4лекция.ppt
- Количество слайдов: 68

Energy of chemical reactions

Basic concepts of thermodynamics Thermodynamics - a science Thermodynamics about mutual transformations of different types of energy.

Chemical thermodynamics studies: l l Transitions chemical energy into other form- thermal, and electrical. etc. What energy effects chemical reactions Opportunities and spontaneously occurring reactions Chemical equilibrium and conditions of its bias.

Basic concepts of thermodynamics The object of study in thermodynamics of a system. System - a collection of substances being in interaction mentally (or actually) apart from the environment.

Basic concepts of thermodynamics SYSTEM HOMOGENEOUSLY comprises one phase Example: KNO 3 solution or water HETEROGENEOUS It consists of two or more phases Example: oversaturated solution, i. e. at the bottom vessel or precipitate water ice

Basic concepts of thermodynamics Phase is part of the system, a uniform at all points on the composition and properties, and is separated from other parts of the system interface.
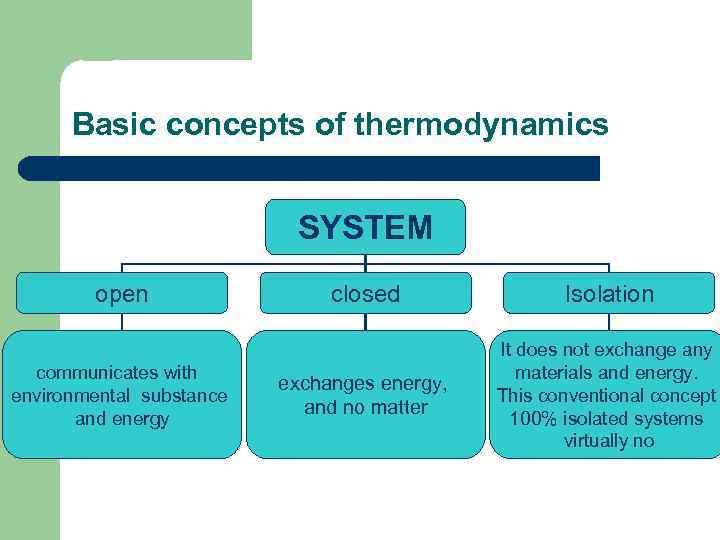
Basic concepts of thermodynamics SYSTEM open communicates with environmental substance and energy closed Isolation exchanges energy, and no matter It does not exchange any materials and energy. This conventional concept 100% isolated systems virtually no

Thermodynamic parameters Temperature - T Pressure - P Density - ρ Concentration - C Heat capacity - c Changing at least one parameter leads to the change of state of the entire system

Parameters of chemical thermodynamics U - internal energy Н – enthalpy S – entropy G - Gibbs energy

The internal energy of the system (U) - it is the total energy, which consists of kinetic and potential energies of molecules, atoms, atomic nuclei and electrons. It does not include the potential energy position of the system in space and the kinetic energy of the motion system as a whole.
![Units of the internal energy: [U] = J, к. J. The absolute value of Units of the internal energy: [U] = J, к. J. The absolute value of](https://present5.com/presentation/3/312893759_437079365.pdf-img/312893759_437079365.pdf-11.jpg)
Units of the internal energy: [U] = J, к. J. The absolute value of the internal energy can not be determined, but it is possible to measure the change ΔU in the transition ΔU from one state to another.

The internal energy of the system Internal energy - a function of the state, which is characterized by a complete energy supply system. The change of the internal energy is not dependent on the route and manner of transition from one state to another. ΔU=U 2 –U 1 U 2 and U 1 - internal energy of the system in the initial and final states, respectively.

Thermodynamic process - this change of state is accompanied by a change in at least one of system parameters over time.

Thermodynamic process Depending on the conditions of transition from one state to another in thermodynamics distinguish the following processes: isothermal Т- const, l isobaric Р-const, l isochoric V-const.

Heat - It is a measure of the energy transferred from one body to another, due to the difference in temperature of these bodies.

Work - It is a measure of the energy transferred from one body to another by moving the mass under the action of any forces.

The first law of thermodynamics It reflects the proportion between the change in internal energy, heat and work: Q=ΔU +A

Law of energy conservation - the energy is not lost does not occur, it is converted from one form to another in a strictly defined, always in equivalent amounts.

Isochoric process The first law of thermodynamics: Q=ΔU +A = ΔU + P×ΔV For isochoric process V=const , then ΔV=0 A=0 We write the first law of TD for isochoric process: Q=ΔU

Isobaric process For isobaric process Р=const. The isobaric processes standard enthalpy of formation is equal to the change in enthalpy (Н). The first law of TD: Qр =ΔU+P× ΔV=(U 2 -U 1) + P(V 2 -V 1)=(U 2+PV 2)-(U 1+PV 1) Denote Н =U+PV Then Qр =H 2 -H 1=ΔН. H enthalpy quantity characterizes the system.

The heat of reaction - is the amount of heat released or absorbed by the system after the chemical reaction

The heat of reaction is equal to the change in enthalpy of the system: l If H 2 > H 1 ΔH= H 2 – H 1 > 0 endothermic reaction Q = Δ H l if H 1 > H 2 ΔH= H 2 – H 1 < 0 exothermic reaction Q = – Δ H

The enthalpy of formation - the amount of heat released or absorbed during the formation of 1 mol of a compound of simple substances.

The standard enthalpy of formation For comparison, the enthalpies of formation of various compounds is determined under the same standard conditions: Т=298 К Р=101, 3 КPа, 1 аtm. , 760 mm Hg l The enthalpy of formation determined under standard conditions is called the standard enthalpy of formation of substance and denoted l

RULE! lenthalpy of formation of simple substances are zero lresistant to simple substances enthalpy zero For example: For a solid iodine enthalpy of formation is zero, and for gaseous iodine is not zero.

The thermochemical equation l is the equation of a chemical reaction in which the specified standard enthalpy of formation and aggregation state.

Features thermochemical equations l in thermochemical equations can be fractional stoichiometric coefficients.

Features thermochemical equations l With thermochemical equations can produce algebraic operations. l They can add, subtract, multiply, any factors with thermal effect

Hess law l Standard enthalpy of formation is determined only by initial and final states of the reactants and do not depend on the path of its course.

Example. Receiving СО 2 1 way: С + О 2 = СО 2 l 2 way: С + 1/2 О 2 = СО СО + 1/2 О 2 = СО 2 l ΔН 1 ΔН 2 ΔН 3 ΔН 1 = ΔН 2 + ΔН 3

The investigation of the law of Hess l The heat of the chemical reaction is the difference between the sum of the enthalpies of formation of the reaction products and the sum of the enthalpies of formation of the starting materials. l It is necessary to take into account the stoichiometric coefficients.

The possibility and the direction of flow chemical reactions

Spontaneous reaction l l l In the study of chemical reactions, it is important to assess the possibility or impossibility of spontaneous flow under these conditions. Spontaneously can occur both exothermic and endothermic reactions. Spontaneous process proceeds without energy from the outside (a mixture of gases, heat transfer from the hot to the cold, the water flows down from the roof)

The second law of thermodynamics Defines the criteria for spontaneous flow process in isolated systems entropy

Entropy l This parameter characterizes the chaotic motion of the particles is a measure of the molecular, atomic and ionic disorder.

Basic provisions l l l Entropiya- is a measure of thermodynamic probability of the state of substances and systems. Any isolated system left to itself, changes in the direction of the state having the maximum probability. All of the processes taking place in an isolated system in the direction of increasing entropy.

The change in entropy ΔS = S 2 –S 1 if ΔS > 0, then the process proceeds in the forward direction, l if ΔS < 0, then the process proceeds in the opposite direction. l

The absolute value of entropy l l l Unlike internal energy and enthalpy can determine the absolute value of the entropy of all the substances, as entropy is the zero point of reference. The entropy of the substance at T = 0 K is zero probability = 1. This macrostate is achieved only microstate. When the phase transitions (melting, boiling) the entropy is growing by leaps and bounds. If the reaction of gaseous substances are involved, then the change in entropy can be measured by changes in the volume of gaseous substances.

Factors uninsulated systems 1. Entalpiyny ΔN- reflects the tendency of the system to the formation of bonds as a result of the mutual attraction of the particles, which leads to their complexity. The energy thus released and ΔH <0. (Desire to move the system to a state with the minimum E, while heat ΔH <0). 2. Entropy (TΔS) - reflects the desire to strengthen the processes of dissociation of the complex particles into simpler and less ordered state as a result of ΔS> 0. (Desire to move the system to a state with a large mess ΔS> 0).

Энергия Гиббса l l Энтропийный и энтальпийный факторы обычно действуют в противоположных направлениях и общее направление реакции определяется влиянием преобладающего фактора. В неизолированных системах критерием является ΔG –энергия Гиббса, ее рассчитывают при разных температурах.
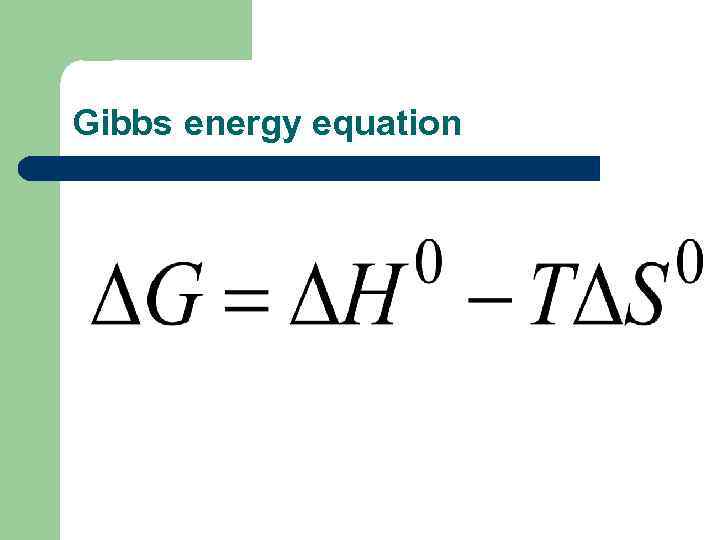
Gibbs energy equation

The magnitude and sign of ΔG provides insights into the fundamental features and direction of the process. ΔG<0 spontaneously flows in the forward direction, l Δ G>0 direct reaction is impossible and flows in the opposite direction, l Δ G=0 the reaction is in equilibrium, i. e. the rate l of the forward reaction is equal to the speed feedback.

The standard Gibbs energy of formation of a chemical compound ΔG ° l Gibbs energy of the reaction is the formation of one mole of the compound is in standard conditions, from simple substances l ΔG ° simple substances as well as ΔH °, ΔS ° equal to zero l Units ΔG ° - k. J / mol l The standard Gibbs energy of formation of a chemical compound ΔG ° is provided in the references

IMPORTANT! Any reaction at constant temperature and pressure occurs spontaneously in the direction of the decrease of Gibbs energy.

KINETICS OF CHEMICAL PROCESSES Chemical kinetics studies the rate and mechanism of chemical reactions. Heterogeneous chemical reactions Speed heterogeneous reaction - is the amount of substance that reacts or formed by the reaction per unit time per unit area of the interface.

Speed homogeneous reaction - is the amount of substance that reacts or formed by the reaction per unit time per unit volume. The rate of reaction in a homogeneous system is a change in the concentration of any of the substances of the system, which occurs per unit of time.

Factors affecting the rate of reaction: 1. the nature of the reactants; 2. concentration substances (C); 3. temperature (T); 4. pressure (P); 5. in the presence of catalysts (Kat); 6. he contact area of the reactants; 7. velocity of the fluid or gas near the surface on which the reaction proceeds.

The dependence of the reaction rate on the concentration of the reactants The law of mass action (K. M. Guldberg, J. Waage in 1867): At constant temperature, the chemical reaction rate is proportional to the product of the concentrations of the reactants to the power of the stoichiometric coefficients facing the formula of these substances in the reaction equation. where k - rate constant.

The magnitude of the rate constant k depends on: 1. The nature of the reactants; 2. Temperature; 3. The presence of a catalyst. But it does not depend on the concentration of substances. С + О 2 = СО 2

The dependence of the reaction rate on the temperature and on the nature of the reactants. The reactive particles with sufficient energy for the elementary act of chemical reaction, called active, and the energy required to overcome the energy barrier - activation energy of the reaction. Grouping of particles in the process of interaction, that is in the process of restructuring bonds, called active complex. 50

Rule van't Hoff: temperature increase per 10 ° C the reaction rate increases about 2 -4 times (temperature coefficient of the reaction rate). Arrhenius (1889 S. Arrhenius): where A - a constant (pre-exponential factor) is determined by the frequency of collisions and proper orientation of the 51 particles.

The dependence of the reaction rate on the catalyst Catalysts - substances that can change (increase or decrease) the rate of a chemical reaction. Reactions in which catalysts are used, called catalyst, and changing the rate of chemical reactions with the catalyst - catalysis. Catalysts - a substance capable of altering the rate of chemical reactions, the number and nature of which after completion of the reaction are the same as before the reaction. 52

Catalysts are divided into three types: homogeneous, heterogeneous and biological. There are two types of catalysis. When the interaction of matter and the catalyst are in the same state of aggregation, say homogeneous catalysis: In heterogeneous catalysis, the catalyst and the reactants are in different states of aggregation, and the reaction proceeds at the interface between the two phases. 53

The dependence of the reaction rate on the catalyst Catalyst - a substance that accelerates the rate of a chemical reaction, but is not consumed in the reaction Inhibitor - a substance that slows the chemical reaction, but is not consumed in the reaction

The dependence of the heterogeneous reaction on the magnitude of the surface area of the reagent Са. СО 3 + 2 НСl Са. Сl 2 + СО 2 + Н 2 О а) Ca. CO 3 - in the form of a piece of chalk or marble, б) Са. СО 3 - in powder form. In case (a) the reaction proceeds more slowly than in the case (b) Conclusion: V = (S)

The dependence of the heterogeneous reaction on the magnitude of the surface area of the reagent Stages of heterogeneous reactions: 1. supply reagent to the particle surface; 2. The chemical reaction at the surface; 3. The removal of reaction products. In general, the reaction rate is determined by the speed of the slowest (limiting) stage.

Chemical equilibrium and equilibrium constant

The state of equilibrium This is the state of the system at which ΔG = 0, and the rate of the forward reaction is equal to the speed feedback: а. А + в. В = с. С + d. D V forward = V feedback

The equilibrium constant describes a quantitative equilibrium

The calculation of the equilibrium constant l To calculate the equilibrium constant of the equilibrium concentrations used. l If the reaction all materials are in a gaseous state, instead of the equilibrium concentrations can use partial pressures.

The partial pressure l it is the pressure of the gas entering into the mixture, which he provided would be if he held the volume that occupies the entire mixture.

Contact Kp and ΔG ° The equilibrium constant is related to the standard Gibbs energy of the following equation: Knowing the value of ΔG ° for the chemical reaction is possible to calculate the equilibrium constant and equilibrium concentrations.

The shift of balance If you change the external conditions change the equilibrium concentrations, there is a shift of balance. The direction of displacement of the chemical equilibrium when the external conditions defined by the rule Le Chatelier.

Le Chatelier's principle When external influence equilibrium shifts towards reducing this impact.

1. The effect of temperature on the equilibrium of a chemical reaction l Higher temperatures - shifts the equilibrium toward the endothermic reaction l Lowering the temperature - shifts the equilibrium toward the exothermic reaction.

2. Effect of pressure on the balance of a chemical reaction l. Increasing the pressure shifts the equilibrium toward the smaller volume. 3 Н 2 + N 2 → 2 NH 3 3 1 → 2 l. If equal volumes, the pressure does not affect the displacement of equilibrium.

3. The impact on the equilibrium concentration of a chemical reaction l. Increasing the concentration of the starting materials shifts the equilibrium reaction towards the products. l. Increasing the concentration of the reaction product shifts the equilibrium toward the starting materials.

4. Effect of the catalyst on the equilibrium of the chemical reaction Introduction of the catalyst does not influence the equilibrium displacement, but accelerates equilibration.
4лекция.ppt