57adb3e01c7b65c100e37f17dc4d1091.ppt
- Количество слайдов: 20
 Efficacy of Prevention of Mother to Child Transmission (PMTCT) of HIV in Rivers State, Nigeria By IBINABO ANITA PORBENI, MD, MSPH Co- Authors: Dr. Emmanuel Keku (MD, MA, MSPH, PHD) Ms. Jackie Lloyd (MS) Dr. Richard Dicker (MD, MS) Dr. William Sawyer (PHD) Dr. Cecilia Hegamin. Younger (PHD)
Efficacy of Prevention of Mother to Child Transmission (PMTCT) of HIV in Rivers State, Nigeria By IBINABO ANITA PORBENI, MD, MSPH Co- Authors: Dr. Emmanuel Keku (MD, MA, MSPH, PHD) Ms. Jackie Lloyd (MS) Dr. Richard Dicker (MD, MS) Dr. William Sawyer (PHD) Dr. Cecilia Hegamin. Younger (PHD)
 HIV in Nigeria; Prevalence of 4. 6%, Rivers State, 7. 3% from http: //www. livingtongues. org/dcs/Baan. pdf http: //www. prisonpastor. com/images/the_new_map_of_nigeria. jpg &
HIV in Nigeria; Prevalence of 4. 6%, Rivers State, 7. 3% from http: //www. livingtongues. org/dcs/Baan. pdf http: //www. prisonpastor. com/images/the_new_map_of_nigeria. jpg &
 Background and Literature Review Ø Rivers state chosen because of high Prevalence of 7. 3% compared with National prevalence of 4. 6% and presence of facilities that carry out PMTCT interventions Ø Evidence shows that effective large scale Prevention of Mother to Child Transmission (PMTCT) of HIV programs can be implemented in settings with limited resources Ø That 15% of new HIV infections each year are caused by mother-to-child-transmission (MTCT) also called Vertical transmission Ø Eliminating HIV infection in infants and young children will accelerate global prevention initiatives United Nations General Assembly Special Session (UNGASS) 2001
Background and Literature Review Ø Rivers state chosen because of high Prevalence of 7. 3% compared with National prevalence of 4. 6% and presence of facilities that carry out PMTCT interventions Ø Evidence shows that effective large scale Prevention of Mother to Child Transmission (PMTCT) of HIV programs can be implemented in settings with limited resources Ø That 15% of new HIV infections each year are caused by mother-to-child-transmission (MTCT) also called Vertical transmission Ø Eliminating HIV infection in infants and young children will accelerate global prevention initiatives United Nations General Assembly Special Session (UNGASS) 2001
 Objectives To clearly outline the different interventions being carried out Ø To see if the interventions are really reducing vertical transmission of HIV/AIDS Ø Ø To see if there any significant differences between interventions Ø To ascertain if the outcomes of interventions are comparable to those being carried out in more developed countries in terms of reducing transmission to as little as 2%. Ø To make recommendations on possible improvements of interventions if applicable and elucidate areas of further study
Objectives To clearly outline the different interventions being carried out Ø To see if the interventions are really reducing vertical transmission of HIV/AIDS Ø Ø To see if there any significant differences between interventions Ø To ascertain if the outcomes of interventions are comparable to those being carried out in more developed countries in terms of reducing transmission to as little as 2%. Ø To make recommendations on possible improvements of interventions if applicable and elucidate areas of further study
 Research Question Ø Do the interventions carried out under Prevention of Mother to Child transmission of HIV (PMTCT) in any way reduce the incidence of vertical transmission of HIV 1 to children in Rivers State and if they do which interventions are the most effective?
Research Question Ø Do the interventions carried out under Prevention of Mother to Child transmission of HIV (PMTCT) in any way reduce the incidence of vertical transmission of HIV 1 to children in Rivers State and if they do which interventions are the most effective?
 Hypothesis I Ø Hypothesis I H 0 The HIV status of children born to women living with HIV is solely due to chance whether they had any intervention or not outcome is the same. HA The HIV status of children born to women living with HIV is dependent on their having been given some form of ARV Prophylaxis Ø Hypothesis II H 0 The HIV status of children born to women living with HIV is independent of the type and time of ARV prophylaxis given HA The HIV status of children born to women living with HIV is dependent on the type and time of ARV Prophylaxis initiated
Hypothesis I Ø Hypothesis I H 0 The HIV status of children born to women living with HIV is solely due to chance whether they had any intervention or not outcome is the same. HA The HIV status of children born to women living with HIV is dependent on their having been given some form of ARV Prophylaxis Ø Hypothesis II H 0 The HIV status of children born to women living with HIV is independent of the type and time of ARV prophylaxis given HA The HIV status of children born to women living with HIV is dependent on the type and time of ARV Prophylaxis initiated
 Hypothesis III Ø H 0 The HIV status of children born to women living with HIV is independent of the mothers HIV treatment history Ø HA The HIV status of children born to women living with HIV is dependent on if the mother has been on HAART before pregnancy or if she had any ARV Prophylaxis.
Hypothesis III Ø H 0 The HIV status of children born to women living with HIV is independent of the mothers HIV treatment history Ø HA The HIV status of children born to women living with HIV is dependent on if the mother has been on HAART before pregnancy or if she had any ARV Prophylaxis.
 Methods and Population Quasi experimental study with Participants drawn from Program reports and Hospital registers. Women were contacted through their HIV support groups consent sought. Ø The study population is the pregnant women living with HIV and the children born from the pregnancy that went through PMTCT interventions or had access to PMTCT services in three local government areas in Rivers State. Ø
Methods and Population Quasi experimental study with Participants drawn from Program reports and Hospital registers. Women were contacted through their HIV support groups consent sought. Ø The study population is the pregnant women living with HIV and the children born from the pregnancy that went through PMTCT interventions or had access to PMTCT services in three local government areas in Rivers State. Ø
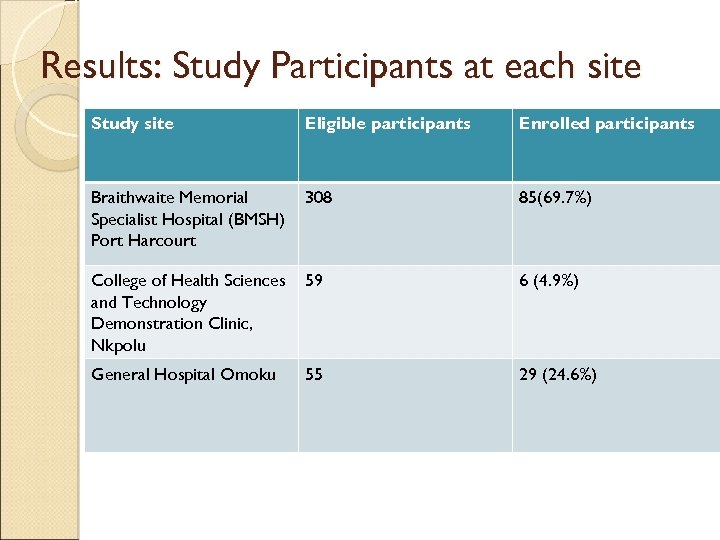 Results: Study Participants at each site Study site Eligible participants Enrolled participants Braithwaite Memorial Specialist Hospital (BMSH) Port Harcourt 308 85(69. 7%) College of Health Sciences and Technology Demonstration Clinic, Nkpolu 59 6 (4. 9%) General Hospital Omoku 55 29 (24. 6%)
Results: Study Participants at each site Study site Eligible participants Enrolled participants Braithwaite Memorial Specialist Hospital (BMSH) Port Harcourt 308 85(69. 7%) College of Health Sciences and Technology Demonstration Clinic, Nkpolu 59 6 (4. 9%) General Hospital Omoku 55 29 (24. 6%)
 Demographics; age of mothers in years and infants in weeks Mean age distribution Median age distribution Modal age 28. 4 28. 5 28 (17. 1%) Range Standard deviation Variance 22 -37 years 3. 164 10. 012 Mean age of children Median Mode 16. 76 17 6 (28. 6%) Range Standard deviation Variance 3 (0. 8%)- 104 (1. 7%) 19. 43 377. 61
Demographics; age of mothers in years and infants in weeks Mean age distribution Median age distribution Modal age 28. 4 28. 5 28 (17. 1%) Range Standard deviation Variance 22 -37 years 3. 164 10. 012 Mean age of children Median Mode 16. 76 17 6 (28. 6%) Range Standard deviation Variance 3 (0. 8%)- 104 (1. 7%) 19. 43 377. 61
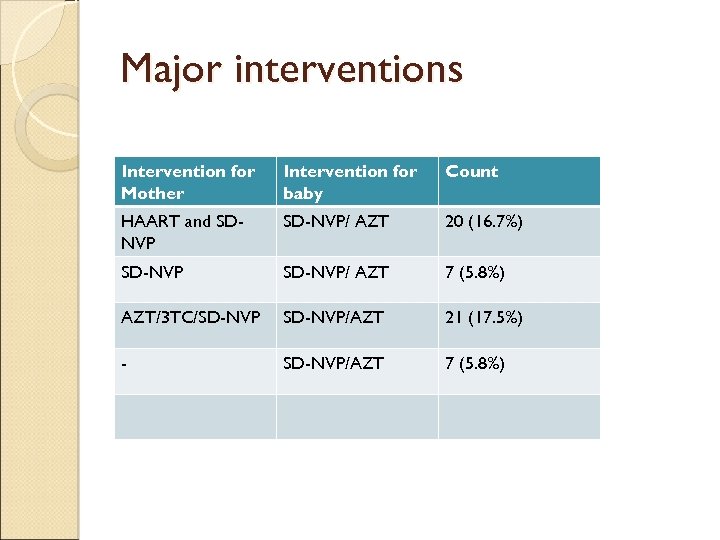 Major interventions Intervention for Mother Intervention for baby Count HAART and SDNVP SD-NVP/ AZT 20 (16. 7%) SD-NVP/ AZT 7 (5. 8%) AZT/3 TC/SD-NVP/AZT 21 (17. 5%) - SD-NVP/AZT 7 (5. 8%)
Major interventions Intervention for Mother Intervention for baby Count HAART and SDNVP SD-NVP/ AZT 20 (16. 7%) SD-NVP/ AZT 7 (5. 8%) AZT/3 TC/SD-NVP/AZT 21 (17. 5%) - SD-NVP/AZT 7 (5. 8%)
 Effect of Interventions on vertical transmission Classification Count Transmission rate Any intervention 60 (50%) 3 (5% ) CL (1. 3%-13%) No intervention 60 (50%) 13 (21. 7%) CL (12. 6%33. 4%)
Effect of Interventions on vertical transmission Classification Count Transmission rate Any intervention 60 (50%) 3 (5% ) CL (1. 3%-13%) No intervention 60 (50%) 13 (21. 7%) CL (12. 6%33. 4%)
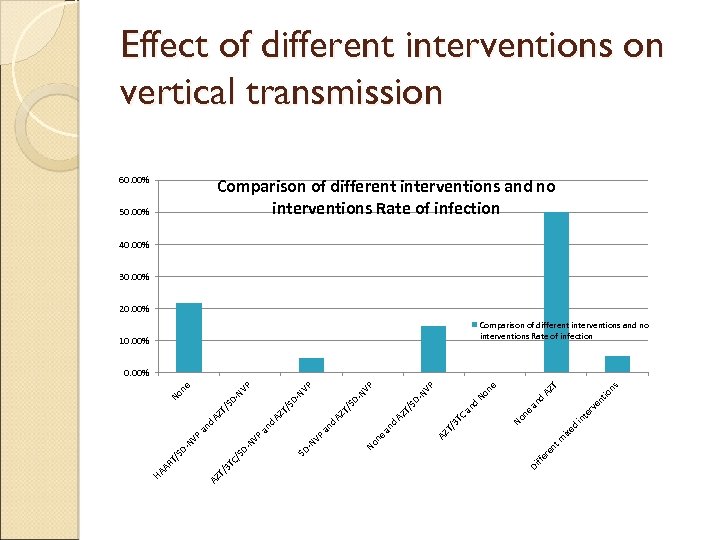 Effect of different interventions on vertical transmission 60. 00% Comparison of different interventions and no interventions Rate of infection 50. 00% 40. 00% 30. 00% 20. 00% Comparison of different interventions and no interventions Rate of infection 10. 00% ns T tio AZ en rv ixe d No in te ne d an m AZ T/ 3 TC AZ an d Di ffe re nt No ne an d No DNV T/ S SD T/ AZ d an P ne P P -N V VP -N SD -N V -N SD TC / T/ 3 AZ HA A RT /S DNV VP P an an d d AZ AZ T/ T/ S SD D- No NV ne P 0. 00%
Effect of different interventions on vertical transmission 60. 00% Comparison of different interventions and no interventions Rate of infection 50. 00% 40. 00% 30. 00% 20. 00% Comparison of different interventions and no interventions Rate of infection 10. 00% ns T tio AZ en rv ixe d No in te ne d an m AZ T/ 3 TC AZ an d Di ffe re nt No ne an d No DNV T/ S SD T/ AZ d an P ne P P -N V VP -N SD -N V -N SD TC / T/ 3 AZ HA A RT /S DNV VP P an an d d AZ AZ T/ T/ S SD D- No NV ne P 0. 00%
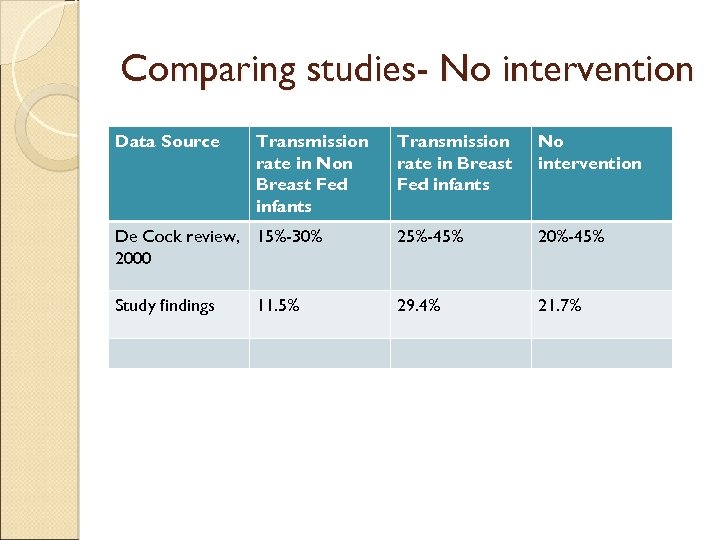 Comparing studies- No intervention Data Source Transmission rate in Non Breast Fed infants Transmission rate in Breast Fed infants No intervention De Cock review, 15%-30% 2000 25%-45% 20%-45% Study findings 29. 4% 21. 7% 11. 5%
Comparing studies- No intervention Data Source Transmission rate in Non Breast Fed infants Transmission rate in Breast Fed infants No intervention De Cock review, 15%-30% 2000 25%-45% 20%-45% Study findings 29. 4% 21. 7% 11. 5%
 Discussion- Highlights Higher rates of transmission amongst lower age groups (p value 0. 000047) More female children were positive 19. 7% as compared with males 7% (RR 2. 8, 95% CI 0. 9 -8. 4 and p value 0. 052). Ø Hypothesis I ü No intervention arm had infants 4. 3 times more likely to be HIV positive than those who had any form of pharmaceutical intervention, (P- value 0. 007)
Discussion- Highlights Higher rates of transmission amongst lower age groups (p value 0. 000047) More female children were positive 19. 7% as compared with males 7% (RR 2. 8, 95% CI 0. 9 -8. 4 and p value 0. 052). Ø Hypothesis I ü No intervention arm had infants 4. 3 times more likely to be HIV positive than those who had any form of pharmaceutical intervention, (P- value 0. 007)
 Discussion: Hypothesis testing; I and II Hypothesis II Ø No significant difference between women who had HAART which Ø predates pregnancy and interventions given within pregnancy and at delivery and after, (P value 0. 21). HAART and SD-NVP for both and AZT had comparable outcomes. Sample size may play a role. Ø Hypothesis III: Ø There was a significant difference between woman who had interventions HAART and short course ARV even if their children had none and women who had none even if their children had, (pvalue 0. 0018). Ø A woman’s treatment history has a lot to do with HIV transmission outcome in her child.
Discussion: Hypothesis testing; I and II Hypothesis II Ø No significant difference between women who had HAART which Ø predates pregnancy and interventions given within pregnancy and at delivery and after, (P value 0. 21). HAART and SD-NVP for both and AZT had comparable outcomes. Sample size may play a role. Ø Hypothesis III: Ø There was a significant difference between woman who had interventions HAART and short course ARV even if their children had none and women who had none even if their children had, (pvalue 0. 0018). Ø A woman’s treatment history has a lot to do with HIV transmission outcome in her child.
 Conclusion and Recommendations Ø PMTCT works and should be offered in every area where ANC is carried out. Ø Even the simplest intervention significantly reduces vertical transmission and should be provided. Ø More research is needed in this area, especially regarding effect of couple counseling and support groups on participant retention in PMTCT programs.
Conclusion and Recommendations Ø PMTCT works and should be offered in every area where ANC is carried out. Ø Even the simplest intervention significantly reduces vertical transmission and should be provided. Ø More research is needed in this area, especially regarding effect of couple counseling and support groups on participant retention in PMTCT programs.
 Contact Details Ibinabo Anita Porbeni- poribi @sgu. edu, ibporbeni@yahoo. com, 6097167359 Emmanuel Keku, ekeku@sgu. edu
Contact Details Ibinabo Anita Porbeni- poribi @sgu. edu, ibporbeni@yahoo. com, 6097167359 Emmanuel Keku, ekeku@sgu. edu
 References AIDS Program Initiative in Nigeria (APIN); www. apin. harvard. edu/images/photo-pmtct. jpg Centers for Disease Control and Prevention (CDC), Division of HIV/AIDS Prevention, National Center HIV/AIDS, Viral Hepatitis, STD and TB Prevention. “One Test, Two Lives. ” Internet webpage http: //www. cdc. gov/hiv/topics/perinatal/1 test 2 lives/ retrieved on the 2 nd of March, 2009 C. I. A World Fact Book (Retrieved March 7 th, 2009) from https: //www. cia. gov/library/publications/the-world-factbook/geos/ni. html De Cock et al 2000, Prevention of mother-to-child HIV transmission in resource-poor countries: translating research into policy and practice. JAMA 2000, 283: 1175 -11826. Federal Republic of Nigeria, 2006 Population Census, Nigeria, Official Gazette (FGP 71/52007/2, 500(OL 24): Legal Notice on Publication of the Details of the Breakdown of the National and State Provisional Totals 2006 Census, http: //www. nigerianstat. gov. ng/Connections/Pop 2006. pdf Federal Ministry of Health (2007); A Technical report on the 2007 HIV/Syphilis sentinel Survey among pregnant women attending antenatal clinics in Nigeria, Nigeria: FMOH. Galadanci Hadiza S, Zubairu Iliyasu, Jamilu Tukur, Mariya Muktar-Yola, Solomon I Adeleke (2008), Uptake of voluntary counseling and testing for HIV by pregnant women in a prevention-of-mother-to-child-transmissio Kroon M and Eley B (2007), Appendix 5: Pediatric HIV in the Western Cape, The Burden of Disease Project, Decreasing the Burden of disease , Final Report, 2007 Leroy V et al 2002, Twenty-four month efficacy of a maternal short-course zidovudine regimen to prevent mother-to-child transmission of HIV-1 in West-Africa. AIDS 2002, 16: 631 -641 Map of Rivers State downloaded from http: //www. livingtongues. org/docs/Baan. pdf, On 25 th March, 2009 Map on Nigeria; http: //www. prisonpastor. com/images/the_new_map_of_nigeria. jp Nigeria Direct, 2005 retrieved on the 7 th of March, 2009 from http: //nigeria. gov. ng/. Nigerian National Population Commission (2006), National census report. Odutolu O, Davies A, Fatusi A, Okonkwo P, Ejembi C (2004, July); 15 th International Conference on AIDS (2004): Bangkok, Thailand). Policy options for the Nigerian PMTCT program. Int. 'l Conf AIDS. 2004 Jul 11 -16; 15: April, 2002; Center for AIDS Prevention Studies (CAPS), Department of Pediatric Immunology, University of California at San Francisco (UCSF), AIDS Research Institute (ARI) River State Ministry of Health (2007) Proposal on “The Acceleration of Prevention in the context of Scale Up”, 2007. Rivers State of Nigeria Government. All rights reserved, downloaded from http: //www. riversstatenigeria. net/index. php? option=com_content&view=article&id=276&Itemid=168 on 25 th March, 2009.
References AIDS Program Initiative in Nigeria (APIN); www. apin. harvard. edu/images/photo-pmtct. jpg Centers for Disease Control and Prevention (CDC), Division of HIV/AIDS Prevention, National Center HIV/AIDS, Viral Hepatitis, STD and TB Prevention. “One Test, Two Lives. ” Internet webpage http: //www. cdc. gov/hiv/topics/perinatal/1 test 2 lives/ retrieved on the 2 nd of March, 2009 C. I. A World Fact Book (Retrieved March 7 th, 2009) from https: //www. cia. gov/library/publications/the-world-factbook/geos/ni. html De Cock et al 2000, Prevention of mother-to-child HIV transmission in resource-poor countries: translating research into policy and practice. JAMA 2000, 283: 1175 -11826. Federal Republic of Nigeria, 2006 Population Census, Nigeria, Official Gazette (FGP 71/52007/2, 500(OL 24): Legal Notice on Publication of the Details of the Breakdown of the National and State Provisional Totals 2006 Census, http: //www. nigerianstat. gov. ng/Connections/Pop 2006. pdf Federal Ministry of Health (2007); A Technical report on the 2007 HIV/Syphilis sentinel Survey among pregnant women attending antenatal clinics in Nigeria, Nigeria: FMOH. Galadanci Hadiza S, Zubairu Iliyasu, Jamilu Tukur, Mariya Muktar-Yola, Solomon I Adeleke (2008), Uptake of voluntary counseling and testing for HIV by pregnant women in a prevention-of-mother-to-child-transmissio Kroon M and Eley B (2007), Appendix 5: Pediatric HIV in the Western Cape, The Burden of Disease Project, Decreasing the Burden of disease , Final Report, 2007 Leroy V et al 2002, Twenty-four month efficacy of a maternal short-course zidovudine regimen to prevent mother-to-child transmission of HIV-1 in West-Africa. AIDS 2002, 16: 631 -641 Map of Rivers State downloaded from http: //www. livingtongues. org/docs/Baan. pdf, On 25 th March, 2009 Map on Nigeria; http: //www. prisonpastor. com/images/the_new_map_of_nigeria. jp Nigeria Direct, 2005 retrieved on the 7 th of March, 2009 from http: //nigeria. gov. ng/. Nigerian National Population Commission (2006), National census report. Odutolu O, Davies A, Fatusi A, Okonkwo P, Ejembi C (2004, July); 15 th International Conference on AIDS (2004): Bangkok, Thailand). Policy options for the Nigerian PMTCT program. Int. 'l Conf AIDS. 2004 Jul 11 -16; 15: April, 2002; Center for AIDS Prevention Studies (CAPS), Department of Pediatric Immunology, University of California at San Francisco (UCSF), AIDS Research Institute (ARI) River State Ministry of Health (2007) Proposal on “The Acceleration of Prevention in the context of Scale Up”, 2007. Rivers State of Nigeria Government. All rights reserved, downloaded from http: //www. riversstatenigeria. net/index. php? option=com_content&view=article&id=276&Itemid=168 on 25 th March, 2009.
 References Sherman, Gayle G, 2005, Dried Blood Spots Improve Access to HIV Diagnosis and Care for Infants in Low-Resource Settings. JAIDS Journal of Acquired Immune Deficiency Syndromes. 38(5): 615 -617, April 15, 2005. Suksomboon N, Poolsup N, Ket-Aim S ( 2007, June): Systematic review of the Efficacy of antiretroviral therapies for reducing the risk of mother-to-child Transmission of HIV infection. J Clin Pharm Ther. ; 32 (3) 293 -31 Taha TE, Kumwenda NI, Gibbons A, Broadhead RL, Fiscus S, Lema V, Liomba G, Nkhoma C, Miotti PG, Hoover DR. ( 2003, October, 11) Lancet. ; 362(9391): 1171 -Short post exposure prophylaxis in newborn babies to reduce mother-to-child transmission of HIV-1: NVAZ randomized clinical trial. Taha E. Taha, Samah Nour, Newton I. Kumwenda, Robin L. Broadhead, Susan A. Fiscus, George Kafula, Chiwawa Nkhoma, Shu Chen and Donald R. Hoover (2005) “Gender Differences in Perinatal HIV Acquisition Among African Infants” Pediatrics 2005; 115; e 167 -e 172 DOI: 10. 1542/peds. 20041590 Thior I, Lockman S, Smeaton LM, Shapiro RL, Wester C, Heymann SJ, Gilbert PB, Stevens L, Peter T, Kim S, van Widenfelt E, Moffat C, Ndase P, Arimi P, Kebaabetswe P, Mazonde P, Makhema J, Mc. Intosh K, Novitsky V, Lee TH, Marlink R, Lagakos S, Essex M; Mashi Study Team (2006 August 16) Tonwe-Gold B, Ekouevi D. K, Viho I, Amani-Bosse C, Toure S, Coffie P. A, Rouet F, Becquet R, Leroy V, El Sadr W. M, Abrams E. J, Dabis F (2007), Antiretroviral treatment and prevention of peripartum and postpartum HIV transmission in West Africa: Evaluation of a two- tiered approach PLo. S Med, 2007 Aug 21; 4 (8): e 257 2008 -09 United Nations Development Project (UNDP), 2007/2008 Human Development UNAIDS/WHO, 2008 and 2009 AIDS Epidemic Update. United Nations General Assembly Special Session (UNGASS) 2001 Report, Country Fact Sheet on Nigeria retrieved on the 7 th of March, 2009 from http: //hdrstats. undp. org/countries/country_fact_sheets/cty_fs_NGA. html Volmink J, Siegfried NL, Van der Merwe L, Brocklehurst P. (2002). Antiretrovirals for Review, updated 2007; (1): CD 003510 reducing the risk of mother-to-child transmission of HIV infection, Cochrane data base Sys. World Health Organization/Joint United Nations on AIDS (UNAIDS, 1999) : HIV in Pregnancy: A Review. 67 p World Health Organization, (2006) WHO Case Definitions of HIV for Surveillance And Revised Clinical Staging and Immunological Classification of HIV-related disease in Adults and Children, August 7, 2006. Downloaded from http: //www. womenchildrenhiv. org/wchiv? page=charts-00 -01 on 4 th of April, 2009. WHO, 2006 Antiretroviral Drugs for Treating Pregnant Women and Preventing HIV Infection in Infants in resource Limited Settings: Towards Universal Access, Recommendations for a public health approach 2006 version WHO/ UNAIDS: AIDS Epidemic update, December, 2007
References Sherman, Gayle G, 2005, Dried Blood Spots Improve Access to HIV Diagnosis and Care for Infants in Low-Resource Settings. JAIDS Journal of Acquired Immune Deficiency Syndromes. 38(5): 615 -617, April 15, 2005. Suksomboon N, Poolsup N, Ket-Aim S ( 2007, June): Systematic review of the Efficacy of antiretroviral therapies for reducing the risk of mother-to-child Transmission of HIV infection. J Clin Pharm Ther. ; 32 (3) 293 -31 Taha TE, Kumwenda NI, Gibbons A, Broadhead RL, Fiscus S, Lema V, Liomba G, Nkhoma C, Miotti PG, Hoover DR. ( 2003, October, 11) Lancet. ; 362(9391): 1171 -Short post exposure prophylaxis in newborn babies to reduce mother-to-child transmission of HIV-1: NVAZ randomized clinical trial. Taha E. Taha, Samah Nour, Newton I. Kumwenda, Robin L. Broadhead, Susan A. Fiscus, George Kafula, Chiwawa Nkhoma, Shu Chen and Donald R. Hoover (2005) “Gender Differences in Perinatal HIV Acquisition Among African Infants” Pediatrics 2005; 115; e 167 -e 172 DOI: 10. 1542/peds. 20041590 Thior I, Lockman S, Smeaton LM, Shapiro RL, Wester C, Heymann SJ, Gilbert PB, Stevens L, Peter T, Kim S, van Widenfelt E, Moffat C, Ndase P, Arimi P, Kebaabetswe P, Mazonde P, Makhema J, Mc. Intosh K, Novitsky V, Lee TH, Marlink R, Lagakos S, Essex M; Mashi Study Team (2006 August 16) Tonwe-Gold B, Ekouevi D. K, Viho I, Amani-Bosse C, Toure S, Coffie P. A, Rouet F, Becquet R, Leroy V, El Sadr W. M, Abrams E. J, Dabis F (2007), Antiretroviral treatment and prevention of peripartum and postpartum HIV transmission in West Africa: Evaluation of a two- tiered approach PLo. S Med, 2007 Aug 21; 4 (8): e 257 2008 -09 United Nations Development Project (UNDP), 2007/2008 Human Development UNAIDS/WHO, 2008 and 2009 AIDS Epidemic Update. United Nations General Assembly Special Session (UNGASS) 2001 Report, Country Fact Sheet on Nigeria retrieved on the 7 th of March, 2009 from http: //hdrstats. undp. org/countries/country_fact_sheets/cty_fs_NGA. html Volmink J, Siegfried NL, Van der Merwe L, Brocklehurst P. (2002). Antiretrovirals for Review, updated 2007; (1): CD 003510 reducing the risk of mother-to-child transmission of HIV infection, Cochrane data base Sys. World Health Organization/Joint United Nations on AIDS (UNAIDS, 1999) : HIV in Pregnancy: A Review. 67 p World Health Organization, (2006) WHO Case Definitions of HIV for Surveillance And Revised Clinical Staging and Immunological Classification of HIV-related disease in Adults and Children, August 7, 2006. Downloaded from http: //www. womenchildrenhiv. org/wchiv? page=charts-00 -01 on 4 th of April, 2009. WHO, 2006 Antiretroviral Drugs for Treating Pregnant Women and Preventing HIV Infection in Infants in resource Limited Settings: Towards Universal Access, Recommendations for a public health approach 2006 version WHO/ UNAIDS: AIDS Epidemic update, December, 2007


