 EURAMOS 1 A randomised trial of the European and American Osteosarcoma Study Group to optimize treatment strategies for resectable osteosarcoma based on histological response to pre-operative chemotherapy Jeremy Whelan for the EURAMOS 1 Trial Management Group, 4 th November 2006
EURAMOS 1 A randomised trial of the European and American Osteosarcoma Study Group to optimize treatment strategies for resectable osteosarcoma based on histological response to pre-operative chemotherapy Jeremy Whelan for the EURAMOS 1 Trial Management Group, 4 th November 2006
 EURAMOS • EURAMOS is the European and American Osteosarcoma Study Group. The study was founded in 2001. § § Children’s Oncology Group Cooperative Osteosarcoma Study Group European Osteosarcoma Intergroup Scandinavian Sarcoma Group
EURAMOS • EURAMOS is the European and American Osteosarcoma Study Group. The study was founded in 2001. § § Children’s Oncology Group Cooperative Osteosarcoma Study Group European Osteosarcoma Intergroup Scandinavian Sarcoma Group
 EURAMOS • Established Oct 2001 • Aim § to improve survival from OS • Objectives § § to carry out large international randomised trials to facilitate biological research to seek new therapeutic approaches to develop common understanding and methodologies for staging, pathology etc.
EURAMOS • Established Oct 2001 • Aim § to improve survival from OS • Objectives § § to carry out large international randomised trials to facilitate biological research to seek new therapeutic approaches to develop common understanding and methodologies for staging, pathology etc.
 EURAMOS 1 • Co-sponsored § MRC in Europe § COG for North America • Co-ordinating Data Centre § MRC Clinical Trials Unit
EURAMOS 1 • Co-sponsored § MRC in Europe § COG for North America • Co-ordinating Data Centre § MRC Clinical Trials Unit
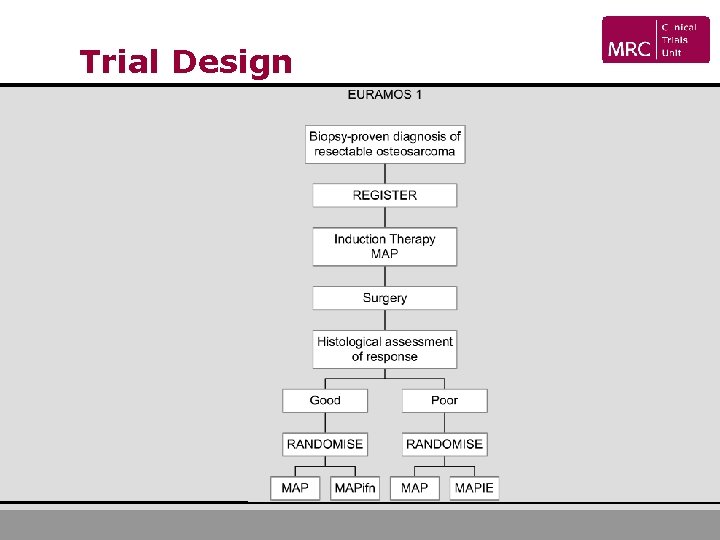 Trial Design
Trial Design
 Trial Design • Key registration criteria § High grade OS of extremity or axial skeleton § Resectable disease § Fit for treatment
Trial Design • Key registration criteria § High grade OS of extremity or axial skeleton § Resectable disease § Fit for treatment
 Trial Design • Key randomisation criteria § Adequate pre-operative chemotherapy § Macroscopically complete resection of primary tumour § Complete removal of all metastases or complete removal planned and feasible § Histological response assessment
Trial Design • Key randomisation criteria § Adequate pre-operative chemotherapy § Macroscopically complete resection of primary tumour § Complete removal of all metastases or complete removal planned and feasible § Histological response assessment
 Trial Design • Primary Objectives § Addition of IE to post-op MAP chemotherapy improves EFS for poor responders § Addition of ifn to post-op MAP chemotherapy improves EFS for good responders
Trial Design • Primary Objectives § Addition of IE to post-op MAP chemotherapy improves EFS for poor responders § Addition of ifn to post-op MAP chemotherapy improves EFS for good responders
 Trial Design • Secondary Objectives § Additional treatment improves OS, short and long term toxicity and quality of life § Additional treatment improves EFS in localised disease § Investigate biological and clinical correlated to histological response and outcome § International cooperation in trials § Examine outcome of the entire cohort of patients
Trial Design • Secondary Objectives § Additional treatment improves OS, short and long term toxicity and quality of life § Additional treatment improves EFS in localised disease § Investigate biological and clinical correlated to histological response and outcome § International cooperation in trials § Examine outcome of the entire cohort of patients
 Trial Design- additional studies • Quality of Life • Biology Studies
Trial Design- additional studies • Quality of Life • Biology Studies
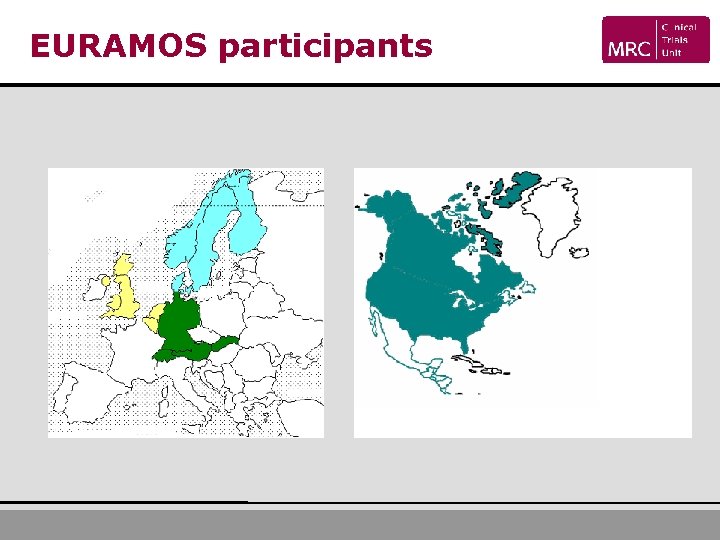 EURAMOS participants
EURAMOS participants
 Progress: open and recruiting USA Registered Randomised 111 40 Germany 87 42 UK 42 17 Netherlands 14 5 Norway 11 4 Canada 10 2 Belgium 9 5 Switzerland 8 5 Sweden 5 3 Demark 2 0 Hungary 1 0 Australia 2 0 302 123 Total
Progress: open and recruiting USA Registered Randomised 111 40 Germany 87 42 UK 42 17 Netherlands 14 5 Norway 11 4 Canada 10 2 Belgium 9 5 Switzerland 8 5 Sweden 5 3 Demark 2 0 Hungary 1 0 Australia 2 0 302 123 Total
 Progress: almost ready • Austria • Finland
Progress: almost ready • Austria • Finland
 Progress: requests to join • • Czech Republic Ireland Poland Israel
Progress: requests to join • • Czech Republic Ireland Poland Israel
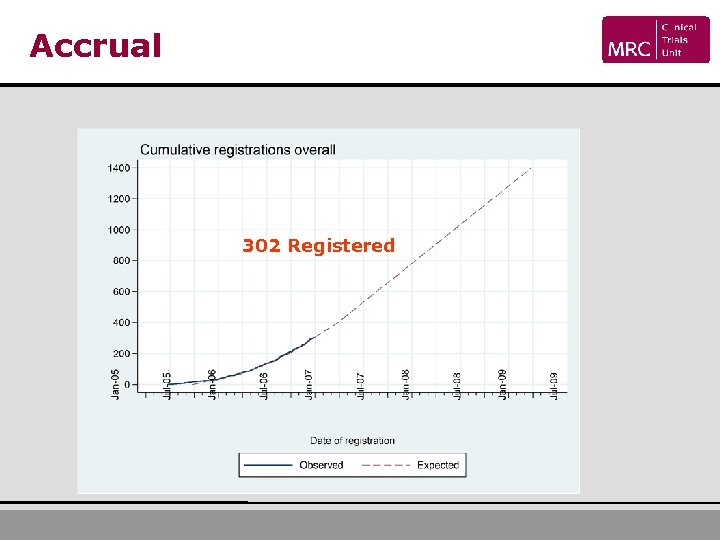 Accrual 302 Registered
Accrual 302 Registered
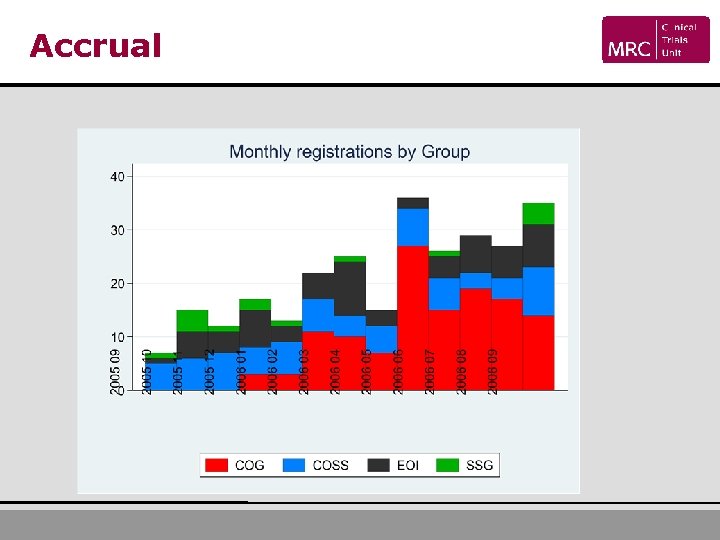 Accrual • 302 Registered
Accrual • 302 Registered
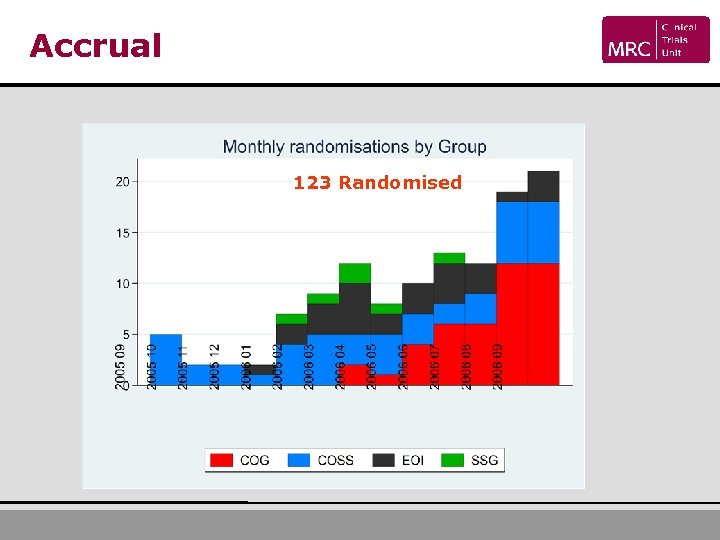 Accrual 123 Randomised
Accrual 123 Randomised
 EURAMOS 1 • Chief Investigators § § § Mark Bernstein (TMG Chair) Neyssa Marina (COG) Stephan Bielack (COSS) Jeremy Whelan (EOI) Sigbjorn Smeland (SSG) • Website § www. euramos. org
EURAMOS 1 • Chief Investigators § § § Mark Bernstein (TMG Chair) Neyssa Marina (COG) Stephan Bielack (COSS) Jeremy Whelan (EOI) Sigbjorn Smeland (SSG) • Website § www. euramos. org