ffea5d350b964887d351358971a8280a.ppt
- Количество слайдов: 22

EUROpean network of transnational collaborative RTD projects in the field of NANOMEDicine www. euronanomed. net

What are the societal needs? - the challenge for Europe Improve public health and welfare – Ageing population – Efficiency of current medical solutions – Emergence of new disease and old diseases are back – Personalized medicine future directions www. euronanomed. net

The global challenge • Therapeutic solutions for only 1/3 known diseases (WHO) • Infections: HIV, TB, malaria, risk of pandemia • Need to increase capability and innovation of medical industry – Rapid and accurate diagnosis for early detection, even prevention – Novel, more efficient therapies with less side effects www. euronanomed. net

Nano from research to patient and industry Nanomedicine: the application of nanotechnology to achieve breakthroughs in healthcare Foto: kaibara 87/www. flickr. com www. euronanomed. net

Emerging nanomedicine ses a Dise tious 009 Infec l 9, 2 t ance ue, vo L ril iss Ap www. euronanomed. net
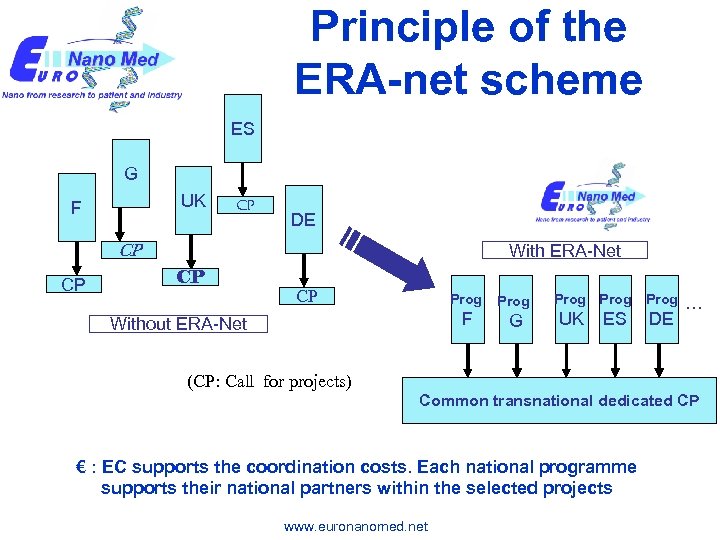
Principle of the ERA-net scheme ES G UK F CP DE CP CP With ERA-Net CP CP Prog F Without ERA-Net Prog G Prog UK ES Prog DE … (CP: Call for projects) Common transnational dedicated CP € : EC supports the coordination costs. Each national programme supports their national partners within the selected projects www. euronanomed. net

Euro. Nano. Med covers 3/4 of European nanomedicine actors • 24 partners from 18 countries/regions • Coordination of funding programmes in nanomedicine • Started January 2009 • EU funding for coordination (ERA-net in FP 7) 2 common joint calls for proposals 2009, 2010 www. euronanomed. net

Euro. Nano. Med: an asset to bridge the gap 2 calls to support RTD projects aiming at – solving an unmet medical need using nanomedicine – promoting a multidisciplinary translational approach with an international focus Euro. Nano. Med will achieve a critical mass of actors !!! www. euronanomed. net

Euro. Nano. Med outputs • Develop innovative diagnostic and therapeutic solutions for the patient, enhancing the competitiveness of the European health industry • Maturation of the field To bring together – academic teams – clinical/public health teams – industrial research teams • Cross-border collaboration between the best researchers/teams in nanomedicine www. euronanomed. net

Why now? What’s the urgency? • Health industry: a key for future growth • European countries invest substantially in nano industry and basic nano research • Nano technologies: a promising field for innovation and patient application Despite these efforts, Europe is lagging behind the US in an increasing competition : Europe needs to act now! www. euronanomed. net

Expected outcome • To move innovation from knowledge to industrial technology and health care applications • Opening the national programmes to support transnational RTD partnerships Long term vision: formula for an Europeanwide integrated programme with a coordinated funding www. euronanomed. net

Contacts • Coordinator Dr. Pierre-Noël LIRSAC Commissariat à l’Energie Atomique (CEA) Pierre-noel. lirsac@cea. fr, Virginie. sivan@cea. fr (+33) 1 69 08 87 99 • Network Steering Committee chair Associate Prof. Karin Forsberg-Nilsson Swedish research Council (SRC) Karin. forsberg. nilsson@vr. se www. euronanomed. net

Euro. Nano. Med ERA Networking towards ERA! www. euronanomed. net
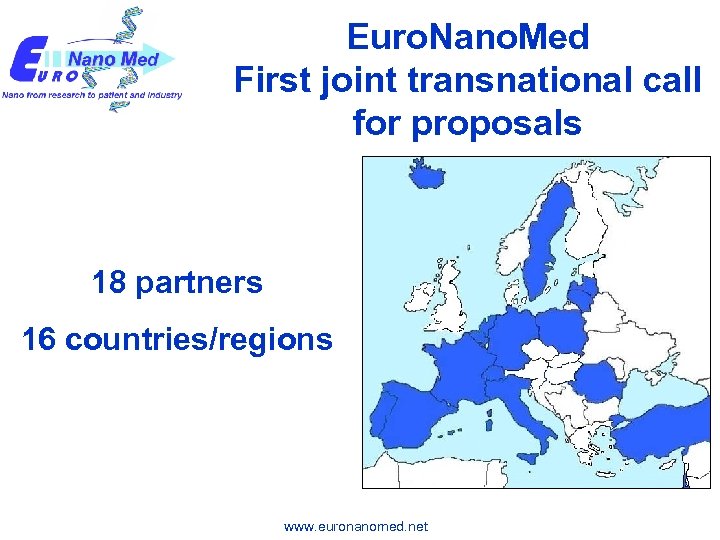
Euro. Nano. Med First joint transnational call for proposals 18 partners 16 countries/regions www. euronanomed. net

Funding organisations in the first call for proposals • Industry, Trade and Tourism Department, Basque Government (ITT), Basque region (Spain) • Swiss National Science Foundation (SNSF), Switzerland • VDI Technologiezentrum Gmb. H, Germany • Instituto de Salud Carlos III (ISCIII), Spain • The French National Research Agency (ANR), France • The Chief Scientist Office, The Ministry of Health (CSO-MOH), Israel • The Icelandic Centre for Research (RANNIS), Iceland • The Latvian Academy of Sciences (LAS), Latvia • Senter. Novem, The Netherlands • National Centre for Research and Development (NCBi. R), Poland • Fundacao para a Ciencia e a Tecnologia (FCT), Portugal • National Center for Programme Management (CNMP), Romania • The Swedish Research Council (SRC)(Vetenskapsrådet), Sweden • The Swedish Governmental Agency for Innovation Systems (VINNOVA ), Sweden • The Scientific and Technological Research Council of Turkey (TUBITAK), Turkey • Science Council of Lithuania (LSC), Lithuania • Economic development, research and innovation Directorate, Veneto region (Italy) • Public Service of Wallonia: Operational General Directorate for Economy, Employment, and Research (DGOEER), Walloon Region (Belgium) www. euronanomed. net

Topics • Diagnostics • Targeted drug delivery • Regenerative medicine www. euronanomed. net

Aim of the 1 st call for proposals • To find new medical solutions • To enable an effective multi-national collaboration on research projects – Promoting multidisciplinary efforts – Encouraging translational research proposals that combine either basic/clinical and/or industrial approach – Having an added value in funding the transnational collaboration over the individual projects • To support novel, ambitious ideas and possibility of application to end users www. euronanomed. net

Eligibility criteria Consortia with room for flexibility – 3 -7 partners from at least three partnering countries – At least one clinical/public health research team and/or one industrial research team per consortium – At least two categories (academic/clinical/industrial teams) – Project consortium partners´ eligibility depends upon national regulation www. euronanomed. net

Funding, 1 st call • Indicative total funding 15 -20 M€ • Funding for up to 3 years • Selected projects receive funding directly from their corresponding national/regional funding bodies after meeting their national regulations www. euronanomed. net

Time lines, 1 st call • Call opens: End of May 2009 • Call closes: September 1 st 2009 • Funding decision: December 2009 www. euronanomed. net

For more information • Euro. Nano. Med Call office Veneto Nanotech s. c. p. a. Via San Crispino, 106 35129 Padova – Italy : +39 049 7705520 email: calloffice@euronanomed. net website: www. euronanomed. net • National/regional contacts www. euronanomed. net

Together we're building nanomedicine for the future! www. euronanomed. net
ffea5d350b964887d351358971a8280a.ppt