dfcad89010dfa8c90a7d6374d5f3e98d.ppt
- Количество слайдов: 36
 Electrochemical Generation of Nanostructures at the Liquid-Liquid Interface Robert A. W. Dryfe School of Chemistry, Univ. of Manchester (U. K. ) robert. dryfe@manchester. ac. uk Leiden, Nov. 2008
Electrochemical Generation of Nanostructures at the Liquid-Liquid Interface Robert A. W. Dryfe School of Chemistry, Univ. of Manchester (U. K. ) robert. dryfe@manchester. ac. uk Leiden, Nov. 2008
 Liquid/Liquid Interfaces in catalysis • Widely used: bi-phasic system, allows for ease of separation of catalysis from reactant mixture. • Electrochemical investigations of phasetransfer catalysis (Schiffrin 1988 [1], Girault 1994 [2]) • Water does not have to be one of the phases = “Fluorous biphase catalysis” (Horvath 1994) [3] • Stable room-temperature ionic liquids: • (Ballantyne 2008 [4]) Leiden, Nov. 2008 H 3 DA TPBF 3 ethylmethylimidazolium ethylsulfate (EMIM Et. SO 4) interface
Liquid/Liquid Interfaces in catalysis • Widely used: bi-phasic system, allows for ease of separation of catalysis from reactant mixture. • Electrochemical investigations of phasetransfer catalysis (Schiffrin 1988 [1], Girault 1994 [2]) • Water does not have to be one of the phases = “Fluorous biphase catalysis” (Horvath 1994) [3] • Stable room-temperature ionic liquids: • (Ballantyne 2008 [4]) Leiden, Nov. 2008 H 3 DA TPBF 3 ethylmethylimidazolium ethylsulfate (EMIM Et. SO 4) interface
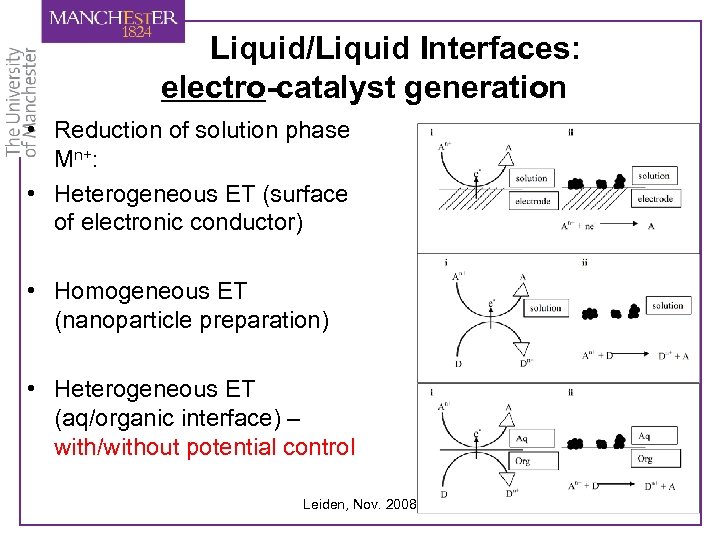 Liquid/Liquid Interfaces: electro-catalyst generation • Reduction of solution phase Mn+: • Heterogeneous ET (surface of electronic conductor) • Homogeneous ET (nanoparticle preparation) • Heterogeneous ET (aq/organic interface) – with/without potential control Leiden, Nov. 2008
Liquid/Liquid Interfaces: electro-catalyst generation • Reduction of solution phase Mn+: • Heterogeneous ET (surface of electronic conductor) • Homogeneous ET (nanoparticle preparation) • Heterogeneous ET (aq/organic interface) – with/without potential control Leiden, Nov. 2008
 Liquid/Liquid Interfaces: electro-catalytic reactions • Questions: • Can the catalyst be used in situ - for catalysis of processes at liquid-liquid phase boundaries? • If so, could catalyst density be controlled (Langmuir trough approach) to optimise reactivity? • Or can catalyst be removed and immobilised on a (conventional) electrode? Leiden, Nov. 2008
Liquid/Liquid Interfaces: electro-catalytic reactions • Questions: • Can the catalyst be used in situ - for catalysis of processes at liquid-liquid phase boundaries? • If so, could catalyst density be controlled (Langmuir trough approach) to optimise reactivity? • Or can catalyst be removed and immobilised on a (conventional) electrode? Leiden, Nov. 2008
 {Liquid-liquid Electrochemistry 1: Distribution potential} • Each ion: distribution equilibrium at the organic/water interface • Define standard Galvani potential of transfer: • Vary potential with common-ion • ratio of ion concentration in each phase (maintained by hydrophilic/hydrophobic counter-ions) “poises” potential • (Nernst-Donnan equilibrium ) • - ion transfer/electron transfer – particularly for SECM @ L/L. Leiden, Nov. 2008
{Liquid-liquid Electrochemistry 1: Distribution potential} • Each ion: distribution equilibrium at the organic/water interface • Define standard Galvani potential of transfer: • Vary potential with common-ion • ratio of ion concentration in each phase (maintained by hydrophilic/hydrophobic counter-ions) “poises” potential • (Nernst-Donnan equilibrium ) • - ion transfer/electron transfer – particularly for SECM @ L/L. Leiden, Nov. 2008
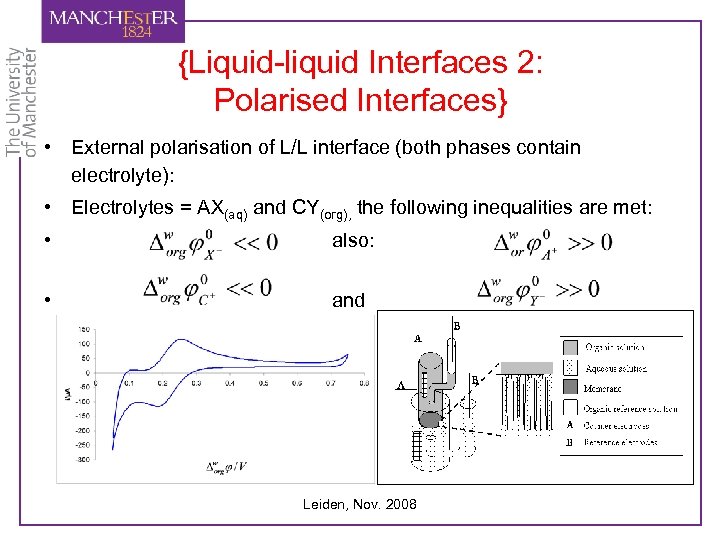 {Liquid-liquid Interfaces 2: Polarised Interfaces} • External polarisation of L/L interface (both phases contain electrolyte): • Electrolytes = AX(aq) and CY(org), the following inequalities are met: • also: • and Leiden, Nov. 2008
{Liquid-liquid Interfaces 2: Polarised Interfaces} • External polarisation of L/L interface (both phases contain electrolyte): • Electrolytes = AX(aq) and CY(org), the following inequalities are met: • also: • and Leiden, Nov. 2008
 Structure of L/L interface • Essentially sharp, even down to molecular scale – nm-scale transition from phase 1 to phase 2. • Interfacial fluctuations (capillary waves): • Competition between thermal motion and interfacial tension • Appear to extend down to molecular scale) = nm scale amplitude • Experimental probes: X-ray scattering, non-linear optical spectroscopy (SFG, SHG), (Schlossman, 2000 [5]), (Richmond 2001 [6]). • => Smooth, reproducible interface. Leiden, Nov. 2008
Structure of L/L interface • Essentially sharp, even down to molecular scale – nm-scale transition from phase 1 to phase 2. • Interfacial fluctuations (capillary waves): • Competition between thermal motion and interfacial tension • Appear to extend down to molecular scale) = nm scale amplitude • Experimental probes: X-ray scattering, non-linear optical spectroscopy (SFG, SHG), (Schlossman, 2000 [5]), (Richmond 2001 [6]). • => Smooth, reproducible interface. Leiden, Nov. 2008
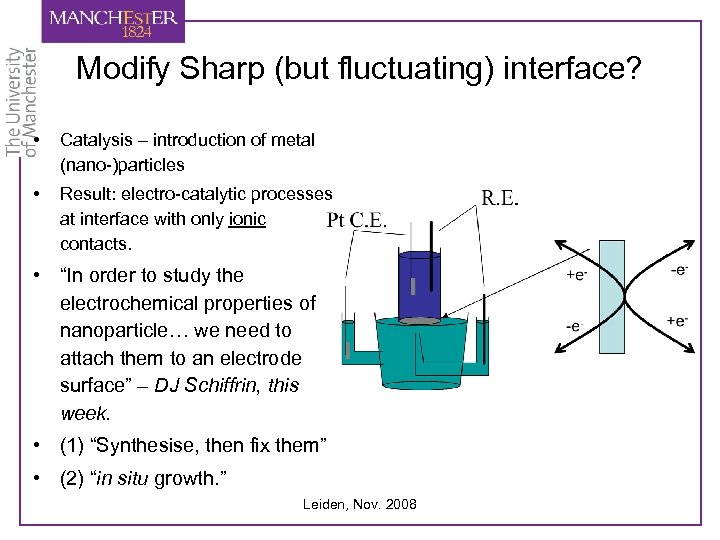 Modify Sharp (but fluctuating) interface? • Catalysis – introduction of metal (nano-)particles • Result: electro-catalytic processes at interface with only ionic contacts. • “In order to study the electrochemical properties of nanoparticle… we need to attach them to an electrode surface” – DJ Schiffrin, this week. • (1) “Synthesise, then fix them” • (2) “in situ growth. ” Leiden, Nov. 2008
Modify Sharp (but fluctuating) interface? • Catalysis – introduction of metal (nano-)particles • Result: electro-catalytic processes at interface with only ionic contacts. • “In order to study the electrochemical properties of nanoparticle… we need to attach them to an electrode surface” – DJ Schiffrin, this week. • (1) “Synthesise, then fix them” • (2) “in situ growth. ” Leiden, Nov. 2008
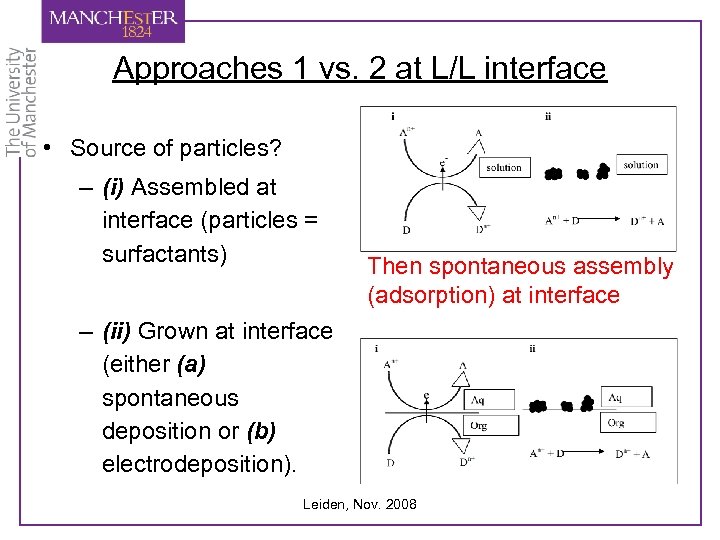 Approaches 1 vs. 2 at L/L interface • Source of particles? – (i) Assembled at interface (particles = surfactants) Then spontaneous assembly (adsorption) at interface – (ii) Grown at interface (either (a) spontaneous deposition or (b) electrodeposition). Leiden, Nov. 2008
Approaches 1 vs. 2 at L/L interface • Source of particles? – (i) Assembled at interface (particles = surfactants) Then spontaneous assembly (adsorption) at interface – (ii) Grown at interface (either (a) spontaneous deposition or (b) electrodeposition). Leiden, Nov. 2008
 (i) Assembly of (pre-formed) particles at L/L interfaces • Method: form hydrosol (organo-sol), particles adsorb interface on introduction of organic (aqueous) phase. • Particles are surfactants, if favourable contact angle, q. • Desorption energy given by: • Particles of given type, will be displaced by those with larger radius (r): • Size segregation effect demonstrated for Cd. Se (Russell, 2003 [7]). Leiden, Nov. 2008
(i) Assembly of (pre-formed) particles at L/L interfaces • Method: form hydrosol (organo-sol), particles adsorb interface on introduction of organic (aqueous) phase. • Particles are surfactants, if favourable contact angle, q. • Desorption energy given by: • Particles of given type, will be displaced by those with larger radius (r): • Size segregation effect demonstrated for Cd. Se (Russell, 2003 [7]). Leiden, Nov. 2008
 (i) Assembly of (pre-formed) particles at L/L interfaces - continued • Other terms in equation: • q can be varied by changing surface chemistry (Vanmaekelbergh, 2003 [8]) – induce assembly of Au NPs by addition of ethanol – contact angle tends 90 o. • Residual surface charge, Au NPs attracted to/from polarised L/L interface – see Figure, from (Fermin, 2004 [9]) • Lippmann equation, interfacial tension is function of applied potential Leiden, Nov. 2008
(i) Assembly of (pre-formed) particles at L/L interfaces - continued • Other terms in equation: • q can be varied by changing surface chemistry (Vanmaekelbergh, 2003 [8]) – induce assembly of Au NPs by addition of ethanol – contact angle tends 90 o. • Residual surface charge, Au NPs attracted to/from polarised L/L interface – see Figure, from (Fermin, 2004 [9]) • Lippmann equation, interfacial tension is function of applied potential Leiden, Nov. 2008
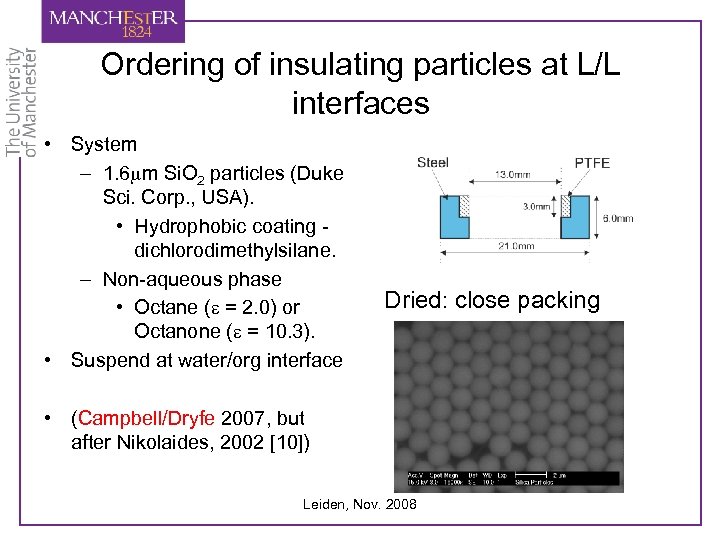 Ordering of insulating particles at L/L interfaces • System – 1. 6 mm Si. O 2 particles (Duke Sci. Corp. , USA). • Hydrophobic coating dichlorodimethylsilane. – Non-aqueous phase • Octane (e = 2. 0) or Octanone (e = 10. 3). • Suspend at water/org interface Dried: close packing • (Campbell/Dryfe 2007, but after Nikolaides, 2002 [10]) Leiden, Nov. 2008
Ordering of insulating particles at L/L interfaces • System – 1. 6 mm Si. O 2 particles (Duke Sci. Corp. , USA). • Hydrophobic coating dichlorodimethylsilane. – Non-aqueous phase • Octane (e = 2. 0) or Octanone (e = 10. 3). • Suspend at water/org interface Dried: close packing • (Campbell/Dryfe 2007, but after Nikolaides, 2002 [10]) Leiden, Nov. 2008
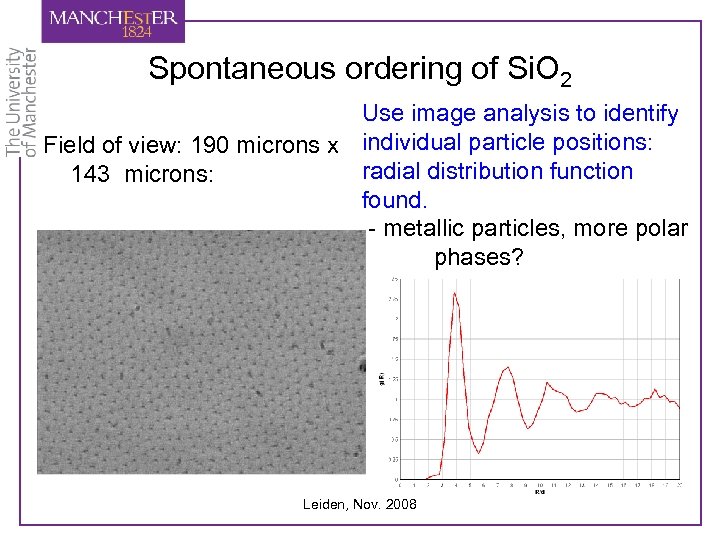 Spontaneous ordering of Si. O 2 Use image analysis to identify Field of view: 190 microns x individual particle positions: radial distribution function 143 microns: found. - metallic particles, more polar phases? Leiden, Nov. 2008
Spontaneous ordering of Si. O 2 Use image analysis to identify Field of view: 190 microns x individual particle positions: radial distribution function 143 microns: found. - metallic particles, more polar phases? Leiden, Nov. 2008
 (ii – a) In situ growth of particles at L/L interfaces: spontaneous chemical reduction • Faraday (1857 [11]): formation of colloidal Au at L/L (water/CS 2) interface • “dark flocculent deposits”, metal in “a fine state of division”. • General problem of particle formation at L/L interface is prevention of aggregation: • e. g. Au deposition @ water/1, 2 dichloroethane interface, fractal structures form: image statistics, growth laws for aggregation process (scale bar = 10 microns) Leiden, Nov. 2008
(ii – a) In situ growth of particles at L/L interfaces: spontaneous chemical reduction • Faraday (1857 [11]): formation of colloidal Au at L/L (water/CS 2) interface • “dark flocculent deposits”, metal in “a fine state of division”. • General problem of particle formation at L/L interface is prevention of aggregation: • e. g. Au deposition @ water/1, 2 dichloroethane interface, fractal structures form: image statistics, growth laws for aggregation process (scale bar = 10 microns) Leiden, Nov. 2008
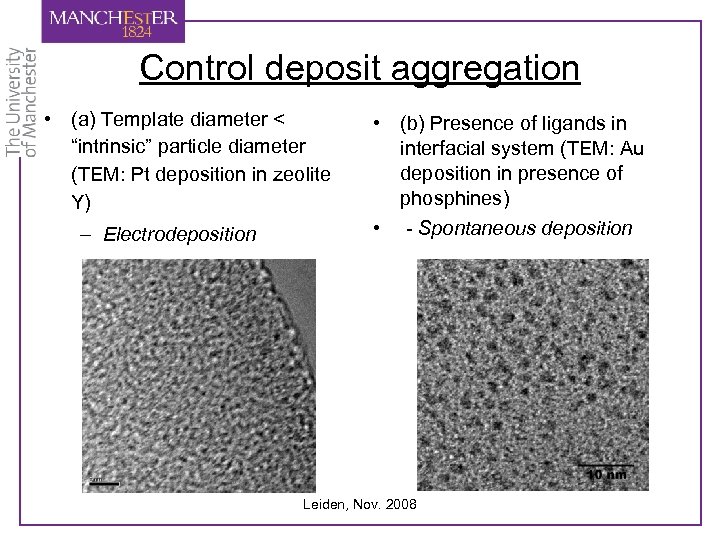 Control deposit aggregation • (a) Template diameter < “intrinsic” particle diameter (TEM: Pt deposition in zeolite Y) – Electrodeposition • (b) Presence of ligands in interfacial system (TEM: Au deposition in presence of phosphines) • - Spontaneous deposition Leiden, Nov. 2008
Control deposit aggregation • (a) Template diameter < “intrinsic” particle diameter (TEM: Pt deposition in zeolite Y) – Electrodeposition • (b) Presence of ligands in interfacial system (TEM: Au deposition in presence of phosphines) • - Spontaneous deposition Leiden, Nov. 2008
 Stabilisation: surface chemistry • Ideal case: modify surfaces to prevent aggregation, but retain catalytic activity. • Brust/Schiffrin (1994, [12]) (+ Faraday? ): thiol stabilisation of Au formed by two-phase reduction • Hutchison (2000 [13]), Rao (2003 [14]) (+ Faraday? ) : phosphine ligands for stabilisation of Au formed at L/L interface. • • Question: for Au deposition, can process (i) = assembly of particles at L/L be related to process (ii) = in situ L/L formation? Leiden, Nov. 2008
Stabilisation: surface chemistry • Ideal case: modify surfaces to prevent aggregation, but retain catalytic activity. • Brust/Schiffrin (1994, [12]) (+ Faraday? ): thiol stabilisation of Au formed by two-phase reduction • Hutchison (2000 [13]), Rao (2003 [14]) (+ Faraday? ) : phosphine ligands for stabilisation of Au formed at L/L interface. • • Question: for Au deposition, can process (i) = assembly of particles at L/L be related to process (ii) = in situ L/L formation? Leiden, Nov. 2008
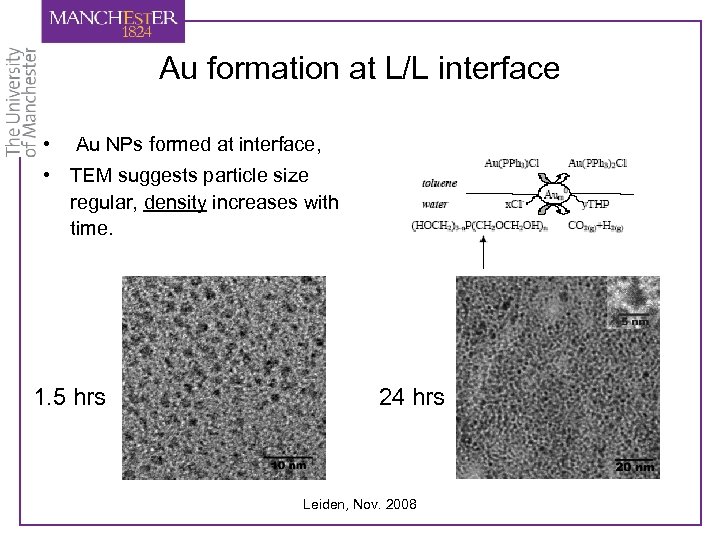 Au formation at L/L interface • Au NPs formed at interface, • TEM suggests particle size regular, density increases with time. 1. 5 hrs 24 hrs Leiden, Nov. 2008
Au formation at L/L interface • Au NPs formed at interface, • TEM suggests particle size regular, density increases with time. 1. 5 hrs 24 hrs Leiden, Nov. 2008
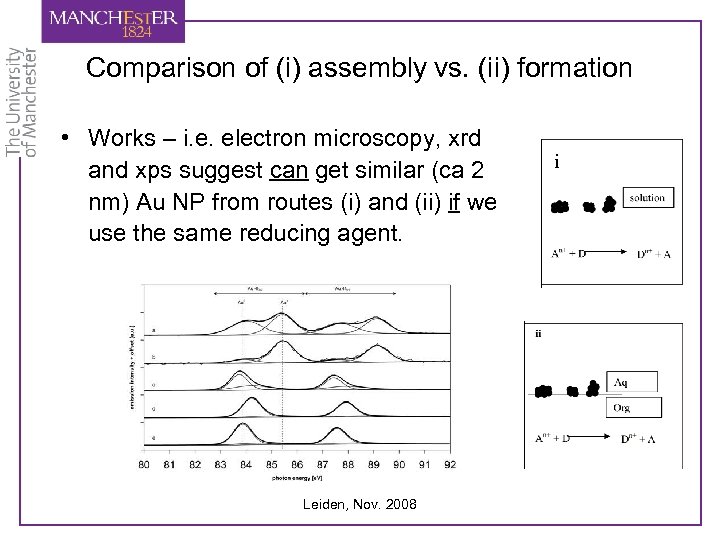 Comparison of (i) assembly vs. (ii) formation • Works – i. e. electron microscopy, xrd and xps suggest can get similar (ca 2 nm) Au NP from routes (i) and (ii) if we use the same reducing agent. Leiden, Nov. 2008 i
Comparison of (i) assembly vs. (ii) formation • Works – i. e. electron microscopy, xrd and xps suggest can get similar (ca 2 nm) Au NP from routes (i) and (ii) if we use the same reducing agent. Leiden, Nov. 2008 i
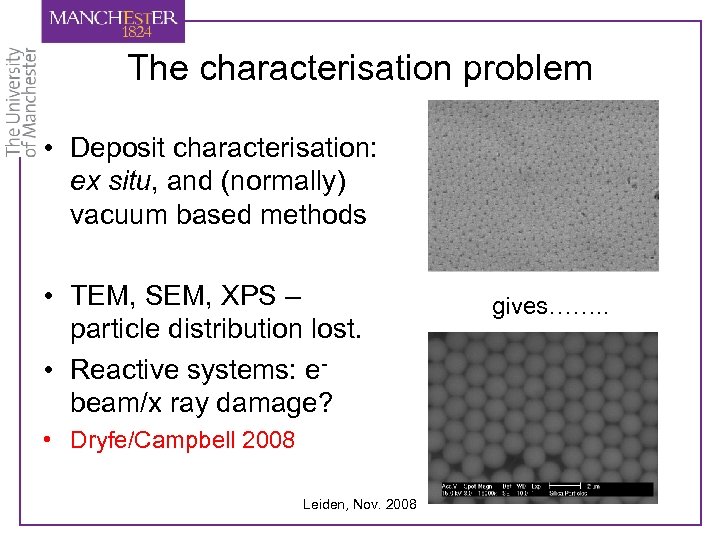 The characterisation problem • Deposit characterisation: ex situ, and (normally) vacuum based methods • TEM, SEM, XPS – particle distribution lost. • Reactive systems: ebeam/x ray damage? • Dryfe/Campbell 2008 Leiden, Nov. 2008 gives……. .
The characterisation problem • Deposit characterisation: ex situ, and (normally) vacuum based methods • TEM, SEM, XPS – particle distribution lost. • Reactive systems: ebeam/x ray damage? • Dryfe/Campbell 2008 Leiden, Nov. 2008 gives……. .
 In situ deposit characterisation: gel or freeze interface • Deposit Au at gel/organic interface: thickness (600 nm) • Approach (ii), deposit Au at L/L interface (org = acrylate and photo-initiator) = photocure interface. • (after Benkoski 2007, approach (i) [15]) • Aim: “freeze” structure of deposit – aggregate of ca 200 nm particles. Dryfe/Ho 2008 Leiden, Nov. 2008
In situ deposit characterisation: gel or freeze interface • Deposit Au at gel/organic interface: thickness (600 nm) • Approach (ii), deposit Au at L/L interface (org = acrylate and photo-initiator) = photocure interface. • (after Benkoski 2007, approach (i) [15]) • Aim: “freeze” structure of deposit – aggregate of ca 200 nm particles. Dryfe/Ho 2008 Leiden, Nov. 2008
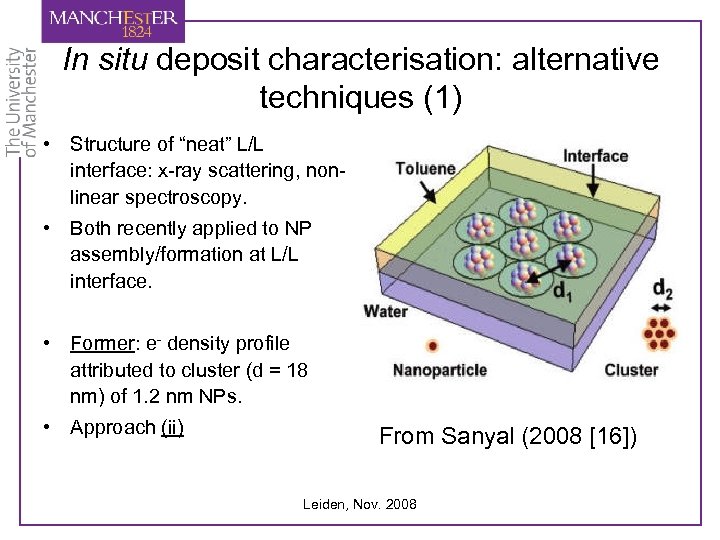 In situ deposit characterisation: alternative techniques (1) • Structure of “neat” L/L interface: x-ray scattering, nonlinear spectroscopy. • Both recently applied to NP assembly/formation at L/L interface. • Former: e- density profile attributed to cluster (d = 18 nm) of 1. 2 nm NPs. • Approach (ii) From Sanyal (2008 [16]) Leiden, Nov. 2008
In situ deposit characterisation: alternative techniques (1) • Structure of “neat” L/L interface: x-ray scattering, nonlinear spectroscopy. • Both recently applied to NP assembly/formation at L/L interface. • Former: e- density profile attributed to cluster (d = 18 nm) of 1. 2 nm NPs. • Approach (ii) From Sanyal (2008 [16]) Leiden, Nov. 2008
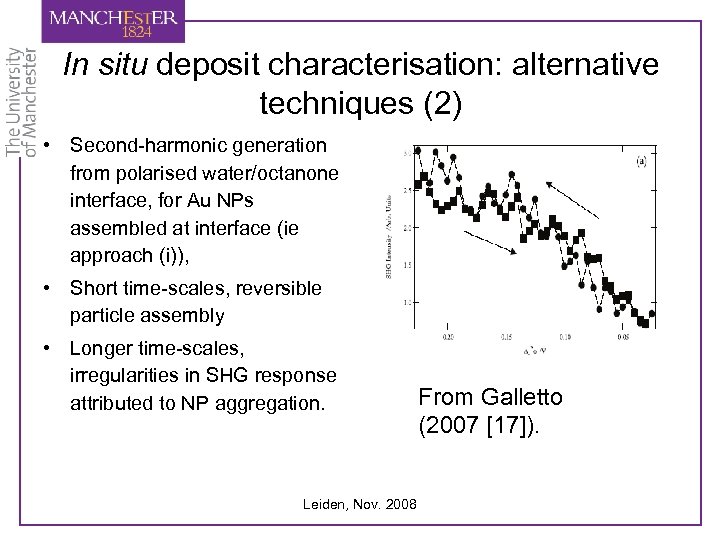 In situ deposit characterisation: alternative techniques (2) • Second-harmonic generation from polarised water/octanone interface, for Au NPs assembled at interface (ie approach (i)), • Short time-scales, reversible particle assembly • Longer time-scales, irregularities in SHG response attributed to NP aggregation. Leiden, Nov. 2008 From Galletto (2007 [17]).
In situ deposit characterisation: alternative techniques (2) • Second-harmonic generation from polarised water/octanone interface, for Au NPs assembled at interface (ie approach (i)), • Short time-scales, reversible particle assembly • Longer time-scales, irregularities in SHG response attributed to NP aggregation. Leiden, Nov. 2008 From Galletto (2007 [17]).
 (ii – b) In situ growth of particles at L/L interfaces: electrochemical reduction • Motivation: apply variable potential difference (4 -electrode methodology): • Study electrochemical growth in absence of solid substrate: – M. Guainazzi (1975 [18]) – Cu, Ag – Schiffrin/Kontturi, (1996 [19]) (Au, Pd) – Unwin, (2003, [20]) - (Ag) – Cunnane, (1998, . [21]) (polymers) – Dryfe, (2006, [22]) (review). • Advantage: Analysis of current response - information on growth. Leiden, Nov. 2008
(ii – b) In situ growth of particles at L/L interfaces: electrochemical reduction • Motivation: apply variable potential difference (4 -electrode methodology): • Study electrochemical growth in absence of solid substrate: – M. Guainazzi (1975 [18]) – Cu, Ag – Schiffrin/Kontturi, (1996 [19]) (Au, Pd) – Unwin, (2003, [20]) - (Ag) – Cunnane, (1998, . [21]) (polymers) – Dryfe, (2006, [22]) (review). • Advantage: Analysis of current response - information on growth. Leiden, Nov. 2008
 What is known at present? • Deposit “units” nm scale, adsorb, tend to aggregate. • (TEM of Pd, scale bar = 100 nm) • Replace single interface with array of micron scale (or smaller) interfaces = template. • g-alumina as template, 200 nm diameter pores (SEM of Pd, scale bar = 100 nm) Leiden, Nov. 2008
What is known at present? • Deposit “units” nm scale, adsorb, tend to aggregate. • (TEM of Pd, scale bar = 100 nm) • Replace single interface with array of micron scale (or smaller) interfaces = template. • g-alumina as template, 200 nm diameter pores (SEM of Pd, scale bar = 100 nm) Leiden, Nov. 2008
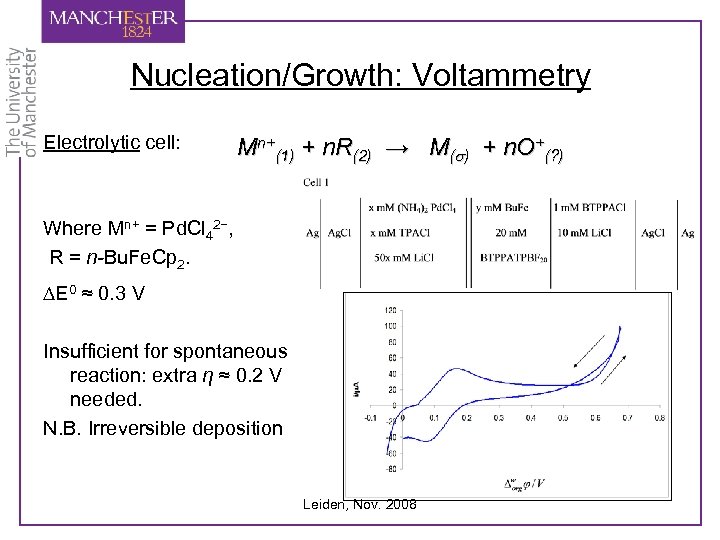 Nucleation/Growth: Voltammetry Electrolytic cell: Mn+(1) + n. R(2) → M(s) + n. O+(? ) Where Mn+ = Pd. Cl 42−, R = n-Bu. Fe. Cp 2. DE 0 ≈ 0. 3 V Insufficient for spontaneous reaction: extra η ≈ 0. 2 V needed. N. B. Irreversible deposition Leiden, Nov. 2008
Nucleation/Growth: Voltammetry Electrolytic cell: Mn+(1) + n. R(2) → M(s) + n. O+(? ) Where Mn+ = Pd. Cl 42−, R = n-Bu. Fe. Cp 2. DE 0 ≈ 0. 3 V Insufficient for spontaneous reaction: extra η ≈ 0. 2 V needed. N. B. Irreversible deposition Leiden, Nov. 2008
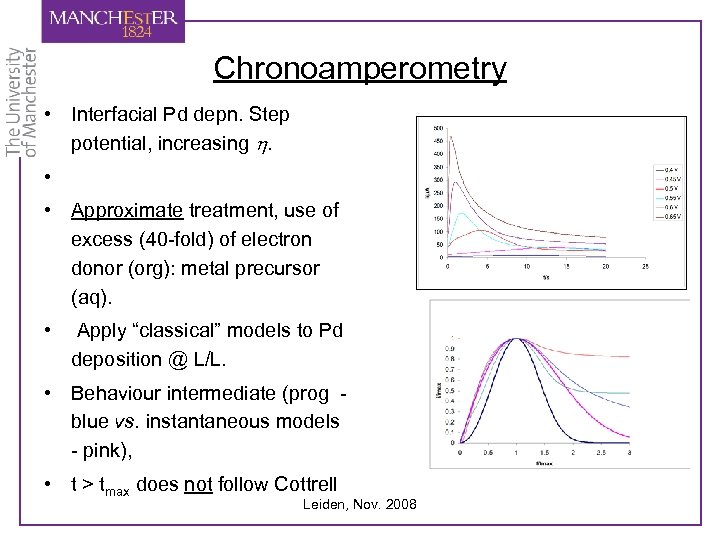 Chronoamperometry • Interfacial Pd depn. Step potential, increasing h. • • Approximate treatment, use of excess (40 -fold) of electron donor (org): metal precursor (aq). • Apply “classical” models to Pd deposition @ L/L. • Behaviour intermediate (prog blue vs. instantaneous models - pink), • t > tmax does not follow Cottrell Leiden, Nov. 2008
Chronoamperometry • Interfacial Pd depn. Step potential, increasing h. • • Approximate treatment, use of excess (40 -fold) of electron donor (org): metal precursor (aq). • Apply “classical” models to Pd deposition @ L/L. • Behaviour intermediate (prog blue vs. instantaneous models - pink), • t > tmax does not follow Cottrell Leiden, Nov. 2008
![Analysis of chronoamperometry • Heerman/Tarallo ≈ Mirkin/Nilov models [23, 24]: Applied overpotential/ V Nucleation Analysis of chronoamperometry • Heerman/Tarallo ≈ Mirkin/Nilov models [23, 24]: Applied overpotential/ V Nucleation](https://present5.com/presentation/dfcad89010dfa8c90a7d6374d5f3e98d/image-27.jpg) Analysis of chronoamperometry • Heerman/Tarallo ≈ Mirkin/Nilov models [23, 24]: Applied overpotential/ V Nucleation Rate constant/ s-1 Nucleation saturation density /cm-2 Diffusion Coefficient/ cm 2 s-1 [Bu. Fc] / m. M 0. 47 0. 29 10063 7. 6× 10 -6 20 0. 52 0. 64 11589 9. 8 × 10 -6 20 0. 57 0. 76 8349 2. 5 × 10 -5 20 0. 62 0. 54 11526 4. 1 × 10 -5 20 Leiden, Nov. 2008
Analysis of chronoamperometry • Heerman/Tarallo ≈ Mirkin/Nilov models [23, 24]: Applied overpotential/ V Nucleation Rate constant/ s-1 Nucleation saturation density /cm-2 Diffusion Coefficient/ cm 2 s-1 [Bu. Fc] / m. M 0. 47 0. 29 10063 7. 6× 10 -6 20 0. 52 0. 64 11589 9. 8 × 10 -6 20 0. 57 0. 76 8349 2. 5 × 10 -5 20 0. 62 0. 54 11526 4. 1 × 10 -5 20 Leiden, Nov. 2008
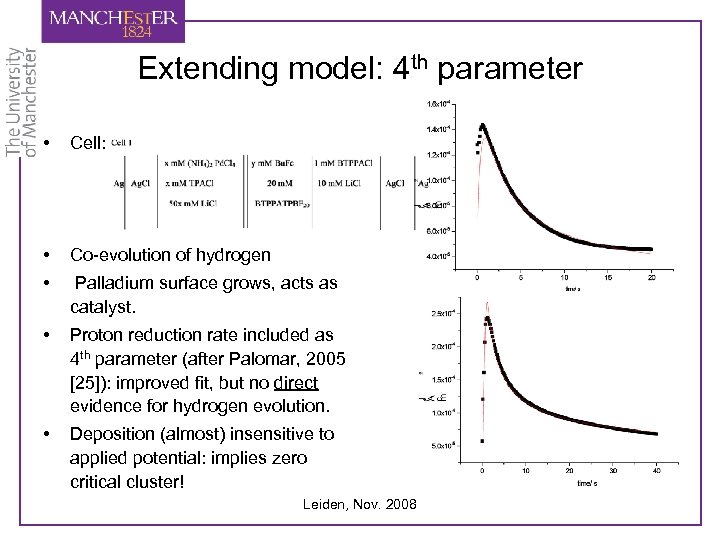 Extending model: 4 th parameter • Cell: • Co-evolution of hydrogen • Palladium surface grows, acts as catalyst. • Proton reduction rate included as 4 th parameter (after Palomar, 2005 [25]): improved fit, but no direct evidence for hydrogen evolution. • Deposition (almost) insensitive to applied potential: implies zero critical cluster! Leiden, Nov. 2008
Extending model: 4 th parameter • Cell: • Co-evolution of hydrogen • Palladium surface grows, acts as catalyst. • Proton reduction rate included as 4 th parameter (after Palomar, 2005 [25]): improved fit, but no direct evidence for hydrogen evolution. • Deposition (almost) insensitive to applied potential: implies zero critical cluster! Leiden, Nov. 2008
 Competitive reactions • p. H dependence of metal deposition? • However, ferrocene oxidation is coupled to H+ transfer (H 2 O 2 generation) • Nernst-Donnan equilibrium dictates interfacial potential, hence extent of H+ transfer. (from Su, Angew. Chem, 2008 [26]) Leiden, Nov. 2008
Competitive reactions • p. H dependence of metal deposition? • However, ferrocene oxidation is coupled to H+ transfer (H 2 O 2 generation) • Nernst-Donnan equilibrium dictates interfacial potential, hence extent of H+ transfer. (from Su, Angew. Chem, 2008 [26]) Leiden, Nov. 2008
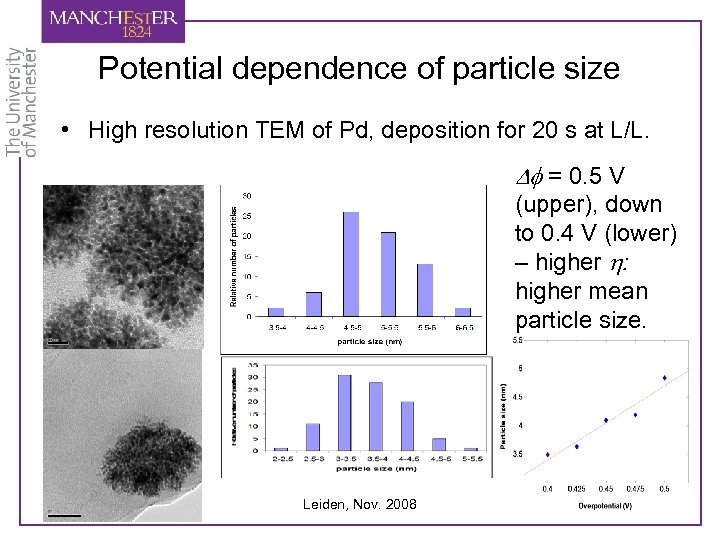 Potential dependence of particle size • High resolution TEM of Pd, deposition for 20 s at L/L. Df = 0. 5 V (upper), down to 0. 4 V (lower) – higher h: higher mean particle size. Leiden, Nov. 2008
Potential dependence of particle size • High resolution TEM of Pd, deposition for 20 s at L/L. Df = 0. 5 V (upper), down to 0. 4 V (lower) – higher h: higher mean particle size. Leiden, Nov. 2008
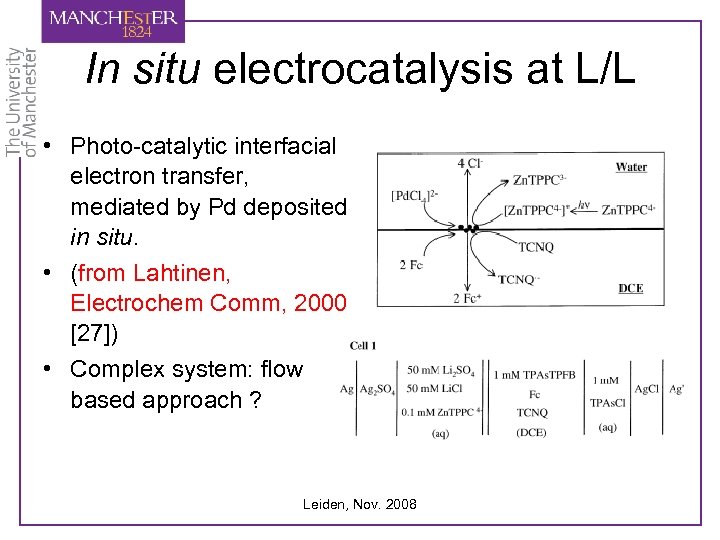 In situ electrocatalysis at L/L • Photo-catalytic interfacial electron transfer, mediated by Pd deposited in situ. • (from Lahtinen, Electrochem Comm, 2000 [27]) • Complex system: flow based approach ? Leiden, Nov. 2008
In situ electrocatalysis at L/L • Photo-catalytic interfacial electron transfer, mediated by Pd deposited in situ. • (from Lahtinen, Electrochem Comm, 2000 [27]) • Complex system: flow based approach ? Leiden, Nov. 2008
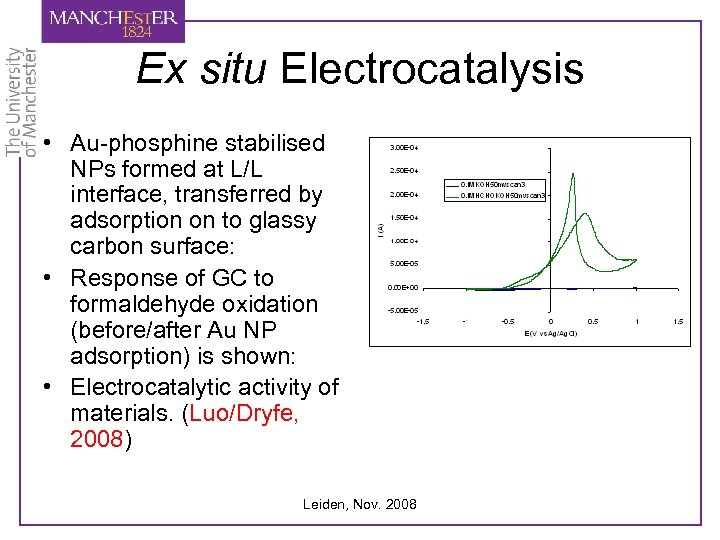 Ex situ Electrocatalysis • Au-phosphine stabilised NPs formed at L/L interface, transferred by adsorption on to glassy carbon surface: • Response of GC to formaldehyde oxidation (before/after Au NP adsorption) is shown: • Electrocatalytic activity of materials. (Luo/Dryfe, 2008) Leiden, Nov. 2008
Ex situ Electrocatalysis • Au-phosphine stabilised NPs formed at L/L interface, transferred by adsorption on to glassy carbon surface: • Response of GC to formaldehyde oxidation (before/after Au NP adsorption) is shown: • Electrocatalytic activity of materials. (Luo/Dryfe, 2008) Leiden, Nov. 2008
 Conclusions • L/L interface offers a ready “contact-less” route to the: • (i) assembly of (catalytically active) particles and • (ii) to the growth of (catalytically active) particles, the latter either by spontaneous or electrochemical approaches. • Issues - Deposit geometry conditions – Applicability of “classical” deposition models • - difficulty/lack of applicability of “standard” nano-scale characterisation techniques • Nano-scale morphology not dictated by strong substrate-deposit attraction but strong substrate(1)-substrate(2) repulsion. • Regularity of particle structure (before aggregation) – uniform flux to each particles? Leiden, Nov. 2008
Conclusions • L/L interface offers a ready “contact-less” route to the: • (i) assembly of (catalytically active) particles and • (ii) to the growth of (catalytically active) particles, the latter either by spontaneous or electrochemical approaches. • Issues - Deposit geometry conditions – Applicability of “classical” deposition models • - difficulty/lack of applicability of “standard” nano-scale characterisation techniques • Nano-scale morphology not dictated by strong substrate-deposit attraction but strong substrate(1)-substrate(2) repulsion. • Regularity of particle structure (before aggregation) – uniform flux to each particles? Leiden, Nov. 2008
 Suggestions for Future Work • Catalytic production of H 2 O 2 at the L/L interface • Photo-catalytic reduction (H 2, CO 2? ? ) at this interface • Does one of the phases have to be H 2 O? • Catalysis as fn(D , p) ? Leiden, Nov. 2008
Suggestions for Future Work • Catalytic production of H 2 O 2 at the L/L interface • Photo-catalytic reduction (H 2, CO 2? ? ) at this interface • Does one of the phases have to be H 2 O? • Catalysis as fn(D , p) ? Leiden, Nov. 2008
 References (1/2) • • • 1. V. J. Cunnane, D. J. Schiffrin, C. Beltran and G. Geblewicz, J. Electroanal. Chem. 247, 203 (1988). 2. S. N. Tan, R. A. W. Dryfe and H. H. Girault, Helv. Chim. Acta, 77, 231 (1994) 3. I. T. Horvath and J. Rabai, Science, 266, 72 (1994) 4. A. D. Ballantyne, A. K. Brisdon and R. A. W. Dryfe, Chem. Comm. , 4980. 5. D. M. Mitrinovic, A. M. Tikhonov, M. Li, Z. Q. Huang and M. L. Schlossman, Phys. Rev. Lett. 85, 582 (2000). 6. L. F. Scatena, M. G. Brown and G. L. Richmond, Science 292, 908 (2001). 7. Y. Lin, H. Skaff, T. Emrick, A. D. Dinsmore and T. P. Russell, Science, 299, 226 (2003). 8. F. Reincke, S. G. Hickey, W. K. Kegel, and D. Vanmaekelbergh, Angew Chem. Int. Ed. , 43, 458 (2004). 9. B. Su, J. P. Abid, D. J. Fermín, H. H. Girault, H. Hoffmannova, P. Krtil, Z. Samec, J. Amer. Chem. Soc. 126, 915 (2004). 10. M. G. Nikolaides, A. R. Bausch, M. F. Hsu, A. D. Dinsmore, M. P. Brenner, C. Gay and D. A. Weitz, Nature 420, 299 (2002). 11. M. Faraday, Philos. Trans. I, 147, 145 (1857). 12. M. Brust, M. Walker, D. J. Schiffrin and R. Whyman, J. Chem. Soc. Chem. Comm. , 801 (1994). 13. W. W. Weare, S. M. Reed, M. G. Warner and J. E. Hutchison, J. Amer. Chem. Soc. , 122, 12890 (2000). Leiden, Nov. 2008
References (1/2) • • • 1. V. J. Cunnane, D. J. Schiffrin, C. Beltran and G. Geblewicz, J. Electroanal. Chem. 247, 203 (1988). 2. S. N. Tan, R. A. W. Dryfe and H. H. Girault, Helv. Chim. Acta, 77, 231 (1994) 3. I. T. Horvath and J. Rabai, Science, 266, 72 (1994) 4. A. D. Ballantyne, A. K. Brisdon and R. A. W. Dryfe, Chem. Comm. , 4980. 5. D. M. Mitrinovic, A. M. Tikhonov, M. Li, Z. Q. Huang and M. L. Schlossman, Phys. Rev. Lett. 85, 582 (2000). 6. L. F. Scatena, M. G. Brown and G. L. Richmond, Science 292, 908 (2001). 7. Y. Lin, H. Skaff, T. Emrick, A. D. Dinsmore and T. P. Russell, Science, 299, 226 (2003). 8. F. Reincke, S. G. Hickey, W. K. Kegel, and D. Vanmaekelbergh, Angew Chem. Int. Ed. , 43, 458 (2004). 9. B. Su, J. P. Abid, D. J. Fermín, H. H. Girault, H. Hoffmannova, P. Krtil, Z. Samec, J. Amer. Chem. Soc. 126, 915 (2004). 10. M. G. Nikolaides, A. R. Bausch, M. F. Hsu, A. D. Dinsmore, M. P. Brenner, C. Gay and D. A. Weitz, Nature 420, 299 (2002). 11. M. Faraday, Philos. Trans. I, 147, 145 (1857). 12. M. Brust, M. Walker, D. J. Schiffrin and R. Whyman, J. Chem. Soc. Chem. Comm. , 801 (1994). 13. W. W. Weare, S. M. Reed, M. G. Warner and J. E. Hutchison, J. Amer. Chem. Soc. , 122, 12890 (2000). Leiden, Nov. 2008
 References (2/2) • • • • 14. C. N. R. Rao, G. U. Kulkarni, P. J. Thomas, V. V. Agrawal and P. Saravanan, J. Phys. Chem. B, 107, 7391 (2003). 15. J. J. Benkoski, R. L. Jones, J. F. Douglas and A. Karim, Langmuir, 23, 3530 (2007). 16. M. J. Sanyal, V. V. Agrawal, M. K. Bera, K. P. Kalyanikutty, J. Daillant, C. Blot, S. Kubowicz, O. Komovalov and C. N. R. Rao, J. Phys. Chem. C, 112, 1739 (2008). 17. P. Galletto, H. H. Girault, C. Gomis-Bas, D. J. Schiffrin, R. Antoine, M. Broyer and P. F. Brevet, J. Phys. Cond. Matt. 19, 375108 (2007). 18 M. Guainazzi, G. Silvestri and G. Serravalle, J. Chem. Soc. Chem. Commun. , 200 (1975). 19 Y. Cheng and D. J. Schiffrin, J. Chem. Soc. Farad. Trans. , 92, 3865 (1996). 20. J. D. Guo, T. Tokimoto, R. Othman and P. R. Unwin, Electrochem. Comm. , 5, 1005 (2003). 21 V. J. Cunnane and U Evans, Chem. Comm. , 2163 (1998). 22. R. A. W. Dryfe, Phys. Chem. Phys. 8, 1869, (2006). 23. L. Heermann and M. Tarallo, J. Electroanal. Chem. , 470, 70 (1999). 24. M. V. Mirkin and E. Nilov, J. Electroanal. Chem. , 283, 35 (1990). 25. M. Palomar-Pardave, B. R. Scharifker, E. M. Arce and M. Romero-Romo, Electrochim. Acta, 50, 4736 (2005). 26. B. Su, R. Partovi Nia, F. Li, M. Hojeij, M. Prudent, C. Corminboeuf, Z. Samec and H. H. Girault, Angew. Chem. Int. Ed. , 47, 4675 (2008). 27. R. M. Lahtinen, D. J. Fermín, H. Jensen, K. Kontturi and H. H. Girault, Electrochem. Comm. 2, 230 (2000). Leiden, Nov. 2008
References (2/2) • • • • 14. C. N. R. Rao, G. U. Kulkarni, P. J. Thomas, V. V. Agrawal and P. Saravanan, J. Phys. Chem. B, 107, 7391 (2003). 15. J. J. Benkoski, R. L. Jones, J. F. Douglas and A. Karim, Langmuir, 23, 3530 (2007). 16. M. J. Sanyal, V. V. Agrawal, M. K. Bera, K. P. Kalyanikutty, J. Daillant, C. Blot, S. Kubowicz, O. Komovalov and C. N. R. Rao, J. Phys. Chem. C, 112, 1739 (2008). 17. P. Galletto, H. H. Girault, C. Gomis-Bas, D. J. Schiffrin, R. Antoine, M. Broyer and P. F. Brevet, J. Phys. Cond. Matt. 19, 375108 (2007). 18 M. Guainazzi, G. Silvestri and G. Serravalle, J. Chem. Soc. Chem. Commun. , 200 (1975). 19 Y. Cheng and D. J. Schiffrin, J. Chem. Soc. Farad. Trans. , 92, 3865 (1996). 20. J. D. Guo, T. Tokimoto, R. Othman and P. R. Unwin, Electrochem. Comm. , 5, 1005 (2003). 21 V. J. Cunnane and U Evans, Chem. Comm. , 2163 (1998). 22. R. A. W. Dryfe, Phys. Chem. Phys. 8, 1869, (2006). 23. L. Heermann and M. Tarallo, J. Electroanal. Chem. , 470, 70 (1999). 24. M. V. Mirkin and E. Nilov, J. Electroanal. Chem. , 283, 35 (1990). 25. M. Palomar-Pardave, B. R. Scharifker, E. M. Arce and M. Romero-Romo, Electrochim. Acta, 50, 4736 (2005). 26. B. Su, R. Partovi Nia, F. Li, M. Hojeij, M. Prudent, C. Corminboeuf, Z. Samec and H. H. Girault, Angew. Chem. Int. Ed. , 47, 4675 (2008). 27. R. M. Lahtinen, D. J. Fermín, H. Jensen, K. Kontturi and H. H. Girault, Electrochem. Comm. 2, 230 (2000). Leiden, Nov. 2008


