ZPE_ANG_2.pptx
- Количество слайдов: 19

Energy sources and energy transformation Fuel properties, combustion and boilers Biomass and renewable sources Ondřej Bartoš ondrej. bartos@fs. cvut. cz

Non-renewable • • Anthracite Hard coal Lignite, Brown coal Peat Shale Crude oil Natural gas Uranium/ Deuterium, Tritium

Fuel composition • Combustible part, Ash, Moisture, Volatile fraction, Carbon content • C+A+W=1 • Ultimate analysis • Heating value
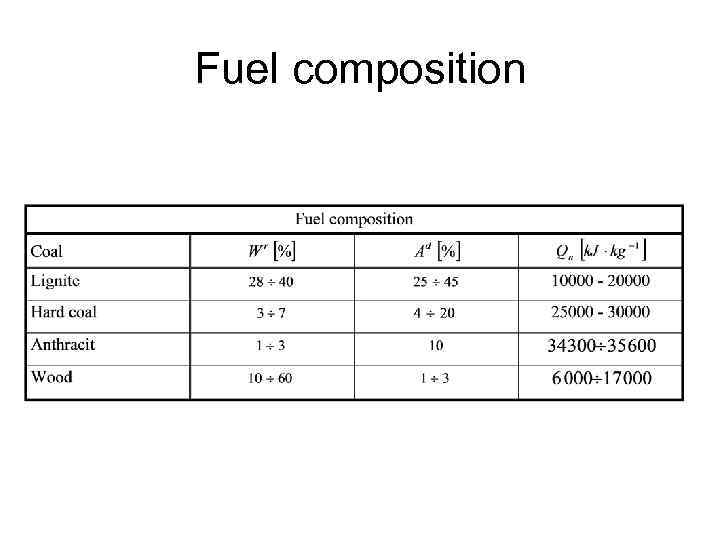
Fuel composition
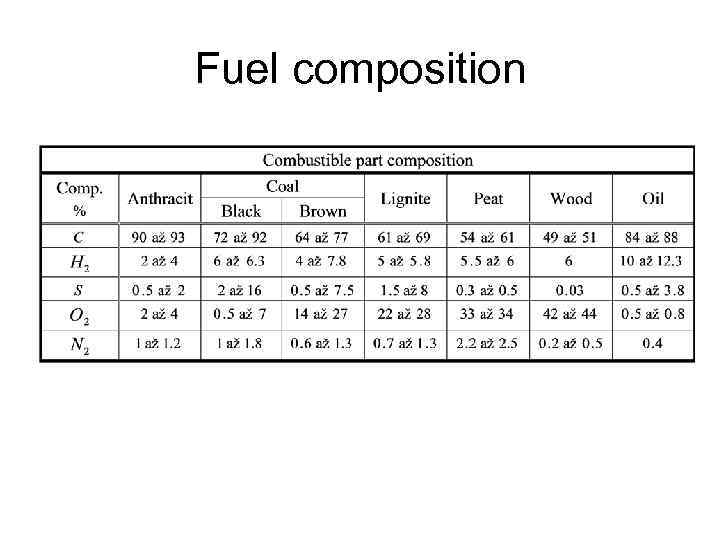
Fuel composition

Fuel caloric value • HHV – higher heating value LHV – lower heating value • HHV -the energy released as heat when a compound undergoes complete combustion with oxygen under standard conditions and the products of combustion are returned to starting condition 20°C • LHV – same but not condensing • Careful - Different definitions

LHV computation • According DULONG: • According VONDRÁČEK:
![Heat content Fuel HHV [k. J/kg] HHV [k. J/m³] (20 °C) Hydrogen 141 900 Heat content Fuel HHV [k. J/kg] HHV [k. J/m³] (20 °C) Hydrogen 141 900](https://present5.com/presentation/85069857_356311040/image-8.jpg)
Heat content Fuel HHV [k. J/kg] HHV [k. J/m³] (20 °C) Hydrogen 141 900 12 760 (g) Methan 55 530 37 520 (g) Ethan 51 900 62 900 (g) Propan 50 350 96 170 (g) Butan 49 510 124 240 (g) Fuel HHV [k. J/kg] HHV [k. J/l] (20 °C) Benzin 47 300 33 580 (l) Diesel 44 800 38 100 (l) Methanol 22 884 18 120 (l) Ethanol 29 847 23 550 (l) Propanol 33 600 26 700 (l) Buthanol 37 334 30 230 (l)

Hard Coal • Plants – Peat – Lignite – subbituminous – Anthracite • 860 Gtons reserves, resources 18000 Gt • 8 Gt year production, 50% China, • Biggest exporter Australia, Importer Japan • Home use, electricity production, • Coke - steel

Lignite • • • Wide range of properties Exclusively for electricity production Year production 1 Gt Spontaneous combustion Open mines
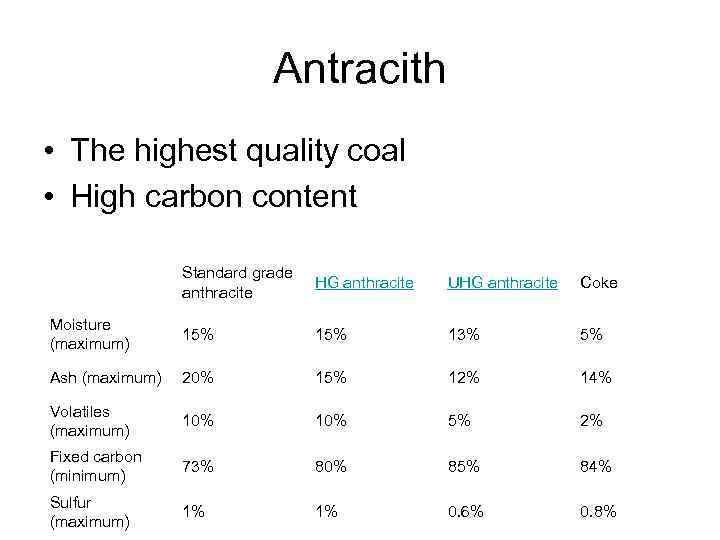
Antracith • The highest quality coal • High carbon content Standard grade anthracite HG anthracite UHG anthracite Coke Moisture (maximum) 15% 13% 5% Ash (maximum) 20% 15% 12% 14% Volatiles (maximum) 10% 5% 2% Fixed carbon (minimum) 73% 80% 85% 84% Sulfur (maximum) 1% 1% 0. 6% 0. 8%

Shale • • Oil shale, HHV 8 – 10 MJ/kg Power plant in Estonia not so effective Shale gas – U. S. 2001 1% production 2010 20%

Peat is a heterogeneous mixture of more or less decomposed plant (humus) material that has accumulated in a water-saturated environment and in the absence of oxygen. Shatura Power Station. Russia has the largest peat power capacity in the world

Coal mining • Deep mining • Open pit
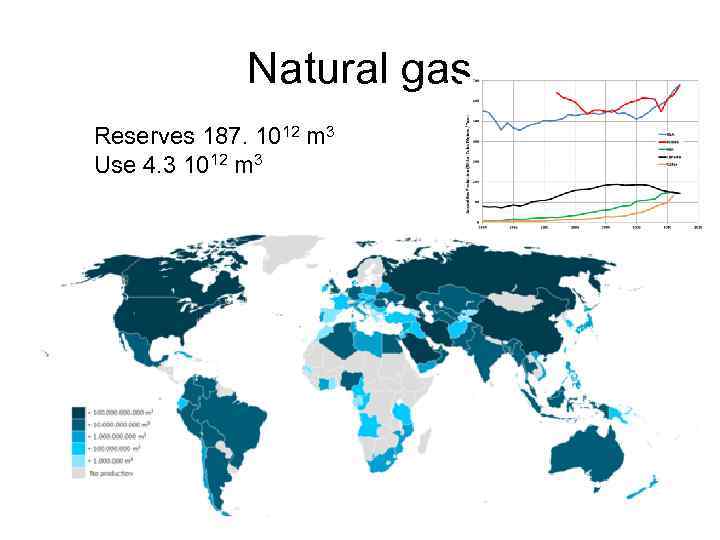
Natural gas Reserves 187. 1012 m 3 Use 4. 3 1012 m 3

Natural gas • Higher heating values of natural gases from various sources • • • Algeria: 42. 00 MJ/m³ Bangladesh: 36. 00 MJ/m³ Canada: 38. 20 MJ/m³ Indonesia: 40. 60 MJ/m³ Netherlands: 33. 32 MJ/m³ Norway: 39. 88 MJ/m³ Pakistan: 34. 90 MJ/m³ Russia: 38. 23 MJ/m³ Saudi Arabia: 38. 00 MJ/m³ United Kingdom: 39. 71 MJ/m³ United States: 38. 42 MJ/m³ Uzbekistan: 37. 89 MJ/m³ • The lower heating value of natural gas is normally about 90 percent of its higher heating val.
![Liquid fuel HHO – Home heating oil Diesel Mazut Fuel HHV [k. J/kg] HHV Liquid fuel HHO – Home heating oil Diesel Mazut Fuel HHV [k. J/kg] HHV](https://present5.com/presentation/85069857_356311040/image-17.jpg)
Liquid fuel HHO – Home heating oil Diesel Mazut Fuel HHV [k. J/kg] HHV [k. J/l] (20 °C) Benzin 47 300 33 580 (l) Diesel 44 800 38 100 (l) Methanol 22 884 18 120 (l) Ethanol 29 847 23 550 (l) Propanol 33 600 26 700 (l) Buthanol 37 334 30 230 (l)

Wood and biomass • Biomass is biological material derived from living, or recently living organisms. It most often refers to plants or plant-based materials which are specifically called lignocellulosic biomass. As an energy source, biomass can either be used directly via combustion to produce heat, or indirectly after converting it to various forms of biofuel. Conversion of biomass to biofuel can be achieved by different methods which are broadly classified into: thermal, chemical, and biochemical methods.

Fundamentals of combustion • Combustion or burning is a high-temperature exothermic chemical reaction between a fuel and an oxidant, usually atmospheric oxygen, that produces oxidized, often gaseous products. Combustion in a fire produces a flame, and the heat produced can make combustion selfsustaining. • • C+O 2 -> C+0. 5 O 2 -> CO+0. 5 O 2 -> H 2 + 0. 5 O 2 -> CO 2 + 393. 5 MJ/kg CO + 110. 5 MJ/kg CO 2 + 283 MJ/kg H 2 O + 285. 9 MJ/kg
ZPE_ANG_2.pptx