977d36e4a455509d2d73c9e09c6d8ea3.ppt
- Количество слайдов: 18
 Engager 6 month outcomes Michael J. Reardon, M. D. Professor of Cardiothoracic Surgery Methodist De. Bakey Heart & Vascular Center
Engager 6 month outcomes Michael J. Reardon, M. D. Professor of Cardiothoracic Surgery Methodist De. Bakey Heart & Vascular Center
 Michael J. Reardon, MD I/we have no real or apparent conflicts of interest to report. Presented on behalf of the Engager investigator group The sponsor (Medtronic) performed the data analysis in this presentation Engager is not approved in the US
Michael J. Reardon, MD I/we have no real or apparent conflicts of interest to report. Presented on behalf of the Engager investigator group The sponsor (Medtronic) performed the data analysis in this presentation Engager is not approved in the US
 D. Holzhey University of Leipzig Heart Center, Leipzig, Germany H. Treede University Heart Center Hamburg, Germany S. Baldus University Heart Center Hamburg, Germany A. Linke University of Leipzig Heart Center, Leipzig, Germany S. Bleiziffer German Heart Center, Munich, Germany J. Börgermann Herz- und Diabeteszentrum, Bad Oeynhausen, Germany J-L. Vanoverschelde Clinique Universitaires Saint Luc, Brussels, Belgium V. Falk University Hospital, Zurich, Switzerland INTERNATIONAL CAUTION: CE Mark Submitted. Not approved in the USA, Canada or Japan Engager CE Pivotal Trial is sponsored by Medtronic
D. Holzhey University of Leipzig Heart Center, Leipzig, Germany H. Treede University Heart Center Hamburg, Germany S. Baldus University Heart Center Hamburg, Germany A. Linke University of Leipzig Heart Center, Leipzig, Germany S. Bleiziffer German Heart Center, Munich, Germany J. Börgermann Herz- und Diabeteszentrum, Bad Oeynhausen, Germany J-L. Vanoverschelde Clinique Universitaires Saint Luc, Brussels, Belgium V. Falk University Hospital, Zurich, Switzerland INTERNATIONAL CAUTION: CE Mark Submitted. Not approved in the USA, Canada or Japan Engager CE Pivotal Trial is sponsored by Medtronic
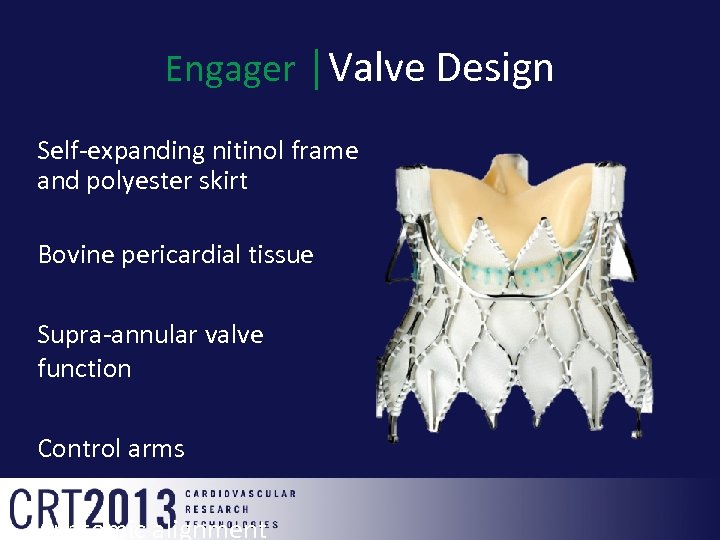 Engager │Valve Design Self-expanding nitinol frame and polyester skirt Bovine pericardial tissue Supra-annular valve function Control arms Anatomic alignment
Engager │Valve Design Self-expanding nitinol frame and polyester skirt Bovine pericardial tissue Supra-annular valve function Control arms Anatomic alignment
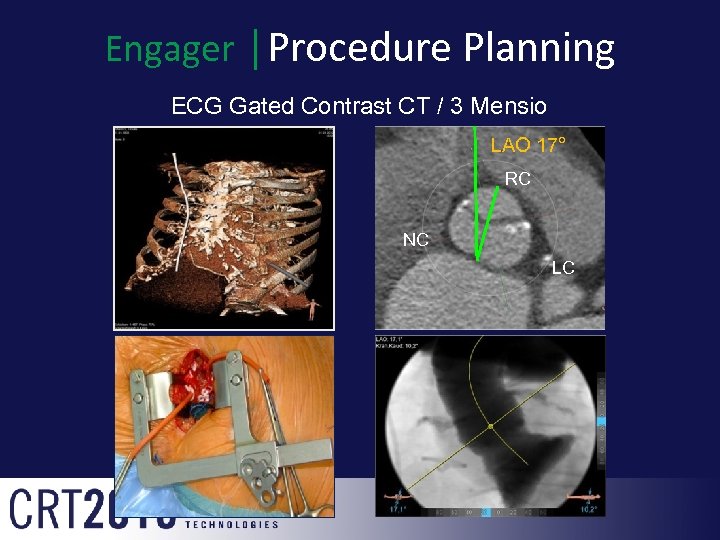 Engager │Procedure Planning ECG Gated Contrast CT / 3 Mensio LAO 17° RC NC LC Cranial 10°
Engager │Procedure Planning ECG Gated Contrast CT / 3 Mensio LAO 17° RC NC LC Cranial 10°
 Engager │System Advantages Control arms capture native leaflets and self-expanding frame conforms to native annulus to minimize PVL Fixation of native leaflets and true commissure to commissure alignment ensures clearance for the coronary ostia Supra-annular valve position facilitates uncompromised valve function even in elliptical annuli Stent design allows less crimping forces and pericardial thickness equal to surgical valves for maximum durability Control arms provide tactile feedback and secure device during deployment 29 Fr
Engager │System Advantages Control arms capture native leaflets and self-expanding frame conforms to native annulus to minimize PVL Fixation of native leaflets and true commissure to commissure alignment ensures clearance for the coronary ostia Supra-annular valve position facilitates uncompromised valve function even in elliptical annuli Stent design allows less crimping forces and pericardial thickness equal to surgical valves for maximum durability Control arms provide tactile feedback and secure device during deployment 29 Fr
 Engager │Procedure Positioning & Deployment Outcomes
Engager │Procedure Positioning & Deployment Outcomes
 Engager CE Pivotal │Methods Primary Endpoint: All-cause mortality at 30 days (ITT analysis) Engager CE Pivotal Trial completed enrollment of 125 patients in December 2012 Interim analysis was conducted on first 61 patients enrolled from September 2011 to May 2012 4 German centers Hamburg (N=25) Leipzig (N=16) Munich (N=10) Bad Oeynhausen (N=10) Focus on 6 -month outcomes from the first 61 patients
Engager CE Pivotal │Methods Primary Endpoint: All-cause mortality at 30 days (ITT analysis) Engager CE Pivotal Trial completed enrollment of 125 patients in December 2012 Interim analysis was conducted on first 61 patients enrolled from September 2011 to May 2012 4 German centers Hamburg (N=25) Leipzig (N=16) Munich (N=10) Bad Oeynhausen (N=10) Focus on 6 -month outcomes from the first 61 patients
 Engager CE Pivotal │Enrolling Sites • Germany • German Heart Center (Munich) • Leipzig University Hospital (Leipzig) • Hamburg University Hospital (Hamburg) • Heart & Diabetes Center (Bad Oeynhausen) • Kerckhoff Clinic (Bad Nauheim) • Switzerland • University Hospital Zurich (Zurich) • Belgium • Cliniques Universitaires Saint-Luc (Brussels) • Israel • Sheba Medical Center (Tel Hashomer)
Engager CE Pivotal │Enrolling Sites • Germany • German Heart Center (Munich) • Leipzig University Hospital (Leipzig) • Hamburg University Hospital (Hamburg) • Heart & Diabetes Center (Bad Oeynhausen) • Kerckhoff Clinic (Bad Nauheim) • Switzerland • University Hospital Zurich (Zurich) • Belgium • Cliniques Universitaires Saint-Luc (Brussels) • Israel • Sheba Medical Center (Tel Hashomer)
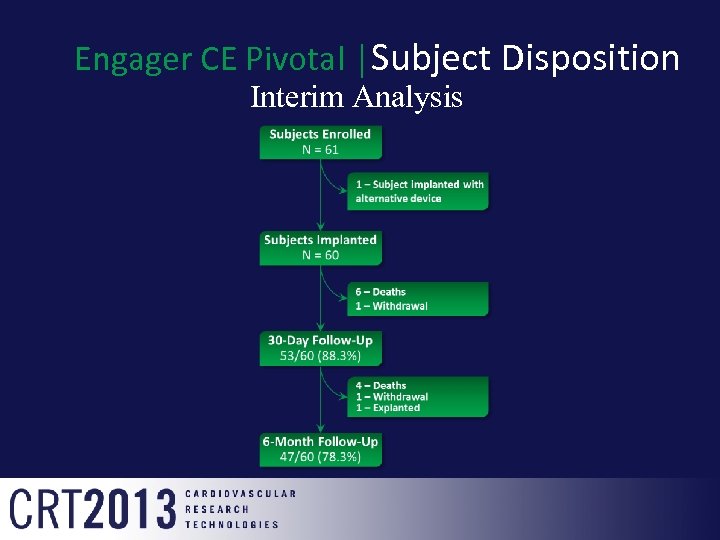 Engager CE Pivotal │Subject Disposition Interim Analysis
Engager CE Pivotal │Subject Disposition Interim Analysis
 Engager CE Pivotal │Baseline Characteristics
Engager CE Pivotal │Baseline Characteristics
 Engager CE Pivotal │Procedural Results * one patient withdrew consent in trial on Day 11 and second patient had trace AR at 30 -day visit
Engager CE Pivotal │Procedural Results * one patient withdrew consent in trial on Day 11 and second patient had trace AR at 30 -day visit
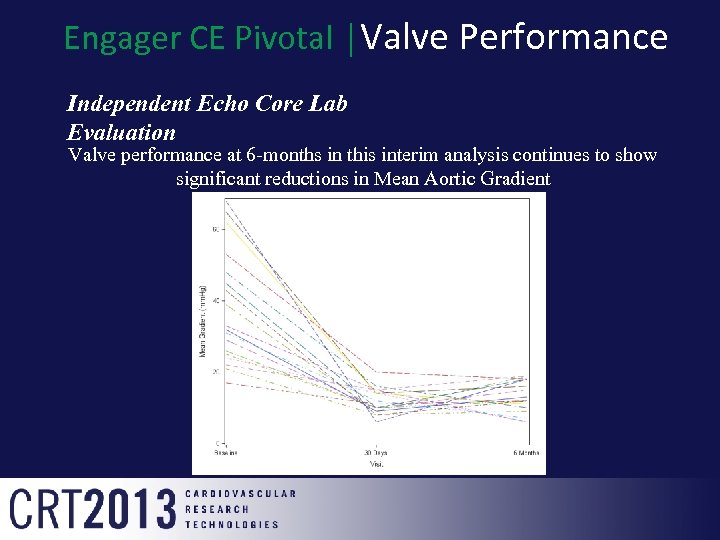 Engager CE Pivotal │Valve Performance Independent Echo Core Lab Evaluation Valve performance at 6 -months in this interim analysis continues to show significant reductions in Mean Aortic Gradient
Engager CE Pivotal │Valve Performance Independent Echo Core Lab Evaluation Valve performance at 6 -months in this interim analysis continues to show significant reductions in Mean Aortic Gradient
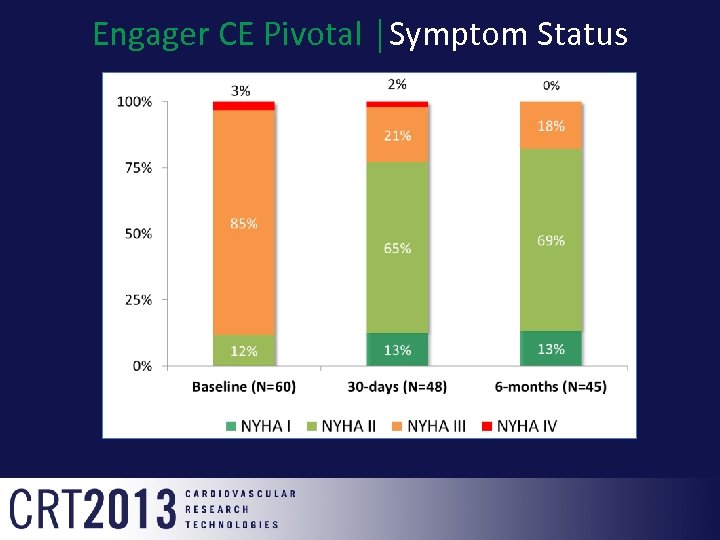 Engager CE Pivotal │Symptom Status
Engager CE Pivotal │Symptom Status
 Engager CE Pivotal │30 -Days & 6 -Month Outcomes
Engager CE Pivotal │30 -Days & 6 -Month Outcomes
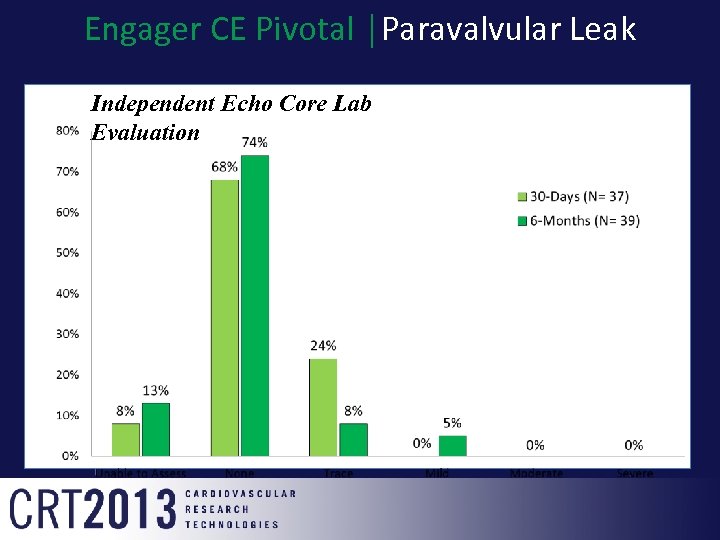 Engager CE Pivotal │Paravalvular Leak Independent Echo Core Lab Evaluation
Engager CE Pivotal │Paravalvular Leak Independent Echo Core Lab Evaluation
 Engager CE Pivotal │Conclusions Engager transcatheter valve system performs as intended with a positive risk/benefit for patients with symptomatic aortic stenosis who are at high risk for surgical aortic valve replacement This multicenter study confirms that transapical implantation of the Engager valve can be performed with a high degree of technical success The absence of positioning related complications such as valve embolization, coronary occlusion, and second valve implantation demonstrates the controlled delivery The low incidence of paravalvular leak is consistent with the design intent of the Engager valve
Engager CE Pivotal │Conclusions Engager transcatheter valve system performs as intended with a positive risk/benefit for patients with symptomatic aortic stenosis who are at high risk for surgical aortic valve replacement This multicenter study confirms that transapical implantation of the Engager valve can be performed with a high degree of technical success The absence of positioning related complications such as valve embolization, coronary occlusion, and second valve implantation demonstrates the controlled delivery The low incidence of paravalvular leak is consistent with the design intent of the Engager valve
 Thank You INTERNATIONAL CAUTION: CE Mark Submitted. Not approved in the USA, Canada or Japan
Thank You INTERNATIONAL CAUTION: CE Mark Submitted. Not approved in the USA, Canada or Japan


