27bd5d5fccd4bfda2131a60796d90496.ppt
- Количество слайдов: 29
 Energy • Energy production and consumption, Agriculture and Chemical Manufacturing underlie most environmental issues • freons-stratospheric O 3 depletion • CO 2 -global warming • Agricultural run off contaminating water ways • Urban smog and aerosols • PCB contamination of the Great Lakes in the US • Acid rain and acid aerosols
Energy • Energy production and consumption, Agriculture and Chemical Manufacturing underlie most environmental issues • freons-stratospheric O 3 depletion • CO 2 -global warming • Agricultural run off contaminating water ways • Urban smog and aerosols • PCB contamination of the Great Lakes in the US • Acid rain and acid aerosols
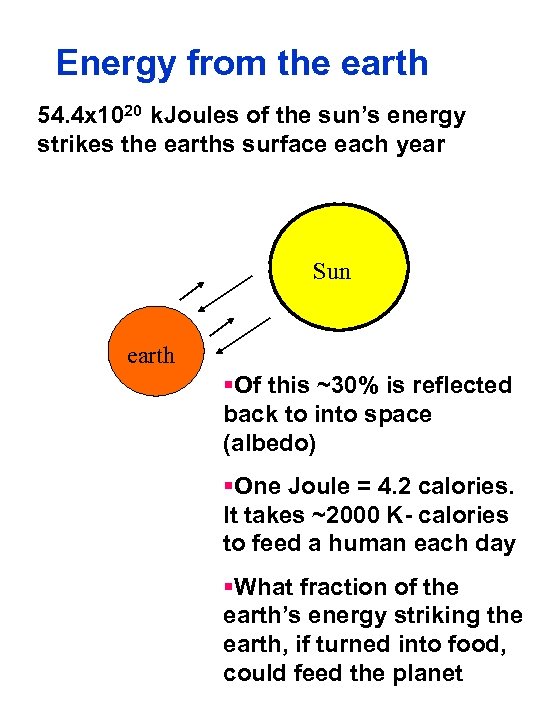 Energy from the earth 54. 4 x 1020 k. Joules of the sun’s energy strikes the earths surface each year Sun earth §Of this ~30% is reflected back to into space (albedo) §One Joule = 4. 2 calories. It takes ~2000 K- calories to feed a human each day §What fraction of the earth’s energy striking the earth, if turned into food, could feed the planet
Energy from the earth 54. 4 x 1020 k. Joules of the sun’s energy strikes the earths surface each year Sun earth §Of this ~30% is reflected back to into space (albedo) §One Joule = 4. 2 calories. It takes ~2000 K- calories to feed a human each day §What fraction of the earth’s energy striking the earth, if turned into food, could feed the planet
 SO what is a joule? ? Force = mass x acceleration f=mxa a = D velocity / D time = dv/dt velocity = D distance / D time a= D distance / D time 2 Work = force x distance W=fxd W= m x a x d and W = m x d 2 /t 2 Work and energy have the same units (The First Law U 2 - U 1 = q - w ) a joule is defined as accelerating 1 kg of mass at 1 meter/sec 2 for a distance of 1 meter A watt is a unit of power = 1 joule/second or energy/time
SO what is a joule? ? Force = mass x acceleration f=mxa a = D velocity / D time = dv/dt velocity = D distance / D time a= D distance / D time 2 Work = force x distance W=fxd W= m x a x d and W = m x d 2 /t 2 Work and energy have the same units (The First Law U 2 - U 1 = q - w ) a joule is defined as accelerating 1 kg of mass at 1 meter/sec 2 for a distance of 1 meter A watt is a unit of power = 1 joule/second or energy/time
 What is the total human energy utilization compared to the Sun’s energy striking the earth? § 54. 4 x 1020 k. Joules of the sun’s energy strikes the earths surface each year §# of k. J striking the earth/year, minus reflection (albedo =0. 3)= total energy 54. 4 x 1020 x 0. 7= 38. 1 x 1020 k. Joules §what fraction of total sun’s energy absorbed by the earth is used by human activity ? § people use 3. 7 x 10 17 k. Joules/year § 3. 7 x 10 17 / 38. 1 x 1020 = 0. 001 = 0. 1% § so if we harnessed 1/1000 th of the sun’s energy we could supply all of our needs
What is the total human energy utilization compared to the Sun’s energy striking the earth? § 54. 4 x 1020 k. Joules of the sun’s energy strikes the earths surface each year §# of k. J striking the earth/year, minus reflection (albedo =0. 3)= total energy 54. 4 x 1020 x 0. 7= 38. 1 x 1020 k. Joules §what fraction of total sun’s energy absorbed by the earth is used by human activity ? § people use 3. 7 x 10 17 k. Joules/year § 3. 7 x 10 17 / 38. 1 x 1020 = 0. 001 = 0. 1% § so if we harnessed 1/1000 th of the sun’s energy we could supply all of our needs
 Worldwide energy use and how do we use fuels (1993) Oil 34. 1% Coal 24. 1% natural gas 17. 4% Biomass 14. 7% Hydro 5. 5% Nuclear 4. 1% energy use/y (1993) Population world 382 x 1018 joules 4. 87 x 109 Indust. world 347 x 1018 joules 1. 22 x 109 Developing world 35 x 1018 joules 3. 65 x 109
Worldwide energy use and how do we use fuels (1993) Oil 34. 1% Coal 24. 1% natural gas 17. 4% Biomass 14. 7% Hydro 5. 5% Nuclear 4. 1% energy use/y (1993) Population world 382 x 1018 joules 4. 87 x 109 Indust. world 347 x 1018 joules 1. 22 x 109 Developing world 35 x 1018 joules 3. 65 x 109
 Where are the global energy reserves oil Figure 1. 5 Spiro. Former USSR page 10 Middle East Asia and Australia including China
Where are the global energy reserves oil Figure 1. 5 Spiro. Former USSR page 10 Middle East Asia and Australia including China
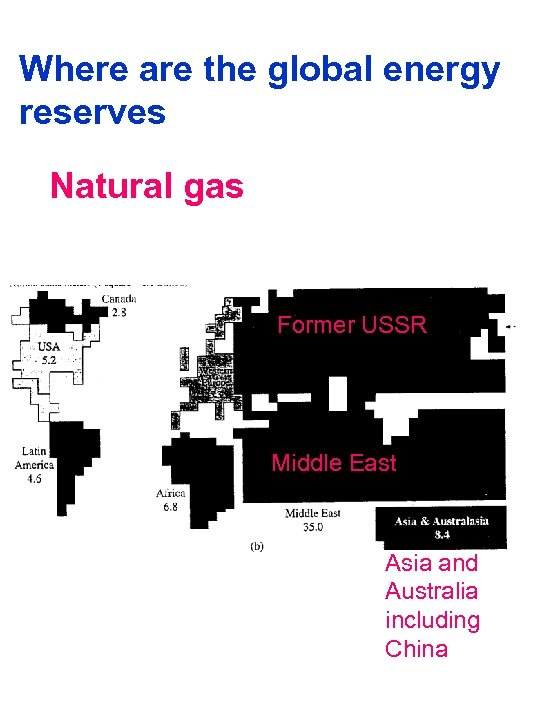 Where are the global energy reserves Natural gas Former USSR Middle East Asia and Australia including China
Where are the global energy reserves Natural gas Former USSR Middle East Asia and Australia including China
 Where are the global energy reserves Coal Former USSR China Australia
Where are the global energy reserves Coal Former USSR China Australia
 Earth’s nonrenewable energy resources (1980) estimated stock x 1021 J consumption (world)/year x 1018 J petroleum 10 135 Natural gas 10 60 coal 250 90 oilshale 2, 000 0 uranium (non-breading water reactors) 20 6. 3 Thorium and Uranium (in breeder react) 10, 000 0 Deuterium and Li in sea water for fusion 1010 0
Earth’s nonrenewable energy resources (1980) estimated stock x 1021 J consumption (world)/year x 1018 J petroleum 10 135 Natural gas 10 60 coal 250 90 oilshale 2, 000 0 uranium (non-breading water reactors) 20 6. 3 Thorium and Uranium (in breeder react) 10, 000 0 Deuterium and Li in sea water for fusion 1010 0
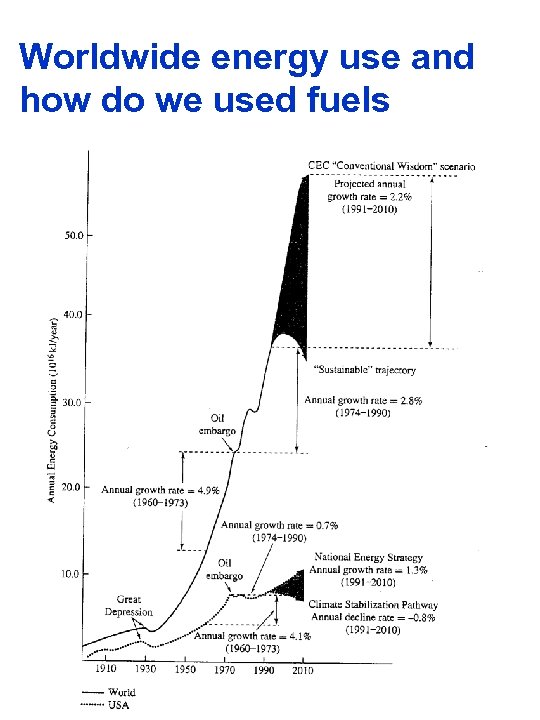 Worldwide energy use and how do we used fuels
Worldwide energy use and how do we used fuels
 how long will the oil last? ? 1980 estimate of reserves Oil 1 x 1022 J Where does a number like this come from? Let’s look at a 2004 Christian Science Monitor article: World wide proven Oil reserves = 1. 1 to 1. 3 x 1012 barrels 1 barrel of oil = 42 US gallons or 158. 99 liters If hydrocarbons have a density of 0. 9 kg/liter 1 barrel = 158. 99 x 0. 9 x 1000 grams = 1. 43 x 105 grams oil /barrel
how long will the oil last? ? 1980 estimate of reserves Oil 1 x 1022 J Where does a number like this come from? Let’s look at a 2004 Christian Science Monitor article: World wide proven Oil reserves = 1. 1 to 1. 3 x 1012 barrels 1 barrel of oil = 42 US gallons or 158. 99 liters If hydrocarbons have a density of 0. 9 kg/liter 1 barrel = 158. 99 x 0. 9 x 1000 grams = 1. 43 x 105 grams oil /barrel
 = 1. 43 x 105 grams oil /barrel We will see later that one gram of oil gives off 44 k. Joules/g when it is burned We said there were 1. 2 x 1012 barrels known reserves This means that we have 1. 43 x 105 x 1. 2 x 1012 x 44 x 10000 joules of oil= ~0. 8 x 1022 joules WE SAID THE 1980 ESTIMATE 1 x 1022 joules
= 1. 43 x 105 grams oil /barrel We will see later that one gram of oil gives off 44 k. Joules/g when it is burned We said there were 1. 2 x 1012 barrels known reserves This means that we have 1. 43 x 105 x 1. 2 x 1012 x 44 x 10000 joules of oil= ~0. 8 x 1022 joules WE SAID THE 1980 ESTIMATE 1 x 1022 joules
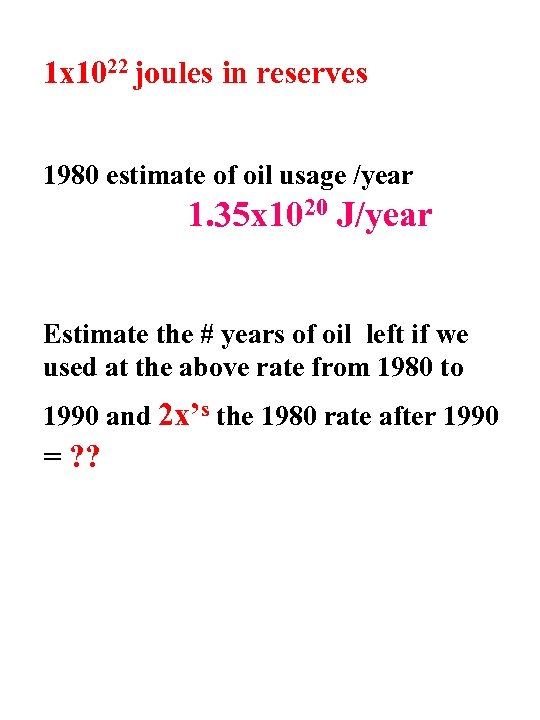 1 x 1022 joules in reserves 1980 estimate of oil usage /year 1. 35 x 1020 J/year Estimate the # years of oil left if we used at the above rate from 1980 to 1990 and 2 x’s the 1980 rate after 1990 = ? ?
1 x 1022 joules in reserves 1980 estimate of oil usage /year 1. 35 x 1020 J/year Estimate the # years of oil left if we used at the above rate from 1980 to 1990 and 2 x’s the 1980 rate after 1990 = ? ?
 New data WE use globally (2004)about 30 x 109 barrels/year If we have 1. 2 x 1012 in reserve or 1200 x 109 in reserve Others say World wide oil reserves have grown 15% between 1999 and 2004 and have grown by a factor of 5 since WWII
New data WE use globally (2004)about 30 x 109 barrels/year If we have 1. 2 x 1012 in reserve or 1200 x 109 in reserve Others say World wide oil reserves have grown 15% between 1999 and 2004 and have grown by a factor of 5 since WWII
 These estimates place the global reserves at ~3 x 1012 barrels and suggest that we have only used 25% of the total oil on the planet What we know is that major importers are not waiting around to see who is right!!! The US, China, Japan are scrambling to tie down interests in Russia, West Africa, Iraq, Iran and Libya
These estimates place the global reserves at ~3 x 1012 barrels and suggest that we have only used 25% of the total oil on the planet What we know is that major importers are not waiting around to see who is right!!! The US, China, Japan are scrambling to tie down interests in Russia, West Africa, Iraq, Iran and Libya
 What happens to the fuel we burn §Burning “old” carbon: fossil fuels add CO 2 to the atmosphere that has been buried as carbon under the earths surface eons ago. §Burning “new”: biomass fuels puts CO 2 in the atmosphere that has just recently been remove from the atmosphere by plants. These kinds of fuels would be considered green house neutral. §Given the exponential increase in our use of fossil fuels, one must ask, how much longer this can go on? §Some people in the automotive industry said in 2000 (conversation of Kamens with D. Schuetzle of Ford) we will see significant shortages by 2015
What happens to the fuel we burn §Burning “old” carbon: fossil fuels add CO 2 to the atmosphere that has been buried as carbon under the earths surface eons ago. §Burning “new”: biomass fuels puts CO 2 in the atmosphere that has just recently been remove from the atmosphere by plants. These kinds of fuels would be considered green house neutral. §Given the exponential increase in our use of fossil fuels, one must ask, how much longer this can go on? §Some people in the automotive industry said in 2000 (conversation of Kamens with D. Schuetzle of Ford) we will see significant shortages by 2015
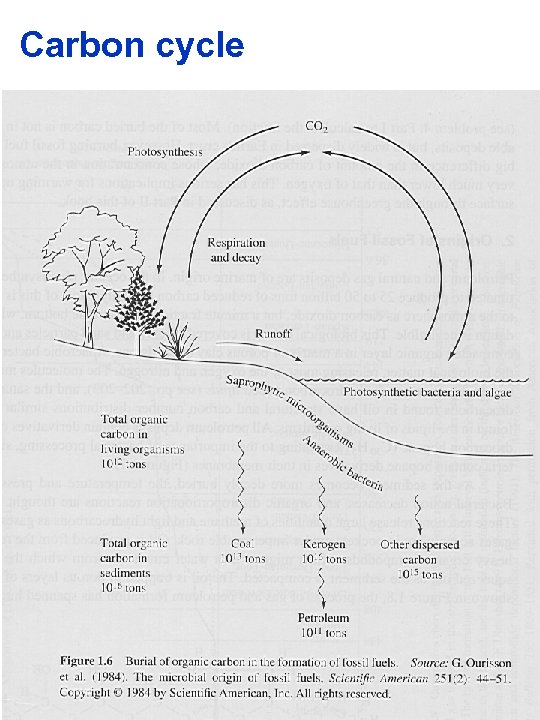 Carbon cycle
Carbon cycle
 Oil §Petroleum and gas deposits come from the seas. §Oceans produce 25 - 50 billion tones of reduced carbon annually. §Most is recycled to the atmosphere as CO 2. A very small fraction settles to the bottom where oxidation is negligible § here it is compacted with clay and sand particles §Anaerobic bacteria digest the bacterial digestible matter, releasing O 2 and N 2. §The hydrocarbons most resistant are the hydrocarbon based lipids and these persist and are found in their cell membranes indicating that bacteria process organic debris in the oceans and over the eons turned it into oil
Oil §Petroleum and gas deposits come from the seas. §Oceans produce 25 - 50 billion tones of reduced carbon annually. §Most is recycled to the atmosphere as CO 2. A very small fraction settles to the bottom where oxidation is negligible § here it is compacted with clay and sand particles §Anaerobic bacteria digest the bacterial digestible matter, releasing O 2 and N 2. §The hydrocarbons most resistant are the hydrocarbon based lipids and these persist and are found in their cell membranes indicating that bacteria process organic debris in the oceans and over the eons turned it into oil
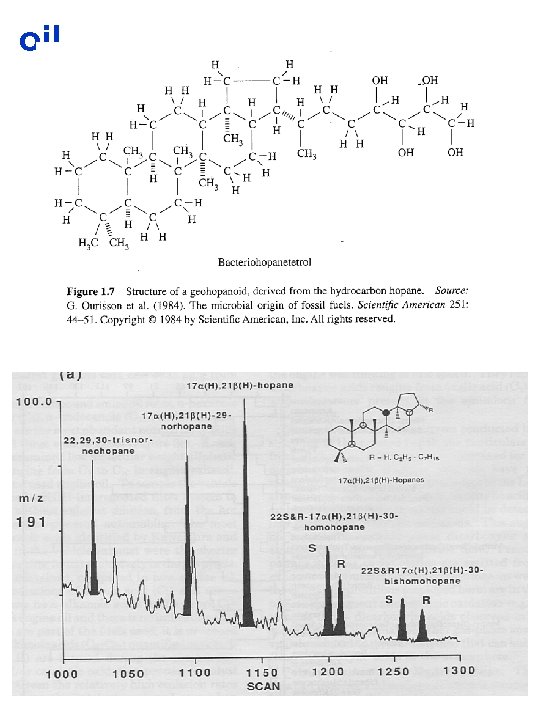 oil
oil
 Coal §Coal formation is land or terrestrial based Woody plants 200 million years ago, as they are now, are composed of cellulose and lignins. Bacteria can digest the cellulose over time but lignins are resistant In swamps the lignins accumulate under water and are compacted into peat Crustal upheavals buried the peat and subjected it to huge pressures and temperatuers peat coal over time
Coal §Coal formation is land or terrestrial based Woody plants 200 million years ago, as they are now, are composed of cellulose and lignins. Bacteria can digest the cellulose over time but lignins are resistant In swamps the lignins accumulate under water and are compacted into peat Crustal upheavals buried the peat and subjected it to huge pressures and temperatuers peat coal over time
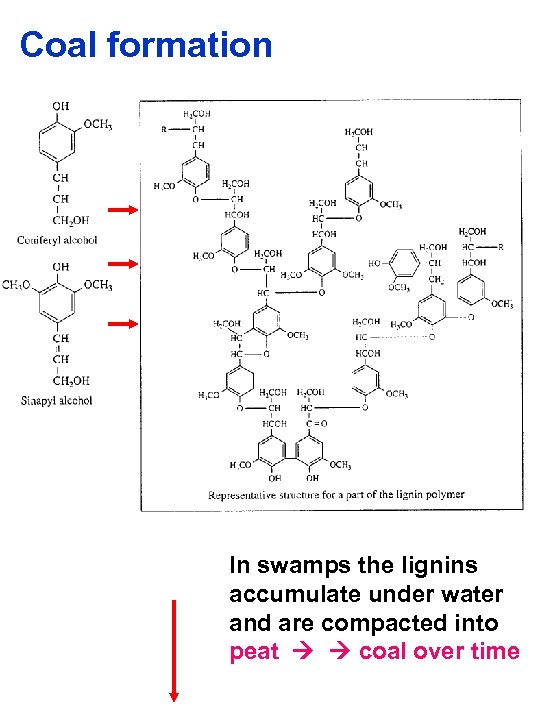 Coal formation In swamps the lignins accumulate under water and are compacted into peat coal over time
Coal formation In swamps the lignins accumulate under water and are compacted into peat coal over time
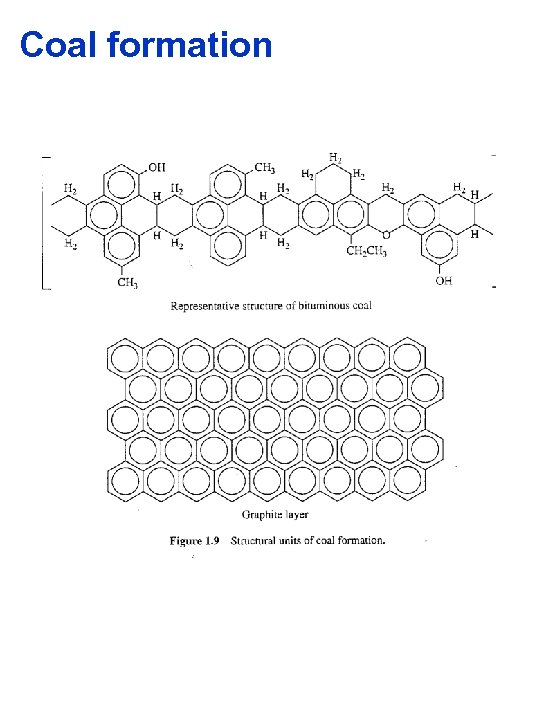 Coal formation
Coal formation
 Fuel energy §When we burn a fuel where does the energy reside? §Let s take hydrogen in water as an example. If we were to react H 2 with O 2 to form water, we would 1 st have to break the hydrogen bonds and the oxygen bonds §This takes energy; in the case of H 2 it takes 432 k. J/mole (~100, 000 calories/mole) for H 2 2 H. § 100, 000 calories will supply you with many minutes of food energy? ? §To break O 2 to O. (O 2 2 O. ) requires 494 k. J/mol §When water forms, however, we get energy back from the formation of H 2 O because new bonds are formed. Which ones? ?
Fuel energy §When we burn a fuel where does the energy reside? §Let s take hydrogen in water as an example. If we were to react H 2 with O 2 to form water, we would 1 st have to break the hydrogen bonds and the oxygen bonds §This takes energy; in the case of H 2 it takes 432 k. J/mole (~100, 000 calories/mole) for H 2 2 H. § 100, 000 calories will supply you with many minutes of food energy? ? §To break O 2 to O. (O 2 2 O. ) requires 494 k. J/mol §When water forms, however, we get energy back from the formation of H 2 O because new bonds are formed. Which ones? ?
 Fuel energy §The equation for the combination of hydrogen and oxygen if say we were to burn hydrogen would be § 2 H 2 + O 2 2 H 2 O §To break a mole of H 2 bonds requires 432 §We need 2 moles of H 2 so this requires 864 joules §To break a mole of O 2 requires 492 k. J/mol; so the total energy required to break 2 H 2 and O 2 apart is 1356 k. J §To form water we need to form two O-H bonds. When one OH bond forms it releases 460 k. J/mole §But there are two water molecules that from = 2 x 460 x 2= 1840 k. J/mole §So how much energy is released?
Fuel energy §The equation for the combination of hydrogen and oxygen if say we were to burn hydrogen would be § 2 H 2 + O 2 2 H 2 O §To break a mole of H 2 bonds requires 432 §We need 2 moles of H 2 so this requires 864 joules §To break a mole of O 2 requires 492 k. J/mol; so the total energy required to break 2 H 2 and O 2 apart is 1356 k. J §To form water we need to form two O-H bonds. When one OH bond forms it releases 460 k. J/mole §But there are two water molecules that from = 2 x 460 x 2= 1840 k. J/mole §So how much energy is released?
 Energy from breaking other bonds (enthalpy) k. J/mole H-H 432 O=O 492 O-H 460 C-H 360 C=O 799 C-C 347 C-C aromatic 519 N=O 632
Energy from breaking other bonds (enthalpy) k. J/mole H-H 432 O=O 492 O-H 460 C-H 360 C=O 799 C-C 347 C-C aromatic 519 N=O 632
 Fuel energy §Let’s do the same thing for burning methane gas; the reaction is methane + oxygen § CH 4 + 2 O 2 CO 2 + 2 H 2 O §We calculated before that to form one mole of H 2 O we get 920 k. J, so for two moles we get 1840 k. J/mole; to form CO 2, which also releases energy, we need to form two C=O bonds {O=C=O} or 2 x 799 k. J. This gives a total formation energy of 1840 + 1598 k. J §But for this process, we 1 st have to break 4 carbon- hydrogen bonds; why? ? This requires 410 k. J/mole/bond or 1640 k. J §The total energy release is the energy forming bonds - break bonds § 3438 k. J – 2628 k. J = 810 k. J excess energy
Fuel energy §Let’s do the same thing for burning methane gas; the reaction is methane + oxygen § CH 4 + 2 O 2 CO 2 + 2 H 2 O §We calculated before that to form one mole of H 2 O we get 920 k. J, so for two moles we get 1840 k. J/mole; to form CO 2, which also releases energy, we need to form two C=O bonds {O=C=O} or 2 x 799 k. J. This gives a total formation energy of 1840 + 1598 k. J §But for this process, we 1 st have to break 4 carbon- hydrogen bonds; why? ? This requires 410 k. J/mole/bond or 1640 k. J §The total energy release is the energy forming bonds - break bonds § 3438 k. J – 2628 k. J = 810 k. J excess energy
 Combustion energies from different fuels (k. J) react. per per moles heat mole gram CO 2 per k. J O 2 fuel 1000 k. J hydrogen 482 2 H 2+O 2 2 H 2 O 482 241 120 0 Gas 810 405 810 CH 4 + 2 O 2 CO 2 +2 H 2 O 52 1. 2 Petroleum 2120 407 610 44 2 (-CH 2 -)+ 3 O 2 2 CO 2 +2 H 2 O 1. 6 Coal 4332 409 512 4 (-CH-)+ 5 O 2 4 CO 2 +2 H 2 O 39 2. 0 Ethanol 1257 419 1257 27 C 2 H 5 OH + 3 O 2 2 CO 2 +3 H 2 O 1. 6 wood 447 447 15 (-CHOH-) + O 2 CO 2 +2 H 2 O 2. 2
Combustion energies from different fuels (k. J) react. per per moles heat mole gram CO 2 per k. J O 2 fuel 1000 k. J hydrogen 482 2 H 2+O 2 2 H 2 O 482 241 120 0 Gas 810 405 810 CH 4 + 2 O 2 CO 2 +2 H 2 O 52 1. 2 Petroleum 2120 407 610 44 2 (-CH 2 -)+ 3 O 2 2 CO 2 +2 H 2 O 1. 6 Coal 4332 409 512 4 (-CH-)+ 5 O 2 4 CO 2 +2 H 2 O 39 2. 0 Ethanol 1257 419 1257 27 C 2 H 5 OH + 3 O 2 2 CO 2 +3 H 2 O 1. 6 wood 447 447 15 (-CHOH-) + O 2 CO 2 +2 H 2 O 2. 2
 A Homework problem Assume as students, that you each use 1000 Watts of power for 12 hours each day (lights, computers, class room air conditioning, etc, travel). Much of this energy in Thailand China is generated from coal. Assume that the process is only 50% efficient, and if you use 1000 Watts it really requires 2000 Watts of coal power. Calculate how much CO 2 is going into the atmosphere to maintain you at this level of energy consumption for each year and put your answer in metric tones/year CO 2.
A Homework problem Assume as students, that you each use 1000 Watts of power for 12 hours each day (lights, computers, class room air conditioning, etc, travel). Much of this energy in Thailand China is generated from coal. Assume that the process is only 50% efficient, and if you use 1000 Watts it really requires 2000 Watts of coal power. Calculate how much CO 2 is going into the atmosphere to maintain you at this level of energy consumption for each year and put your answer in metric tones/year CO 2.
 Hint 1 watt = 1 joule/sec Estimate the total number of joules used per year if you are using 2000 watts for 12 hours each day The combustion table gives you the mole of CO 2 evolved from burring 1000 k. J of coal. Convert this to the total moles of CO 2 given off per year for 2000 watts at 12 hours each day Convert to metric tonnes of CO 2 per year One metic tonne equals 1000 kg
Hint 1 watt = 1 joule/sec Estimate the total number of joules used per year if you are using 2000 watts for 12 hours each day The combustion table gives you the mole of CO 2 evolved from burring 1000 k. J of coal. Convert this to the total moles of CO 2 given off per year for 2000 watts at 12 hours each day Convert to metric tonnes of CO 2 per year One metic tonne equals 1000 kg


