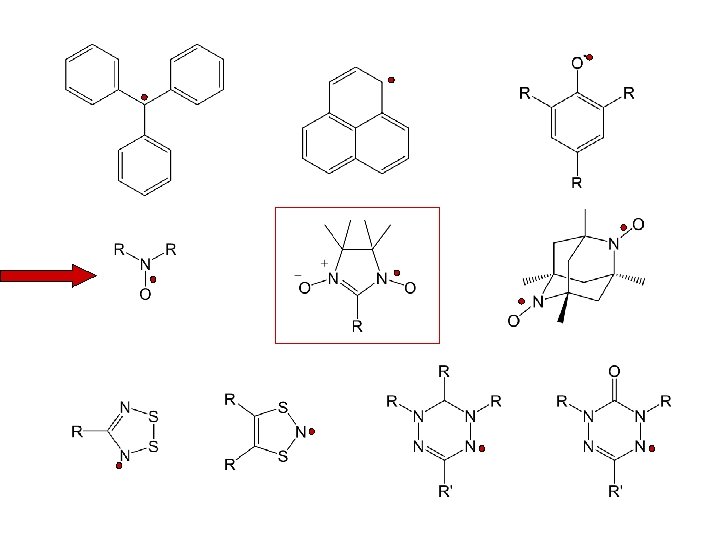
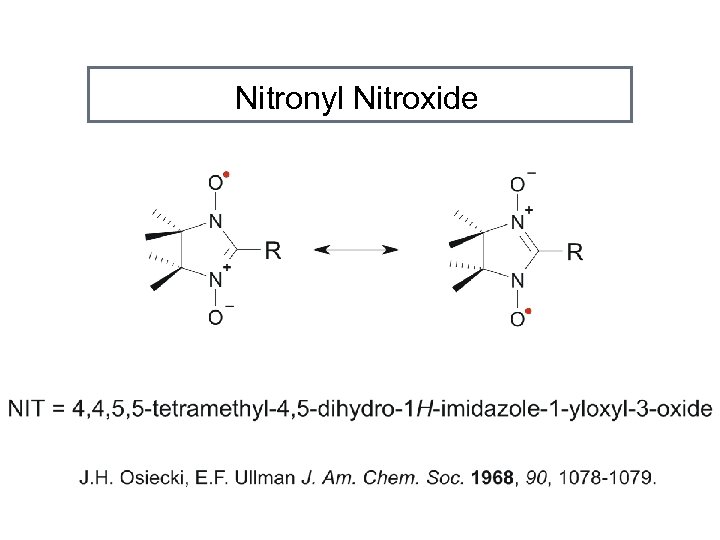 Nitronyl Nitroxide
Nitronyl Nitroxide
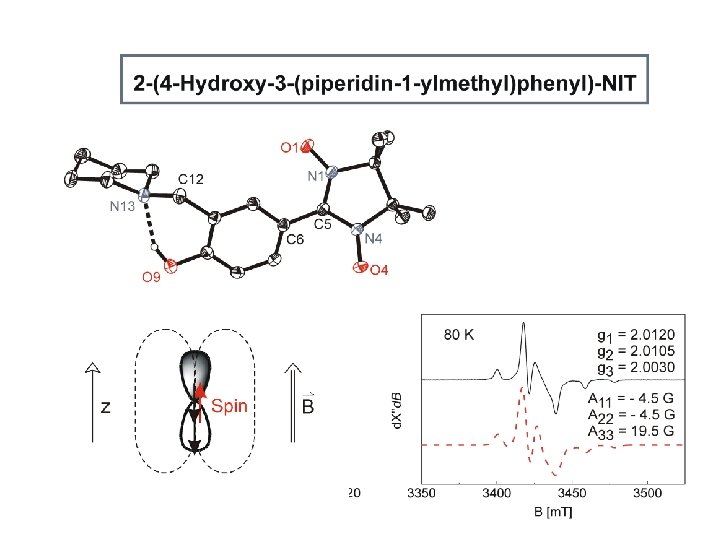
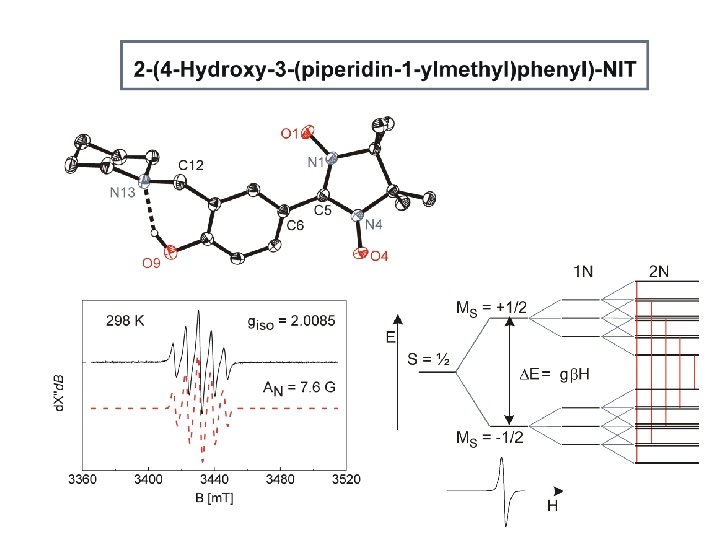 N 1 – O 1 1. 285(1) Å N 4 – O 4 1. 287(1) Å N – O 1. 143 Å N = O 1. 316 Å C 5 – N 1. 353(1) Å C – N 1. 438 Å C = N 1. 260 Å
N 1 – O 1 1. 285(1) Å N 4 – O 4 1. 287(1) Å N – O 1. 143 Å N = O 1. 316 Å C 5 – N 1. 353(1) Å C – N 1. 438 Å C = N 1. 260 Å
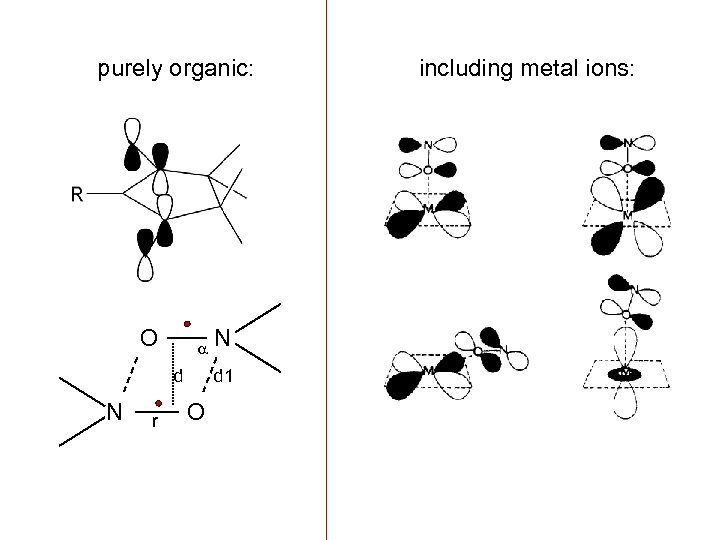 purely organic: O d N r N d 1 O including metal ions:
purely organic: O d N r N d 1 O including metal ions:
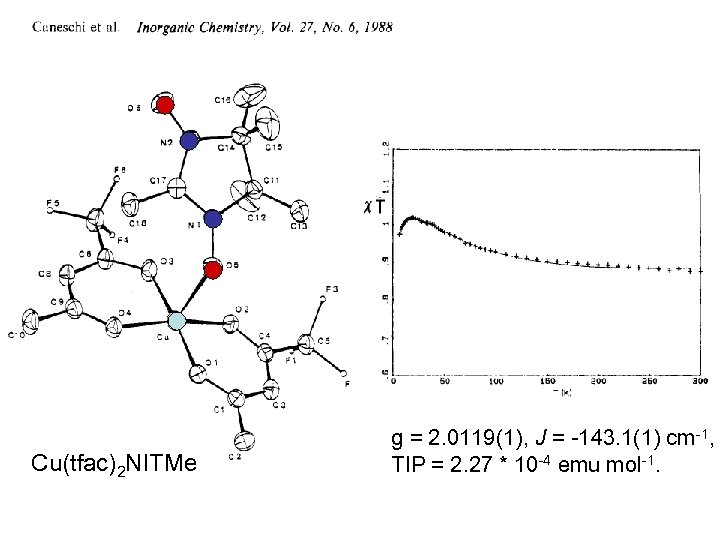 Cu(tfac)2 NITMe g = 2. 0119(1), J = -143. 1(1) cm-1, TIP = 2. 27 * 10 -4 emu mol-1.
Cu(tfac)2 NITMe g = 2. 0119(1), J = -143. 1(1) cm-1, TIP = 2. 27 * 10 -4 emu mol-1.
 Typical Values of the Magnetic Coupling Constant J for Metal-Nitroside Complexes metal ion type of coupling copper(II)b copper(II)c nickel(II) cobalt(II) manganese(II) AF F AF AF AF J, a cm-1 500 -10 to 70 500 300 150 -300 a Positive J means antiferromagnetic coupling. The energy separation between singlet and triplet is J. b The nitroxide is an equatorial site. c The nitroxide in an axial site. Note: H = J S 1. S 2
Typical Values of the Magnetic Coupling Constant J for Metal-Nitroside Complexes metal ion type of coupling copper(II)b copper(II)c nickel(II) cobalt(II) manganese(II) AF F AF AF AF J, a cm-1 500 -10 to 70 500 300 150 -300 a Positive J means antiferromagnetic coupling. The energy separation between singlet and triplet is J. b The nitroxide is an equatorial site. c The nitroxide in an axial site. Note: H = J S 1. S 2
 rel. weak interaction between the magnetic orbitals the metal-radical overlap is small, energy separation between the two orbitals is large molecular orbitals 1 and 2 mainly localized on metal and on the radical fragment, respectively. Since ( 2 - 2)1/2 S JAF ( 2 - 2)1/2 S JAF is determined by the variation of the squared overlap between the magnetic orbitals. J = 2 k + 4 S J 2 J S 2
rel. weak interaction between the magnetic orbitals the metal-radical overlap is small, energy separation between the two orbitals is large molecular orbitals 1 and 2 mainly localized on metal and on the radical fragment, respectively. Since ( 2 - 2)1/2 S JAF ( 2 - 2)1/2 S JAF is determined by the variation of the squared overlap between the magnetic orbitals. J = 2 k + 4 S J 2 J S 2
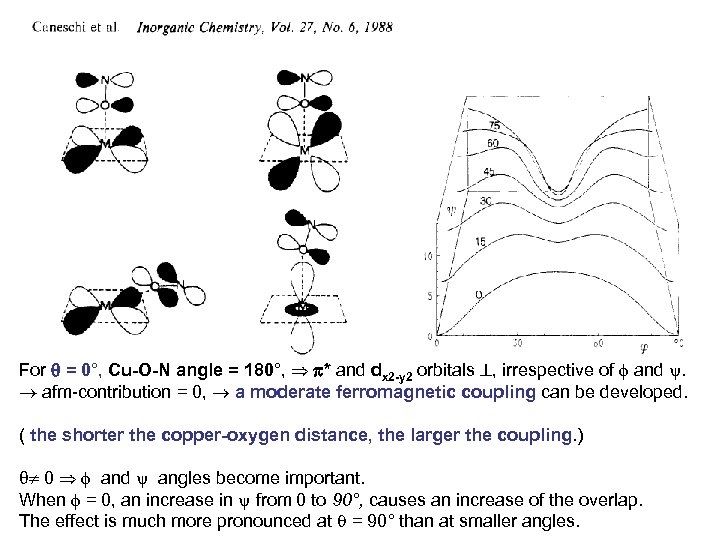 For = 0°, Cu-O-N angle = 180°, * and dx 2 -y 2 orbitals , irrespective of and . afm-contribution = 0, a moderate ferromagnetic coupling can be developed. ( the shorter the copper-oxygen distance, the larger the coupling. ) 0 and angles become important. When = 0, an increase in from 0 to 90°, causes an increase of the overlap. The effect is much more pronounced at = 90° than at smaller angles.
For = 0°, Cu-O-N angle = 180°, * and dx 2 -y 2 orbitals , irrespective of and . afm-contribution = 0, a moderate ferromagnetic coupling can be developed. ( the shorter the copper-oxygen distance, the larger the coupling. ) 0 and angles become important. When = 0, an increase in from 0 to 90°, causes an increase of the overlap. The effect is much more pronounced at = 90° than at smaller angles.
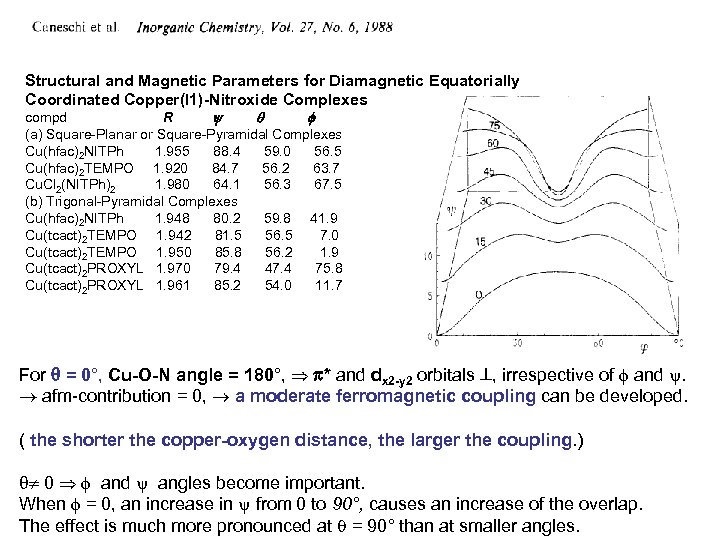 Structural and Magnetic Parameters for Diamagnetic Equatorially Coordinated Copper(I 1)-Nitroxide Complexes compd R (a) Square-Planar or Square-Pyramidal Complexes Cu(hfac)2 NITPh 1. 955 88. 4 59. 0 56. 5 Cu(hfac)2 TEMPO 1. 920 84. 7 56. 2 63. 7 Cu. Cl 2(NITPh)2 1. 980 64. 1 56. 3 67. 5 (b) Trigonal-Pyramidal Complexes Cu(hfac)2 NITPh 1. 948 80. 2 59. 8 41. 9 Cu(tcact)2 TEMPO 1. 942 81. 5 56. 5 7. 0 Cu(tcact)2 TEMPO 1. 950 85. 8 56. 2 1. 9 Cu(tcact)2 PROXYL 1. 970 79. 4 47. 4 75. 8 Cu(tcact)2 PROXYL 1. 961 85. 2 54. 0 11. 7 For = 0°, Cu-O-N angle = 180°, * and dx 2 -y 2 orbitals , irrespective of and . afm-contribution = 0, a moderate ferromagnetic coupling can be developed. ( the shorter the copper-oxygen distance, the larger the coupling. ) 0 and angles become important. When = 0, an increase in from 0 to 90°, causes an increase of the overlap. The effect is much more pronounced at = 90° than at smaller angles.
Structural and Magnetic Parameters for Diamagnetic Equatorially Coordinated Copper(I 1)-Nitroxide Complexes compd R (a) Square-Planar or Square-Pyramidal Complexes Cu(hfac)2 NITPh 1. 955 88. 4 59. 0 56. 5 Cu(hfac)2 TEMPO 1. 920 84. 7 56. 2 63. 7 Cu. Cl 2(NITPh)2 1. 980 64. 1 56. 3 67. 5 (b) Trigonal-Pyramidal Complexes Cu(hfac)2 NITPh 1. 948 80. 2 59. 8 41. 9 Cu(tcact)2 TEMPO 1. 942 81. 5 56. 5 7. 0 Cu(tcact)2 TEMPO 1. 950 85. 8 56. 2 1. 9 Cu(tcact)2 PROXYL 1. 970 79. 4 47. 4 75. 8 Cu(tcact)2 PROXYL 1. 961 85. 2 54. 0 11. 7 For = 0°, Cu-O-N angle = 180°, * and dx 2 -y 2 orbitals , irrespective of and . afm-contribution = 0, a moderate ferromagnetic coupling can be developed. ( the shorter the copper-oxygen distance, the larger the coupling. ) 0 and angles become important. When = 0, an increase in from 0 to 90°, causes an increase of the overlap. The effect is much more pronounced at = 90° than at smaller angles.
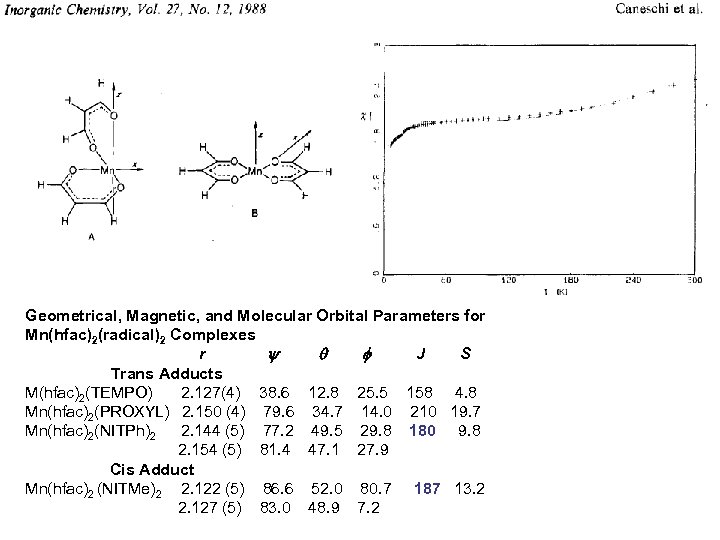 Geometrical, Magnetic, and Molecular Orbital Parameters for Mn(hfac)2(radical)2 Complexes r J S Trans Adducts M(hfac)2(TEMPO) 2. 127(4) 38. 6 12. 8 25. 5 158 4. 8 Mn(hfac)2(PROXYL) 2. 150 (4) 79. 6 34. 7 14. 0 210 19. 7 Mn(hfac)2(NITPh)2 2. 144 (5) 77. 2 49. 5 29. 8 180 9. 8 2. 154 (5) 81. 4 47. 1 27. 9 Cis Adduct Mn(hfac)2 (NITMe)2 2. 122 (5) 86. 6 52. 0 80. 7 187 13. 2 2. 127 (5) 83. 0 48. 9 7. 2
Geometrical, Magnetic, and Molecular Orbital Parameters for Mn(hfac)2(radical)2 Complexes r J S Trans Adducts M(hfac)2(TEMPO) 2. 127(4) 38. 6 12. 8 25. 5 158 4. 8 Mn(hfac)2(PROXYL) 2. 150 (4) 79. 6 34. 7 14. 0 210 19. 7 Mn(hfac)2(NITPh)2 2. 144 (5) 77. 2 49. 5 29. 8 180 9. 8 2. 154 (5) 81. 4 47. 1 27. 9 Cis Adduct Mn(hfac)2 (NITMe)2 2. 122 (5) 86. 6 52. 0 80. 7 187 13. 2 2. 127 (5) 83. 0 48. 9 7. 2
 bridging Nitronyl Nitroxide radicals
bridging Nitronyl Nitroxide radicals
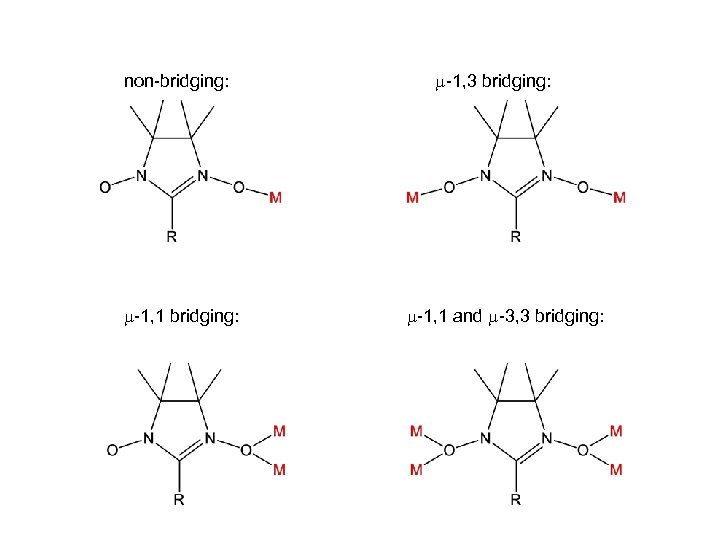 non-bridging: -1, 1 bridging: -1, 3 bridging: -1, 1 and -3, 3 bridging:
non-bridging: -1, 1 bridging: -1, 3 bridging: -1, 1 and -3, 3 bridging:
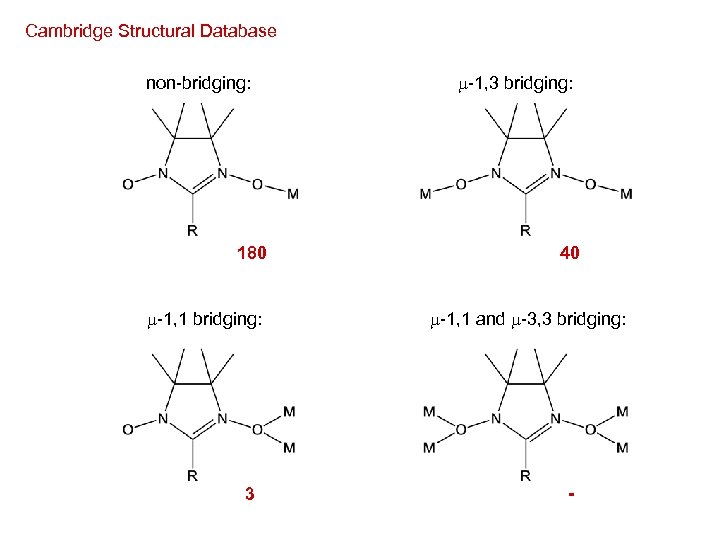 Cambridge Structural Database non-bridging: 180 -1, 1 bridging: 3 -1, 3 bridging: 40 -1, 1 and -3, 3 bridging: -
Cambridge Structural Database non-bridging: 180 -1, 1 bridging: 3 -1, 3 bridging: 40 -1, 1 and -3, 3 bridging: -
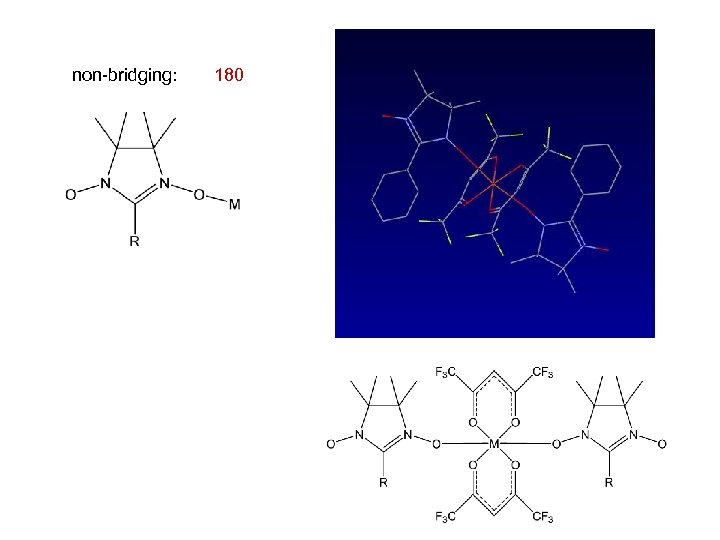 non-bridging: 180
non-bridging: 180
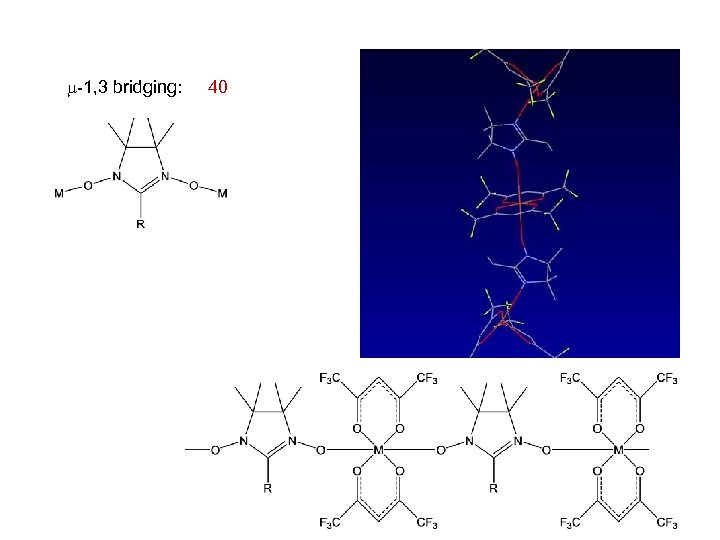 -1, 3 bridging: 40
-1, 3 bridging: 40
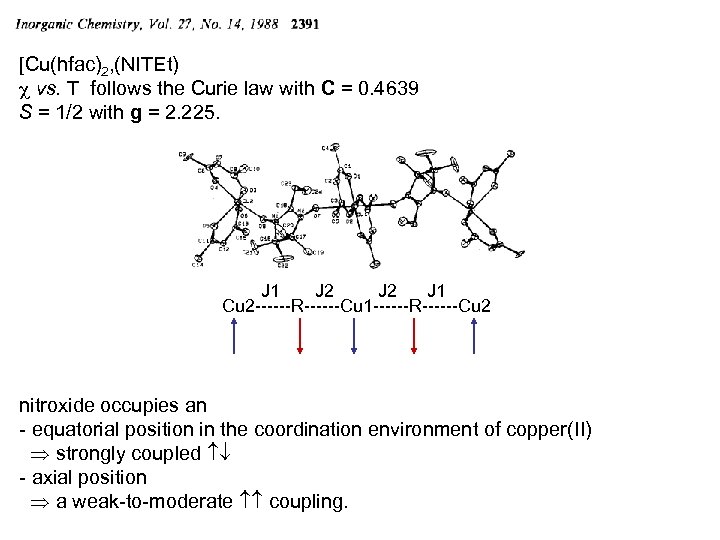 [Cu(hfac)2, (NITEt) c vs. T follows the Curie law with C = 0. 4639 S = 1/2 with g = 2. 225. J 1 J 2 J 1 Cu 2 ------R------Cu 1 ------R------Cu 2 nitroxide occupies an - equatorial position in the coordination environment of copper(II) strongly coupled - axial position a weak-to-moderate coupling.
[Cu(hfac)2, (NITEt) c vs. T follows the Curie law with C = 0. 4639 S = 1/2 with g = 2. 225. J 1 J 2 J 1 Cu 2 ------R------Cu 1 ------R------Cu 2 nitroxide occupies an - equatorial position in the coordination environment of copper(II) strongly coupled - axial position a weak-to-moderate coupling.
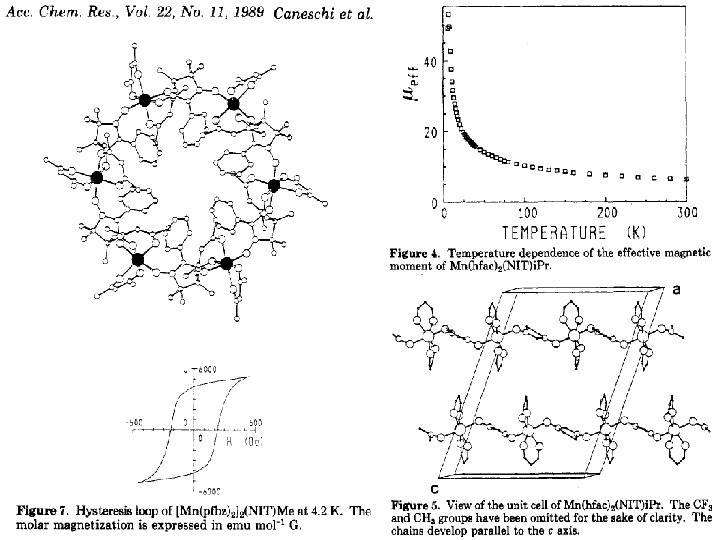
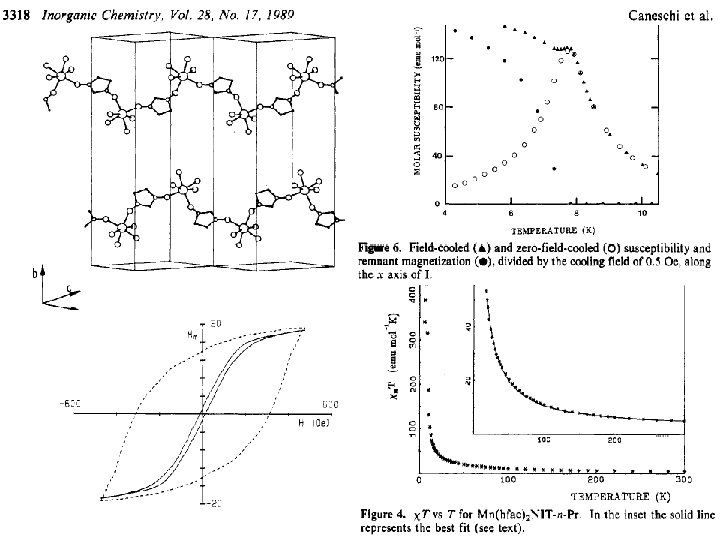
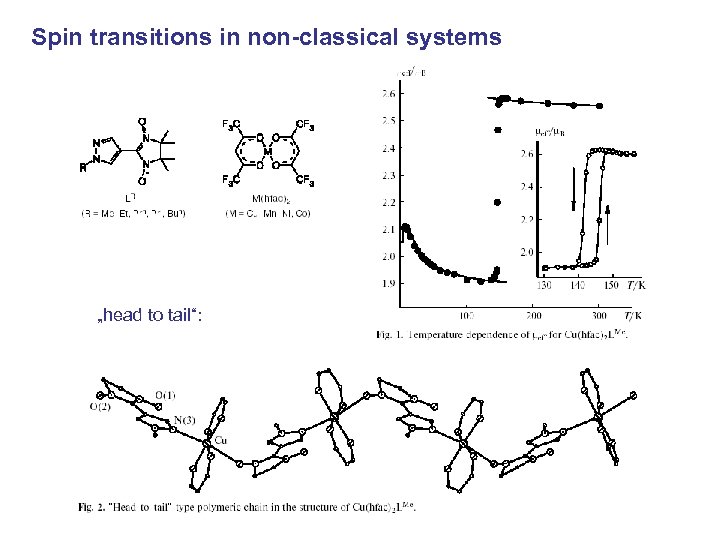 Spin transitions in non-classical systems „head to tail“:
Spin transitions in non-classical systems „head to tail“:
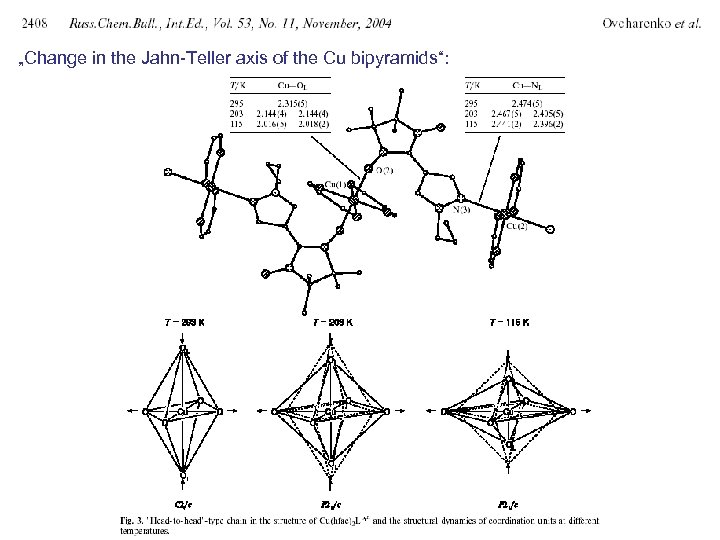 „Change in the Jahn-Teller axis of the Cu bipyramids“:
„Change in the Jahn-Teller axis of the Cu bipyramids“:
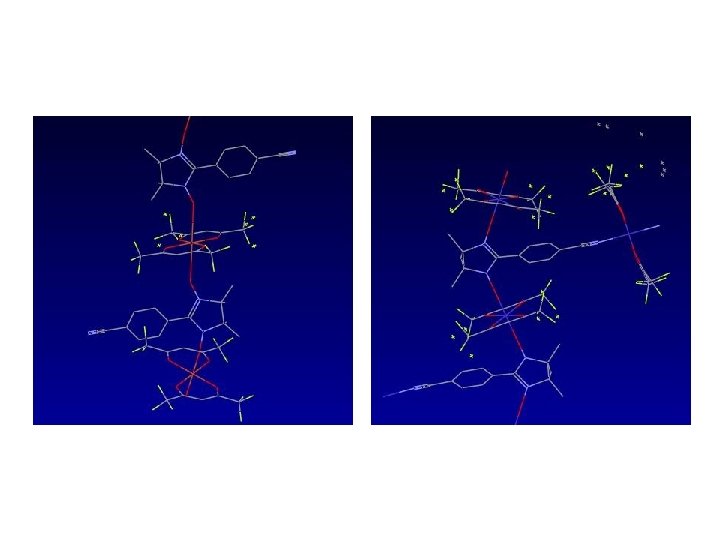
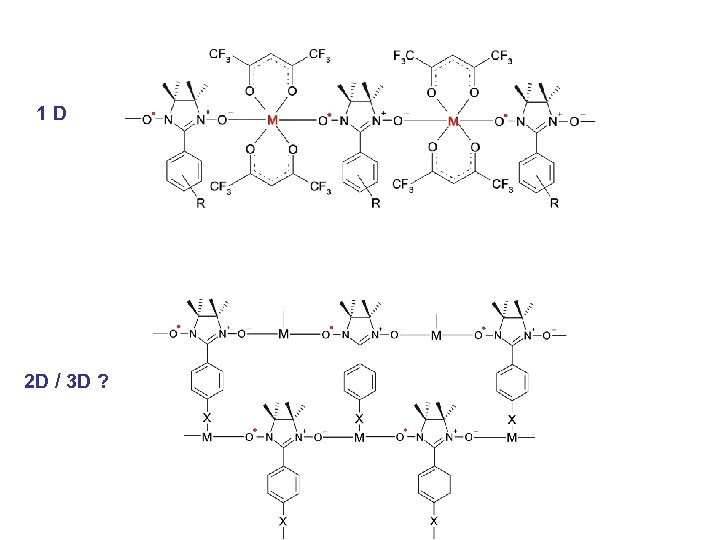 1 D 2 D / 3 D ?
1 D 2 D / 3 D ?
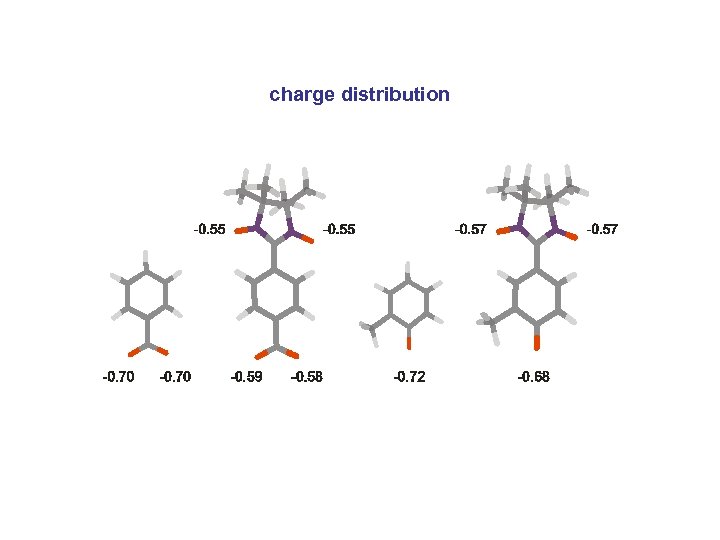 charge distribution
charge distribution
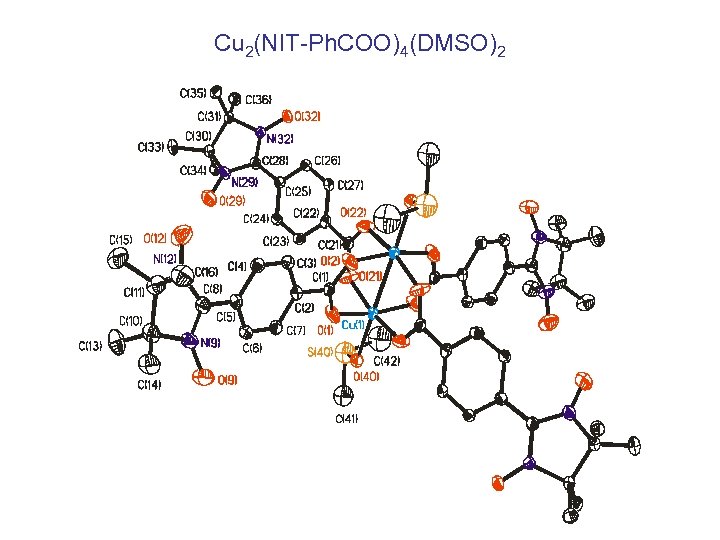 Cu 2(NIT-Ph. COO)4(DMSO)2
Cu 2(NIT-Ph. COO)4(DMSO)2
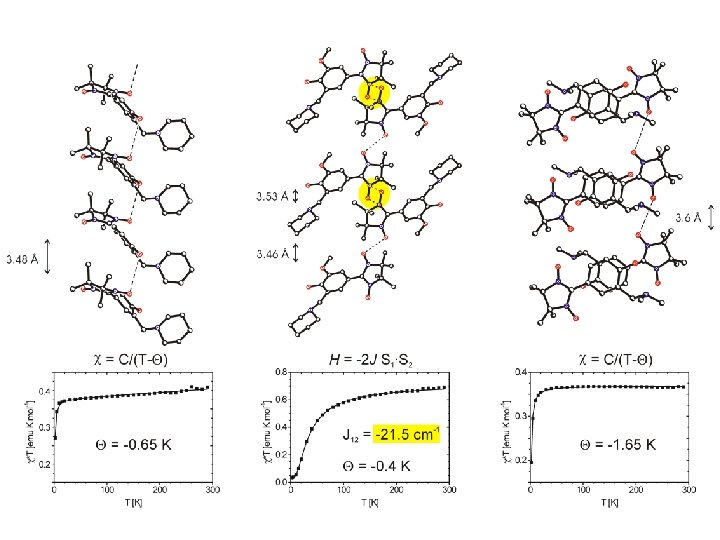
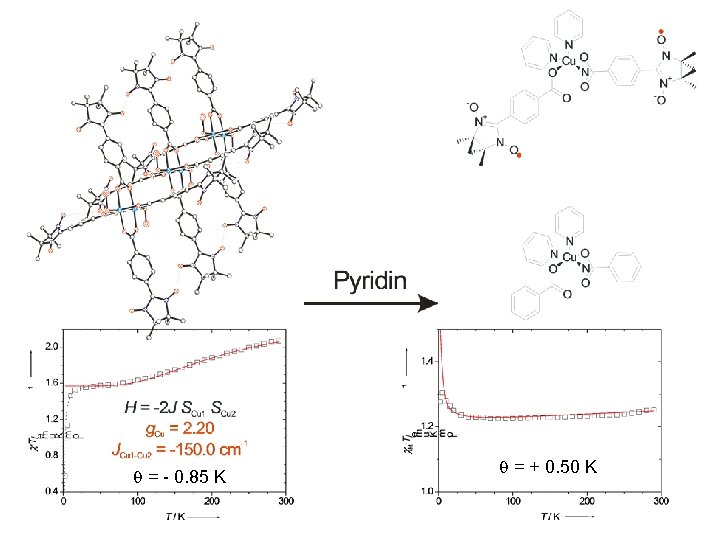 = - 0. 85 K = + 0. 50 K
= - 0. 85 K = + 0. 50 K
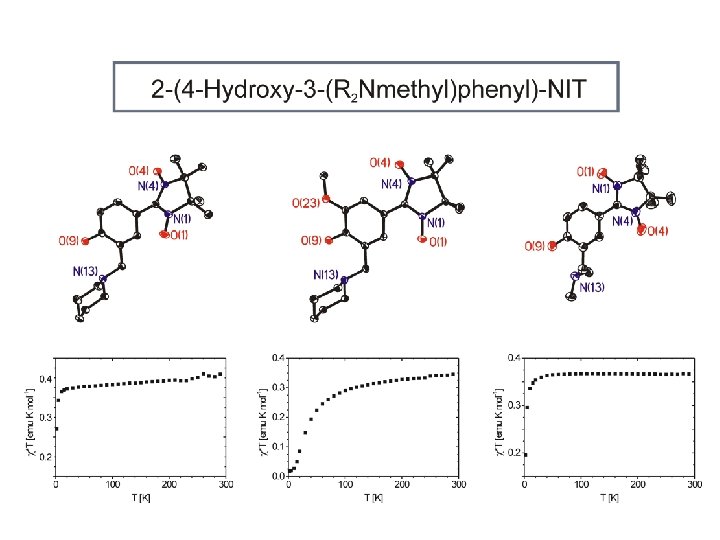
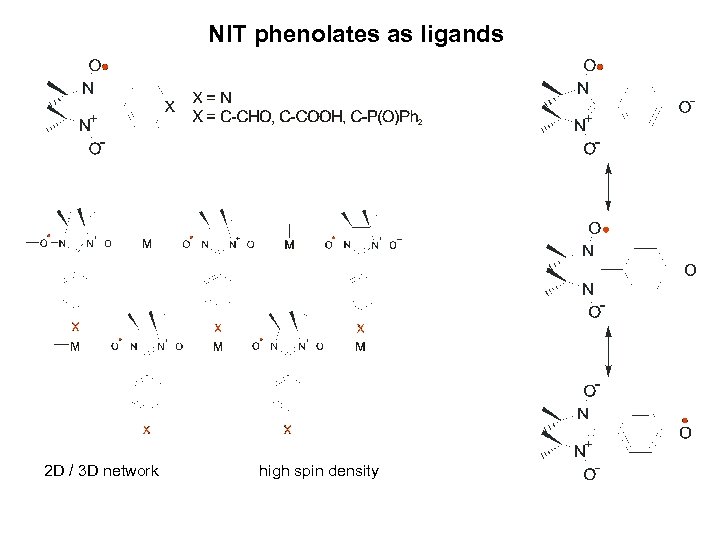 NIT phenolates as ligands 2 D / 3 D network high spin density
NIT phenolates as ligands 2 D / 3 D network high spin density
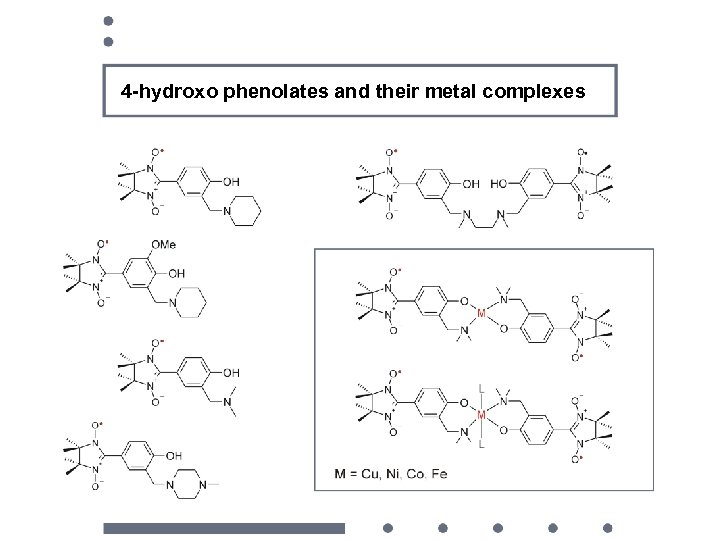 4 -hydroxo phenolates and their metal complexes
4 -hydroxo phenolates and their metal complexes
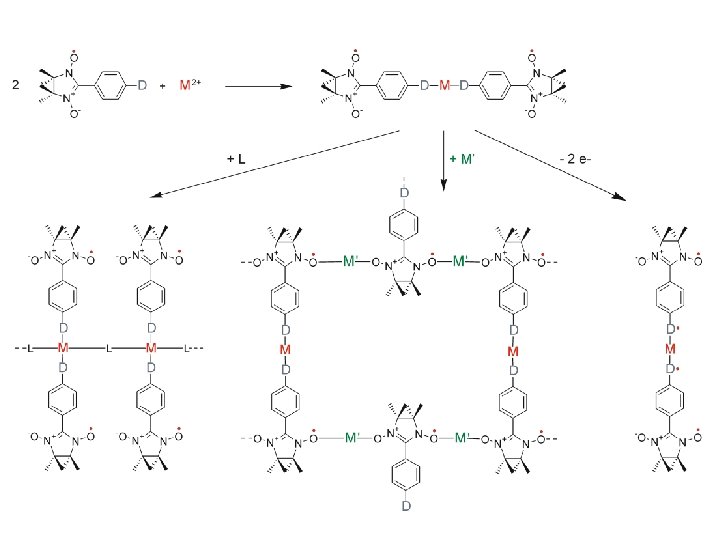
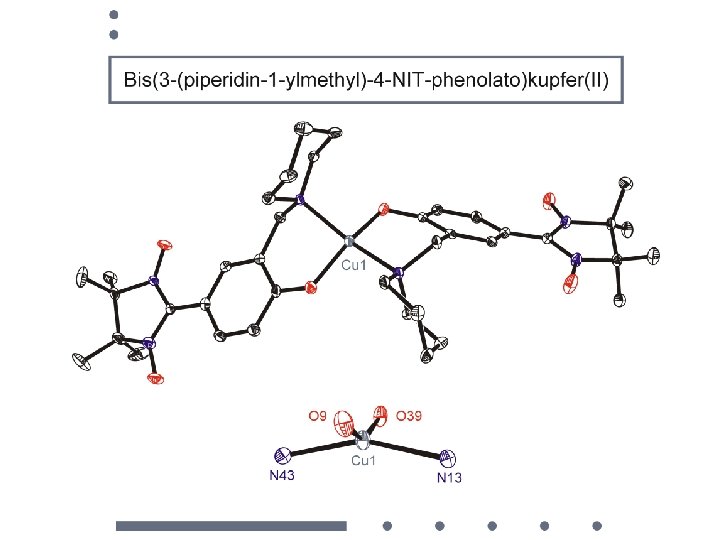
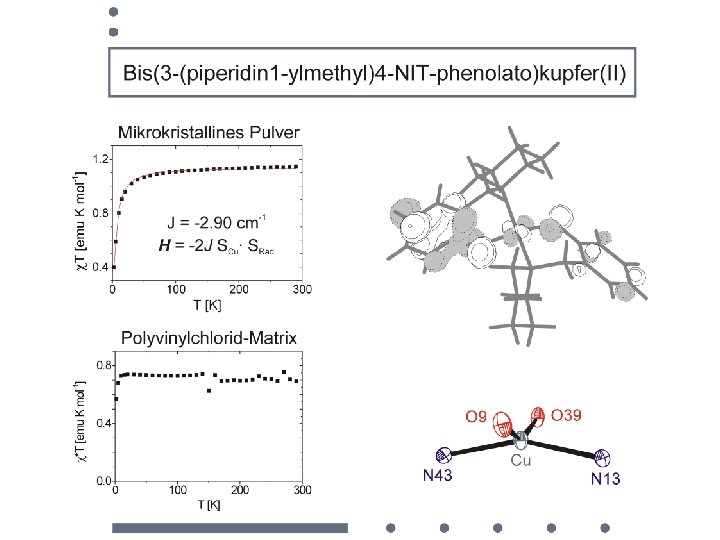
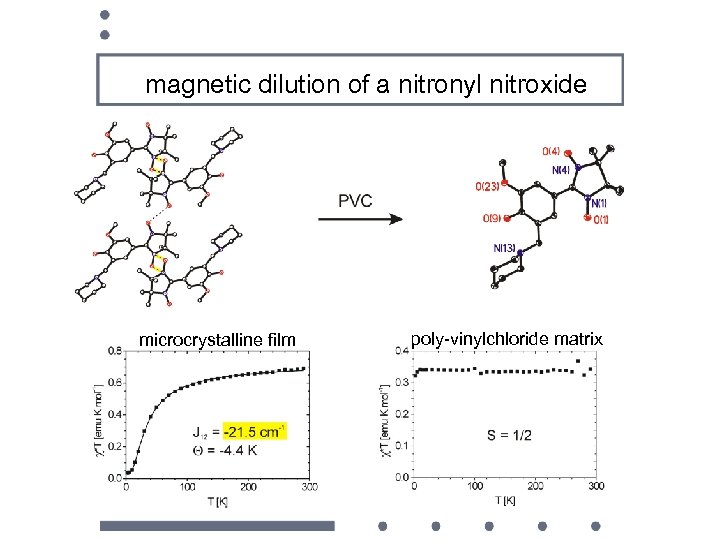 magnetic dilution of a nitronyl nitroxide microcrystalline film poly-vinylchloride matrix
magnetic dilution of a nitronyl nitroxide microcrystalline film poly-vinylchloride matrix
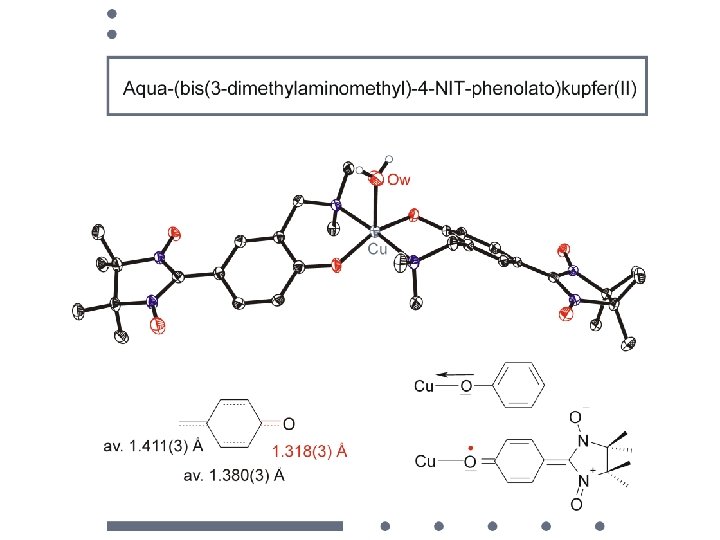
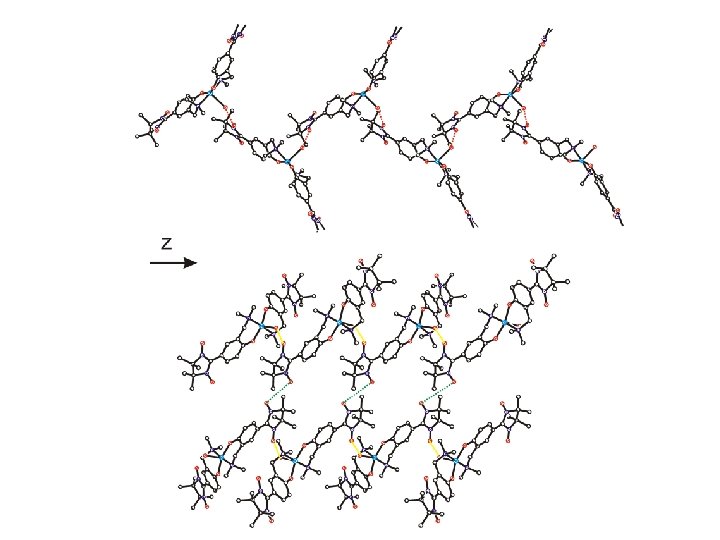
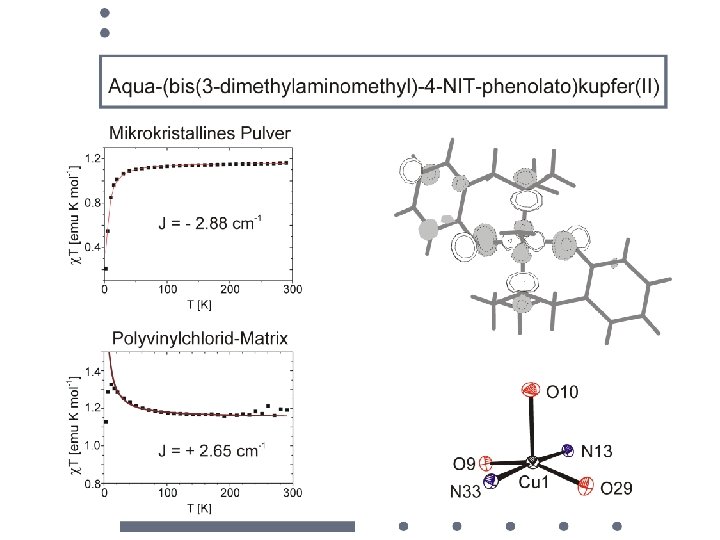
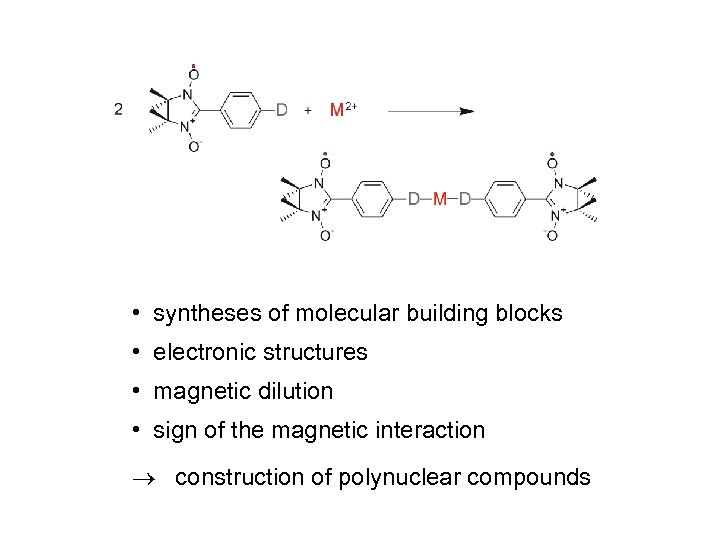 • syntheses of molecular building blocks • electronic structures • magnetic dilution • sign of the magnetic interaction construction of polynuclear compounds
• syntheses of molecular building blocks • electronic structures • magnetic dilution • sign of the magnetic interaction construction of polynuclear compounds
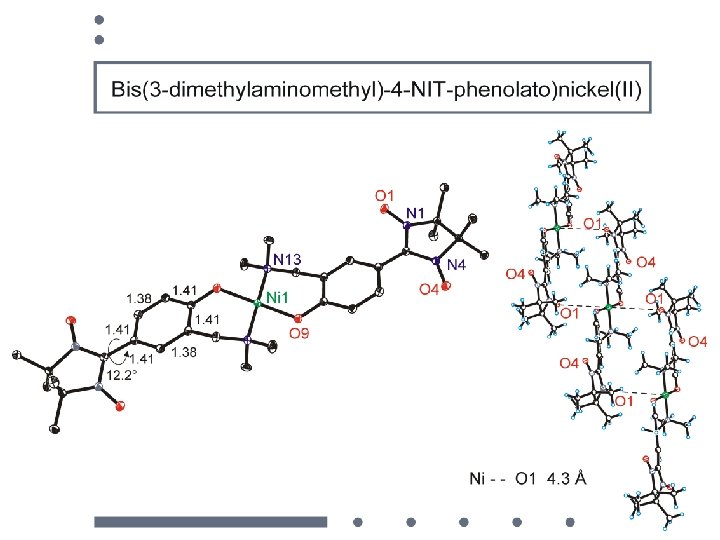
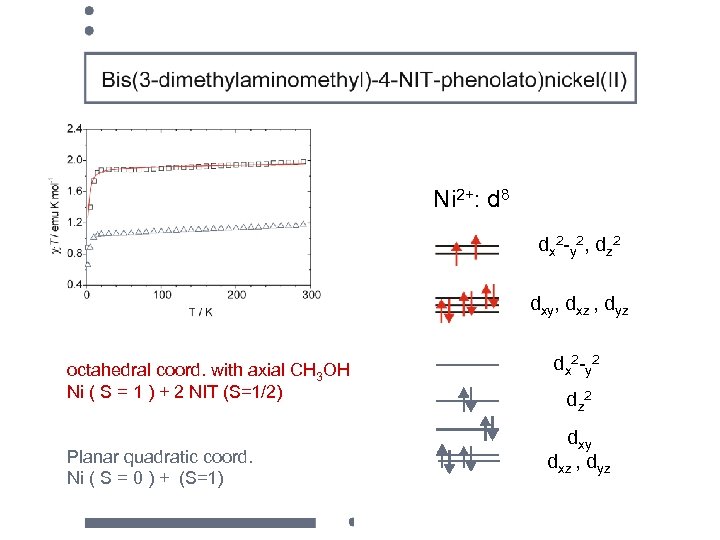 Ni 2+: d 8 dx 2 -y 2, dz 2 dxy, dxz , dyz octahedral coord. with axial CH 3 OH Ni ( S = 1 ) + 2 NIT (S=1/2) Planar quadratic coord. Ni ( S = 0 ) + (S=1) d x 2 -y 2 dz 2 dxy dxz , dyz
Ni 2+: d 8 dx 2 -y 2, dz 2 dxy, dxz , dyz octahedral coord. with axial CH 3 OH Ni ( S = 1 ) + 2 NIT (S=1/2) Planar quadratic coord. Ni ( S = 0 ) + (S=1) d x 2 -y 2 dz 2 dxy dxz , dyz
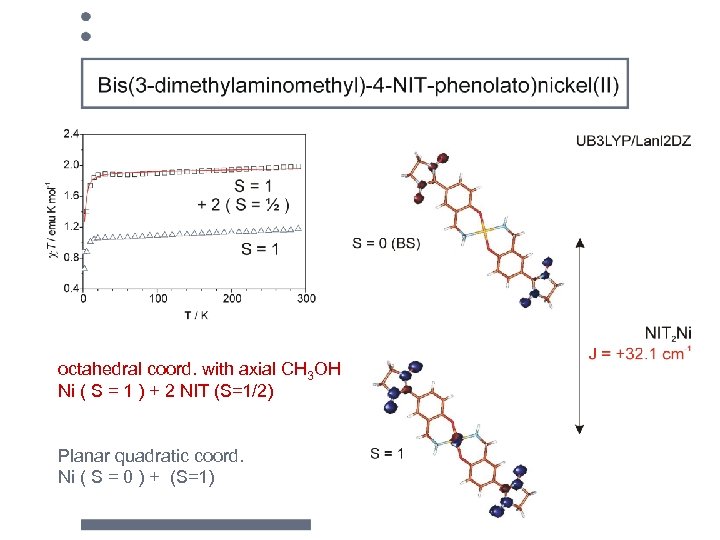 octahedral coord. with axial CH 3 OH Ni ( S = 1 ) + 2 NIT (S=1/2) Planar quadratic coord. Ni ( S = 0 ) + (S=1)
octahedral coord. with axial CH 3 OH Ni ( S = 1 ) + 2 NIT (S=1/2) Planar quadratic coord. Ni ( S = 0 ) + (S=1)
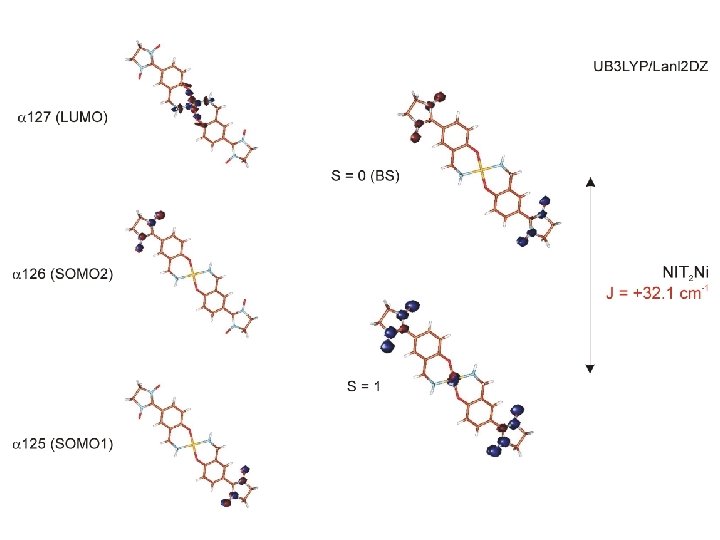
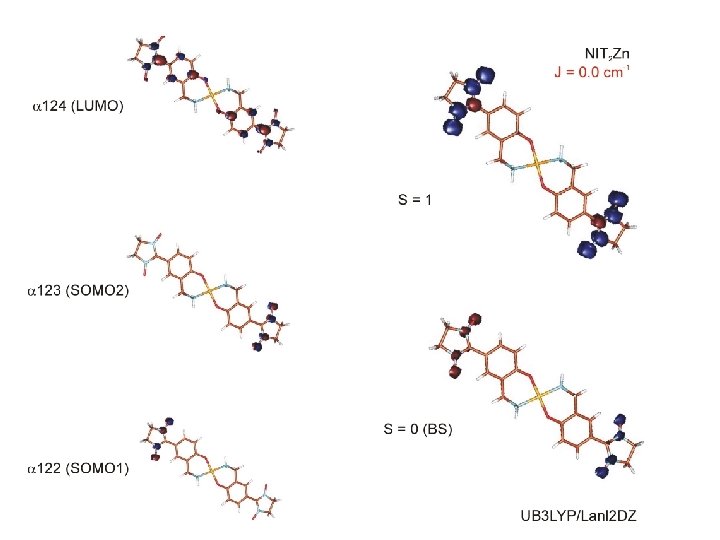
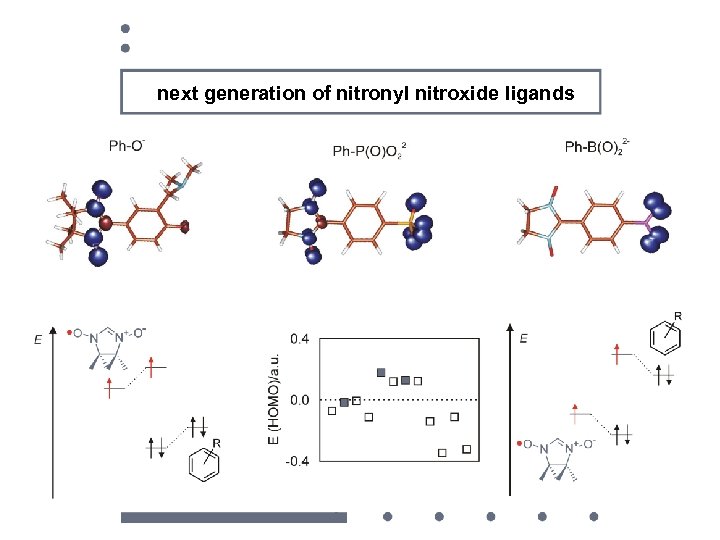 next generation of nitronyl nitroxide ligands
next generation of nitronyl nitroxide ligands
 Multifunctional Radicals
Multifunctional Radicals
 Chelating Radicals
Chelating Radicals
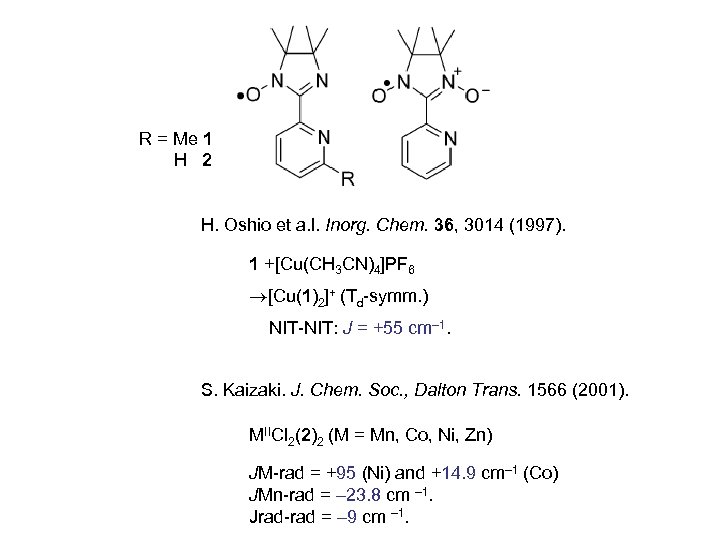 R = Me 1 H 2 H. Oshio et a. l. Inorg. Chem. 36, 3014 (1997). 1 +[Cu(CH 3 CN)4]PF 6 [Cu(1)2]+ (Td-symm. ) NIT-NIT: J = +55 cm– 1. S. Kaizaki. J. Chem. Soc. , Dalton Trans. 1566 (2001). MIICl 2(2)2 (M = Mn, Co, Ni, Zn) JM-rad = +95 (Ni) and +14. 9 cm– 1 (Co) JMn-rad = – 23. 8 cm – 1. Jrad-rad = – 9 cm – 1.
R = Me 1 H 2 H. Oshio et a. l. Inorg. Chem. 36, 3014 (1997). 1 +[Cu(CH 3 CN)4]PF 6 [Cu(1)2]+ (Td-symm. ) NIT-NIT: J = +55 cm– 1. S. Kaizaki. J. Chem. Soc. , Dalton Trans. 1566 (2001). MIICl 2(2)2 (M = Mn, Co, Ni, Zn) JM-rad = +95 (Ni) and +14. 9 cm– 1 (Co) JMn-rad = – 23. 8 cm – 1. Jrad-rad = – 9 cm – 1.
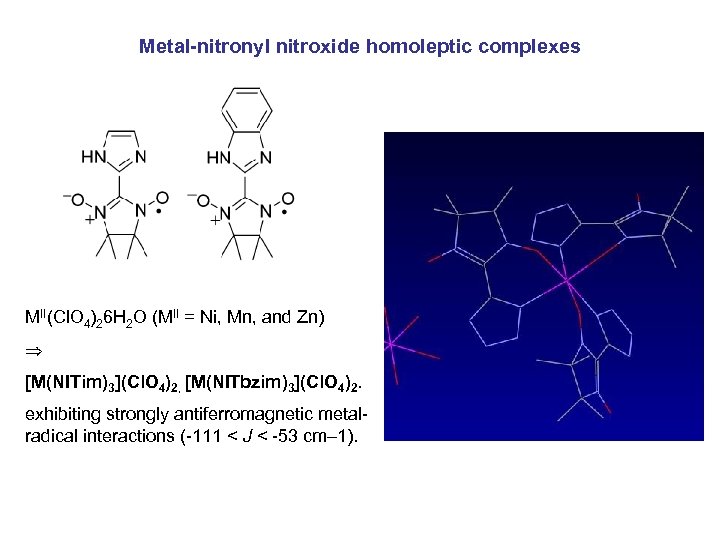 Metal-nitronyl nitroxide homoleptic complexes MII(Cl. O 4)26 H 2 O (MII = Ni, Mn, and Zn) [M(NITim)3](Cl. O 4)2, [M(NITbzim)3](Cl. O 4)2. exhibiting strongly antiferromagnetic metalradical interactions (-111 < J < -53 cm– 1).
Metal-nitronyl nitroxide homoleptic complexes MII(Cl. O 4)26 H 2 O (MII = Ni, Mn, and Zn) [M(NITim)3](Cl. O 4)2, [M(NITbzim)3](Cl. O 4)2. exhibiting strongly antiferromagnetic metalradical interactions (-111 < J < -53 cm– 1).
![coordination polymers [Mn(NITIm)(NITIm. H)]Cl. O 4 Ziessel [Mn 2(NITIm)3]Cl. O 4 Rey and Luneau coordination polymers [Mn(NITIm)(NITIm. H)]Cl. O 4 Ziessel [Mn 2(NITIm)3]Cl. O 4 Rey and Luneau](https://present5.com/presentation/697dfd0f03644e598eb4dd728e8d0b88/image-61.jpg) coordination polymers [Mn(NITIm)(NITIm. H)]Cl. O 4 Ziessel [Mn 2(NITIm)3]Cl. O 4 Rey and Luneau Ferromagnetic ordering temperatures 1. 4 K [Mn 2(NITIm)3]Cl. O 4 40 K [Mn 2(NITBz. Im)3]Cl. O 4
coordination polymers [Mn(NITIm)(NITIm. H)]Cl. O 4 Ziessel [Mn 2(NITIm)3]Cl. O 4 Rey and Luneau Ferromagnetic ordering temperatures 1. 4 K [Mn 2(NITIm)3]Cl. O 4 40 K [Mn 2(NITBz. Im)3]Cl. O 4
 Chelating Radicals
Chelating Radicals
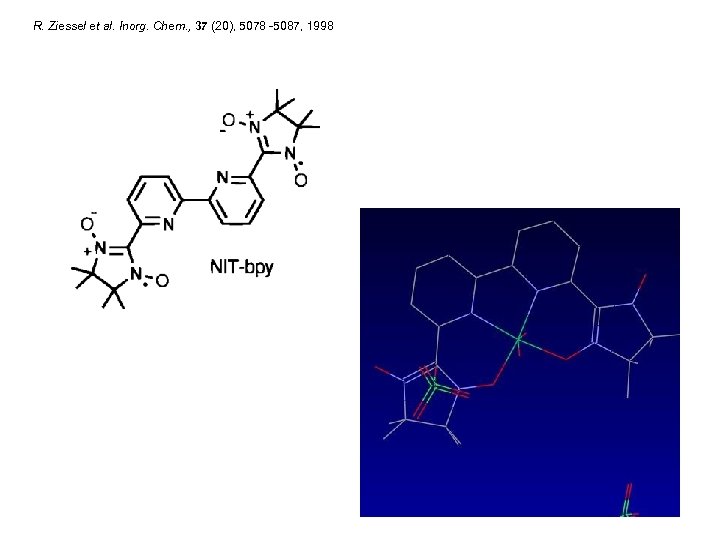 R. Ziessel et al. Inorg. Chem. , 37 (20), 5078 -5087, 1998
R. Ziessel et al. Inorg. Chem. , 37 (20), 5078 -5087, 1998
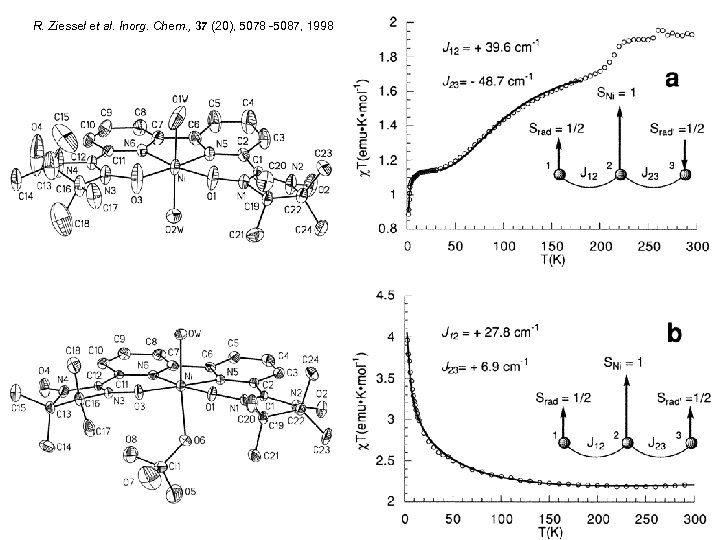 R. Ziessel et al. Inorg. Chem. , 37 (20), 5078 -5087, 1998
R. Ziessel et al. Inorg. Chem. , 37 (20), 5078 -5087, 1998
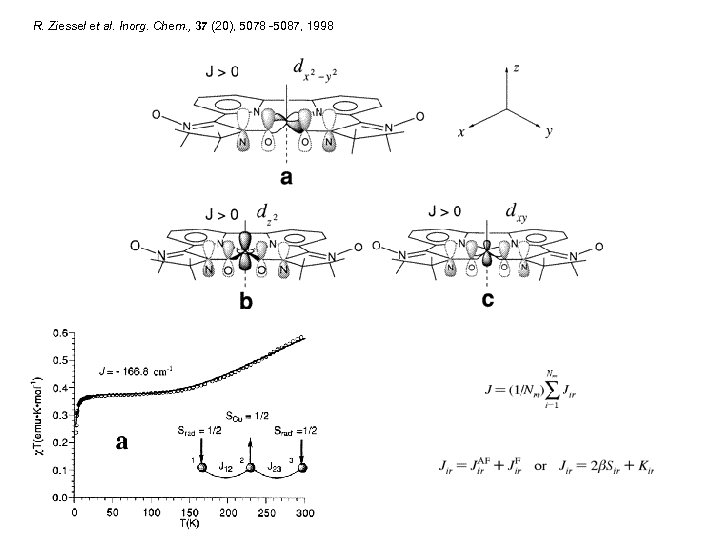 R. Ziessel et al. Inorg. Chem. , 37 (20), 5078 -5087, 1998
R. Ziessel et al. Inorg. Chem. , 37 (20), 5078 -5087, 1998
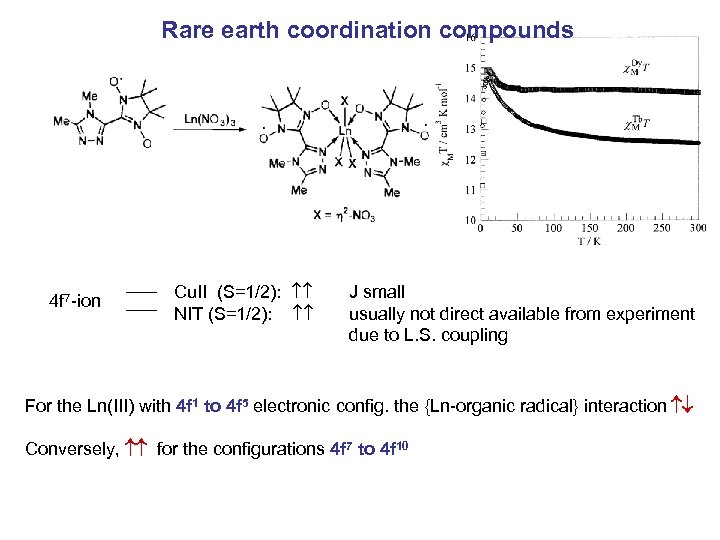 Rare earth coordination compounds 4 f 7 -ion Cu. II (S=1/2): NIT (S=1/2): J small usually not direct available from experiment due to L. S. coupling For the Ln(III) with 4 f 1 to 4 f 5 electronic config. the {Ln-organic radical} interaction Conversely, for the configurations 4 f 7 to 4 f 10
Rare earth coordination compounds 4 f 7 -ion Cu. II (S=1/2): NIT (S=1/2): J small usually not direct available from experiment due to L. S. coupling For the Ln(III) with 4 f 1 to 4 f 5 electronic config. the {Ln-organic radical} interaction Conversely, for the configurations 4 f 7 to 4 f 10
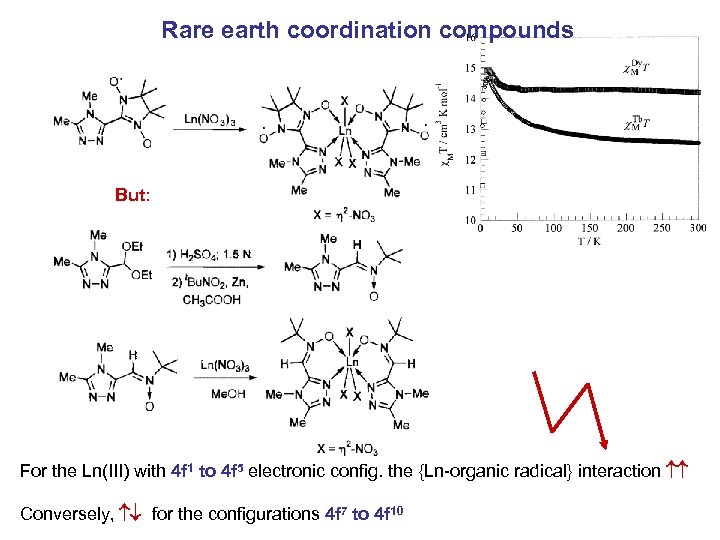 Rare earth coordination compounds But: For the Ln(III) with 4 f 1 to 4 f 5 electronic config. the {Ln-organic radical} interaction Conversely, for the configurations 4 f 7 to 4 f 10
Rare earth coordination compounds But: For the Ln(III) with 4 f 1 to 4 f 5 electronic config. the {Ln-organic radical} interaction Conversely, for the configurations 4 f 7 to 4 f 10
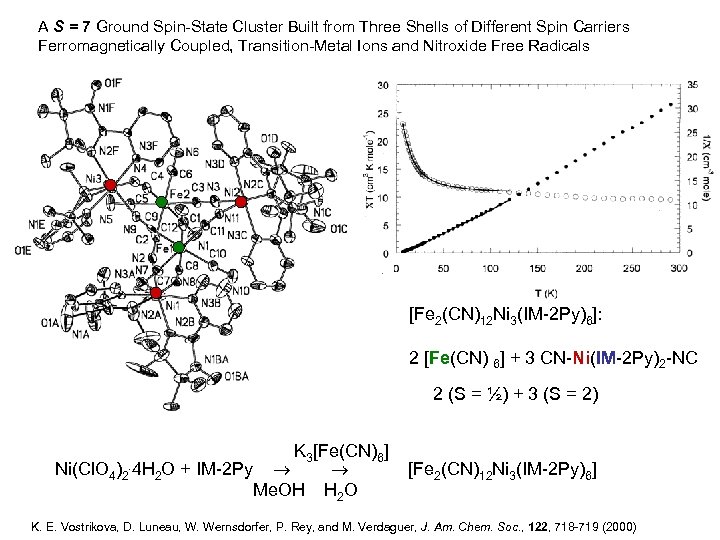 A S = 7 Ground Spin-State Cluster Built from Three Shells of Different Spin Carriers Ferromagnetically Coupled, Transition-Metal Ions and Nitroxide Free Radicals [Fe 2(CN)12 Ni 3(IM-2 Py)6]: 2 [Fe(CN) 6] + 3 CN-Ni(IM-2 Py)2 -NC 2 (S = ½) + 3 (S = 2) Ni(Cl. O 4)2 . 4 H K 3[Fe(CN)6] 2 O + IM-2 Py [Fe 2(CN)12 Ni 3(IM-2 Py)6] Me. OH H 2 O K. E. Vostrikova, D. Luneau, W. Wernsdorfer, P. Rey, and M. Verdaguer, J. Am. Chem. Soc. , 122, 718 -719 (2000)
A S = 7 Ground Spin-State Cluster Built from Three Shells of Different Spin Carriers Ferromagnetically Coupled, Transition-Metal Ions and Nitroxide Free Radicals [Fe 2(CN)12 Ni 3(IM-2 Py)6]: 2 [Fe(CN) 6] + 3 CN-Ni(IM-2 Py)2 -NC 2 (S = ½) + 3 (S = 2) Ni(Cl. O 4)2 . 4 H K 3[Fe(CN)6] 2 O + IM-2 Py [Fe 2(CN)12 Ni 3(IM-2 Py)6] Me. OH H 2 O K. E. Vostrikova, D. Luneau, W. Wernsdorfer, P. Rey, and M. Verdaguer, J. Am. Chem. Soc. , 122, 718 -719 (2000)
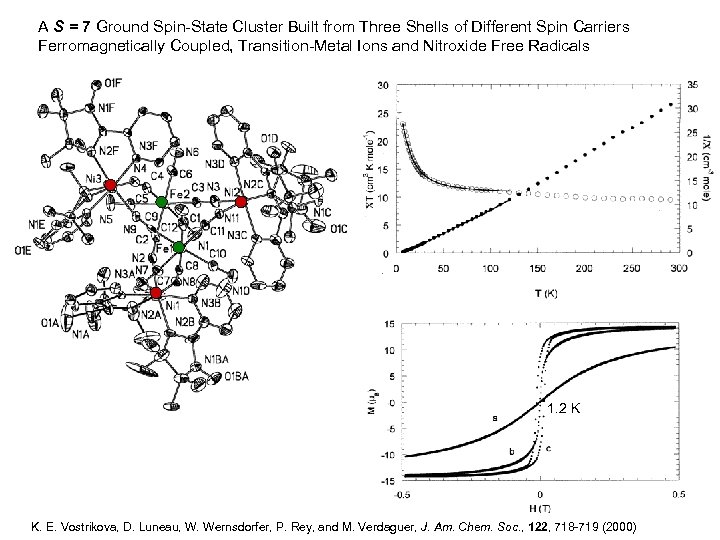 A S = 7 Ground Spin-State Cluster Built from Three Shells of Different Spin Carriers Ferromagnetically Coupled, Transition-Metal Ions and Nitroxide Free Radicals 1. 2 K K. E. Vostrikova, D. Luneau, W. Wernsdorfer, P. Rey, and M. Verdaguer, J. Am. Chem. Soc. , 122, 718 -719 (2000)
A S = 7 Ground Spin-State Cluster Built from Three Shells of Different Spin Carriers Ferromagnetically Coupled, Transition-Metal Ions and Nitroxide Free Radicals 1. 2 K K. E. Vostrikova, D. Luneau, W. Wernsdorfer, P. Rey, and M. Verdaguer, J. Am. Chem. Soc. , 122, 718 -719 (2000)
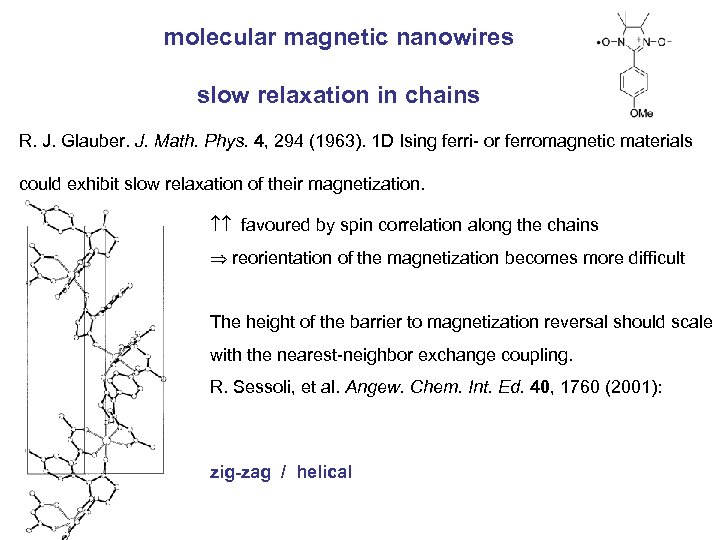 molecular magnetic nanowires slow relaxation in chains R. J. Glauber. J. Math. Phys. 4, 294 (1963). 1 D Ising ferri- or ferromagnetic materials could exhibit slow relaxation of their magnetization. favoured by spin correlation along the chains reorientation of the magnetization becomes more difficult The height of the barrier to magnetization reversal should scale with the nearest-neighbor exchange coupling. R. Sessoli, et al. Angew. Chem. Int. Ed. 40, 1760 (2001): zig-zag / helical
molecular magnetic nanowires slow relaxation in chains R. J. Glauber. J. Math. Phys. 4, 294 (1963). 1 D Ising ferri- or ferromagnetic materials could exhibit slow relaxation of their magnetization. favoured by spin correlation along the chains reorientation of the magnetization becomes more difficult The height of the barrier to magnetization reversal should scale with the nearest-neighbor exchange coupling. R. Sessoli, et al. Angew. Chem. Int. Ed. 40, 1760 (2001): zig-zag / helical
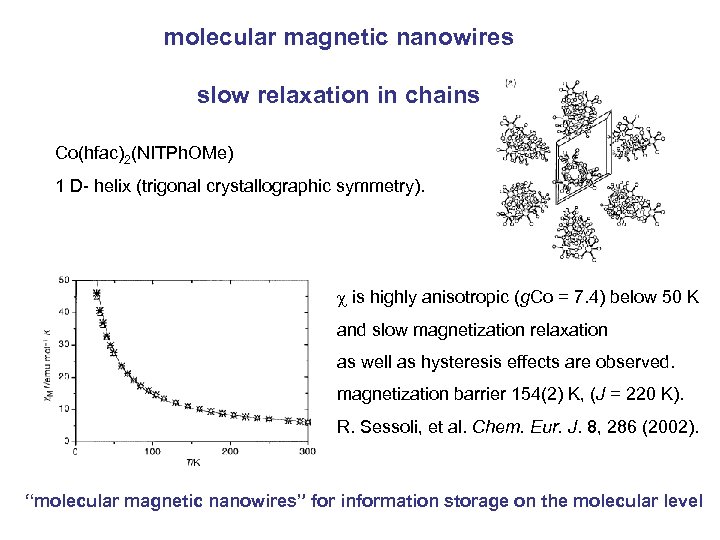 molecular magnetic nanowires slow relaxation in chains Co(hfac)2(NITPh. OMe) 1 D- helix (trigonal crystallographic symmetry). c is highly anisotropic (g. Co = 7. 4) below 50 K and slow magnetization relaxation as well as hysteresis effects are observed. magnetization barrier 154(2) K, (J = 220 K). R. Sessoli, et al. Chem. Eur. J. 8, 286 (2002). “molecular magnetic nanowires” for information storage on the molecular level
molecular magnetic nanowires slow relaxation in chains Co(hfac)2(NITPh. OMe) 1 D- helix (trigonal crystallographic symmetry). c is highly anisotropic (g. Co = 7. 4) below 50 K and slow magnetization relaxation as well as hysteresis effects are observed. magnetization barrier 154(2) K, (J = 220 K). R. Sessoli, et al. Chem. Eur. J. 8, 286 (2002). “molecular magnetic nanowires” for information storage on the molecular level
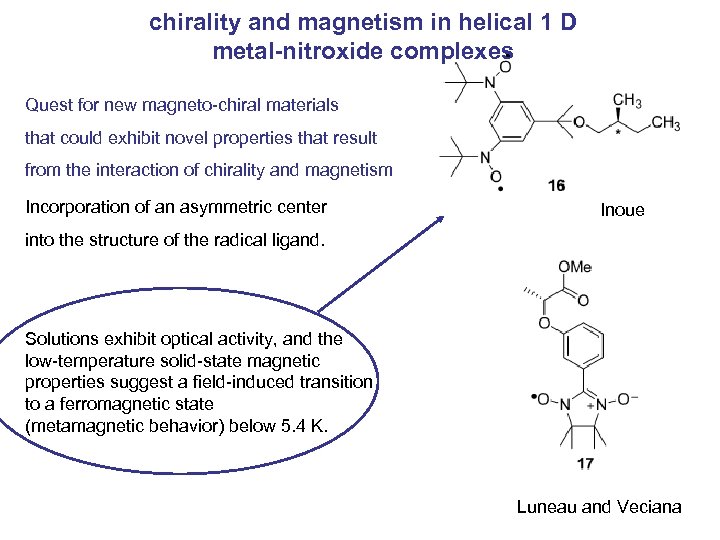 chirality and magnetism in helical 1 D metal-nitroxide complexes Quest for new magneto-chiral materials that could exhibit novel properties that result from the interaction of chirality and magnetism Incorporation of an asymmetric center Inoue into the structure of the radical ligand. Solutions exhibit optical activity, and the low-temperature solid-state magnetic properties suggest a field-induced transition to a ferromagnetic state (metamagnetic behavior) below 5. 4 K. Luneau and Veciana
chirality and magnetism in helical 1 D metal-nitroxide complexes Quest for new magneto-chiral materials that could exhibit novel properties that result from the interaction of chirality and magnetism Incorporation of an asymmetric center Inoue into the structure of the radical ligand. Solutions exhibit optical activity, and the low-temperature solid-state magnetic properties suggest a field-induced transition to a ferromagnetic state (metamagnetic behavior) below 5. 4 K. Luneau and Veciana
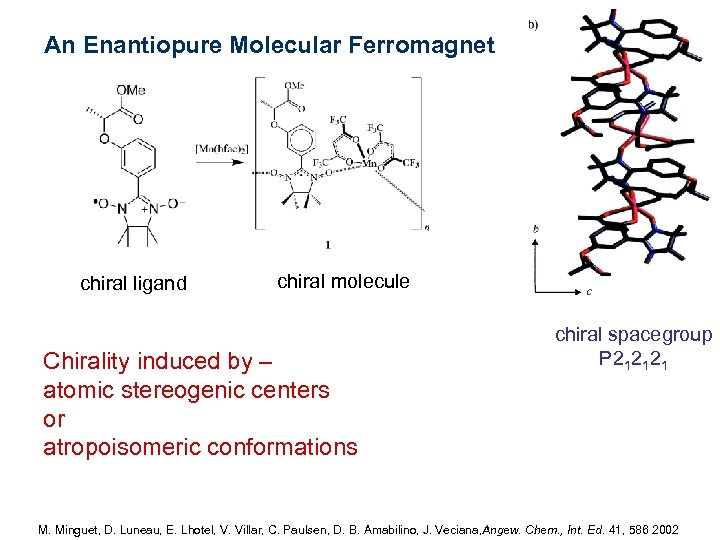 An Enantiopure Molecular Ferromagnet chiral ligand chiral molecule Chirality induced by – atomic stereogenic centers or atropoisomeric conformations chiral spacegroup P 212121 M. Minguet, D. Luneau, E. Lhotel, V. Villar, C. Paulsen, D. B. Amabilino, J. Veciana, Angew. Chem. , Int. Ed. 41, 586 2002
An Enantiopure Molecular Ferromagnet chiral ligand chiral molecule Chirality induced by – atomic stereogenic centers or atropoisomeric conformations chiral spacegroup P 212121 M. Minguet, D. Luneau, E. Lhotel, V. Villar, C. Paulsen, D. B. Amabilino, J. Veciana, Angew. Chem. , Int. Ed. 41, 586 2002
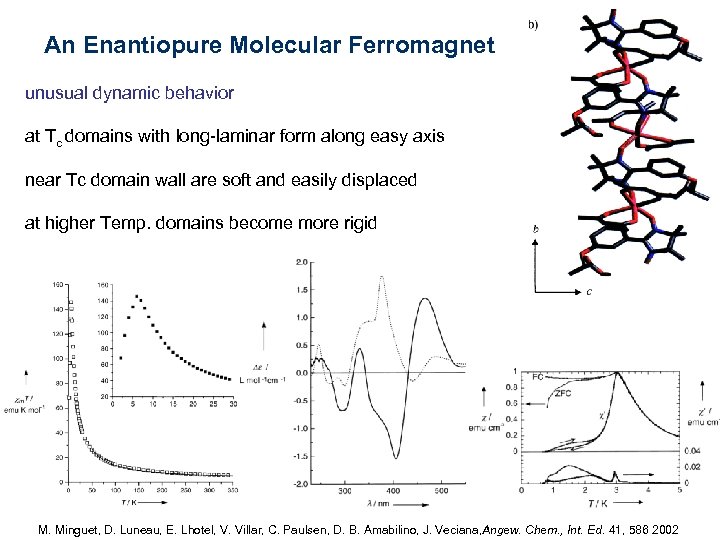 An Enantiopure Molecular Ferromagnet unusual dynamic behavior at Tc domains with long-laminar form along easy axis near Tc domain wall are soft and easily displaced at higher Temp. domains become more rigid M. Minguet, D. Luneau, E. Lhotel, V. Villar, C. Paulsen, D. B. Amabilino, J. Veciana, Angew. Chem. , Int. Ed. 41, 586 2002
An Enantiopure Molecular Ferromagnet unusual dynamic behavior at Tc domains with long-laminar form along easy axis near Tc domain wall are soft and easily displaced at higher Temp. domains become more rigid M. Minguet, D. Luneau, E. Lhotel, V. Villar, C. Paulsen, D. B. Amabilino, J. Veciana, Angew. Chem. , Int. Ed. 41, 586 2002
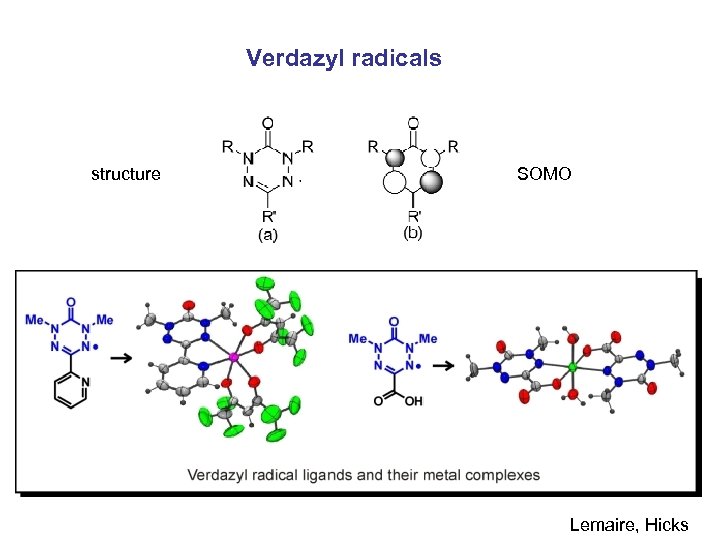 Verdazyl radicals structure SOMO Lemaire, Hicks
Verdazyl radicals structure SOMO Lemaire, Hicks
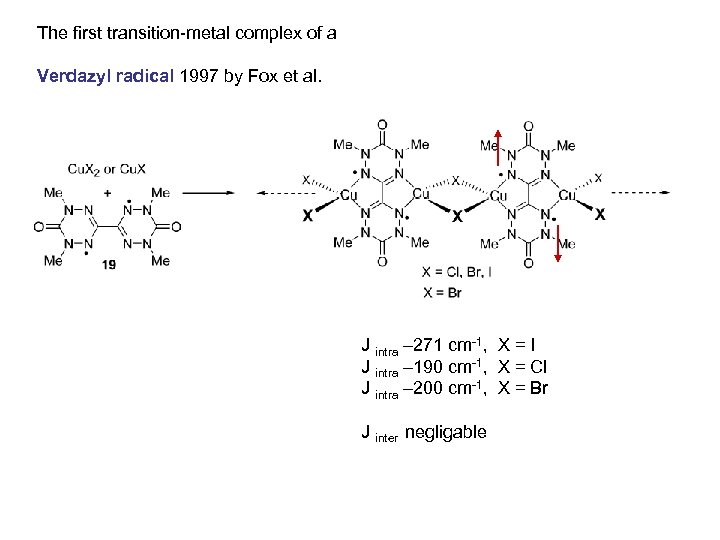 The first transition-metal complex of a Verdazyl radical 1997 by Fox et al. J intra – 271 cm-1, X = I J intra – 190 cm-1, X = Cl J intra – 200 cm-1, X = Br J inter negligable
The first transition-metal complex of a Verdazyl radical 1997 by Fox et al. J intra – 271 cm-1, X = I J intra – 190 cm-1, X = Cl J intra – 200 cm-1, X = Br J inter negligable
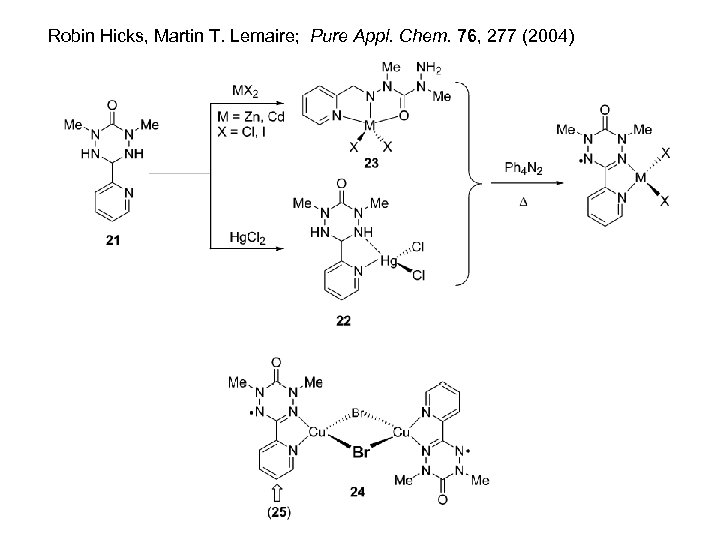 Robin Hicks, Martin T. Lemaire; Pure Appl. Chem. 76, 277 (2004)
Robin Hicks, Martin T. Lemaire; Pure Appl. Chem. 76, 277 (2004)
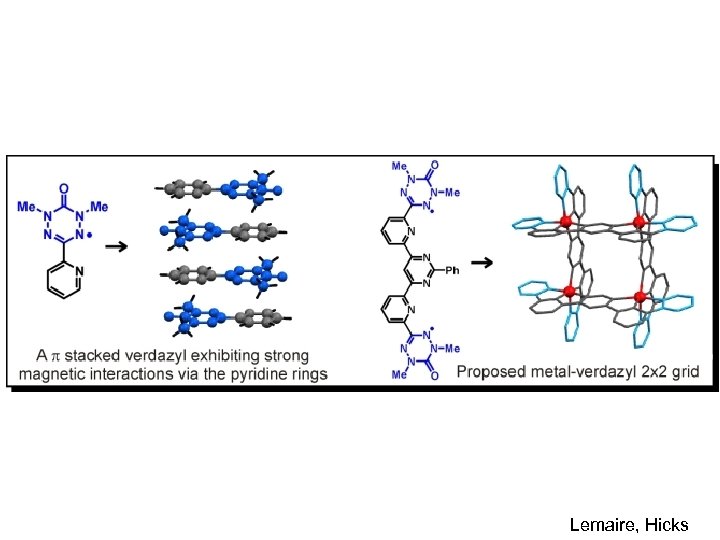 Lemaire, Hicks
Lemaire, Hicks
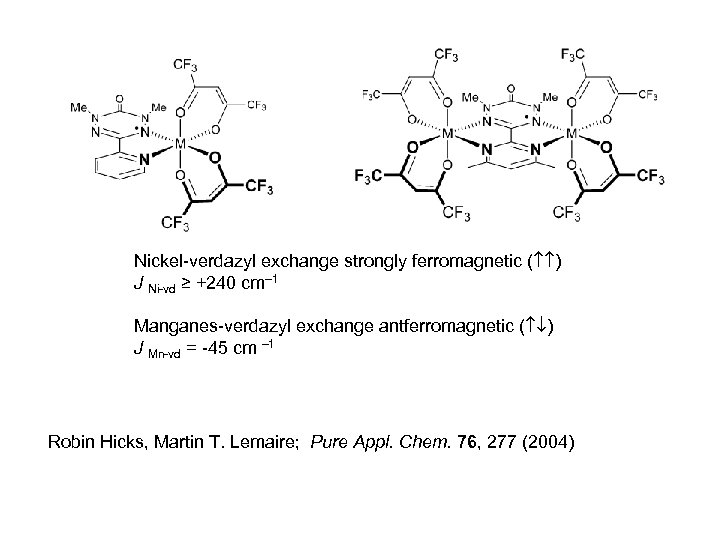 Nickel-verdazyl exchange strongly ferromagnetic ( ) J Ni-vd ≥ +240 cm– 1 Manganes-verdazyl exchange antferromagnetic ( ) J Mn-vd = -45 cm – 1 Robin Hicks, Martin T. Lemaire; Pure Appl. Chem. 76, 277 (2004)
Nickel-verdazyl exchange strongly ferromagnetic ( ) J Ni-vd ≥ +240 cm– 1 Manganes-verdazyl exchange antferromagnetic ( ) J Mn-vd = -45 cm – 1 Robin Hicks, Martin T. Lemaire; Pure Appl. Chem. 76, 277 (2004)
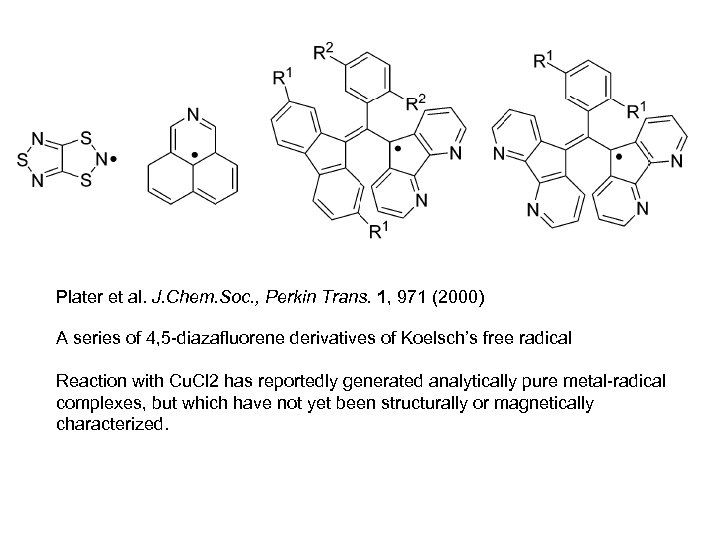 Plater et al. J. Chem. Soc. , Perkin Trans. 1, 971 (2000) A series of 4, 5 -diazafluorene derivatives of Koelsch’s free radical Reaction with Cu. Cl 2 has reportedly generated analytically pure metal-radical complexes, but which have not yet been structurally or magnetically characterized.
Plater et al. J. Chem. Soc. , Perkin Trans. 1, 971 (2000) A series of 4, 5 -diazafluorene derivatives of Koelsch’s free radical Reaction with Cu. Cl 2 has reportedly generated analytically pure metal-radical complexes, but which have not yet been structurally or magnetically characterized.
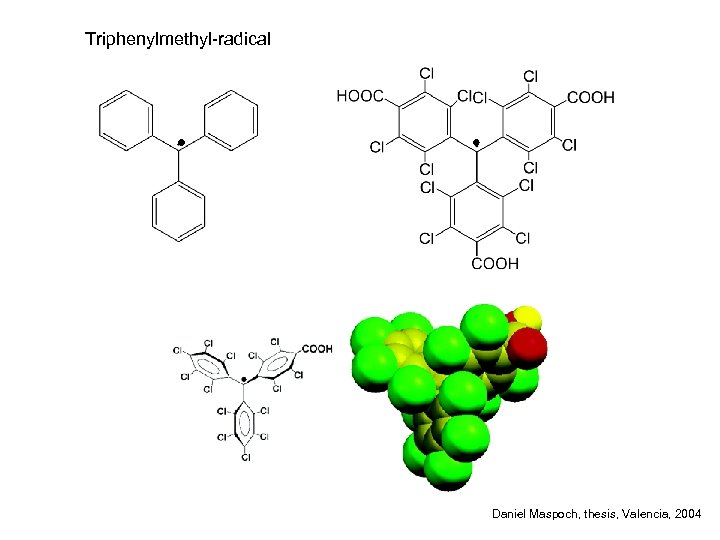 Triphenylmethyl-radical Daniel Maspoch, thesis, Valencia, 2004
Triphenylmethyl-radical Daniel Maspoch, thesis, Valencia, 2004
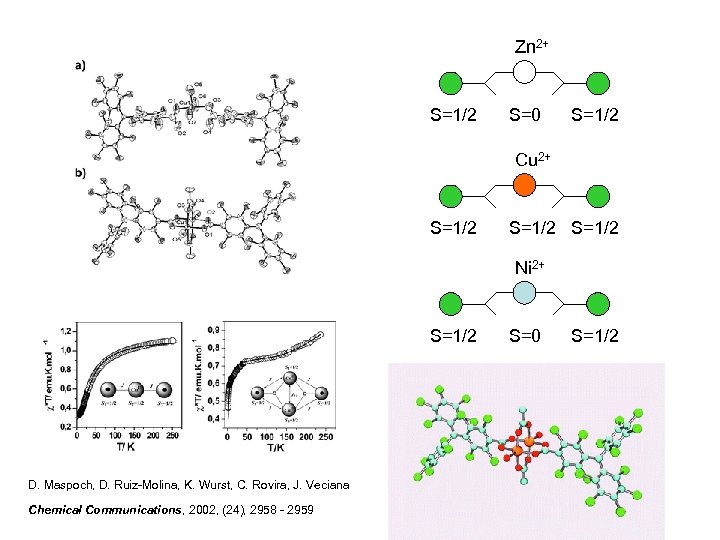 Zn 2+ S=1/2 S=0 S=1/2 Cu 2+ S=1/2 Ni 2+ S=1/2 D. Maspoch, D. Ruiz-Molina, K. Wurst, C. Rovira, J. Veciana Chemical Communications, 2002, (24), 2958 - 2959 S=0 S=1/2
Zn 2+ S=1/2 S=0 S=1/2 Cu 2+ S=1/2 Ni 2+ S=1/2 D. Maspoch, D. Ruiz-Molina, K. Wurst, C. Rovira, J. Veciana Chemical Communications, 2002, (24), 2958 - 2959 S=0 S=1/2
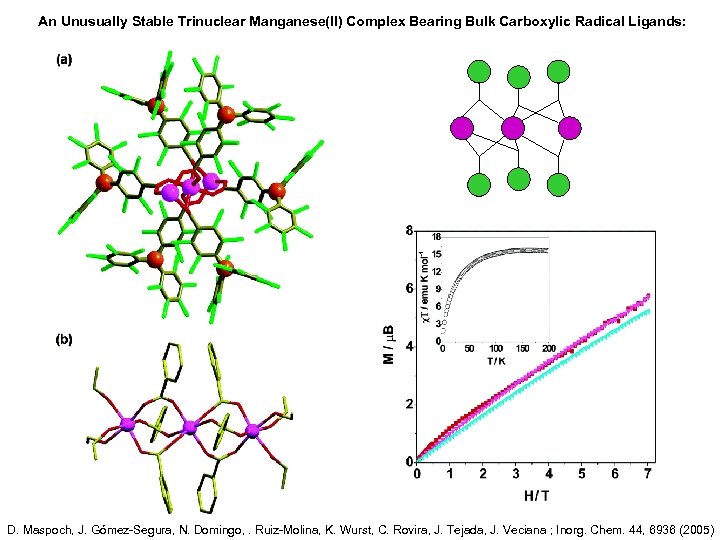 An Unusually Stable Trinuclear Manganese(II) Complex Bearing Bulk Carboxylic Radical Ligands: D. Maspoch, J. Gómez-Segura, N. Domingo, . Ruiz-Molina, K. Wurst, C. Rovira, J. Tejada, J. Veciana ; Inorg. Chem. 44, 6936 (2005)
An Unusually Stable Trinuclear Manganese(II) Complex Bearing Bulk Carboxylic Radical Ligands: D. Maspoch, J. Gómez-Segura, N. Domingo, . Ruiz-Molina, K. Wurst, C. Rovira, J. Tejada, J. Veciana ; Inorg. Chem. 44, 6936 (2005)
![Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ = Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ =](https://present5.com/presentation/697dfd0f03644e598eb4dd728e8d0b88/image-84.jpg) Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ = 7, 7, 8, 8 -tetracyano-p-quinodimethane, TCNE = tetracyanoethene
Charge Transfer Salts [Fe. Cp*2]+. [TCNQ] -. Bis(ethylenedithio)tetrathiafulvalene [Fe. Cp*2]+. [TCNE] -. TCNQ = 7, 7, 8, 8 -tetracyano-p-quinodimethane, TCNE = tetracyanoethene
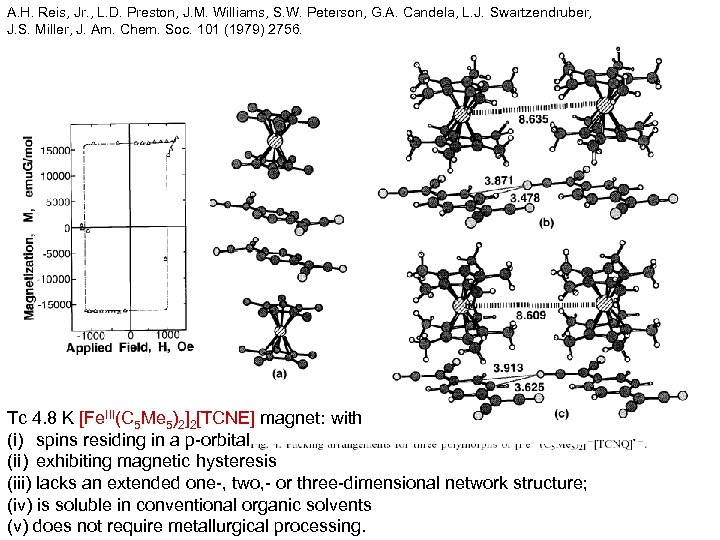 A. H. Reis, Jr. , L. D. Preston, J. M. Williams, S. W. Peterson, G. A. Candela, L. J. Swartzendruber, J. S. Miller, J. Am. Chem. Soc. 101 (1979) 2756. Tc 4. 8 K [Fe. III(C 5 Me 5)2]2[TCNE] magnet: with (i) spins residing in a p-orbital (ii) exhibiting magnetic hysteresis (iii) lacks an extended one-, two, - or three-dimensional network structure; (iv) is soluble in conventional organic solvents (v) does not require metallurgical processing.
A. H. Reis, Jr. , L. D. Preston, J. M. Williams, S. W. Peterson, G. A. Candela, L. J. Swartzendruber, J. S. Miller, J. Am. Chem. Soc. 101 (1979) 2756. Tc 4. 8 K [Fe. III(C 5 Me 5)2]2[TCNE] magnet: with (i) spins residing in a p-orbital (ii) exhibiting magnetic hysteresis (iii) lacks an extended one-, two, - or three-dimensional network structure; (iv) is soluble in conventional organic solvents (v) does not require metallurgical processing.
![Variation of Tc with increasing spin number per metal in [MIII(C 5 Me 5)2]+[TCNE]-. Variation of Tc with increasing spin number per metal in [MIII(C 5 Me 5)2]+[TCNE]-.](https://present5.com/presentation/697dfd0f03644e598eb4dd728e8d0b88/image-86.jpg) Variation of Tc with increasing spin number per metal in [MIII(C 5 Me 5)2]+[TCNE]-.
Variation of Tc with increasing spin number per metal in [MIII(C 5 Me 5)2]+[TCNE]-.
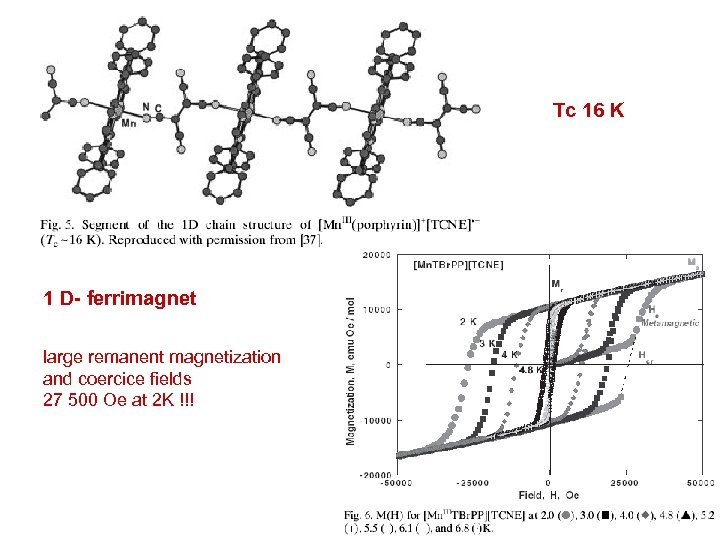 Tc 16 K 1 D- ferrimagnet large remanent magnetization and coercice fields 27 500 Oe at 2 K !!!
Tc 16 K 1 D- ferrimagnet large remanent magnetization and coercice fields 27 500 Oe at 2 K !!!
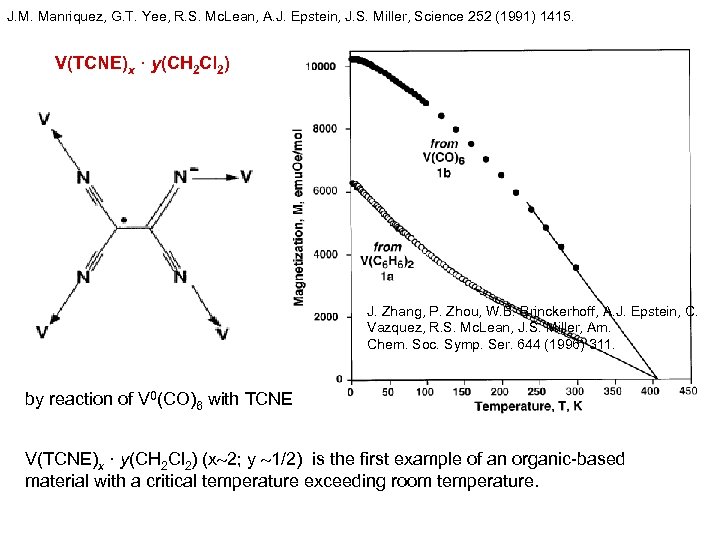 J. M. Manriquez, G. T. Yee, R. S. Mc. Lean, A. J. Epstein, J. S. Miller, Science 252 (1991) 1415. V(TCNE)x · y(CH 2 Cl 2) J. Zhang, P. Zhou, W. B. Brinckerhoff, A. J. Epstein, C. Vazquez, R. S. Mc. Lean, J. S. Miller, Am. Chem. Soc. Symp. Ser. 644 (1996) 311. by reaction of V 0(CO)6 with TCNE V(TCNE)x · y(CH 2 Cl 2) (x 2; y 1/2) is the first example of an organic-based material with a critical temperature exceeding room temperature.
J. M. Manriquez, G. T. Yee, R. S. Mc. Lean, A. J. Epstein, J. S. Miller, Science 252 (1991) 1415. V(TCNE)x · y(CH 2 Cl 2) J. Zhang, P. Zhou, W. B. Brinckerhoff, A. J. Epstein, C. Vazquez, R. S. Mc. Lean, J. S. Miller, Am. Chem. Soc. Symp. Ser. 644 (1996) 311. by reaction of V 0(CO)6 with TCNE V(TCNE)x · y(CH 2 Cl 2) (x 2; y 1/2) is the first example of an organic-based material with a critical temperature exceeding room temperature.

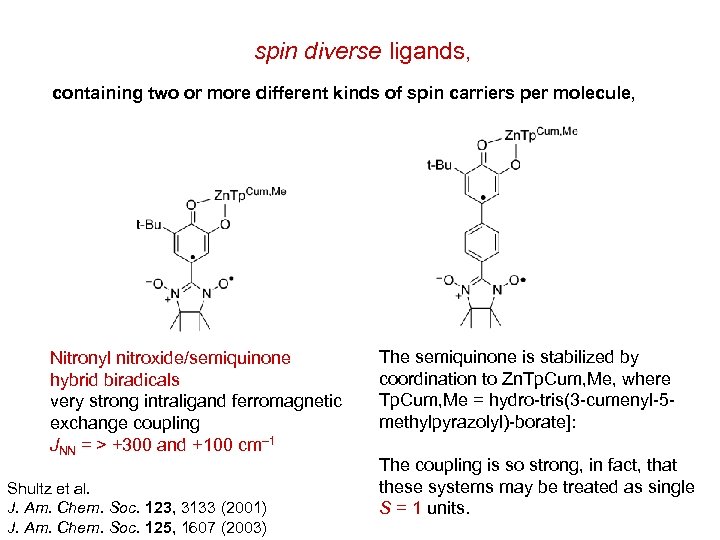 spin diverse ligands, containing two or more different kinds of spin carriers per molecule, Nitronyl nitroxide/semiquinone hybrid biradicals very strong intraligand ferromagnetic exchange coupling JNN = > +300 and +100 cm– 1 Shultz et al. J. Am. Chem. Soc. 123, 3133 (2001) J. Am. Chem. Soc. 125, 1607 (2003) The semiquinone is stabilized by coordination to Zn. Tp. Cum, Me, where Tp. Cum, Me = hydro-tris(3 -cumenyl-5 methylpyrazolyl)-borate]: The coupling is so strong, in fact, that these systems may be treated as single S = 1 units.
spin diverse ligands, containing two or more different kinds of spin carriers per molecule, Nitronyl nitroxide/semiquinone hybrid biradicals very strong intraligand ferromagnetic exchange coupling JNN = > +300 and +100 cm– 1 Shultz et al. J. Am. Chem. Soc. 123, 3133 (2001) J. Am. Chem. Soc. 125, 1607 (2003) The semiquinone is stabilized by coordination to Zn. Tp. Cum, Me, where Tp. Cum, Me = hydro-tris(3 -cumenyl-5 methylpyrazolyl)-borate]: The coupling is so strong, in fact, that these systems may be treated as single S = 1 units.
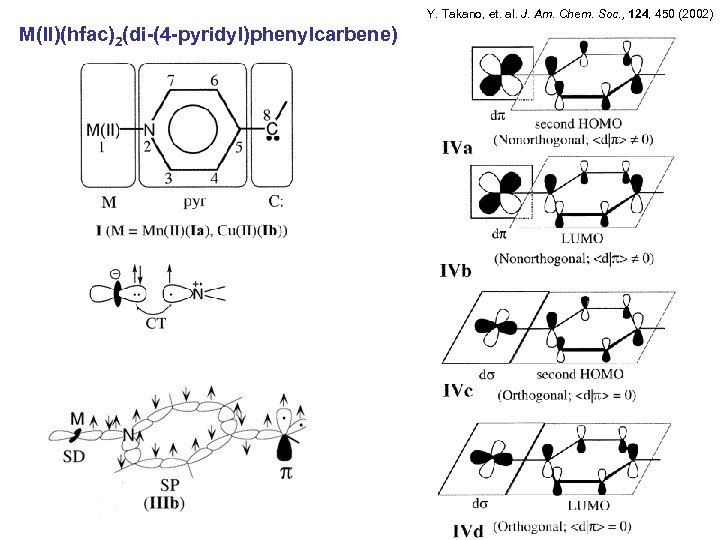 Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002) M(II)(hfac)2(di-(4 -pyridyl)phenylcarbene)
Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002) M(II)(hfac)2(di-(4 -pyridyl)phenylcarbene)
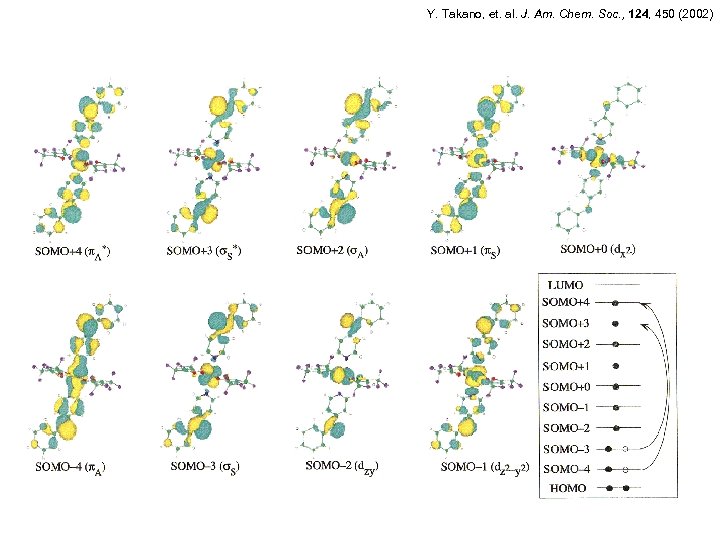 Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002)
Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002)
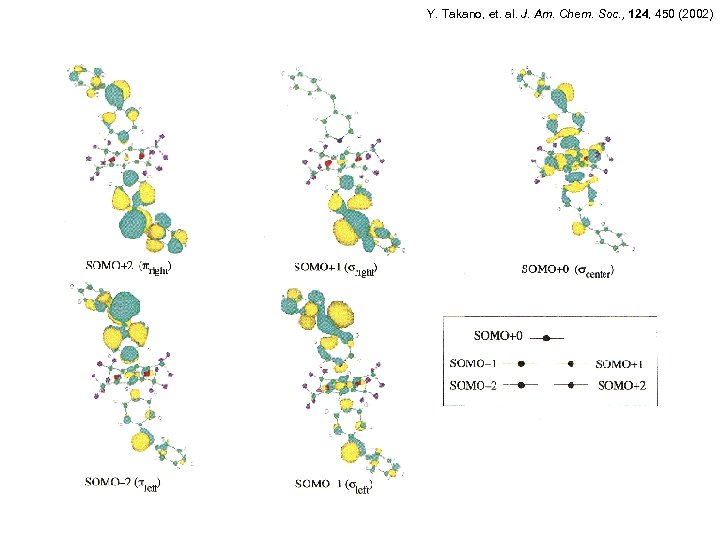 Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002)
Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002)
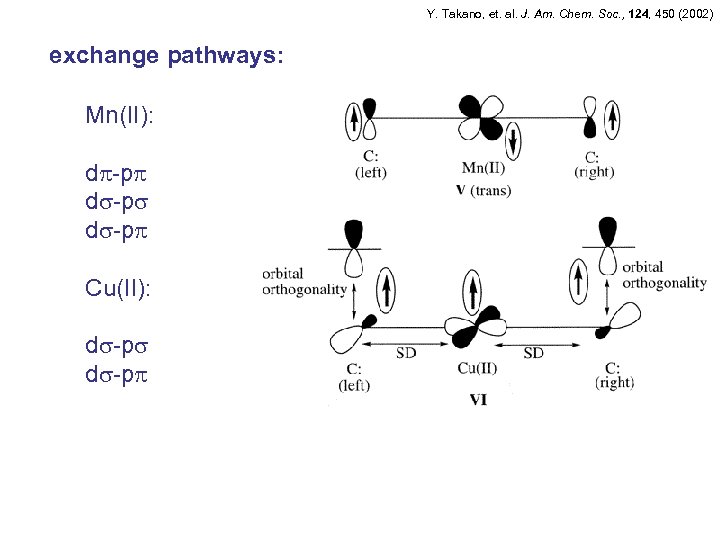 Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002) exchange pathways: Mn(II): d -p Cu(II): d -p
Y. Takano, et. al. J. Am. Chem. Soc. , 124, 450 (2002) exchange pathways: Mn(II): d -p Cu(II): d -p
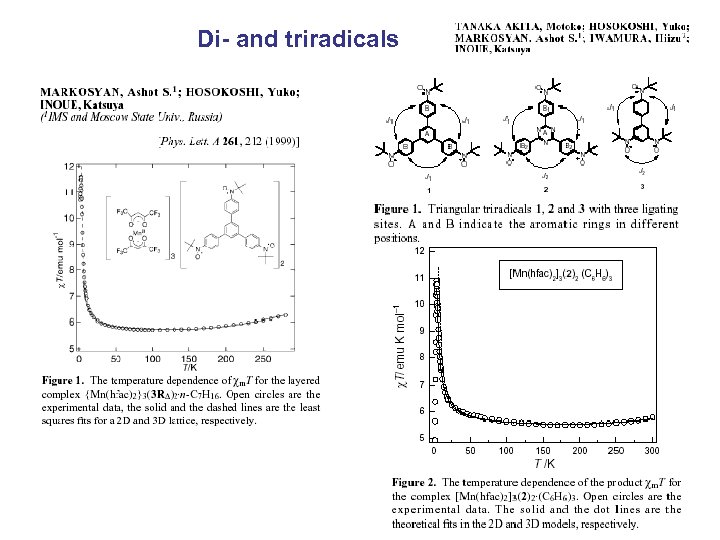 Di- and triradicals
Di- and triradicals
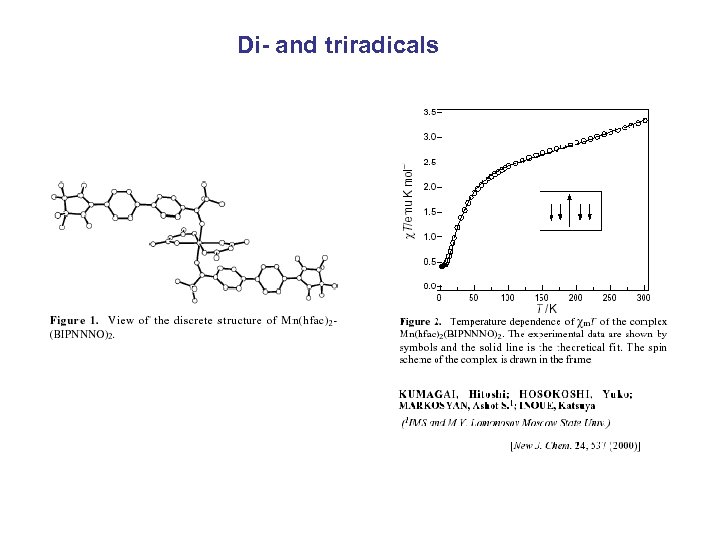 Di- and triradicals
Di- and triradicals
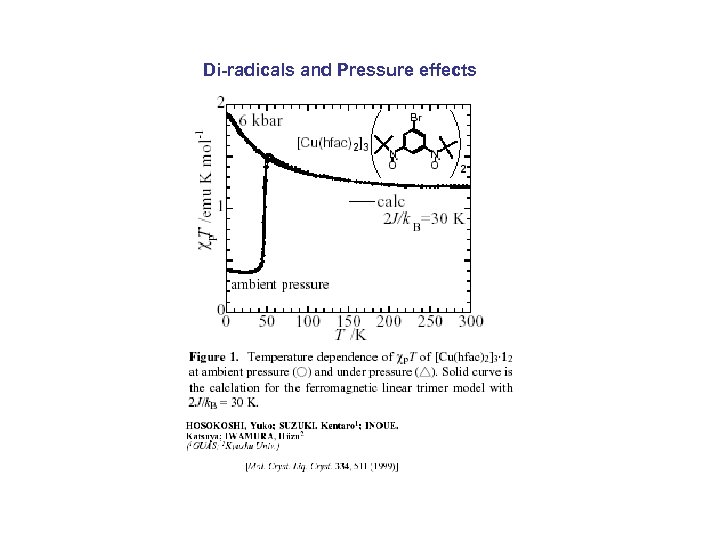 Di-radicals and Pressure effects
Di-radicals and Pressure effects


