97a5c0c062e65268bbf7df55390a7cef.ppt
- Количество слайдов: 30

Energy Levels and Orbitals An investigation into electrons and their location and behavior within the atom Learning Targets: A. Describe the process of excitation and emission of energy by an electron. B. Write electron configurations for elements or ions (incl. noble gas config. ) C. Draw orbital energy diagrams for elements or ions.
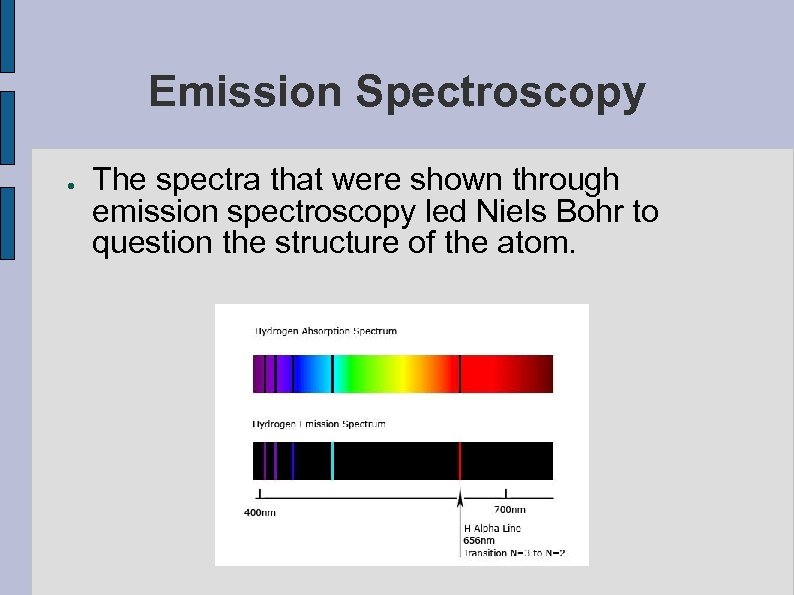
Emission Spectroscopy ● The spectra that were shown through emission spectroscopy led Niels Bohr to question the structure of the atom.
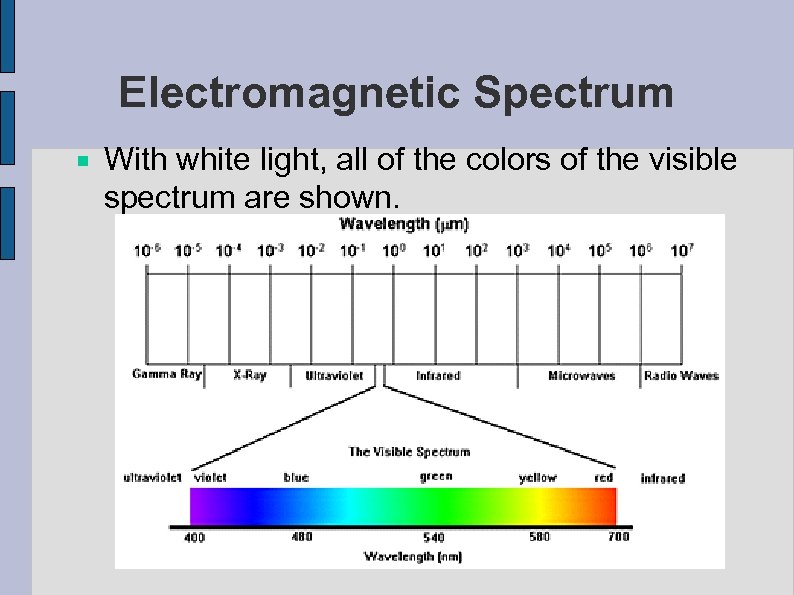
Electromagnetic Spectrum With white light, all of the colors of the visible spectrum are shown.

Emission Spectroscopy ● Since that was NOT what the spectra of elements looked like, Bohr began to look at why only certain wavelengths of color appeared.
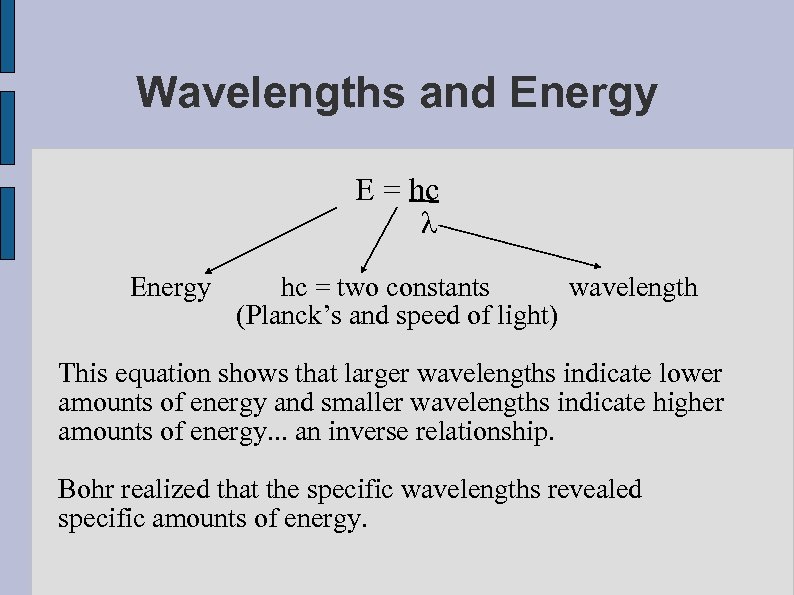
Wavelengths and Energy E = hc λ Energy hc = two constants wavelength (Planck’s and speed of light) This equation shows that larger wavelengths indicate lower amounts of energy and smaller wavelengths indicate higher amounts of energy. . . an inverse relationship. Bohr realized that the specific wavelengths revealed specific amounts of energy.

The Bohr Model According to Niels Bohr, an electron can circle the nucleus in orbits of only certain distances from the nucleus. Bohr called these orbits, or energy levels. An electron cannot be in-between energy levels (i. e. it is either on the first level or the second). Therefore, energy is quantized.

Exciting electrons. . . Niels Bohr realized that the spectra were being created as electrons moved between these energy levels: ● ● If an electron absorbs energy, it may jump to a higher energy level. When an electron is at a higher energy level we say that the electron is in its “excited” state. When the electron releases energy in the form of radiation, we say that the electron has returned to its “ground” state. The type of radiation that is emitted depends on the amount of energy released (more on that in a moment)
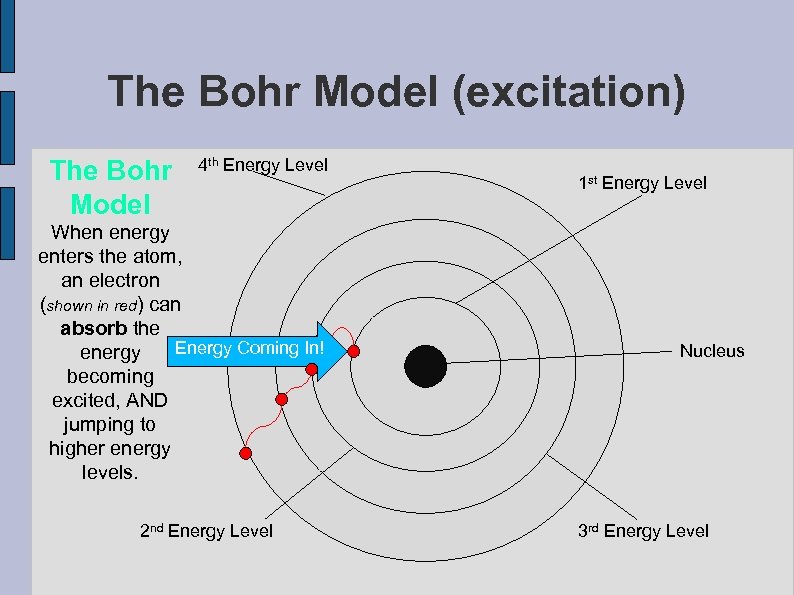
The Bohr Model (excitation) The Bohr Model 4 th Energy Level When energy enters the atom, an electron (shown in red) can absorb the Energy Coming In! energy becoming excited, AND jumping to higher energy levels. 2 nd Energy Level 1 st Energy Level Nucleus 3 rd Energy Level
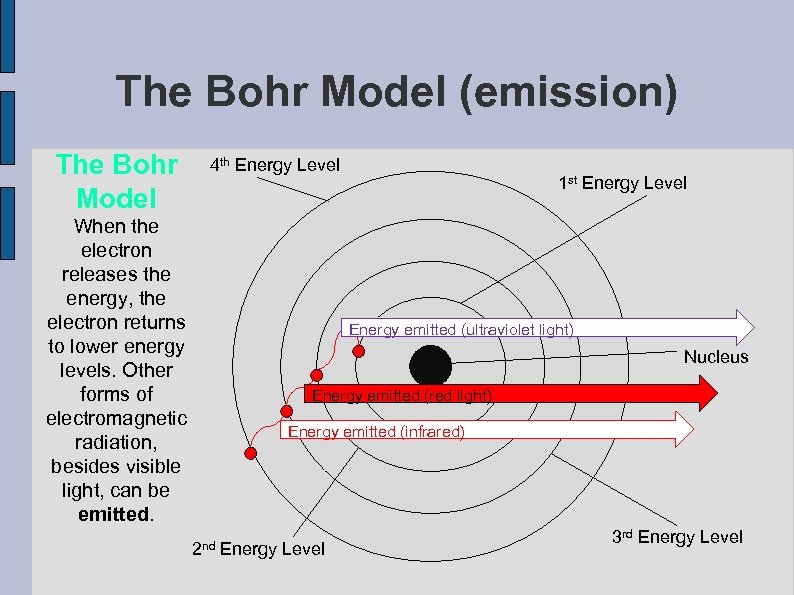
The Bohr Model (emission) The Bohr Model 4 th Energy Level When the electron releases the energy, the electron returns to lower energy levels. Other forms of electromagnetic radiation, besides visible light, can be emitted. 1 st Energy Level Energy emitted (ultraviolet light) Nucleus Energy emitted (red light) Energy emitted (infrared) 2 nd Energy Level 3 rd Energy Level
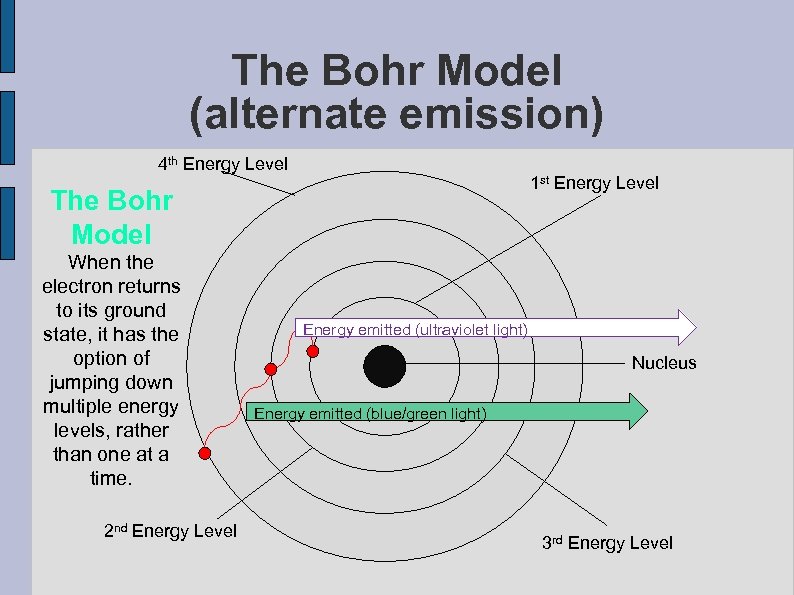
The Bohr Model (alternate emission) 4 th Energy Level 1 st Energy Level The Bohr Model When the electron returns to its ground state, it has the option of jumping down multiple energy levels, rather than one at a time. 2 nd Energy Level Energy emitted (ultraviolet light) Nucleus Energy emitted (blue/green light) 3 rd Energy Level

Types of Radiation The following are types of electromagnetic radiation, listed from highest energy to the lowest: ● ● ● Gamma rays: cosmic radiation, very high energy Ultraviolet rays (UV): solar radiation, high energy Infrared rays (IR): thermal radiation, remote controls, low energy ● Visible Light (more to follow) ● Microwave rays: microwave oven, very low energy ● Radio: lowest energy waves
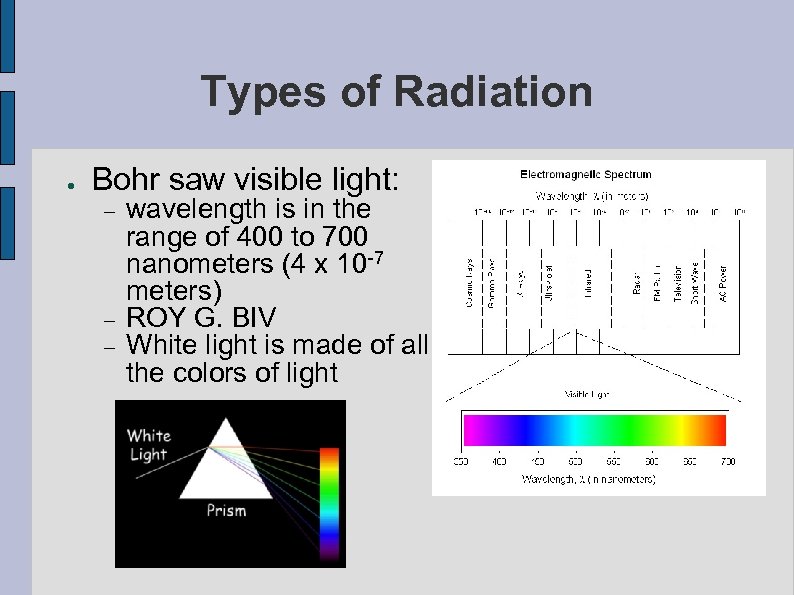
Types of Radiation ● Bohr saw visible light: wavelength is in the range of 400 to 700 nanometers (4 x 10 -7 meters) ROY G. BIV White light is made of all the colors of light

Energy Levels and Spectra Electrons release certain types of electromagnetic radiation as they fall to specific energy levels Energy Level Spectra Emission Change 2 --> 1 3 --> 1 4 --> 1 3 --> 2 4 --> 2 5 --> 2 4 --> 3 Ultraviolet Visible Red Visible Blue/Green Visible Blue Infrared
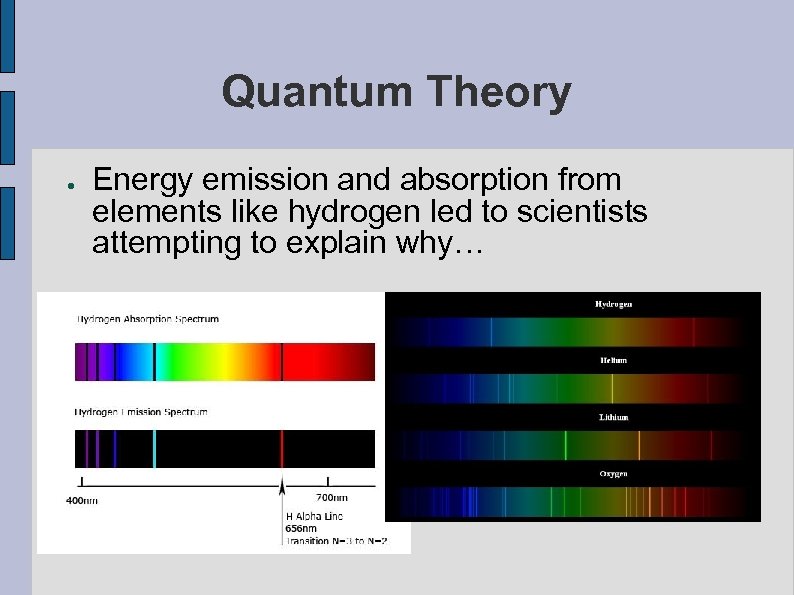
Quantum Theory ● Energy emission and absorption from elements like hydrogen led to scientists attempting to explain why…

Quantum Mechanical Model ● In addition to knowing that there were energy levels in the atom, three scientists began to notice other things. . . Heisenberg – impossible to know the exact position and exact speed of an electron at the same time De Broglie – electrons have wave-like properties, as in they move in wave patterns Schroedinger – developed probability of finding each electron in a given location
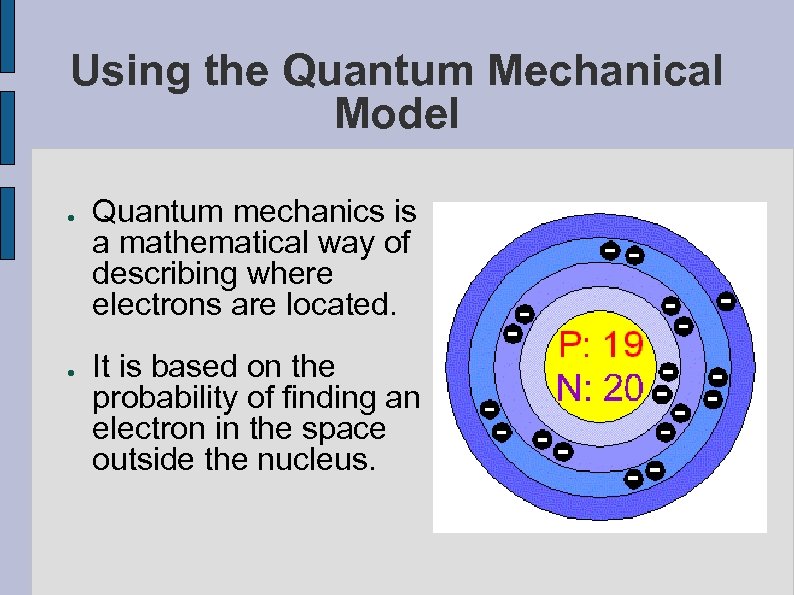
Using the Quantum Mechanical Model ● ● Quantum mechanics is a mathematical way of describing where electrons are located. It is based on the probability of finding an electron in the space outside the nucleus.

Why Quantum Numbers? ● The quantum numbers are like an address: ● State City Street House Number Each piece of information is needed to describe the location, and each one tells more specific information about where the electron is located.
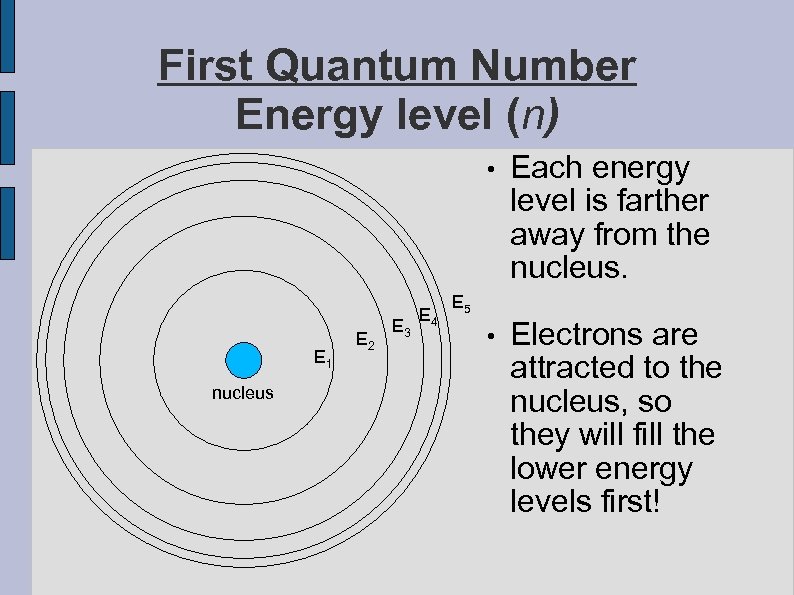
First Quantum Number Energy level (n) • E 1 nucleus E 2 E 3 E 4 Each energy level is farther away from the nucleus. • Electrons are attracted to the nucleus, so they will fill the lower energy levels first! E 5

Second Quantum Number Subshell (l) As the energy levels increase, so do the number of subshells that are needed to cover all the space around the atom. The first energy level (n=1) has 1 subshell (s) The second energy level (n=2) has 2 subshells (s & p) The third energy level (n=3) has 3 subshells (s, p, & d) The fourth energy level (n=4) has 4 subshells (s, p, d, & f)

Extension… ● How many subshells would be present in energy level 5? ● Answer: 5! s, p, d, f, and g How many subshells would be present in energy level 6? Answer: 6! s, p, d, f, g, and h
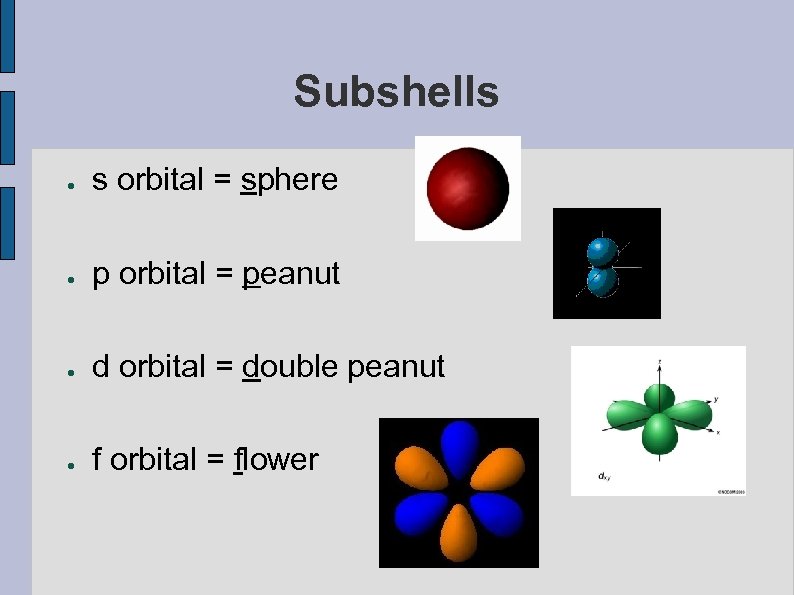
Subshells ● s orbital = sphere ● p orbital = peanut ● d orbital = double peanut ● f orbital = flower

Quantum Mechanical Model ● To recap: ● Energy level 1 = 1 subshell (s) Energy level 2 = 2 subshells (s and p) Energy level 3 = 3 subshells (s, p, and d) Energy level 4 = 4 subshells (s, p, d, and f) etc. Each energy level is larger than the previous. As a result, there are more possible locations for where an electron could reside. Why are more subshells present?
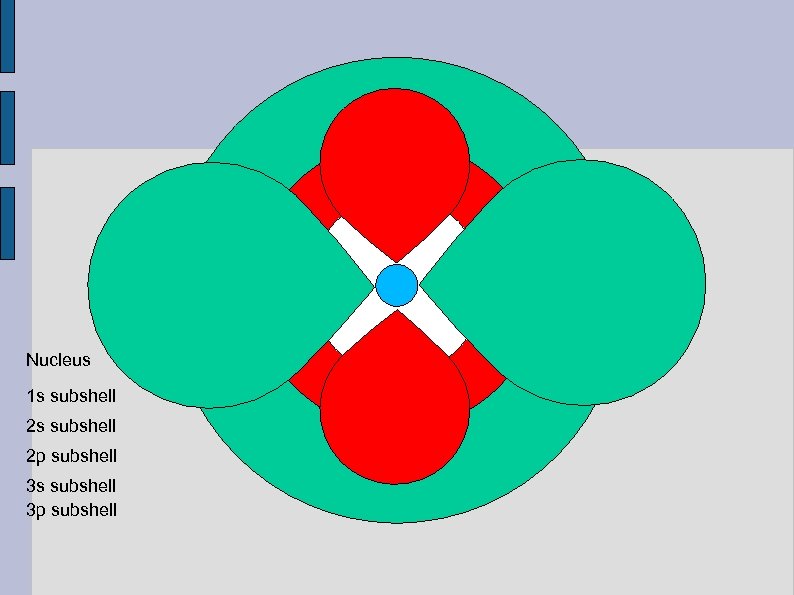
Nucleus 1 s subshell 2 p subshell 3 s subshell 3 p subshell
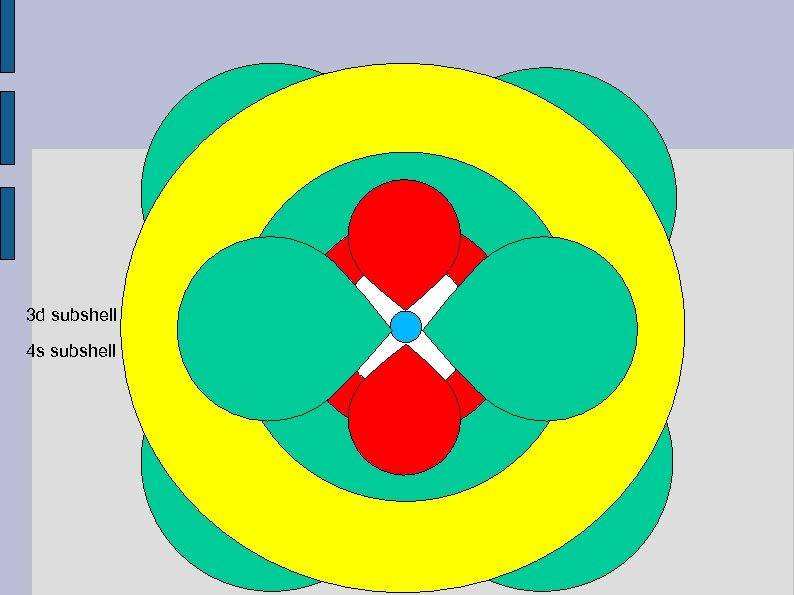
3 d subshell 4 s subshell

Third Quantum Number Atomic Orbitals ( ml ) The atomic orbital essentially describes how many of that shape of subshell are needed to cover all the space around the nucleus. • The more complicated the shape, the more orbitals are needed to cover all the space. •

Third Quantum Number Atomic Orbitals ( ml ) • “s” has 1 orbital (just 1 type of s) • “p” has 3 orbitals (px, py, pz) • “d” has 5 orbitals (dxy, dxz, dyz, dz , dx -y ) • “f” has 7 orbitals (etc. , ) 2 2 2
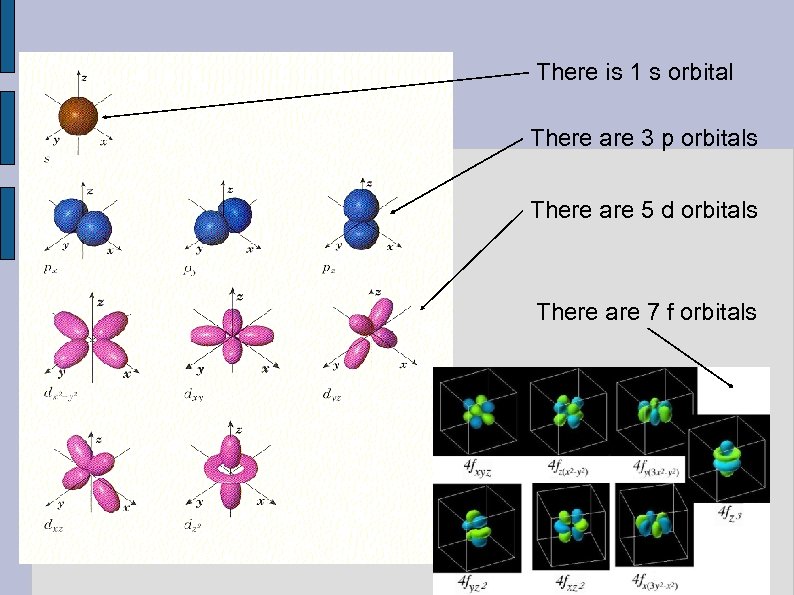
There is 1 s orbital There are 3 p orbitals There are 5 d orbitals There are 7 f orbitals

Fourth Quantum Number Electron Spin ( ms ) Each electron can be spin up (+1/2) or spin down (-1/2) No two electrons in the same orbital orientation can have the same spin. With only one spin up and one spin down, the maximum number of electrons that can fit into any given orbital orientation is two. This is called the Pauli Exclusion Principle.
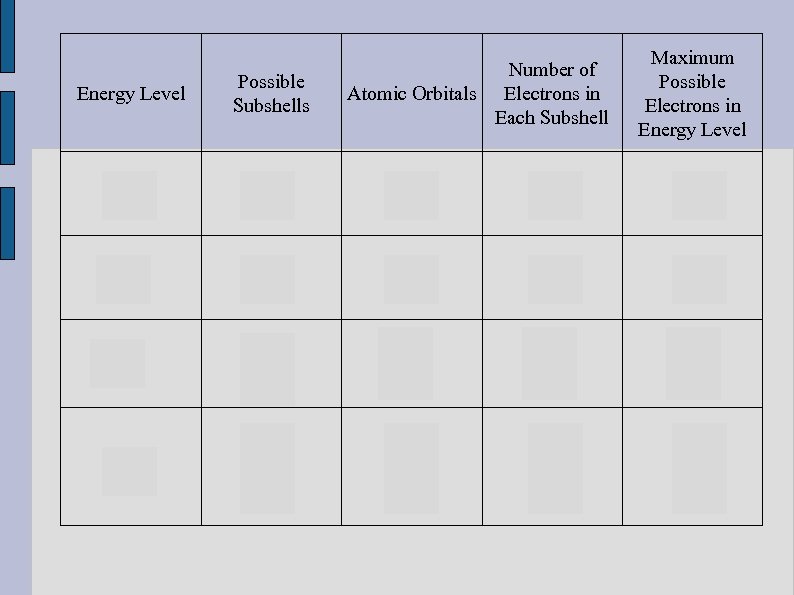
Number of Atomic Orbitals Electrons in Each Subshell Maximum Possible Electrons in Energy Level Possible Subshells 1 2 2 2 s p 1 3 2 6 8 3 s p d 1 3 5 2 6 10 18 4 s p d f 1 3 5 7 2 6 10 14 32

Aufbau Principle / Hunds’ Rule Aufbau: Fill from the ground up Hund’s Rule: When choosing between equivalent orbitals, fill the empty orbitals first
97a5c0c062e65268bbf7df55390a7cef.ppt