f31c834cb86e0c716709460608ffc8a6.ppt
- Количество слайдов: 21
 Eurocan. Platform A Platform of European Cancer Research Centres for Translational Research Ulrik Ringborg Cancer. Center Karolinska
Eurocan. Platform A Platform of European Cancer Research Centres for Translational Research Ulrik Ringborg Cancer. Center Karolinska
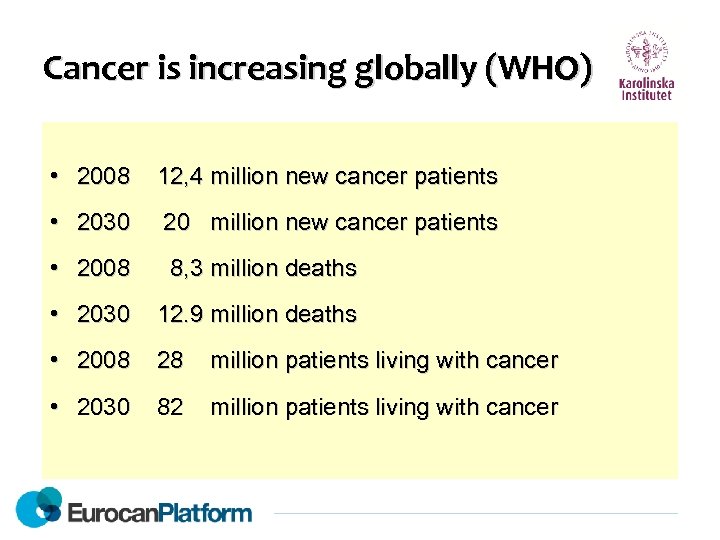 Cancer is increasing globally (WHO) • 2008 12, 4 million new cancer patients • 2030 20 million new cancer patients • 2008 8, 3 million deaths • 2030 12. 9 million deaths • 2008 28 million patients living with cancer • 2030 82 million patients living with cancer
Cancer is increasing globally (WHO) • 2008 12, 4 million new cancer patients • 2030 20 million new cancer patients • 2008 8, 3 million deaths • 2030 12. 9 million deaths • 2008 28 million patients living with cancer • 2030 82 million patients living with cancer
 Conclusions from epidemiological analyses • Cancer is one of the main chronic diseases • Present time trends of improvement of the mortality rate in Europe is not enough to decrease the number of cancer deaths over the next decades • At the same time, cancer biology & modern technologies offer new possibilities to change the negative trends
Conclusions from epidemiological analyses • Cancer is one of the main chronic diseases • Present time trends of improvement of the mortality rate in Europe is not enough to decrease the number of cancer deaths over the next decades • At the same time, cancer biology & modern technologies offer new possibilities to change the negative trends
 Strategies to decrease the problem • Improve prevention • Increase cure rate • Early detection is the bridge between prevention & therapeutics
Strategies to decrease the problem • Improve prevention • Increase cure rate • Early detection is the bridge between prevention & therapeutics
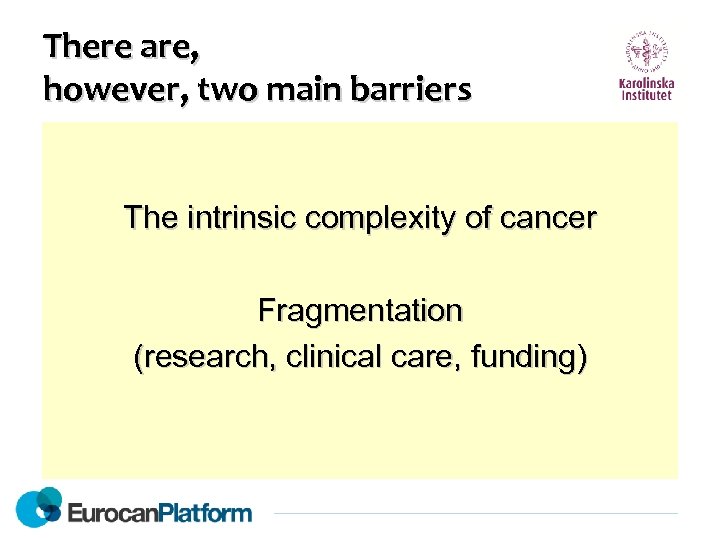 There are, however, two main barriers The intrinsic complexity of cancer Fragmentation (research, clinical care, funding)
There are, however, two main barriers The intrinsic complexity of cancer Fragmentation (research, clinical care, funding)
 Fragmentation of cancer research – main reasons Suboptimal translational cancer resesarch Bridging between basic and clinical research – early translational research can be improved Bridging between clinical research and implementation and evaluation in the clinical care – late translational research should be improved Lack of critical mass Eurocan project
Fragmentation of cancer research – main reasons Suboptimal translational cancer resesarch Bridging between basic and clinical research – early translational research can be improved Bridging between clinical research and implementation and evaluation in the clinical care – late translational research should be improved Lack of critical mass Eurocan project
 Critical mass • The Comprehensive Cancer Center (CCC) to guarantee the critical mass for clinical care & infrastructures for research & education Integration of care, research and education – the linkage between research & innovation • Network of CCCs to guarantee the critical mass for translational cancer research - Patients - Biological materials - Technological resources - Competences
Critical mass • The Comprehensive Cancer Center (CCC) to guarantee the critical mass for clinical care & infrastructures for research & education Integration of care, research and education – the linkage between research & innovation • Network of CCCs to guarantee the critical mass for translational cancer research - Patients - Biological materials - Technological resources - Competences
 A key message • Collaboration between individual research groups is no longer the solution! • Collaboration between centres is mandatory to guarantee infrastructure support, critical mass of expertise & resources, as well as to improve coordination.
A key message • Collaboration between individual research groups is no longer the solution! • Collaboration between centres is mandatory to guarantee infrastructure support, critical mass of expertise & resources, as well as to improve coordination.
 A European platform for translational cancer research The platform will bring together Comprehensive Cancer Centres with a strong research agenda with basic/preclinical Cancer Centres in an integrated network to collaborate and share resources to optimize the translational process and to increase global competitiveness * Such a platform of centres is the only possible way to reach the critical mass and sustainability that is necessary to innovate and deliver in all areas of cancer research * The Stockholm Declaration, Molecular Oncology, 2, 2008
A European platform for translational cancer research The platform will bring together Comprehensive Cancer Centres with a strong research agenda with basic/preclinical Cancer Centres in an integrated network to collaborate and share resources to optimize the translational process and to increase global competitiveness * Such a platform of centres is the only possible way to reach the critical mass and sustainability that is necessary to innovate and deliver in all areas of cancer research * The Stockholm Declaration, Molecular Oncology, 2, 2008
 Eurocan. Platform - No. E Structuring translational cancer research between cancer research centres in Europe A structure for innovative translational research on prevention, early detection and therapeutics The whole cancer research continuum is covered – 16 WPs * Duration of project – 5 years * 12 million euros * 28 participants from 11 countries
Eurocan. Platform - No. E Structuring translational cancer research between cancer research centres in Europe A structure for innovative translational research on prevention, early detection and therapeutics The whole cancer research continuum is covered – 16 WPs * Duration of project – 5 years * 12 million euros * 28 participants from 11 countries
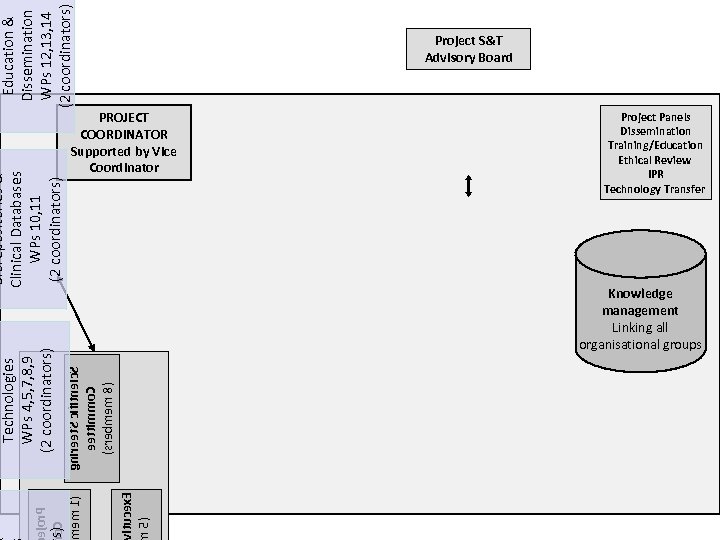 vitucex. E m 5( cejor. P ms)C o bmem 1( gnireet. S cifitneic. S eettimmo. C )srebmem 8( Technologies WPs 4, 5, 7, 8, 9 (2 coordinators) Biorepositories & Clinical Databases WPs 10, 11 (2 coordinators) Education & Dissemination WPs 12, 13, 14 (2 coordinators) Project S&T Advisory Board PROJECT COORDINATOR Supported by Vice Coordinator Project Panels Dissemination Training/Education Ethical Review IPR Technology Transfer Knowledge management Linking all organisational groups
vitucex. E m 5( cejor. P ms)C o bmem 1( gnireet. S cifitneic. S eettimmo. C )srebmem 8( Technologies WPs 4, 5, 7, 8, 9 (2 coordinators) Biorepositories & Clinical Databases WPs 10, 11 (2 coordinators) Education & Dissemination WPs 12, 13, 14 (2 coordinators) Project S&T Advisory Board PROJECT COORDINATOR Supported by Vice Coordinator Project Panels Dissemination Training/Education Ethical Review IPR Technology Transfer Knowledge management Linking all organisational groups
 Project Steering Committee • Each partner presented by one person • WP-leader not representing the partner may be nonvoting member • Majority required for decision • Meetings twice annualy
Project Steering Committee • Each partner presented by one person • WP-leader not representing the partner may be nonvoting member • Majority required for decision • Meetings twice annualy
 Role of the Project Steering Committee • Delegate certain areas of responsibility to the CO or ad hoc groups of consortium members. • Provide overall project management policy. • Make formal decisions on the project & project strategy. • Define, allocate and develop tasks. • Check the progress of the work. • Ensure full integration and coordination of the research teams. • Plan for change and risk management.
Role of the Project Steering Committee • Delegate certain areas of responsibility to the CO or ad hoc groups of consortium members. • Provide overall project management policy. • Make formal decisions on the project & project strategy. • Define, allocate and develop tasks. • Check the progress of the work. • Ensure full integration and coordination of the research teams. • Plan for change and risk management.
 Role of the Project Steering Committee, cont • • • Author and implement the Quality Assurance Plan. Coordinate the preparation of technical reports. Define the budgets for the individual parts of the project. Reviewing and proposing budget reallocations Identify potential new project partners. Permit formal exchanges of information between the partners. • Ensure optimal use of project results. • Develop decision-making structures.
Role of the Project Steering Committee, cont • • • Author and implement the Quality Assurance Plan. Coordinate the preparation of technical reports. Define the budgets for the individual parts of the project. Reviewing and proposing budget reallocations Identify potential new project partners. Permit formal exchanges of information between the partners. • Ensure optimal use of project results. • Develop decision-making structures.
 Management • Co-ordinator Ulrik Ringborg • Vice Co-ordinator Rolf Lewensohn • Project Manager Christina von Gertten • Project Administrator Evelyn Göransson • Support – Grants Office KI, Miles Davies – Legal Council at KI
Management • Co-ordinator Ulrik Ringborg • Vice Co-ordinator Rolf Lewensohn • Project Manager Christina von Gertten • Project Administrator Evelyn Göransson • Support – Grants Office KI, Miles Davies – Legal Council at KI
 Executive committee • • • Ulrik Ringborg Rolf Lewensohn Anton Berns Anne-Lise Börresen-Dale Julio Celis Day-to-day responsbilities delegated by the Project Steering Committee
Executive committee • • • Ulrik Ringborg Rolf Lewensohn Anton Berns Anne-Lise Börresen-Dale Julio Celis Day-to-day responsbilities delegated by the Project Steering Committee
 Scientific Steering Committee Anton Berns (chairman) Alexander Eggermont René Bernards Marco Pierotti Carlos Caldas Ulrik Ringborg Julio Celis Thomas Tursz
Scientific Steering Committee Anton Berns (chairman) Alexander Eggermont René Bernards Marco Pierotti Carlos Caldas Ulrik Ringborg Julio Celis Thomas Tursz
 Role of Scientific Steering Committee • • Scientific co-ordination of the project Propose & evaluate research projects Propose sharing of research resources Support the scientific integration in the Eurocan. Platform Deal with legal & ethical issues Participate in discussions with the industry Prioritize training & exchange of researchers Work for research support & sustainability
Role of Scientific Steering Committee • • Scientific co-ordination of the project Propose & evaluate research projects Propose sharing of research resources Support the scientific integration in the Eurocan. Platform Deal with legal & ethical issues Participate in discussions with the industry Prioritize training & exchange of researchers Work for research support & sustainability
 Scientific Advisory Board • Hedvig Hricak • Mark Israel • John Mendelsohn • Richard Schilsky
Scientific Advisory Board • Hedvig Hricak • Mark Israel • John Mendelsohn • Richard Schilsky
 A Comprehensive Platform for Translational Cancer Research • Increase the interaction between cancer biology & clinical research • New strategies for research on prevention, early detection & therapeutics • Increase infrastructure support & critical mass overall • Cover the cancer research continuum including late translational research • Involve methods to measure innovation prevention & cancer care
A Comprehensive Platform for Translational Cancer Research • Increase the interaction between cancer biology & clinical research • New strategies for research on prevention, early detection & therapeutics • Increase infrastructure support & critical mass overall • Cover the cancer research continuum including late translational research • Involve methods to measure innovation prevention & cancer care
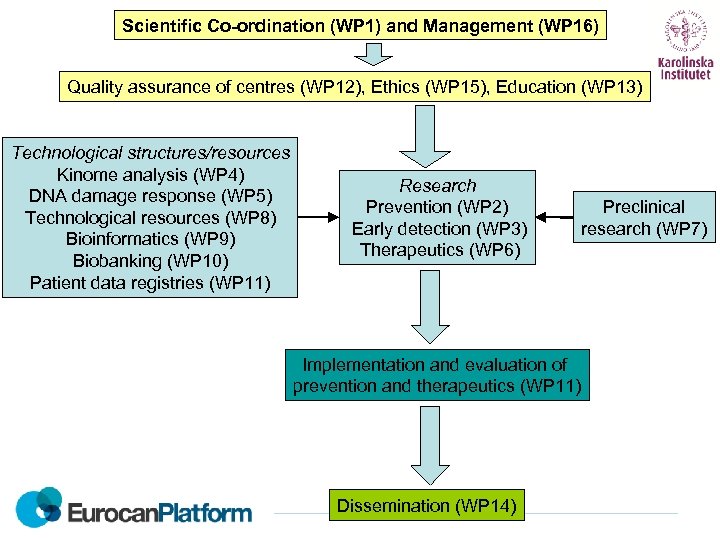 Scientific Co-ordination (WP 1) and Management (WP 16) Quality assurance of centres (WP 12), Ethics (WP 15), Education (WP 13) Technological structures/resources Kinome analysis (WP 4) DNA damage response (WP 5) Technological resources (WP 8) Bioinformatics (WP 9) Biobanking (WP 10) Patient data registries (WP 11) Research Prevention (WP 2) Early detection (WP 3) Therapeutics (WP 6) Implementation and evaluation of prevention and therapeutics (WP 11) Dissemination (WP 14) Preclinical research (WP 7)
Scientific Co-ordination (WP 1) and Management (WP 16) Quality assurance of centres (WP 12), Ethics (WP 15), Education (WP 13) Technological structures/resources Kinome analysis (WP 4) DNA damage response (WP 5) Technological resources (WP 8) Bioinformatics (WP 9) Biobanking (WP 10) Patient data registries (WP 11) Research Prevention (WP 2) Early detection (WP 3) Therapeutics (WP 6) Implementation and evaluation of prevention and therapeutics (WP 11) Dissemination (WP 14) Preclinical research (WP 7)


