f543e2fe240a4e549bfb3484a25e0893.ppt
- Количество слайдов: 16

EQASs for Blood Borne Virus Genomes and BSE Prions from Cattle Brain and INSTAND Reference Materials H. -P. Grunert 1, 2, K. -O. Habermehl 1, 2 , V. Lindig 1 and H. Zeichhardt 1, 3 1 Charité University Medicine Berlin, Campus Benjamin Franklin, Institute for Infectious Diseases Medicine, Dept. Virology, Berlin 2 Institute for Biotechnological Diagnostics (GBD), Berlin 3 Institute for Standardization and Documentation in Medical Laboratory (INSTAND), Düsseldorf (WHO Collaborating Center) Collaborating Centers of International Consortium for Blood Safety (ICBS), New York

National and WHO International EQASs in Immunology/Virology Serology PCR/NAT HIV-1/2 Cytomegalovirus HIV-1 (RNA) Enterov. (PCR/NAT) HIV-1 p 24 Epstein-Barr Virus Hepatitis B Virus Adenov. (PCR/NAT) Hepatitis A Virus Varicella Zoster V. Hepatitis C Virus Hepatitis B V. Prg. I Herpes simplex V. Cytomegalovirus Hepatitis B V. Prg. II Measlesvirus Varicella Zoster V. Hepatitis C Virus Mumpsvirus Herpes simpl. V. 1 Hepatitis D Virus Resp. Sync. V. AG Herpes simpl. V. 2 Hepatitis E Virus Influenzav. A/B AG Influenzavirus A/B Rubellavirus TBE Virus Parvovirus B 19 BSE (Pr. Psc) HPV (PCR/NAT) Samples total: > 549. 000 Grunert, So. GAT, Bethesda, 2005 2

EQASs in Virology Virus serology since 1988: > 446, 000 samples total (20 programs) Virus genome detection since 1997: > 103, 000 samples total (12 programs) Organization INSTAND (Düsseldorf) Authorized by Bundesärztekammer (BÄK) World Health Organization (WHO) International Consortium for Blood Safety (ICBS) Scientific societies Deutsche Vereinigung zur Bekämpfung der Viruskrankheiten (DVV) Gesellschaft für Virologie (Gf. V) Deutsche Gesellschaft für Hygiene und Mikrobiologie (DGHM) Grunert, So. GAT, Bethesda, 2005 3

Experiences from EQAS for HIV, HCV and HBV: Evaluation of quantitative PCR/NAT by median +/- 0. 5 log 10 IU/ml versus copies/ml Grunert, So. GAT, Bethesda, 2005 4
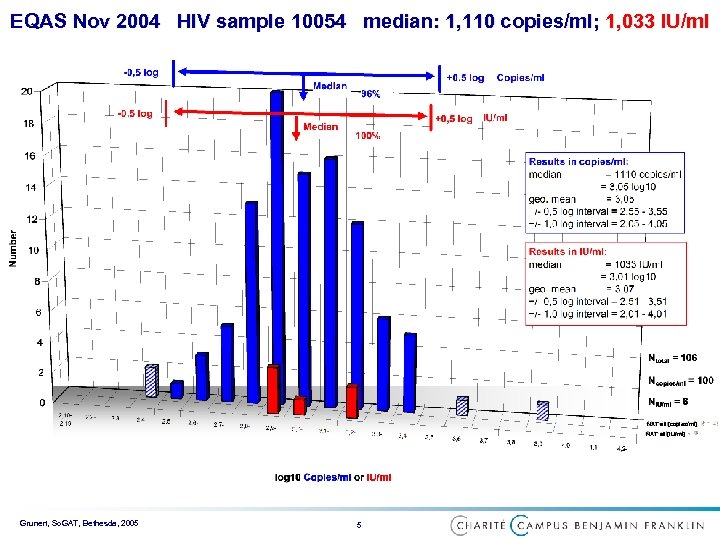
EQAS Nov 2004 HIV sample 10054 median: 1, 110 copies/ml; 1, 033 IU/ml NAT all [copies/ml] NAT all [IU/ml] Grunert, So. GAT, Bethesda, 2005 5
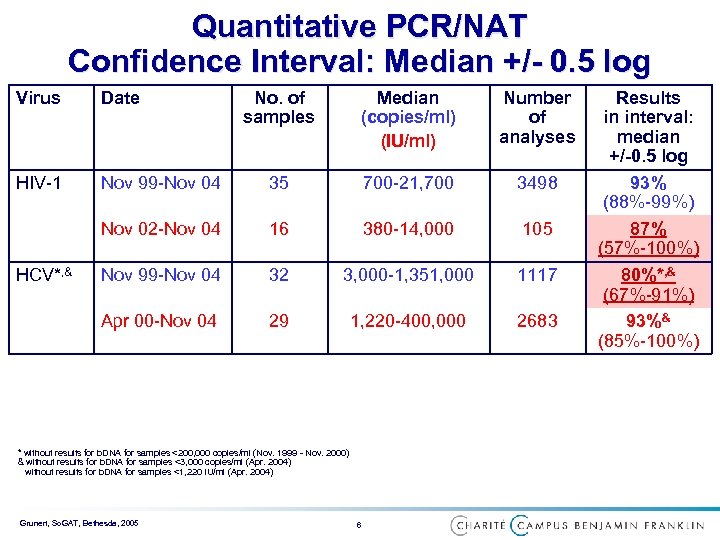
Quantitative PCR/NAT Confidence Interval: Median +/- 0. 5 log Virus Date HIV-1 Median (copies/ml) (IU/ml) Number of analyses Nov 99 -Nov 04 35 700 -21, 700 3498 Nov 02 -Nov 04 16 380 -14, 000 105 Nov 99 -Nov 04 32 3, 000 -1, 351, 000 1117 Apr 00 -Nov 04 HCV*, & No. of samples 29 1, 220 -400, 000 2683 * without results for b. DNA for samples <200, 000 copies/ml (Nov. 1999 - Nov. 2000) & without results for b. DNA for samples <3, 000 copies/ml (Apr. 2004) without results for b. DNA for samples <1, 220 IU/ml (Apr. 2004) Grunert, So. GAT, Bethesda, 2005 6 Results in interval: median +/-0. 5 log 93% (88%-99%) 87% (57%-100%) 80%*, & (67%-91%) 93%& (85%-100%)
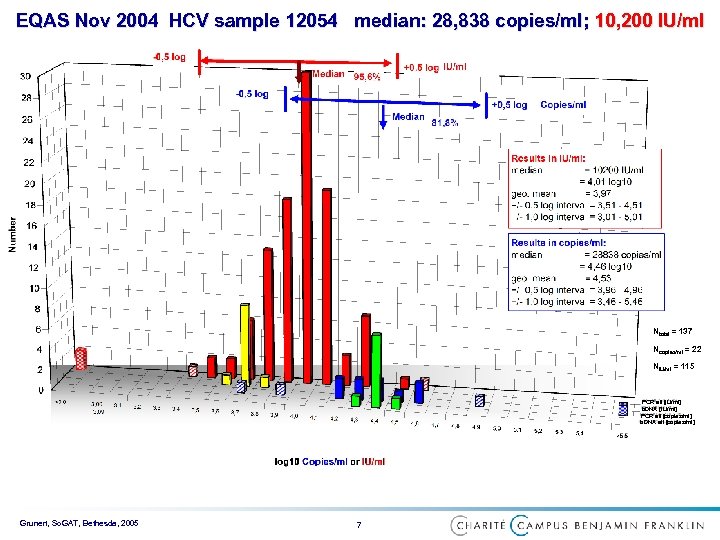
EQAS Nov 2004 HCV sample 12054 median: 28, 838 copies/ml; 10, 200 IU/ml Ntotal = 137 Ncopies/ml = 22 NIU/ml = 115 PCR all [IU/ml] b. DNA [IU/ml] PCR all [copies/ml] b. DNA all [copies/ml] Grunert, So. GAT, Bethesda, 2005 7
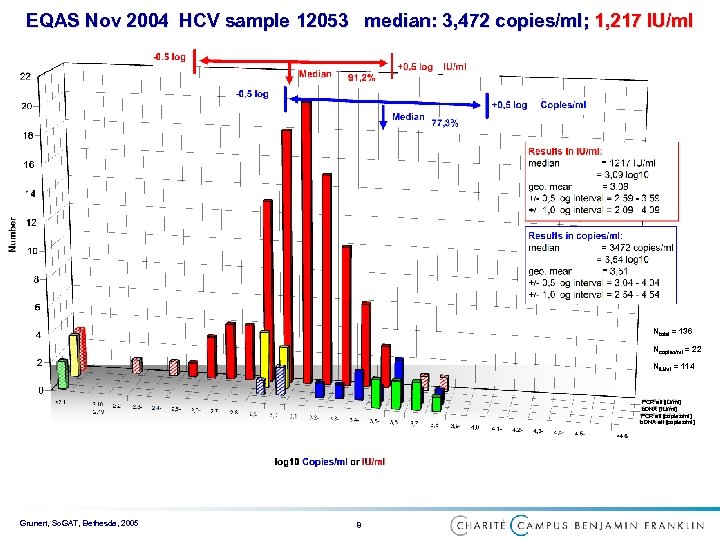
EQAS Nov 2004 HCV sample 12053 median: 3, 472 copies/ml; 1, 217 IU/ml Ntotal = 136 Ncopies/ml = 22 NIU/ml = 114 PCR all [IU/ml] b. DNA [IU/ml] PCR all [copies/ml] b. DNA all [copies/ml] Grunert, So. GAT, Bethesda, 2005 8
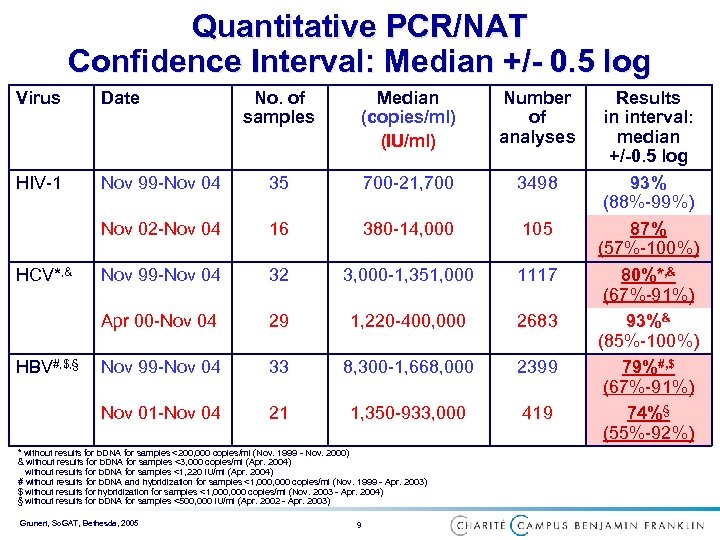
Quantitative PCR/NAT Confidence Interval: Median +/- 0. 5 log Virus Date HIV-1 Number of analyses Nov 99 -Nov 04 35 700 -21, 700 3498 16 380 -14, 000 105 Nov 99 -Nov 04 32 3, 000 -1, 351, 000 1117 Apr 00 -Nov 04 29 1, 220 -400, 000 2683 Nov 99 -Nov 04 33 8, 300 -1, 668, 000 2399 Nov 01 -Nov 04 HBV#, $, § Median (copies/ml) (IU/ml) Nov 02 -Nov 04 HCV*, & No. of samples 21 1, 350 -933, 000 419 * without results for b. DNA for samples <200, 000 copies/ml (Nov. 1999 - Nov. 2000) & without results for b. DNA for samples <3, 000 copies/ml (Apr. 2004) without results for b. DNA for samples <1, 220 IU/ml (Apr. 2004) # without results for b. DNA and hybridization for samples <1, 000 copies/ml (Nov. 1999 - Apr. 2003) $ without results for hybridization for samples <1, 000 copies/ml (Nov. 2003 - Apr. 2004) § without results for b. DNA for samples <500, 000 IU/ml (Apr. 2002 - Apr. 2003) Grunert, So. GAT, Bethesda, 2005 9 Results in interval: median +/-0. 5 log 93% (88%-99%) 87% (57%-100%) 80%*, & (67%-91%) 93%& (85%-100%) 79%#, $ (67%-91%) 74%§ (55%-92%)
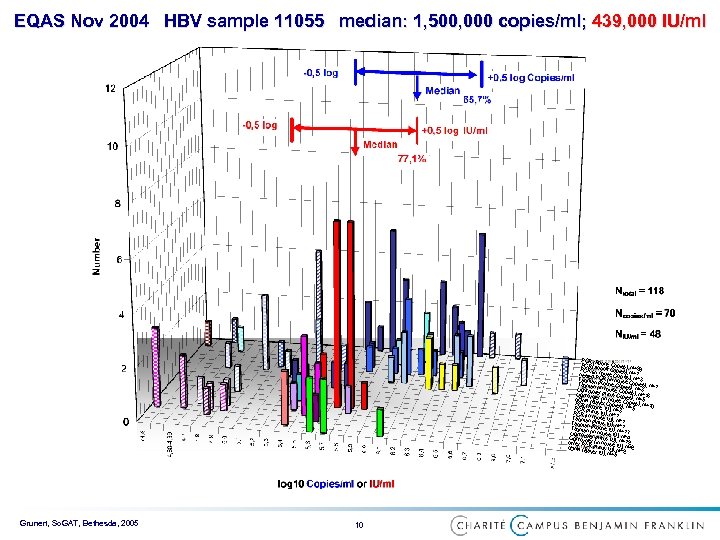
EQAS Nov 2004 HBV sample 11055 median: 1, 500, 000 copies/ml; 439, 000 IU/ml PCR (R PCR (A oche Copies) PCR (in bbott Copies); ; N=29 nested house Copies)N=1 P Taqman CR (in house ; N=3 Taqman (Roche Copies); N=1 Lightcyc (in house Cop s); N=3 Lightcyc ler (Artus Cop ies); N=13 b. DNA (Bler (in house ies); N=5 PCR (R ayer Copies); N=10 PCR (A oche IU); N=3 ; N=5 PCR (in rtus IU); N=1 Taqman house IU); N=1 Taqman (Artus IU); N =1 Taqman (Roche IU); N Lightcyc (in house IU); =17 Lightcyc ler (Artus IU); N=4 other P ler (in house N=12 C b. DNA (B R (Artus IU); N=2 N ayer IU ); N=5 =2 Grunert, So. GAT, Bethesda, 2005 10
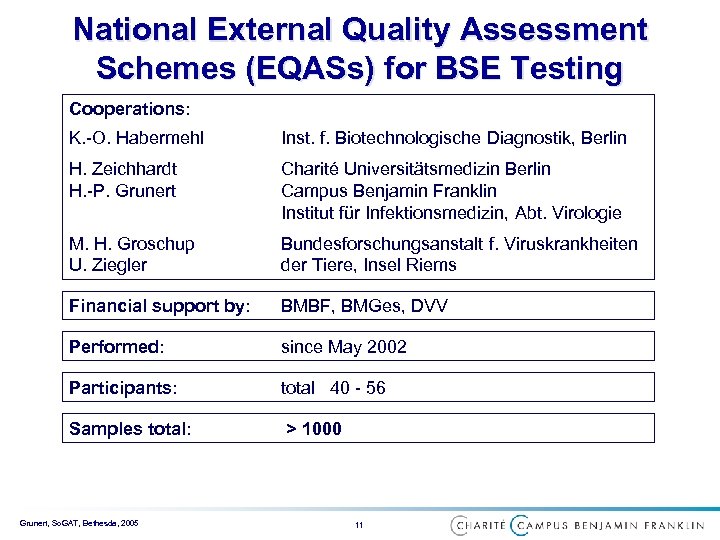
National External Quality Assessment Schemes (EQASs) for BSE Testing Cooperations: K. -O. Habermehl Inst. f. Biotechnologische Diagnostik, Berlin H. Zeichhardt H. -P. Grunert Charité Universitätsmedizin Berlin Campus Benjamin Franklin Institut für Infektionsmedizin, Abt. Virologie M. H. Groschup U. Ziegler Bundesforschungsanstalt f. Viruskrankheiten der Tiere, Insel Riems Financial support by: BMBF, BMGes, DVV Performed: since May 2002 Participants: total 40 - 56 Samples total: > 1000 Grunert, So. GAT, Bethesda, 2005 11
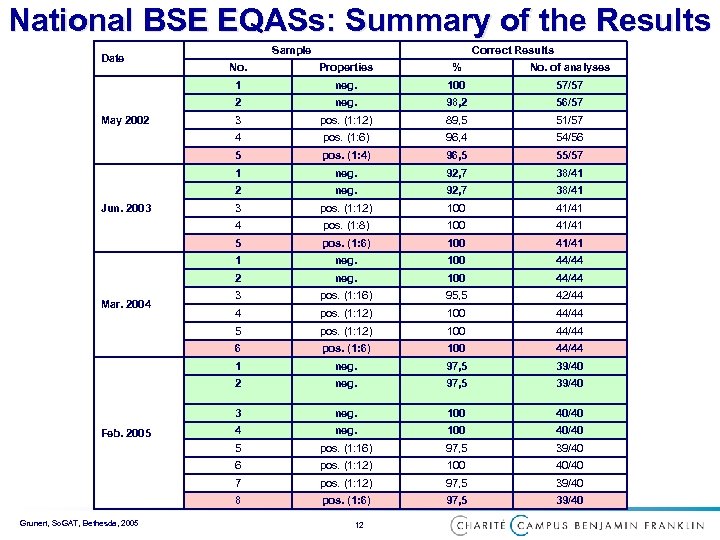
National BSE EQASs: Summary of the Results Date Sample Correct Results 100 57/57 neg. 98, 2 56/57 3 pos. (1: 12) 89, 5 51/57 pos. (1: 6) 96, 4 54/56 pos. (1: 4) 96, 5 55/57 1 neg. 92, 7 38/41 2 neg. 92, 7 38/41 3 pos. (1: 12) 100 41/41 4 pos. (1: 8) 100 41/41 5 pos. (1: 6) 100 41/41 1 neg. 100 44/44 2 neg. 100 44/44 3 pos. (1: 16) 95, 5 42/44 4 pos. (1: 12) 100 44/44 5 pos. (1: 12) 100 44/44 6 pos. (1: 6) 100 44/44 1 neg. 97, 5 39/40 2 neg. 97, 5 39/40 3 neg. 100 40/40 4 neg. 100 40/40 5 pos. (1: 16) 97, 5 39/40 6 pos. (1: 12) 100 40/40 7 pos. (1: 12) 97, 5 39/40 8 Grunert, So. GAT, Bethesda, 2005 neg. 5 Feb. 2005 No. of analyses 4 Mar. 2004 % 2 Jun. 2003 Properties 1 May 2002 No. pos. (1: 6) 97, 5 39/40 12
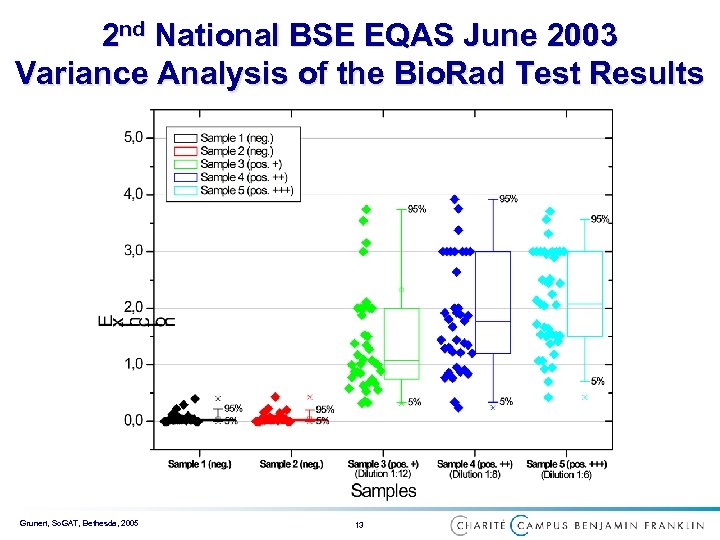
2 nd National BSE EQAS June 2003 Variance Analysis of the Bio. Rad Test Results Grunert, So. GAT, Bethesda, 2005 13

Global Network for External and Internal Quality Control External Quality Control (EQASs) • EQASs (Germany, Europe, WHO) with >990 laboratories with all routine tests • 3 x per year (2006: 4 x per year) Internal Quality Control Surplus samples from EQASs • "Run" controls • daily National reference materials • "Run" controls in coop. with PEI • daily Identical controls for: Grunert, So. GAT, Bethesda, 2005 Diagnostic labs Blood banks Industries Licensing organizations (e. g. PEI) Scientific labs 14
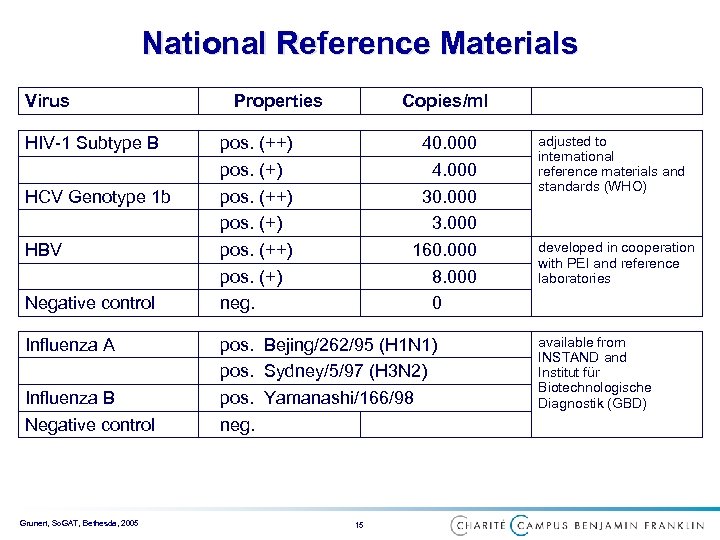
National Reference Materials Virus HIV-1 Subtype B HCV Genotype 1 b HBV Negative control Influenza A Influenza B Negative control Grunert, So. GAT, Bethesda, 2005 Properties Copies/ml pos. (++) pos. (+) neg. 40. 000 4. 000 30. 000 3. 000 160. 000 8. 000 0 pos. Bejing/262/95 (H 1 N 1) pos. Sydney/5/97 (H 3 N 2) pos. Yamanashi/166/98 neg. 15 adjusted to international reference materials and standards (WHO) developed in cooperation with PEI and reference laboratories available from INSTAND and Institut für Biotechnologische Diagnostik (GBD)

INSTAND Reference Materials (www. instand-ev. de) INSTAND INSTITUTE FOR STANDARDIZATION AND DOCUMENTATION IN MEDICAL LABORATORIES e. V. P. O. Box 250211, D-40093 Düsseldorf, Germany Tel. : +49 -211 -159213 0, Fax: +49 -211 -159213 30, E-mail: instand@instand-ev. de Samples for Test Assessment of Virus Diagnostics National Reference Samples (National Working References) Surplus Samples of External Quality Assessment Schemes in Virus Immunology and Virus PCR/NAT As of February 2005 Grunert, So. GAT, Bethesda, 2005 16
f543e2fe240a4e549bfb3484a25e0893.ppt