c54b02b7bccc64be36d668fd72280f88.ppt
- Количество слайдов: 18
 Enzimologija Nastavnik: Tanja Ćirković Veličković
Enzimologija Nastavnik: Tanja Ćirković Veličković
 Pregled n n n Opšte informacie o predmetu i načinu ocenjivanja Literatura Opšti pojmovi Nomenklatura enzima Uvodna znanja
Pregled n n n Opšte informacie o predmetu i načinu ocenjivanja Literatura Opšti pojmovi Nomenklatura enzima Uvodna znanja
 Program i organizacija vežbi Teorijske vezbe – izračunavanja u enzimologiji n Laboratorijske vežbe: osnovna odredjivanja u enzimologiji n Laboratorijske vežbe: Razni aspekti primene enzima Fond časova predavanja i vežbi: 3 + 3 (teorijske vežbe) Asistent: Marija Stojadinovic i Dragana Stanic n
Program i organizacija vežbi Teorijske vezbe – izračunavanja u enzimologiji n Laboratorijske vežbe: osnovna odredjivanja u enzimologiji n Laboratorijske vežbe: Razni aspekti primene enzima Fond časova predavanja i vežbi: 3 + 3 (teorijske vežbe) Asistent: Marija Stojadinovic i Dragana Stanic n
 Način ocenjivanja n n n Predavanja: 10 bodova Kolokvijum 1: 20 bodova Kolokvijum 2: 20 bodova Seminarski rad: 5 bodova Problemski zadatak: 20 bodova Usmeni deo ispita: 25 bodova
Način ocenjivanja n n n Predavanja: 10 bodova Kolokvijum 1: 20 bodova Kolokvijum 2: 20 bodova Seminarski rad: 5 bodova Problemski zadatak: 20 bodova Usmeni deo ispita: 25 bodova
 Literatura n n n Structure and mechanism in protein science, Alan Fersht Enzimologija: Laboratorijski priručnik, Radivoje Prodanović i Tanja Ćirković Veličković Zbirka zadataka iz enzimologije, Ivanka Karadžić
Literatura n n n Structure and mechanism in protein science, Alan Fersht Enzimologija: Laboratorijski priručnik, Radivoje Prodanović i Tanja Ćirković Veličković Zbirka zadataka iz enzimologije, Ivanka Karadžić
 Enzyme catalysis, not different, just better! Katalizator je supstanca koja dovodi do ubrzanja hemijske reakcije, tako da se pri tom promena G ne menja (IUPAC, 1981. ) n Biološki katalizatori: Proteini (enzimi, abzimi) RNK (ribozimi) Mimetici enzima - Šta enzim radi? Struktura proteina – enzima Hemijska kinetika – enzimska kinetika n
Enzyme catalysis, not different, just better! Katalizator je supstanca koja dovodi do ubrzanja hemijske reakcije, tako da se pri tom promena G ne menja (IUPAC, 1981. ) n Biološki katalizatori: Proteini (enzimi, abzimi) RNK (ribozimi) Mimetici enzima - Šta enzim radi? Struktura proteina – enzima Hemijska kinetika – enzimska kinetika n
 n Sukcinat dehidrogenaza sa vezanim hemom (prostetičnom grupom) Enzim • • Monomeran Homo/hetero dimeran, trimeran etc Oligomeran Multienzimski kompleksi • Holoenzim (apoenzim + kofaktor/koenzim) • • • Kofaktor Koenzim Prosteticna grupa (čvrsto vezan kofaktor)
n Sukcinat dehidrogenaza sa vezanim hemom (prostetičnom grupom) Enzim • • Monomeran Homo/hetero dimeran, trimeran etc Oligomeran Multienzimski kompleksi • Holoenzim (apoenzim + kofaktor/koenzim) • • • Kofaktor Koenzim Prosteticna grupa (čvrsto vezan kofaktor)
 Nomenklatura n n Trivijalna imena enzima E. C. nomenklatura
Nomenklatura n n Trivijalna imena enzima E. C. nomenklatura
 E. C. Nomenklatura enzima Recommendations of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology on the Nomenclature and Classification of Enzymes by the Reactions they Catalyse http: //www. chem. qmul. ac. uk/iubmb/en zyme/ n n
E. C. Nomenklatura enzima Recommendations of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology on the Nomenclature and Classification of Enzymes by the Reactions they Catalyse http: //www. chem. qmul. ac. uk/iubmb/en zyme/ n n
 n n n n EC 1. 1 Oksidoreduktaze Acting on the CH-OH group of donors Contents EC 1. 1. 1 With NAD or NADP as acceptor EC 1. 1. 2 With a cytochrome as acceptor EC 1. 1. 3 With oxygen as acceptor EC 1. 1. 4 With a disulfide as acceptor EC 1. 1. 5 With a quinone or similar compound as acceptor EC 1. 1. 99 With other acceptors EC 2 Transferaze EC 3 Hidrolaze EC 4 Liaze EC 5 Izomeraze EC 6 Ligaze
n n n n EC 1. 1 Oksidoreduktaze Acting on the CH-OH group of donors Contents EC 1. 1. 1 With NAD or NADP as acceptor EC 1. 1. 2 With a cytochrome as acceptor EC 1. 1. 3 With oxygen as acceptor EC 1. 1. 4 With a disulfide as acceptor EC 1. 1. 5 With a quinone or similar compound as acceptor EC 1. 1. 99 With other acceptors EC 2 Transferaze EC 3 Hidrolaze EC 4 Liaze EC 5 Izomeraze EC 6 Ligaze
 n n n n n n EC 1 Oxidoreductases EC 1. 1 Acting on the CH-OH group of donors EC 1. 2 Acting on the aldehyde or oxo group of donors EC 1. 3 Acting on the CH-CH group of donors EC 1. 4 Acting on the CH-NH 2 group of donors EC 1. 5 Acting on the CH-NH group of donors EC 1. 6 Acting on NADH or NADPH EC 1. 7 Acting on other nitrogenous compounds as donors EC 1. 8 Acting on a sulfur group of donors EC 1. 9 Acting on a heme group of donors EC 1. 10 Acting on diphenols and related substances as donors EC 1. 11 Acting on a peroxide as acceptor EC 1. 12 Acting on hydrogen as donor EC 1. 13 Acting on single donors with incorporation of molecular oxygen ( oxygenases) EC 1. 14 Acting on paired donors, with incorporation or reduction of molecular oxygen EC 1. 15 Acting on superoxide radicals as acceptor EC 1. 16 Oxidising metal ions EC 1. 17 Acting on CH or CH 2 groups EC 1. 18 Acting on iron-sulfur proteins as donors EC 1. 19 Acting on reduced flavodoxin as donor EC 1. 20 Acting on phosphorus or arsenic in donors EC 1. 21 Acting on X-H and Y-H to form an X-Y bond EC 1. 97 Other oxidoreductases
n n n n n n EC 1 Oxidoreductases EC 1. 1 Acting on the CH-OH group of donors EC 1. 2 Acting on the aldehyde or oxo group of donors EC 1. 3 Acting on the CH-CH group of donors EC 1. 4 Acting on the CH-NH 2 group of donors EC 1. 5 Acting on the CH-NH group of donors EC 1. 6 Acting on NADH or NADPH EC 1. 7 Acting on other nitrogenous compounds as donors EC 1. 8 Acting on a sulfur group of donors EC 1. 9 Acting on a heme group of donors EC 1. 10 Acting on diphenols and related substances as donors EC 1. 11 Acting on a peroxide as acceptor EC 1. 12 Acting on hydrogen as donor EC 1. 13 Acting on single donors with incorporation of molecular oxygen ( oxygenases) EC 1. 14 Acting on paired donors, with incorporation or reduction of molecular oxygen EC 1. 15 Acting on superoxide radicals as acceptor EC 1. 16 Oxidising metal ions EC 1. 17 Acting on CH or CH 2 groups EC 1. 18 Acting on iron-sulfur proteins as donors EC 1. 19 Acting on reduced flavodoxin as donor EC 1. 20 Acting on phosphorus or arsenic in donors EC 1. 21 Acting on X-H and Y-H to form an X-Y bond EC 1. 97 Other oxidoreductases
 E. C. vs. Trivijalna nomenklatura n EC 1. 1 alcohol dehydrogenase EC 1. 1. 1. 2 alcohol dehydrogenase (NADP+) EC 1. 1. 1. 3 homoserine dehydrogenase EC 1. 1. 1. 4 (R, R)-butanediol dehydrogenase EC 1. 1. 1. 5 acetoin dehydrogenase EC 1. 1. 1. 6 glycerol dehydrogenase EC 1. 1. 1. 7 propanediol-phosphate dehydrogenase EC 1. 1. 1. 8 glycerol-3 -phosphate dehydrogenase (NAD+) EC 1. 1. 1. 9 D-xylulose reductase EC 1. 10 L-xylulose reductase
E. C. vs. Trivijalna nomenklatura n EC 1. 1 alcohol dehydrogenase EC 1. 1. 1. 2 alcohol dehydrogenase (NADP+) EC 1. 1. 1. 3 homoserine dehydrogenase EC 1. 1. 1. 4 (R, R)-butanediol dehydrogenase EC 1. 1. 1. 5 acetoin dehydrogenase EC 1. 1. 1. 6 glycerol dehydrogenase EC 1. 1. 1. 7 propanediol-phosphate dehydrogenase EC 1. 1. 1. 8 glycerol-3 -phosphate dehydrogenase (NAD+) EC 1. 1. 1. 9 D-xylulose reductase EC 1. 10 L-xylulose reductase
 n http: //www. chem. qmul. ac. uk/iubmb/ enzyme/search. html
n http: //www. chem. qmul. ac. uk/iubmb/ enzyme/search. html
 n n Searching the Enzyme list This search form looks at IUBMB Enzyme Nomenclature with a URL starting http: //www. chem. qmul. ac. uk/iubmb/enzyme/ All recommended Enzymes are listed on the web. This search excludes other biochemical recommendations on enzyme kinetics, biochemical thermodynamics, and recommendations made by IUBMB only. To search these click here or for more chemical recommendations and other IUPAC recommendations click here. Search: PEPSIN Some search terms cause problems: For (S)-stylopine use stylopine otherwise all references with S as an initial are included. For germacrene A use "germacrene A"
n n Searching the Enzyme list This search form looks at IUBMB Enzyme Nomenclature with a URL starting http: //www. chem. qmul. ac. uk/iubmb/enzyme/ All recommended Enzymes are listed on the web. This search excludes other biochemical recommendations on enzyme kinetics, biochemical thermodynamics, and recommendations made by IUBMB only. To search these click here or for more chemical recommendations and other IUPAC recommendations click here. Search: PEPSIN Some search terms cause problems: For (S)-stylopine use stylopine otherwise all references with S as an initial are included. For germacrene A use "germacrene A"
 n n n n n Web Results 1 - 10 of about 32 for pepsin [definition]. (0. 06 seconds) EC 3. 4. 23. Aspartic endopeptidases. Contents. EC 3. 4. 23. 1 pepsin A EC 3. 4. 23. 2 pepsin B EC 3. 4. 23. 3 gastricsin EC 3. 4. 23. 4 chymosin EC 3. 4. 23. 5 cathepsin D. . . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/ - 5 k - Cached - Similar pages EC 3. 4. 23. 1 Human pepsin A occurs in five molecular forms. Pig pepsin D [1, 2] is . . . Lee, D. and Ryle, A. P. Pepsin D. A minor component of commercial pepsin. . . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/1. html - 5 k - Cached - Similar pages EC 3. 4. 23. 18 In peptidase family A 1 (pepsin A family). Formerly included in EC 3. 4. 23. 6. . . proteinase of Aspergillus awamori - an analog of penicillopepsin and pepsin. . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/18. html - 7 k - Cached - Similar pages EC 3. 4. 23. 3 Other names: pepsin C; pig parapepsin II; parapepsin II. . . In peptidase family A 1 (pepsin A family). Formerly EC 3. 4. 4. 22. Links to other databases: BRENDA. . . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/3. html - 4 k - Cached - Similar pages
n n n n n Web Results 1 - 10 of about 32 for pepsin [definition]. (0. 06 seconds) EC 3. 4. 23. Aspartic endopeptidases. Contents. EC 3. 4. 23. 1 pepsin A EC 3. 4. 23. 2 pepsin B EC 3. 4. 23. 3 gastricsin EC 3. 4. 23. 4 chymosin EC 3. 4. 23. 5 cathepsin D. . . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/ - 5 k - Cached - Similar pages EC 3. 4. 23. 1 Human pepsin A occurs in five molecular forms. Pig pepsin D [1, 2] is . . . Lee, D. and Ryle, A. P. Pepsin D. A minor component of commercial pepsin. . . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/1. html - 5 k - Cached - Similar pages EC 3. 4. 23. 18 In peptidase family A 1 (pepsin A family). Formerly included in EC 3. 4. 23. 6. . . proteinase of Aspergillus awamori - an analog of penicillopepsin and pepsin. . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/18. html - 7 k - Cached - Similar pages EC 3. 4. 23. 3 Other names: pepsin C; pig parapepsin II; parapepsin II. . . In peptidase family A 1 (pepsin A family). Formerly EC 3. 4. 4. 22. Links to other databases: BRENDA. . . www. chem. qmul. ac. uk/iubmb/enzyme/EC 3/4/23/3. html - 4 k - Cached - Similar pages
 n n n n IUBMB Enzyme Nomenclature EC 3. 4. 23. 1 Accepted name: pepsin A Reaction: Preferential cleavage: hydrophobic, preferably aromatic, residues in P 1 and P 1' positions. Cleaves Phe 1 Val, Gln 4 His, Glu 13 Ala, Ala 14 Leu, Leu 15 Tyr, Tyr 16 Leu, Gly 23 Phe, Phe 24 Phe and Phe 25 Tyr bonds in the B chain of insulin Other names: pepsin; lactated pepsin; pepsin fortior; fundus-pepsin; elixir lactate of pepsin; P I; lactated pepsin elixir; P II; pepsin R; pepsin D Comments: The predominant endopeptidase in the gastric juice of vertebrates, formed from pepsinogen A by limited proteolysis. Human pepsin A occurs in five molecular forms. Pig pepsin D [1, 2] is unphosphorylated pepsin A. Type example of peptidase family A 1. Formerly EC 3. 4. 4. 1 Links to other databases: BRENDA, EXPASY, GTD, MEROPS, PDB, CAS registry number: 9001 -75 -6 References: 1. Lee, D. and Ryle, A. P. Pepsinogen D. A fourth proteolytic zymogen from pig gastric mucosa. Biochem. J. 104 (1967) 735 -741. [PMID: 4167464] 2. Lee, D. and Ryle, A. P. Pepsin D. A minor component of commercial pepsin preparations. Biochem. J. 104 (1967) 742 -748. [PMID: 4860638] 3. Foltmann, R. Gastric proteinases -structure, function, evolution and mechanism of action. Essays Biochem. 17 (1981) 52 -84. 3. Foltmann, R. Gastric proteinases -structure, function, evolution and mechanism of action. Biochem. [PMID: 6795036] 4. James, M. N. G. and Sielecki, A. R. Molecular structure of an aspartic proteinase zymogen, porcine pepsinogen, at 1. 8 Å resolution. Nature 319 (1986) 33 -38. [PMID: 3941737] 5. Fruton, J. S. Aspartyl proteinases. In New Comprehensive Biochemistry Vol. 16, Hydrolytic Enzymes (Neuberger, A. and 5. Fruton, J. S. Aspartyl proteinases. In New Vol. 16, Hydrolytic Brocklehurst, K. , eds), pp. 1 -38 (1987) Elsevier, Amsterdam 6. Tang, J. and Wong, R. N. S. Evolution in the structure and function of aspartic proteases. J. Cell. Biochem. 33 (1987) 53 -63. Biochem. [PMID: 3546346] 7. Pohl, J. and Dunn, B. M. Secondary enzyme-substrate interactions: kinetic evidence for ionic interactions between substrate side chains and the pepsin active site. Biochemistry 27 (1988) 4827 -4834. [PMID: 3139029]
n n n n IUBMB Enzyme Nomenclature EC 3. 4. 23. 1 Accepted name: pepsin A Reaction: Preferential cleavage: hydrophobic, preferably aromatic, residues in P 1 and P 1' positions. Cleaves Phe 1 Val, Gln 4 His, Glu 13 Ala, Ala 14 Leu, Leu 15 Tyr, Tyr 16 Leu, Gly 23 Phe, Phe 24 Phe and Phe 25 Tyr bonds in the B chain of insulin Other names: pepsin; lactated pepsin; pepsin fortior; fundus-pepsin; elixir lactate of pepsin; P I; lactated pepsin elixir; P II; pepsin R; pepsin D Comments: The predominant endopeptidase in the gastric juice of vertebrates, formed from pepsinogen A by limited proteolysis. Human pepsin A occurs in five molecular forms. Pig pepsin D [1, 2] is unphosphorylated pepsin A. Type example of peptidase family A 1. Formerly EC 3. 4. 4. 1 Links to other databases: BRENDA, EXPASY, GTD, MEROPS, PDB, CAS registry number: 9001 -75 -6 References: 1. Lee, D. and Ryle, A. P. Pepsinogen D. A fourth proteolytic zymogen from pig gastric mucosa. Biochem. J. 104 (1967) 735 -741. [PMID: 4167464] 2. Lee, D. and Ryle, A. P. Pepsin D. A minor component of commercial pepsin preparations. Biochem. J. 104 (1967) 742 -748. [PMID: 4860638] 3. Foltmann, R. Gastric proteinases -structure, function, evolution and mechanism of action. Essays Biochem. 17 (1981) 52 -84. 3. Foltmann, R. Gastric proteinases -structure, function, evolution and mechanism of action. Biochem. [PMID: 6795036] 4. James, M. N. G. and Sielecki, A. R. Molecular structure of an aspartic proteinase zymogen, porcine pepsinogen, at 1. 8 Å resolution. Nature 319 (1986) 33 -38. [PMID: 3941737] 5. Fruton, J. S. Aspartyl proteinases. In New Comprehensive Biochemistry Vol. 16, Hydrolytic Enzymes (Neuberger, A. and 5. Fruton, J. S. Aspartyl proteinases. In New Vol. 16, Hydrolytic Brocklehurst, K. , eds), pp. 1 -38 (1987) Elsevier, Amsterdam 6. Tang, J. and Wong, R. N. S. Evolution in the structure and function of aspartic proteases. J. Cell. Biochem. 33 (1987) 53 -63. Biochem. [PMID: 3546346] 7. Pohl, J. and Dunn, B. M. Secondary enzyme-substrate interactions: kinetic evidence for ionic interactions between substrate side chains and the pepsin active site. Biochemistry 27 (1988) 4827 -4834. [PMID: 3139029]
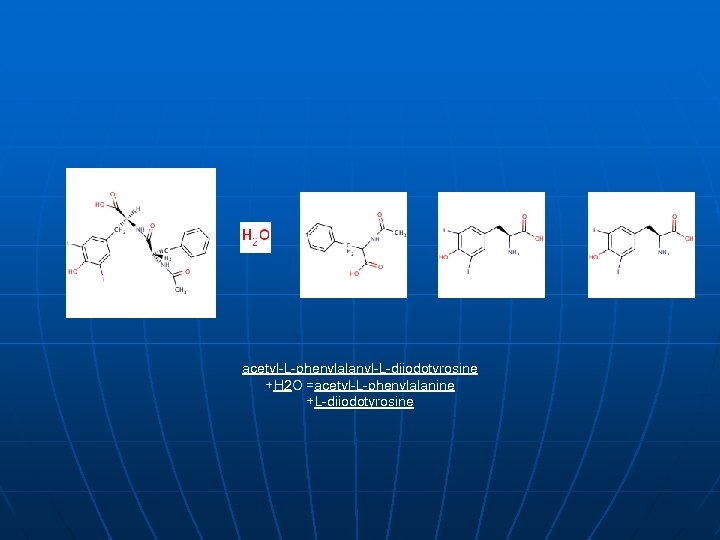 acetyl-L-phenylalanyl-L-diiodotyrosine +H 2 O =acetyl-L-phenylalanine +L-diiodotyrosine
acetyl-L-phenylalanyl-L-diiodotyrosine +H 2 O =acetyl-L-phenylalanine +L-diiodotyrosine
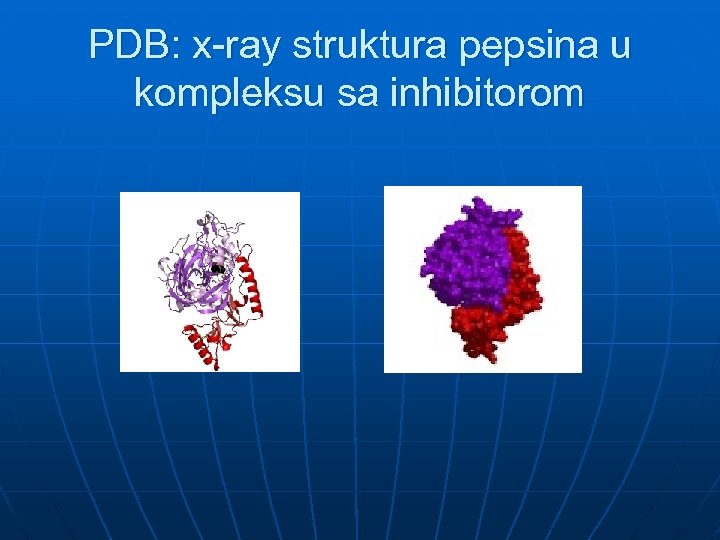 PDB: x-ray struktura pepsina u kompleksu sa inhibitorom
PDB: x-ray struktura pepsina u kompleksu sa inhibitorom


